Abstract
Immunoassays have long been widely used in a variety of applications, such as for medical diagnostics, pharmaceutical analysis, environmental, food safety testing, and for basic scientific investigations because of its simplicity, sensitivity, and specificity. Microfluidic systems, also well known as a “lab-on-a-chip” or a “micro-total-analysis-system” have attracted a lot of attention in the past two decades because of advantages associated with miniaturization, integration, and automation. A promising platform for the combination of these two technologies, microfluidic immunoassays, has been extensively explored in recent years. The aim of this article is to review recent advancements in microfluidic immunoassays. A brief introduction to immunoassays and microfluidic devices will include a literature review, followed by an in-depth discussion of essential techniques in designing a microfluidic-based immunoassay from different perspectives, including device substrates, sample/reagent transportation, surface modification, immobilization, and detection schemes. Finally, future perspectives on microfluidic immunoassays will be provided. These developments with microfluidic immunoassays may provide a promising tool for automatic, sensitive, and selective measurements in practical applications.
Introduction
Immunoassays
Antibodies (Abs) are proteins produced in animals and human bodies by immunological responses to the presence of allochthonous substances called antigens (Ags). They have a highly specific affinity for these Ags in nature. Each Ab has a unique structure recognized by a corresponding Ag in a lock-and-key mechanism. Immunoassays have a variety of formats, all of which make use of the sensitivity and specificity of this Ab—Ag interaction, which allows for the quantification and monitoring of small molecules, such as drugs and metabolites, 1 large proteins, 2 nucleic acids, 3 and even whole pathogens. 4 They have been widely used in clinical analysis, food safety and environmental monitoring, and basic bio-technological investigations.
Immunoassays can be used to detect either Abs or Ags according to the needs of the experiments. Generally, immunoassays can be classified as either a competitive or a noncompetitive format. 5 In the competitive format as shown in Eqs. (1) and (2), the unlabeled Ags competes with labeled Ags (Ag*) for a limited number of Ab-binding sites. As the amount of unlabeled Ags in a sample increases, the amount of labeled Ags bound to the Abs decreases, resulting in a decrease in the detection signal if the Ab-bound Ags (Ag*—Ab) are detected, or an increase in signal, if the labeled free Ags are detected.

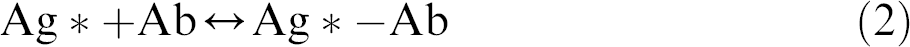
Alternatively, in the noncompetitive format, as shown in Eq. (3), Ags in a sample conjugate with an excessive amount of labeled Abs (Ab*) to form a complex that is strongly dependent on the number of Ags, resulting an increase in the detection signal as the Ags in the sample increases.
Immunoassays can also be divided into heterogeneous and homogeneous formats. 5 In a heterogeneous format, Abs or Ags are immobilized on a solid substrate where the complex forms. On the other hand, in a homogeneous format, the conjugation takes place in the solution phase. Heterogeneous immunoassays take advantage of the high surface area/volume ratio and result in good performance in sensitivity. However, inextricable steps are sometimes required for the immobilization of Abs or Ags on the solid substrate. Homogeneous immunoassays benefit from the multiplexing and fast electrophoretic separations. However, preconcentration steps are usually required because the surface area of the substrate is not used for immobilization of Abs or Ags. Both formats have been extensively studied and can be easily implemented in microfluidic devices. This section only provides a brief introduction to immunoassays. Detailed information about immunoassays can be found in the literature. 6 –8
Microfluidics
Microfluidic systems fabricated by microelectromechanical systems (MEMS) technology are now usually referred to as“lab-on-a-chip” (LOC),“biochips,” or“micro-total-analysis-system.” They are often envisioned as miniaturized versions of their large-scale counterparts. These miniaturized systems can carry out entire protocols traditionally performed in a laboratory. Sample pretreatment, sample/reagent transport, mixing, reaction, separation, detection, and product collection can all be performed automatically on a single LOC system. Functional microfluidic devices, such as micropumps, microvalves, microfilters, microreactors, and microseparators can be microfabricated and even integrated to perform a specific assay. The advantages of these developed LOC systems include less sample/reagent consumption, a reduced risk of contamination, enhanced sensitivity, less unit cost, lower power consumption, and a higher reliability and functionality. More importantly, portability arising from their compact form is a key factor for point-of-care (POC) applications. Despite these advantages, there are still potential limitations, such as bubbles formation and dead volume in microfluidics that need to be addressed. The bubbles formed in microdevices disrupt the continuity of the liquids and may result in poor performance. This is particularly serious in electrophoresis-based microdevices because the bubbles disturb the applied electric field. 9 Dead volume is another key issue in microfluidics. In the microscale environments, dead volume may cause serious contaminations and hence poor results. 10 The works in LOC have grown rapidly in the past two decades, and many review articles are available. 11 –15 In this article, only recent advancements in microfluidic immunoassays are covered here.
Immunoassays in Microfluidic Systems
The process in most immunoassays includes a series of washing, mixing, and incubation steps, which are labor intensive and time consuming, which often takes several hours, sometimes even up to 2 days to perform one single assay. Most of the time required in a long immunoassay is mostly because of the long incubation time attributed to inefficient mass transport for the immunoagents to move from a solution to the surface where the conjugation occurs because the immunoreaction itself is relatively rapid. 16 Moreover, the immunoagents used in immunoassays are relatively expensive. The consumption of the immunoagents can be greatly reduced if the system is miniaturized. Therefore, there is a demand to develop an automated and miniaturized platform for immunoassays. Such a platform must be capable of simplifying procedures, reducing the assay time and sample/reagent consumption and enhancing the reaction efficiency. The advantages of the microfluidic systems described previously fulfill these important criteria for immunoassays. Therefore, extensive investigations using microfluidics for performing immunoassays have been reported recently. The following sections introduce some representative original articles in microfluidic immunoassays published between the years 2005 and 2009. It should provide readers a comprehensive understanding of this promising technology.
Substrate Materials for Microfluidic Immunoassays
Microfluidic devices for immunoassays can be fabricated from a variety of materials. The most commonly used substrate materials are silicon, glass, and polymers. Each of these materials has its own advantages and limitations. In this section, a brief introduction to these materials is first given and a comparison of material properties is listed in Table 1.
Comparison of silicon, glass, and polymer as materials for microfluidic immunoassays

Although glass and polymers are optically transparent, they may adsorb light in the UV spectrum.
Although all the materials are capable of being mass produced, the fabrication processes for silicon and glass are relatively slow. For polymers, replication methods for PDMS are not as convenient as those used for PC, PS, and PMMA in practical and commercial aspects.
Silicon
Silicon is used as a material for fabricating microfluidic devices because of the well-established microfabrication processes developed by the microelectronic and the MEMS industry and from extensively studied surface chemistry. Furthermore, it possesses good thermal conductivity and is resistant to high temperatures; therefore, is suitable for applications requiring a relatively high operating temperature, such as for a polymerase chain reaction (PCR) and for bioreactions. The most popular fabrication method for silicon-based microfluidic structures is wet etching, 17 usually resulting in microstructures with low aspect ratio features that may be a limitation in most applications. This issue may be addressed by using dry etching, 18 which is a relatively expensive microfabrication process. Moreover, silicon is not optically transparent and is electrically conductive, thus resulting in limitations for optical and electrochemical detection and for electrokinetic transportation, which are key issues with microfluidic immunoassays. More importantly, silicon substrates are relatively expensive when compared with other materials, such as glass and polymers. Furthermore, the fabrication process for silicon-based microfluidic devices involving substrate cleaning, resist coating, photolithography, development, and wet/dry etching is relatively time consuming and costly. These limitations hinder its practical applications in commercial immunoassays.
Glass
In contrast to silicon, glass substrates are less expensive, optically transparent throughout the visible spectrum, and not electrically conductive. These properties overcome some of the problems associated with silicon-based microfluidic devices. Therefore, glass seems to be an ideal substrate for performing microfluidic immunoassays. However, glass-based microfluidic devices are usually fabricated by a wet etching method, 19 which still limits the etched microstructures to low aspect ratio features. Again, some expensive dry-etching processes for glass can be applied to address this limitation. 20
Polymers
Polymers are alternative materials to silicon and glass in microfluidic immunoassays because of their relatively low cost and simple fabrication process. Many polymers have been used in microfluidic applications, such as polymethyl methacrylate (PMMA), 21 polycarbonate, 22 polystyrene (PS), 23 and polydimethylsiloxane (PDMS). 24 Among these polymers, PDMS is one of the most commonly used materials for microfluidic immunoassays in recent studies because of its desirable characteristics, such as flexibility, optical transparency (down to 230 nm), and biocompatibility. PDMS is a polymer composed of repeating OSi(CH3)2 monomers, resulting in a hydrophobic surface, which is the major limitation when used with immunoassays because of the nonspecific adsorption of proteins and other molecules, poor wettability with aqueous solutions, and the formation of bubbles. 25 This disadvantage, however, can be easily overcome by bulk/surface modification and well-developed functionalization techniques. 26 –31
Heterogeneous Substrate Materials
Recently, heterogeneous materials formed by using two different types of materials for microfluidic devices have been extensively explored. For example, PDMS/glass 32 and silicon/glass 33 are commonly used for forming microfluidic devices. These hybrid microfluidic devices provide advantages attributed to their component materials. For example, a microchip made of PDMS and glass can fulfill the requirements of fast fabrication, low cost, flexibility, biocompatibility, and good thermal conductivity.
Sample/Reagent Transport
Liquid transport plays an important role in microfluidic immunoassays because the performance of the liquid driving system can directly affect the results of the assays. Many strategies for on-chip liquid transport in microfluidic immunoassays have been reported in the literature, such as electroosmotic, 34,35 electrowetting, 36 –38 pneumatic, 39 –42 centrifugal, 43 –48 power-free, 49 piezoelectric, 50 and thermopneu-matic 51 approaches. A comparison of these on-chip liquid transport strategies is listed in Table 2. External power sources, valve integration, and flow rates for each pumping method are also listed.
Comparison of strategies for on-chip sample/reagent transportation in microfluidic immunoassays

Although on-chip valves are not integrated in the electrowetting-based transport method, liquids can be immediately stopped when the electric voltage is not applied.
The flow rate for electrowetting is normally expressed as a flow velocity, and ranges from 0.01 to 0.1 m/s for a droplet with a radius less than 10−3 m.
Electroosmotic Pumping
Electroosmotic pumping based on electroosmotic flow (EOF) 34 is a commonly used liquid transport strategy in microfluidic systems because of the ease of automation and no moving parts are required. EOF is the bulk flow resulting from the effect of the electric field on the solution double layer at the channel wall. Counterions (typically, cations) building up near the wall surface to maintain charge balance form the double layer and create a potential difference very close to the wall surface, which is known as the zeta potential. The zeta potential is determined by the surface charge on the channel wall. Because this charge is strongly pH dependent, the magnitude of the EOF varies with pH. The zeta potential is also dependent on the ionic strength of the buffer. Increased ionic strength results in double-layer compression, decreased zeta potential, and hence reduced EOF. As the electric field is applied, the cations forming the diffuse double layer are attracted toward the cathode. Using this mechanism, the steps for flow switching, sequencing, and stopping can be easily achieved by controlling the applied electric field. For example, Gao et al. 35 developed a microchip for simultaneous detection of multiple microbial Ags based on electroosmotic pumping. As shown in Figure 1, Ags were first adsorbed onto a PDMS-coated glass slide by using a microfluidic network, followed by bonding to another PDMS slab-bearing H-shaped microchannels designed for delivering solutions of the primary Ab, the washing buffer, and the secondary Ab. The sequential pumping of these solutions was achieved by using a programmable high voltage sequencer equipped with eight independent outputs. Using this method, the determination for
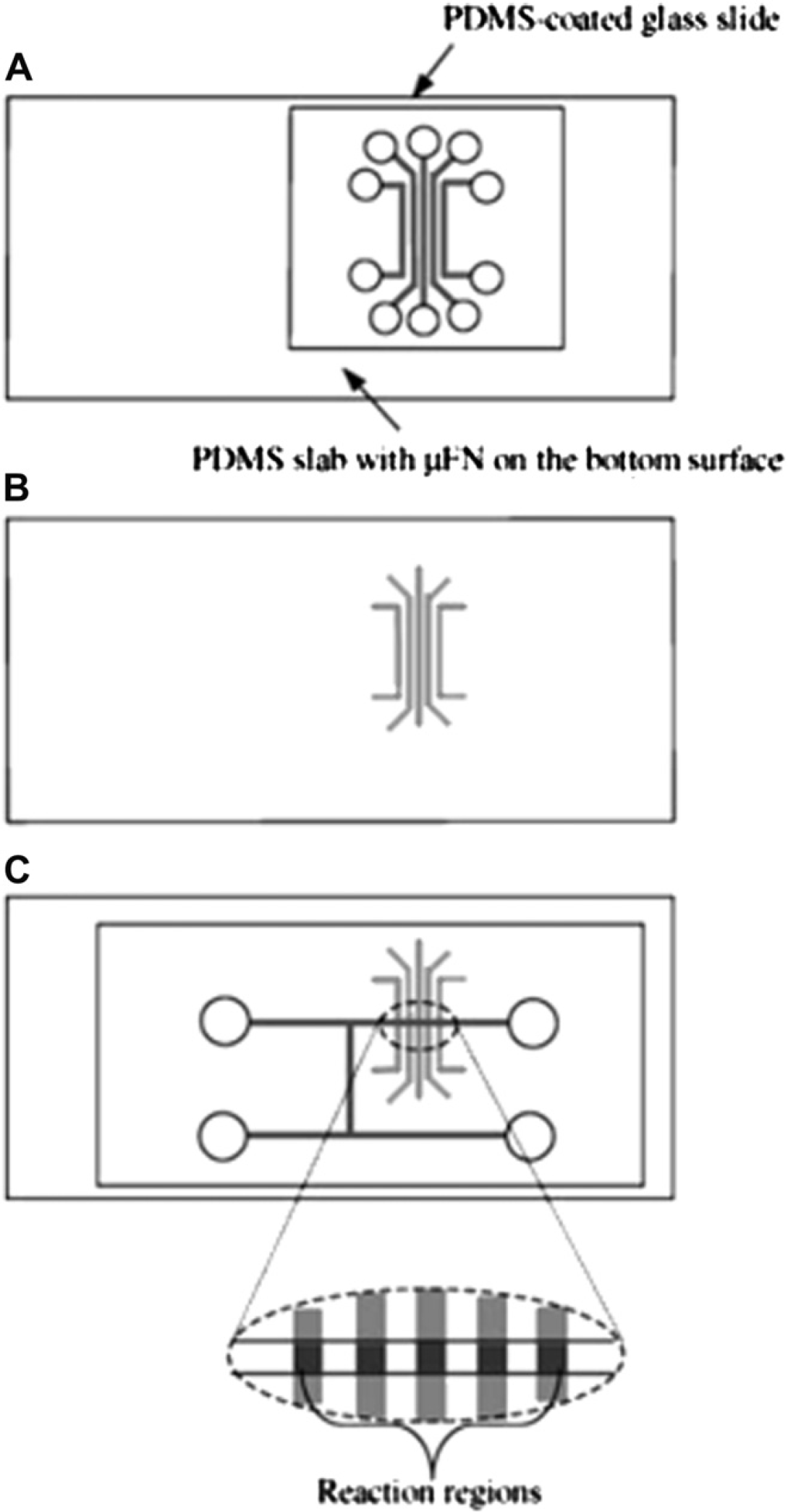
The fabrication process of the microchip for simultaneous detection of multiple microbial antigens based on electrokinetic pumping. (A-B) Antigen immobilization and (C) bonding with a PDMS slab-bearing H-shaped microchannels and forming the reaction regions. Reprinted from Ref. 35 with permission from Springer Science+Business Media (Copyright 2009 Springer Science+Business Media).
Electrowetting Force
Electrowetting is a liquid transport strategy whereby liquid droplets are manipulated in the presence of programmed voltage sequences applied to an electrode array. 36 It can be a promising platform for performing immunoassays. For instance, combining electrowetting and magnetic microbead-based immunoassays, Sista et al. 37 demonstrated a microfluidic chip for performing heterogeneous immunoassays. As shown in Figure 2, a sample droplet and a reagent droplet containing capture Ab-conjugated magnetic beads, reporter-conjugated Abs, and blocking proteins were dispensed on the surface of the chip. These two droplets were then merged, mixed, and incubated by electrowetting-based transport, followed by applying a magnetic field to capture the sandwiched microbead complexes. Then, unbound components were split from the merged droplet, followed by the introduction of the washing buffer. The splitting and washing steps were repeated several times to thoroughly remove unwanted components. Finally, a reagent droplet was added for chemiluminescent detection. Using this electrowetting system, a sandwiched heterogeneous immunoassay on human insulin and interleukin-6 (IL-6) has been successfully performed within 7 min. This electrowetting-based microfluidic platform can be also successfully used for the identification of a cardiac marker, troponin I, from whole blood samples, 36 sample preparation for bacterial pathogens indicating infectious diseases, 36 and the extraction of human genomic DNA. 38 It is believed that the electrowetting-based microfluidic immunoassay is adaptable for POC applications because of its relatively low cost and simple instrumentation.

Merging (A), incubation (B), immobilization (C), splitting (D), and washing (E) processes in an electrowetting-based immunoassay. Reprinted from Ref. 37 with permission from the Royal Society of Chemistry (RSC) (Copyright 2009 The Royal Society of Chemistry).
Micropneumatic Pumping
The micropneumatic pump is composed of a liquid flow microchannel, flexible PDMS membranes, and air chambers to pneumatically deflect the PDMS membranes in sequence via the introduction of compressed air. 39 –42 As shown in Figure 3, the peristaltic effect driven by the time-phased deflection of PDMS membranes along the microchannel can be used to deliver liquid in the microchannel. It has been demonstrated that micropneumatic pumps, incorporated with microvalves, can be used to determine the presence of Abs specific to hepatitis C virus and syphilis from serum samples. 39 In this work, the Ag was first coated on the surface of a detection area. Then, serum samples, a washing buffer, a horseradish peroxidase (HRP)-labeled secondary Ab, a developing buffer, and a stopping buffer in individual reservoirs were sequentially transported to the detection area by the micropneumatic pump, followed by optical detection via the measurement of absorbance. Usually it takes at least three electromagnetic valves to control the sequential deflection of the PDMS membranes. Alternatively, similar micropneumatic pumps have been reported, in which different-sized air chambers were interconnected by air channels to transport liquids in microchannels. 40 Because the interconnecting air channels allowed for the sequential actuation of the air chambers, this generated a peristaltic-like activation of the PDMS membranes, only one electromagnetic valve was required to perform the pumping. By using this micropneumatic pump, bead-based flow cytometry using immunoassays was performed in a microchip. 41 The delivery of virus samples, a washing buffer, virus-specific-Ab-conjugated magnetic beads, dye-labeled Abs, and sheath flows for focusing the sample stream were all transported by the micropneumatic pump. Using this micro flow cytometer chip, the entire process, including sample/reagent delivery, mixing, incubation, washing, detection, and the collection of beads was completed within 40 min. Recently, another micropneumatic pump was reported, which was incorporated with a normally closed valve for generating high pumping rates and to prevent any backflow from the destination wells after releasing the compressed air. 42 Using this liquid transport method, it avoided contamination from the waste chambers, and this design is expected to be applicable for many future microfluidic applications.

The sequential deflection of PDMS membranes in a micropneumatic pump.
Centrifugal Force
The centrifugal force has been used for liquid transport in compact disc (CD)-based immunoassays in recent years, and some review articles are available. 43,44 Immunoassays on a CD are a promising platform to realize many POC applications by embedding many analytical functions performed in a laboratory, such as sample/reagent transport, metering, dilution, mixing, separation, and detection. All these functions can be performed automatically in a disc format. It has been expected to be a powerful platform for medical and clinical diagnostics. It has great commercial potential because of simple instrumentation, ease for economic mass production, and easy adaptation to existing detection methods, particularly those based on optical detection. Moreover, with the ability to perform simultaneous and identical functions in parallel layouts, immunoassays on a CD are an ideal platform for high throughput screening of analytes. Figure 4 gives a schematic illustration of a CD-based sandwich enzyme-linked immunosorbent assay (ELISA). The key concept is to overcome the capillary force, which prevents the liquid in one chamber from moving out, by using the centrifugal force controlled by the rotational speed of the CD. Furthermore, the increasing centrifugal force from the center toward the edge of the CD is used for flow sequencing which replaces the stepwise procedures in a conventional ELISA. Not only can ELISA be performed on this CD-based platform but also other immunoassays can be realized using a very similar approach. For more detailed information such as the operating theory and instrumentation, readers can refer to various review articles. 43,44

ELISA in a lab-on-a-CD. (A) The 4-array CD-ELISA design, and (B) a single assay. Chambers 2, 4, 6, and 8 are loaded with washing buffer, whereas chambers W, D, 1, 3, 5, 7, 9 are designed for waste, detection, first antibody, blocking solution, sample, second antibody, and substrate, respectively. As the CD rotates, the solutions in chambers 1 to 9 flow out sequentially because of the decreasing centrifugal force and then complete the ELISA procedures. The most common valves are hydrophobic and capillary valves. Hydrophobic valves stop the flow by the hydrophobic surfaces created in the microchannel, and capillary valves stop the flow by a capillary pressure barrier at junctions where the channel diameter suddenly expands.
This section will focus on some representative works on CD-based immunoassays published between the years 2005 and 2009. The first centrifugal fluidic platform was developed in 1969 45 and has been extensively studied since then. Companies, such as Gyros AB, Tecan, and Burstein Technologies also developed commercial products. Recently, Gyros AB developed a CD-based immunoassay for the determination of α-fetoprotein (AFP), IL-6, and carcinoembryonic antigen (CEA). The disc structure is an intricate web of microchannels and reaction chambers packed with streptavidin-coated PS microparticles for conjugating with biotinylated Abs against AFP, IL-6, and CEA. Alexa 647-labeled bovine serum albumin (BSA) was used as a detection Ab. All the flow control is through the rotation of the CD mounted on a workstation equipped with a detection system (laser-induced fluorescence, LIF) and robotic arms for dispensing liquids into the inlets on the CD. The experimental results showed that the detection limits for AFP, IL-6, and CEA were 0.15, 1.25, and 1.31 pmol/L, respectively. 46 The inter-and intra-assay coefficients of variation were less than 10% and 20%, respectively. This disc also demonstrated an excellent analytical efficiency (104 assays in 50 min) when compared with the performance of traditional 96-well ELISA plates (96 assays, a few hours). Moreover, the relationship between the fluorescent imaging profiles of the column structure and the kinetic properties of the Ag and Ab was also investigated.
Although the disc developed by Gyros AB gives an excellent analytical efficiency within acceptable variations, an expensive robotic unit is inevitably required. For practical applications, a cheap and fully automated CD immunoassay without a robotic system is required. To achieve this, valves capable of precisely controlling the flow sequences in an immunoassay are required. For instance, a disc with superhydrophobic capillary valves was reported to perform the sequential flow steps. 47 A capillary valve is the most popular valving component in CD immunoassays because it requires no moving parts or external actuation. Unfortunately, proteins present in solution may adsorb onto conventional capillary valves to gradually make them hydrophilic, leading to the failure of the valve. To solve this problem, polyaniline nanofibers along with a noncrystalline fluorine coating to create superhydrophobic nanostructures as valves have been reported. 47 Measurements showed that the contact angle before and after protein blocking were 170° and 165°, respectively, indicating that these valves maintain their superhydrophobic properties after treatment with protein solutions. This excellent result is because of the increased surface roughness and decreased surface energy attributed to the coating of polyaniline nanofibers and noncrystalline fluorine, respectively. Furthermore, to improve the detection sensitivity of the assay, the detection surface was sequentially treated by an oxygen plasma, polyethyleneimine (PEI), tyrosinase, and protein A to increase its binding capacity based on orientation-controlled immobilization. This disc integrates functions, including pumping, valving, fluid splitting, washing, and mixing. Automation, without using a robotic unit, was achieved because the primary Ab and blocking protein were preimmobilized onto the detection area. The substrate, conjugate, washing, secondary Ab, and Ag solution were loaded into corresponding wells for flow sequencing. Comparing with a conventional 96-well ELISA plate, this disc has advantages that included minimized sample/regent consumption, shorter assay time, and a broader dynamic range.
Another strategy to control the transport of liquid in a CD immunoassay is to use active valves. For example, a disc with a laser irradiated ferrowax microvalve (LIFM) that can be actively controlled to open and close by laser irradiation was reported. 48 LIFMs are valves made of nano-composite materials in which nanosized iron oxide particles are dispersed in paraffin wax. They are phase change-based microvalves and can be fabricated as normally open, normally closed, and reversible microvalves. Low-intensity laser light can melt the paraffin wax with the embedded iron oxide nanoparticles, whereas laser light with a high intensity cannot melt the wax. Using this mechanism, the LIFMs can be actively opened or closed by the irradiation of laser light during the assay process. This disc contains plasma separation channels and chambers for a substrate solution, washing buffer, stopping buffer, mixing, waste, and detection. These chambers are connected by microchannels where LIFMs are fabricated in a normally open or normally closed state. Figure 5 shows a schematic illustration of the disc design and the reaction steps involving in the active opening and closing of the LIFMs. In this disc, valves 3, 6, 9, and 12 are the normally open LIFMs where all other valves are the normally closed LIFMs. Using these LIFMs together with precoated PS microbeads, the concentrations of the antibody to the hepatitis B surface antigen (anti-HBs) and hepatitis B virus surface antigen (HBsAg) from whole blood samples can be determined in an automated manner within 30 min without using robotic arms, which is a much shorter time when compared with conventional ELISA plates (longer than 90 and 120 min for anti-HBs and HBsAg, respectively).

(A) Schematic illustration of the disk design. (B—G) The reaction principle of the CD immunoassay to detect anti-HBs. The lightning symbols indicate the laser irradiation on the LIFMs. Reprinted from Ref. 48 with permission from the Royal Society of Chemistry (RSC) (Copyright 2009 The Royal Society of Chemistry).
Power-Free Transport
Most of the liquid transport devices require external power sources to drive the liquids. For example, the electro-wetting method requires the application of a voltage, the micropneumatic pump needs compressed air generated by a bulky pump to deflect the membranes, and the centrifugal force-based transport method uses a rotary motor to spin the CD. Taking into consideration the demands of POC applications, a power-free liquid transport method, such as that used in commercial immunoassay strips, is also a promising alternative. For example, Hosokawa et al. 49 developed a power-free injection method for a microfluidic immunoassay by air evacuation. As shown in Figure 6, a microchip composed of PDMS microchannels and a glass bottom plate was first degassed in a vacuum system to store the pumping energy, followed by covering them with a piece of adhesive tape. When this microchip was brought back to atmospheric conditions, air dissolved into the PDMS flows again through the walls of the microchannels and reservoirs. Once an aliquot was pipetted at the inlet of the microchannel, the dissolution of air reduces air pressure in the microchannelreservoir system, and hence pumps the solution into the microchannel. Because the inlet of the microchannel is narrow and wettable, a capillary retention valve is naturally formed to prevent the microchannel from drying out. When a second aliquot was pipetted at the inlet of the microchannel, the pumping effect occurred again. Using this method, sample and reagents required for performing an immunoassay were sequentially injected into the microchannels for immobilization, blocking, washing, reaction, and detection. It has been used to demonstrate the detection of rabbit IgG and human C-reactive protein (CRP) with a detection limit of 0.21 and 0.42 nM, respectively, which can be performed within 20 min. Power-free liquid transportation is promising because one of the ultimate goals for microfluidic immunoassays is to perform these chips for POC applications outside the laboratory with minimum support equipment.

Schematic illustration of a microchip based on power-free liquid transport. (A) The chip design, and (B) the pumping mechanism. Reprinted from Ref. 49 with permission from the Royal Society of Chemistry (RSC) (Copyright 2006 The Royal Society of Chemistry).
Heterogeneous Microfluidic Immunoassays
Immobilization is a key step in heterogeneous immunoassays because it greatly influences the specificity and sensitivity. Briefly, the surface of these microfluidic chips has to be treated so that proteins can be immobilized for further detection. In general, heterogeneous microfluidic immunoassays can be classified as surface-based and bead-based immobilization methods. In heterogeneous microfluidic immunoassays, the Abs or Ags are usually immobilized on the surfaces of the microchannels or microbeads. Immobilization on surfaces of microchannels requires additional steps during the microfabrication process and may suffer from poor reproducibility and reliability. In contrast, microbead-based immobilization can be performed outside the microchip and offers a significantly large surface for immobilization, and hence improved performance of the immunoassays. 52,54
Surface-Based Immobilization
Many surface-based immobilization strategies have been developed for microfluidic immunoassays. Different substrate materials, such as PDMS, 55–59 cycloolefin-copolymer, 60 PS, 61,62 silicon nitride (SN), 63 and PMMA, 64 have been reported for use in microfluidic devices. Among them, surface immobilization on PDMS will be emphasized in this section because it is now the most popular material for fabricating microfluidic devices. For example, a method called“macro-molecules to PDMS transfer” was reported to directly entrap macromolecules for capturing target analytes during the PDMS polymerization step. 55 In this work, rabbit IgG or anti-CRP solutions were spotted on a Teflon master by a piezoelectric arrayer, followed by pouring liquid PDMS on top. The PDMS membrane was then peeled off after curing, and the molecules (rabbit IgG or anti-CRP) were thus transferred to the PDMS membrane. The rabbit IgG and anti-CRP-conjugated PDMS membranes were then used to capture rheumatoid factor (RF) and free CRP in human serum samples, respectively, followed by conjugating with peroxidaselabeled detection Abs for chemiluminescence detection. Using this method, the sensitivities for CRP and RF measurements were 12.5μg/L and 5.3 IU/mL, respectively. Due to its simplicity and efficiency for immobilization, this method has a great potential to be applied to fabricate PDMS-based microchip for immunoassays.
Alternatively, Hashimoto et al. 56 developed an electrochemical method to perform localized immobilization of proteins within the channels of a microchip. In this work, a layer of PDMS containing micropost structures was first attached to the inner surface of an upper glass substrate by an oxygen plasma treatment before assembling with a bottom glass substrate where Pt electrodes were deposited. After assembly, a solution containing PEI and heparin was introduced to electrostatically form a PEI/heparin multilayer on the wall of the channel. To obtain an area for protein immobilization, the channel was filled with a solution of potassium bromide, followed by applying an electrical pulse to the patterned electrodes to locally produce Br2 and HBrO. The oxidation action of HBrO then caused the PEI/heparin multilayer to locally detach from the substrate, and the exposed area (PDMS) was thus ready for localized immobilization because heparin is resistant to the adhesion of many proteins. Using this method, antiglutathione peroxidase (GPX) was locally immobilized to capture GPX, followed by conjugation with Cy3-labeled anti-GPX for fluorescent detection. This breakthrough work provides a simple method for immobilizing proteins in predefined areas and is believed to be promising for surface immobilization.
Supported bilayer membranes (SBMs), such as phospholipid bilayer membranes are biomimetic membranes capable of providing long-term hydrophilic and protein-resistant properties. Therefore, they have been reported for surface immobilization. For example, Phillips and Cheng 57 used phosphatidylcholine membranes assembled on PDMS as a supported membrane for protein immobilization. In this work, a phosphatidylcholine membrane was first assembled on an oxygen plasma-treated PDMS surface, followed by vesicle fusion to form cell surface ligand GM1-integrated SBMs for capturing the cholera toxin (CT). Then, rabbit anti-CT and a dye-labeled goat antirabbit Ab were sequentially conjugated for fluorescent detection. Using this method, the entire assay can be completed within 25 min with a detection limit of 8 fmol for the measurement of CT. The SBMs, however, may peel away from the solid support when exposed to air. Therefore, reinforced SBMs (r-SBMs) were developed to solve this problem and sensing of
Another method for immobilization is to use dextran, which is a biocompatible polymer. The immobilization of dextran on a microchip surface by introducing linkers for protein immobilization is a less toxic and inexpensive process. More importantly, the hydrophilicity and immobilization efficiency can be enhanced because of the abundant oxidation-resultant aldehyde groups. For instance, Yu et al. 59 used this dextran-based immobilization to simultaneously detect three biomarkers. In this work, a solution containing H2O and HCl was first introduced to oxidize the surface of the PDMS channels, followed by the introduction of a solution containing 3-aminopropyltriethoxylsilane (APTES) and ethanol to make the surface abundant with amine groups. Then, the partially oxidized dextran (with aldehyde groups) solution was introduced to conjugate onto the channel surface via the formation of shiff's base between the amines and aldehydes, followed by treating with NaIO4 to oxidize the dextrans again. The surface is now aldehyde abundant and is ready to react with the amine groups of proteins. Capture Abs, such as anti-IL5, anti-HBsAg, and anti-rabbit IgG were immobilized in different channels to capture IL5, HBsAg, and rabbit IgG, respectively, followed by introducing HRP-labeled detection Abs for colorimetric detection. Using this method, a detection limit of 100 pg/mL was obtained for these three markers.
In addition to PDMS, dextran can be also used in a cyclo-olefin-copolymer substrate for protein immobilization. For instance, önsson et al. 60 first treated a copolymer chip with an oxygen plasma and APTES to expose amine groups, followed by a sequentially treatment with partially oxidized dextran and NaIO4 to make the channel surface aldehyde abundant. Then, capture Abs for CRP were immobilized for performing immunoassays. Using Alexa 647-labeled detection Abs for fluorescent detection, a detection limit of 2.6 ng/mL was obtained. These dextran-based immobilizations are long-term stable, inexpensive, and capable of being produced in large quantities, resulting in the possibility for commercialization.
Other materials, such as PS, SN, and PMMA have also been used as substrates for microfluidic immunoassays. For example, Darain et al. 61,62 used different strategies to immobilize proteins on PS substrates. In one work, 61 the PS substrate was sequentially treated with ultraviolet (UV)/O3 (to introduce OH group), APTES (to introduce NH2 group), and glutaraldehyde (to introduce CHO group), followed by conjugating with antimyoglobin for capturing myoglobin. In another work, 62 the PS substrate was sequentially treated with sputtered gold (to introduce a Au surface), 16-mercapto-hexadeconic acid (to introduce COOH groups), and N-hydroxy succinimide ester and 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide hydrochloride (to introduce active NSH esters), followed by conjugating with antihorse IgG, for capturing horse IgG. After fluorescent detection, the detection limit for myoglobin and horse IgG was obtained as 16 ng/mL and 0.71μg/mL, respectively. These two PS-based immobilization strategies can be extended for the measurement of other important biomarkers for diagnostic applications.
SN has been widely used in the semiconductor industry because of its excellent chemical, electrical, and optical properties. It can also be used as a substrate for microfluidic immunoassays. For instance, He et al. 63 used SN as the substrate for immunosensing of CRP in two formats. The first format is a two-layer system in which anti-CRP was directly patterned on a SN-coated silicon wafer as the first layer to capture dye-labeled CRP; the second format is a four-layer system in which biotinylated BSA was first adsorbed on a SN-coated silicon wafer, followed by sequential conjugation with avidin, biotinylated anti-CRP, and dye-labeled CRP. The biotinylated BSA works as the first adhesion layer and as a blocking material as well. Therefore, another blocking solution is not required in this format. Using 1.76μM of CRP as the analyte, the fluorescence intensity obtained from the four-layer system is 3.2-fold stronger than that obtained from a two-layer system.
PMMA has been used as a substrate for microfluidic devices because of its relatively low cost and optical transparency. For example, Wang et al. 64 used PMMA microchannels filled with an Al2O3 sol—gel network to encapsulate capture Abs for immunoassays. In this work, the inner surface of the PMMA microchannel was first treated with a copolymer to bear silanol groups, followed by introducing a solution containing anti-IgG and Al2O3 sol gel to allow polycondensation between the sol gel and the silanol groups. The anti-IgG was thus encapsulated in the network for capturing target IgG. After conjugating with alkaline phosphatase (ALP)-labeled secondary Abs, electrochemical detection was used with a detection limit of 1 pg/mL. The excellent sensitivity is attributed to the high surface area-to-volume ratio of the network. Table 3 summarizes the aforementioned surface-based immobilization strategies. The substrate selection, target analyte, immobilization scheme, detection strategy, and detection limits have all been summarized.
Summary of surface immobilization strategies in microfluidic immunoassays

Bead-Based Immobilization
As mentioned previously, microbead-based immobilization offers a large surface area and hence may improve the performance of the immunoassays. In the following sections, bead-based immobilization methods classified as either nonmagnetic or magnetic microbeads in microfluidic immunoassays will be reviewed and discussed.

Schematic illustration of the detection method based on immunogold silver staining. Reprinted from Ref. 65 with permission from Wiley InterScience (Copyright 2008 Wiley-VCH Verlag GmbH & Co. KGaA).
Alternatively, Lee et al. 66 used glass beads to detect a bacterial pathogen. In this work, glass beads were sequentially treated with 3-aminopropyl trimethoxysilane, glutaraldehyde, and a capture Ab (anti-O157 Ab). These beads were then packed in spatially isolated microchambers, followed by introducing l-lysine solution for blocking unreacted surfaces. Samples containing
Most of the previous works only used the outer surface of the beads for immobilization. To improve the sensitivity of an immunoassay, Yang et al. 67 used superporous agarose beads (superpores with diameters ranging from 10 to 80 μm) as a solid support for performing microfluidic immunoassays. In this work, superporous agarose beads were sequentially treated with protein A and anti-goat IgG to capture target goat IgG, followed by treatment with ALP-conjugated anti-goat IgG and 5-bromo-4-chloro-3-indolyphosphate/nitroblue terazolium for colorimetric detection. Using these superporous beads as immobilization materials, a detection limit of 100 pg/mL of goat IgG was obtained, which is 10 times more sensitive than conventional bead-based microfluidic immunoassays. 68 The improved sensitivity is mainly because of the large surface area attributed to the inner matrices of the super-porous beads.
Most nonmagnetic bead-based microfluidic immunoassays require the fabrication of microstructures to retain the beads for immunoreactions. Alternatively, Sivagnanam et al. 69 used a brilliant strategy to retain the beads. In this work, positively charged APTES were patterned on a glass substrate, followed by the electrostatic assembly of negatively charged streptavidin-coated microbeads. These self-assembled streptavidin-coated microbeads were then used to conjugate with a biotinylated capture Ab to capture the target Ags. Using this method, a target mouse IgG Ag was detected with a detection limit of 15 ng/mL and 250 pg/mL in a stop-flow and a continuous-flow mode, respectively. This approach requires no microstructures to retain beads and is promising for patterning complicated immobilization beads for multiplexed applications.
Bead-based immunoassays have also been used in flow cytometry since the 1980s, 70 with an improved sensitivity, specificity, and dynamic range. 71,72 Recently, Holmes et al. 53 developed a bead-based immunoassay performed on a microchip-based flow cytometer. In this work, commercial glycidyl methacrylate microbeads were sequentially treated with succinic anhydride, protein A, and human IgG or rabbit IgG to form human IgG or rabbit IgG-conjugated microbeads, respectively. These microbeads were then reacted with samples containing Cy3-labeled anti-human IgG and Cy5-labeled anti-rabbit IgG to form the complexes, followed by analysis in a micro flow cytometer. The micro flow cytometer used negative dielectrophoresis to focus the microbeads into a particle stream, and the impedance and fluorescence signals were measured for indicating particle size and for obtaining scatter plots, respectively. The results were comparable to those obtained by a commercial flow cytometer. Although the data from measurements of particle sizes were not used (accurate sizing of beads with diameters ranging from 2 to 10 μm), it has a great potential for bead encoding. Combining the bead encoding with fluorescence detection in a micro flow cytometer is believed to be a promising platform for multiplexed diagnosis.
There are a lot of studies on magnetic bead-based microfluidic immunoassays. Readers can refer to several articles for different applications, such as the determination of AFP, 78 cholera toxin subunit B, 79 and multiplexed immunoassays. 80
Homogeneous Microfluidic Immunoassays
Unlike heterogeneous immunoassays performed in microfluidic devices, a concentration step is usually required in microchip-based homogeneous immunoassays because the surface of the microchannel is not used for immobilization. Homogeneous microfluidic immunoassays are mostly performed in an electrophoresis-based microchip format, and many concentration strategies, such as the use of a preconcentration membrane or isotachophoresis (ITP) to stack the target analytes have been used for improving the sensitivity of detection. For instance, Meagher et al. 81 developed an electrophoretic immunoassay-based microchip incorporated with a photopolymerized membrane for preconcentration and polyacrylamide gel-filled microchannels for sample/reagent loading and Ab/complex separation. As shown in Figure 8, protein biotoxins (sample) were loaded through a larger pore-size polyacrylamide gel to upstream of the membrane, followed by trapping for preconcentration under the continuous application of an electric field. The capability of the membrane to perform preconcentration arises from its porous structure that allows the transport of buffer ions but excludes the proteins. Then, dye-labeled Abs were loaded and concentrated in the same area. After mixing and conjugating, unbound dye-labeled Abs and complexes were separated in the separation channel in which a smaller pore-size polyacrylamide gel was filled on switching the direction of the electric field. Using this method, the entire immunoassay can be performed within 20 min with a minimal sample volume of less than 10 μL and a detection limit of less than 10 pM for SEB determination. This microchip was also used for the determination of collagen-cleaving enzyme matrix metalloproteinase-8 for rapid saliva-based clinical diagnostics. 82 Although the aforementioned microchip performs very well for preconcentration and analysis of target analytes, a microchannel for the separation of unbound labeled Abs and the complexes was required. In another work, Reichmuth et al. 83 fabricated a membrane with proper pores (about 10 nm) in the microchannel. Due to these larger pores, this membrane allows the transport of both buffer ions and unbound Abs but excludes the viral particles (about 80 nm). Using this membrane-based concentration and filtration method, rapid detection of swine influenza virus was successfully performed within 6 min with a detection limit of 610 Truckee—Carson Irrigation District (TCID) 50/mL without an electrophoretic separation.
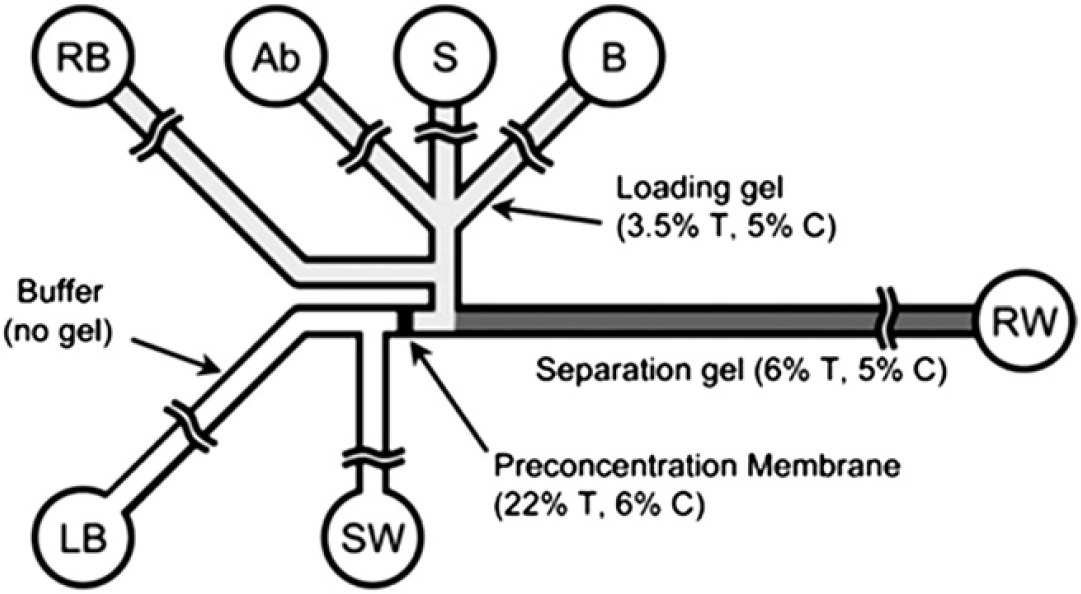
Schematic illustration of the electroosmotic microchip incorporated with a preconcentration membrane and a separation channel. S = sample, SW = sample waste, Ab = antibody, B = buffer (not used here), LB = loading buffer, RB = running buffer, RW = running waste. Reprinted from Ref. 81 with permission from the Royal Society of Chemistry (RSC) (Copyright 2008 The Royal Society of Chemistry).
ITP 84 is a commonly used preconcentration method in capillary electrophoresis and microchip electrophoresis. For instance, Kawabata et al. 85 developed an“electrokinetic analyte transport assay” microchip in which ITP was performed for on-chip concentration, mixing, and reaction, followed by electrophoretic separation and detection in the downstream channel. As shown in Figure 9, an electrophoretic microchip was designed for the formation of plugs of the tailing buffer, the first Ab, the sample, the secondary Ab, and the leading buffer in the channel by the application of vacuum pressure at the waste wells. On the application of an electric field between reservoirs 1 and 6, the first Ab was concentrated by ITP and migrated into the sample plug to form the first Ab—sample complex. This complex then migrated into a dye labeled-secondary Ab plug to form the sandwich complex (first Ab—sample-labeled secondary Ab). This sandwich complex was then separated from the other components by using a microchip gel electrophoresis through the application of an electric field between reservoirs 5 and 6. Using this ITP-integrated electrophoretic microfluidic immunoassay chip, AFP was successfully detected within 136 s with a detection limit of 5 pM. Similarly, Kagebayashi et al. 86 also used a microchip together with a desktop instrument containing components functionalized for chip loading/transportation/storage, pipetting, vacuum applications, conductivity monitoring, electric field generation, sample and reagent storage, and LIF detection to determine total AFP from serum samples. Good correlation with a commercially available reference method (LiBASys) for 68 patient serum samples was obtained. In summary, these on-chip electrophoretic immunoassays using membranes or ITP-based concentration and gel separation have a great potential to provide a rapid, sensitive, portable, and cost-effective method for both clinical and research applications.

Schematic illustration of the ITP-integrated microfluidic immunoassay chip. W = waste, 1 = tailing buffer, 2 = first antibody (DNA coupled anti-AFP WA1 antibody), 3 = sample (AFP), 4 = second antibody (Hilyte-labeled anti-AFP WA2 antibody), 5 = handoff well, 6 = leading buffer. Reprinted from Ref. 85 with permission from Wiley InterScience (Copyright 2008 Wiley-VCH Verlag GmbH & Co. KGaA)
Detection in Microfluidic Immunoassays
Generally speaking, in an immunoassay, one of the immunoagents must be conjugated with specific labeling molecules to facilitate the detection of the analytes. The most common detection techniques used in microfluidic immunoassays are fluorescent 41,46,47,49,53,56 –58,60 –63,65,69,77,81 –83,86 and lumines-cencent 37,55 detections because of their excellent sensitivity. Electrochemical detection is also popular because of its capability for miniaturization and simple instrumentation. 64,65 Readers can refer to the recommended references for more information about these detection methods. In the following sections, recent works in microfluidic immunoassays using different detection strategies, except those previously aforementioned detection schemes, will be reviewed and discussed. We mainly focus on label-free detection methods, including surface plasmon resonance (SPR), a thermal lens microscope (TLM), quartz crystal microbalance (QCM), and wavelength-interrogated optical sensor (WIOS) schemes. Detection methods based on the labeling of gold nanoparticles (colorimetric detection) and magnetic microbeads (magnetoelectric detection) are also introduced. These methods are classified as optical (SPR, TLM, WIOS, colorimetric detection) and nonoptical (QCM, magnetoelectronic detection) detections in the following sections.
Optical Detection

(A) Schematic illustration of a microfluidic system integrated with a 2-D SPR phase imaging system. (B) Schematic illustration of the microfluidic device comprising of a fluidic transport module and a temperature control module. Reprinted from Ref. 87 with permission from Elsevier (Copyright 2007 Elsevier).

A multiple micro ELISA system composed of valving and immunoreaction capillaries and a PDMS microchannel network. Reprinted from Ref. 91 with permission from Elsevier (Copyright 2007 Elsevier).

The sensing principle of a WIOS. Reprinted from Ref. 94 with permission from the Royal Society of Chemistry (RSC) (Copyright 2009 The Royal Society of Chemistry).
Nonoptical Detection
Conclusions and Future Perspectives
Microfluidic immunoassays may play an important role in the areas of clinical diagnostics, pharmaceutical analysis, environmental and food safety surveillance, and basic scientific investigations because of the capabilities for miniaturization, integration, and automation. However, to achieve this objective, integrating knowledge from fields, such as material characterization, fabrication methods, liquid transportation, surface modification, immobilization, and detection schemes need to be optimized and integrated. Therefore, critical issues in the following fields are important and should be addressed properly.
Mass Production
For commercial and disposability considerations, polymeric substrates are the dominant candidates in this field. Although PDMS has been widely used for fabricating prototypes of microfluidic devices for immunoassays, the fabrication process is still limited to a replication method involving several hours of curing time. Materials with similar properties and capabilities for mass production by techniques, such as injection molding and embossing are in great demand for practical applications.
Multiplicity
An important feature of microfluidic immunoassays is the ability to perform a multiplexed assay in a single chip. Recently, Zhao et al. 105 and Sun et al. 106 developed a suspension array for a multiplexed immunoassay using silica colloidal crystal beads (SCCBs) exhibiting different reflection spectra as carriers. After the reaction between a sample solution containing fluorescence dye-labeled anti-hIgG and anti-mouse IgG with a SCCBs suspension containing human IgG or mouse IgG-conjugated SCCBs, a multiplexed immunoassay can be successfully performed by a color-encoded method. By combining microfluidic devices with SCCBs-based multiplex immunoassays, it is believed to be a potential platform for clinical applications. The multiplexed assay will continue to be the dominant method for commercialization of these microfluidic immunoassays.
Surface Modification and Immobilization
Nonspecific adsorption or binding to molecules rather than analytes is a key concern in immunoassays because it may greatly impair the sensitivity and selectivity. This is a challenging issue in microfluidic immunoassays. Fortunately, surface chemistry for the functional modification of commonly used materials including device substrates and microbeads/microspheres has been well studied and developed and is expected to provide a solid foundation in microfluidic immunoassays. However, surface modification and immobilization of these new materials still requires tremendous research efforts.
Purification and Concentration
Due to the complexity and the low trace of analytes in biosamples, purification and concentration steps are often required in microfluidic immunoassays. Currently, micro-or nanoparticles and magnetic beads have been widely used for improving the sensitivity by performing a purification process. With a high surface area and ease of manipulation, these beads are expected to provide a promising method for one-step purification and concentration by combing these two techniques together (e.g., beads-on-beads configuration) 107 in microfluidic immunoassays.
Detection
Detection systems for microfluidic immunoassays are usually bulky and expensive, in contrast to other microcompo-nents, such as micropumps, microvalves, micromixers, and microheaters. Although some works on integrated detection system 108 have been developed, the cost, sensitivity, and fabrication processes still hinder their practical applications. Therefore, developing and integrating miniaturized, compact, portable, and inexpensive detection systems with an acceptable sensitivity onto microfluidic devices are still in great demand.
Integration, Packaging, and Cost down Issues
The ultimate goal for practical commercialization is to develop fully integrated, well packaged, disposable, and cheap microfluidic systems for immunoassays. Although many microfluidic devices have been demonstrated, most of them are operated by well-trained personnel in the laboratories and off-chip peripheral units, such as power sources, controllers, and detection systems are used. Moreover, the device cost is still relatively high because they are not massively produced. These factors greatly hinder their commercial applications. Fortunately, some efforts are made to address these problems in recent years. For example, a small-sized blood analyzer comprising a disc loader, a disc positioning, and rotation controller, a laser position controller, an optical detection unit, a temperature controller, and a user interface controller was developed by Lee et al. 48 Similarly, a prototype incorporating electronics, optics, and chip architecture for high throughput toxin diagnostics was developed by Meagher et al. 81 A desktop instrument containing the LIF detection system, mechanisms for chip loading and transport, electrophoresis power supply, a means for monitoring the buffer conductivity, chip storage cassettes, and refrigerated reagent storage compartment was developed by Kagebayashi et al. 86
It is envisioned that an integrated low-cost microfluidic immunoassays with a multiplexed detection function can be realized in the near future.
Storage of Reagents
Although many portable microfluidic devices have been well developed and applied in immunoassays in recent years, the long-term storage of bioreagents in portable microfluidic devices is rarely discussed. This is one important issue in portable devices because most bioreagents are not durable under room temperature and some of them require special environments for storage. Therefore, further efforts are required to address these demands.
Acknowledgment
The authors gratefully acknowledge the financial support provided by the National Science Council in Taiwan (NSC 96-2628-E-006-238-MY3).
