Abstract
An innovative lab-on-a-chip device based on iridium oxide (IrOx) nanowires was designed, fabricated, and developed for real-time “point-of-care” diagnostics. The turnaround time in detection and the need for expensive equipment for analysis have considverably limited rapid and “point-of-care” diagnosis. This research demonstrates the potential of IrOx nanowires toward early disease diagnosis by detecting proteins that are disease markers. The device designed is based on electrical detection of protein biomarkers wherein a single capture immunoassay is built onto a vertically aligned IrOx nanowire platform. Detection of two inflammatory proteins, C-reactive protein (CRP) and myeloperoxidase (MPO) (Calbiochem, La Jolla, CA), that are biomarkers of cardiovascular diseases is demonstrated individually and concurrently. The performance metrics of the device in response to the two biomarkers in pure form and in serum samples were evaluated and compared with standard enzyme-linked immunosorbent assay (ELISA). The methodology that has been adopted is based on measuring real-time impedance changes because of the protein binding event through electrochemical impedance spectroscopy and calibrating its change in magnitude with concentration of proteins. We have demonstrated limit of detection up to 500 pg/mL for protein MPO and I ng/mL for CRP with cross-reactivity of less than 8% of selective binding of these two inflammatory biomarkers.

Introduction
“Point-of-care” diagnostic testing, at or near the site of patient care, has become a highly active area of research with the significant advancements in chip design and fabrication technologies. With the emergence of nanostructured materials that enable nanoscale biomolecule confinement, a new class of ultrasensitive portable diagnostics tools has been developed for clinical health care applications. 1,2 However, identifying and matching new materials and technologies for a specific application have been the real challenge in designing and prototyping cost-effective, lab-on-a-chip-based diagnostics platforms. This research succeeds in demonstrating a potentially disruptive technology with the use of iridium oxide (IrOx) nanowires toward “real-time” point-of-care testing.
Many proteins act as disease markers, essentially marking the physiological condition of the disease or health disorder. Monitoring these proteins, called biomarkers, continuously with time, has been identified as a method for rapid and early disease diagnosis. 3,4 Label-free methods do not use labels and monitor the target protein directly, hence minimizing sample contamination and at the same time enabling high portability suitable for point-of-care detection when compared with labeled detection methods such as ELISA.
Electrical detection methodologies have an edge over other label-free methodologies because of the detectable electrical and dielectric properties of proteins. 5,6 The ionic charges primarily from the many ionizable groups on the side chains of their amino acids, makes proteins highly sensitive toward electrical detection. Nanomaterials, including nanowires, offer the unique ability of size-based trapping and localization of proteins along with highly increased surface area of interaction for protein binding. 7 This leads to higher sensitivity of detection and at the same time reduces the device footprint toward prototyping highly portable devices. IrOx nanowires, with its high sensitivity to surface charge variations and its conducive physisorption properties makes it a highly suitable nanomaterial for electrical monitoring of biomolecule binding events.
In this article, we demonstrate the design and fabrication of a real-time electrical immunoassay based on IrOx nanowires, also known as the IrOx nanomonitor—for disease diagnosis through time-continuous monitoring of protein biomarkers. We have tested the nanomonitor in detecting two inflammatory proteins, C-reactive protein (CRP) and myeloperoxidase (MPO) (Calbiochem, La Jolla, CA) that have been identified as biomarkers for cardiovascular diseases. 8,9 The design, fabrication approach, and device principles have been discussed in the following sections.
Design and Fabrication
The IrOx nanomonitor fabrication comprises of three steps: (1) photolithographic patterning and fabrication of base platform, (2) fabrication of the nanotextured protein binding and detection area, that is, vertical arrays of IrOx nanowires, and (3) passivation and packaging with microfluidic flow system on a chip to arrive at a lab-on-a-chip device.
Base Platform Fabrication
The base platform is based on a working electrode (WE)—counter electrode arrangement wherein change in capacitance will be measured across these electrodes. 10 The WE acts as the sensing electrode, whereas the counter electrode acts as a pseudo-reference, hence acting like a differential circuit arrangement. The electrodes were made circular in shape to maximize the surface area of interaction between the electrodes and avoid possible edge effects. The diameter of the counter electrode is 30 μm and that of the WE is 8 μm. These electrodes are separated by a gap of 1 μm as shown in Figure 1A. The physical design of the electrodes was based on achieving highest sensitivity toward change in capacitance. Hence, the gap between the electrodes was made small (1 μm) and the area of the counter electrode was made 50 times the area of the WE so that it acts like a differential reference. Interconnects were patterned to link these electrodes to input/output (I/O) pads located along the perimeter of the chip.
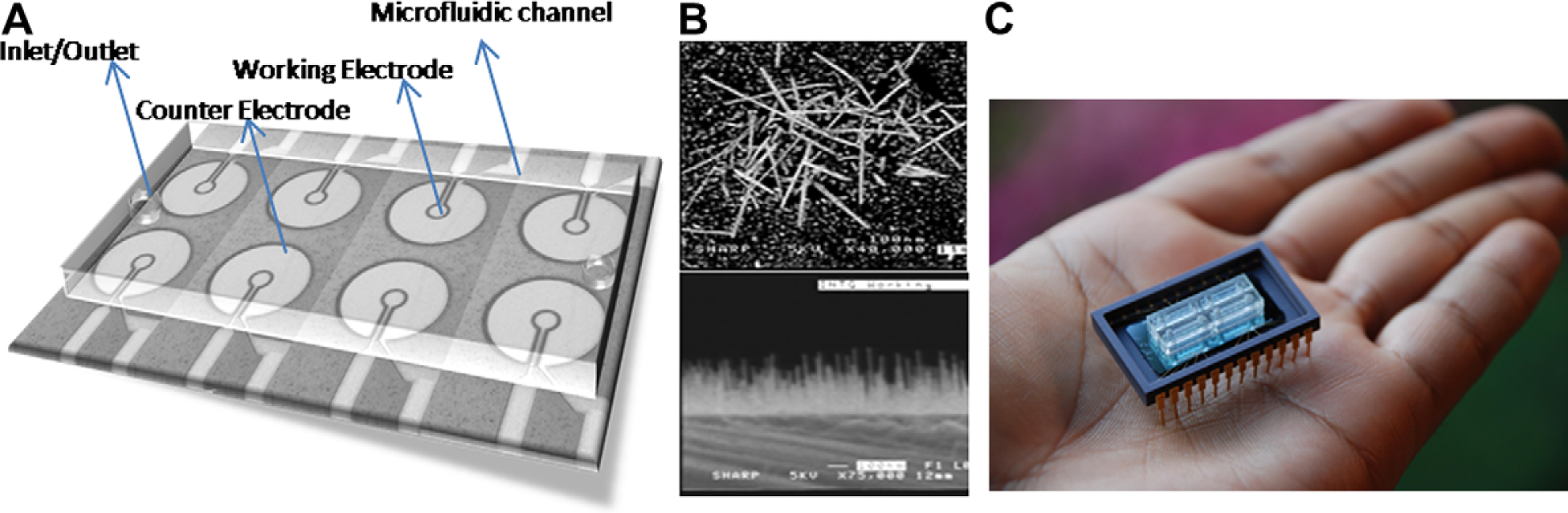
(A) Three-dimensional view of the fabricated electrodes inside a microfluidic channel with interconnects leading to I/O pads. (B) SEM micrograph of the iridium oxide nanowires grown on the WE. (C) Integrated lab-on-a-chip device with external pin layout.
A standard photolithographic process flow was carried out on a silicon wafer using an appropriate design mask to fabricate this device. After patterning the electrodes, Ir thin film of thickness 1–5 nm was deposited on the electrodes followed by selective wet etching. Ir film was chosen because it offers the noncontinuous surface necessary for IrOx nanowire growth while being highly conductive. IrOx nanowires were grown only on the WE to maximize protein binding.
Fabrication of IrOx Nanowires
IrOx nanowires were grown on the Ir thin film with noncontinuous surfaces on a silicon substrate through the metalorganic chemical vapor deposition (MOCVD) process (Fig. 1B). Oxygen and (methylcyclopentadienyl) (1,5-cyclooctadiene) iridium (I) were used as precursors in the MOCVD process to promote the growth of IrOx nanowires on the growth promotion film surfaces. The IrOx nanowires have a diameter of about 10–100 nm and lengths in the range of 100–1000 nm, an aspect ratio (length to width) of greater than 50:1. The growth length, density, and vertical orientation can be controlled by temperature, pressure, flow, substrates, and time. Detailed description of the growth of IrOx nanowires is given elsewhere. 11,12 The use of nanowires highly increases the effective surface area of interaction for proteins when compared with the alumina membranes in previous research. 10 IrOx nanowires with Ir film was used in comparison with gold electrodes in the previous system and the results shown in later sections showcase the significant advance in performance metrics.
Packaging to a Lab-on-a-Chip Device
After completely fabricating the sensing sites, the substrate was subjected to patterned passivation where the complete surface of the device except the electrodes and the I/O pads was covered with an insulating layer of silicon nitride or silicon dioxide to eliminate noise from background and environment and any capacitive interference between interconnects.
After all steps of fabrication, the substrate was mounted on a chip with the I/O pads wire bonded to the external pin layout of the chip as shown in Figure 1C. Microfluidic channels made from polydimethylsiloxane (PDMS) with an inlet and an outlet were fixed on top of the device for easy manual injection of samples with a pipette and to maintain a constant sample volume of 20 μL during analysis. It measures around 2 × 0.5 × 0.5 cm in physical dimensions and it insulates the sample from any external noises.
Experimental Methods
The device works on the principle of formation and perturbation of the electrical double layer. 13,14 An electrical double layer is the charged interface that is formed on a liquid/metal interface with an array of opposite charges on each side of the interface. The buffer solution used in the IrOx nanomonitor is 1x phosphate buffered saline (PBS). The typical conductivity of 1× PBS at 25°C was found to be around 0.0159 S/cm with a pH of 7.4. An electrical double layer occurs as charged particles and oriented dipoles of the buffer solution get localized at the liquid/metal interface.
When the Ir electrode surface and subsequently the IrOx nanowires are charged, it attracts oppositely charged species and forms a neutral region around the electrode. This essentially acts like a parallel plate capacitor and the associated capacitance will hence be called Cdl. At pH 7.4, after determining the isoelectric point (pI) of a protein, the resultant charge is calculated for its parity and magnitude. For CRP, the pI is estimated to be 5.62, hence the resultant charge at 7.4 pH is around −4.2, whereas the pI of MPO is highly cationic (> 10) and hence a resultant charge of +17.6. Therefore, a negative direct current (DC) bias is applied to the electrode for MPO detection, whereas a positive bias is applied for CRP to attract the proteins to adsorb faster to the nanowires and increase throughput. However, irrespective of the magnitude of charge, a maximum of ±200 mV is applied to avoid any possible denaturing of proteins.
When the buffer solution is spiked with CRP, these negatively charged proteins bind to the IrOx nanowires at the nanowire/liquid interface and hence perturb the surface charge distribution at the inner Helmholtz layer that is typically around 10–30 nm thick. With more proteins binding at the interface, the associated surface charges also change significantly. These perturbations in the double layer essentially in turn perturb the double layer capacitance formed at the interface. Hence, the quantification of the concentration of protein binding is done by measuring the electrochemical change in this double layer capacitance. To ensure selective binding of the proteins onto the nanowires in the presence of other competing proteins, monoclonal protein-specific receptors (antibodies) are coated on the nanowire surface.
Equivalent Circuit
Impedance is measured between the WE and counter electrode while they form an electrochemical cell with PBS buffer as the electrolytic medium. The model that has been adopted is called the Randles cell model (Fig. 2). 14 When protein binds to the nanowires, it perturbs the electrical double layer to change Cdl significantly at lower frequencies (<10 kHz). The dielectric capacitance, Cde is always constant and compensated by the impedance analyzer through control. This is because all the proteins are aliquoted using the same PBS buffer; hence the ionic conductance and the dielectric medium remain the same throughout.
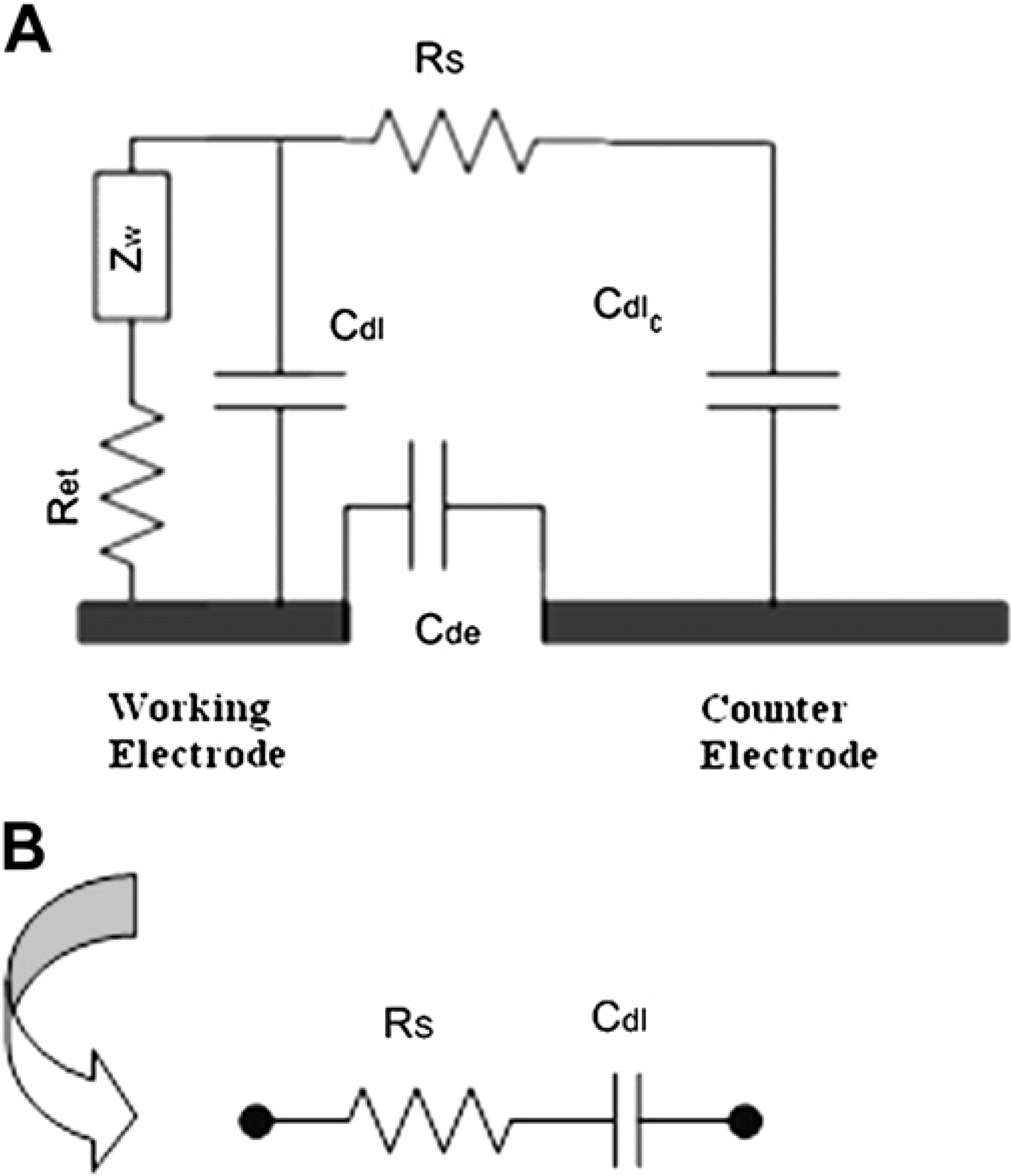
(A) Equivalent electrical circuit across the electrodes following Randles cell model. (B) Equivalent circuit simplification.
As the Warburg impedance, ZW is very high at frequencies lower than 100 kHz, it virtually blocks the path for any current flow. Hence, the equivalent circuit impedance gets simplified to a series combination of the solution resistance, RS and Cdl, where RS remains constant at lower frequencies. Therefore, the change in impedance is completely attributed to the change in the double layer capacitance at frequencies (<10 kHz).
Cdl was found to change the most at 1 kHz. With increasing background and external interferences at lower frequencies and decreasing capacitance with higher frequencies, 1 kHz was found to be the optimum frequency for detection. The change in impedance at 1 kHz was analyzed using electrochemical impedance spectroscopy technique for the quantification of protein concentration. The impedance analyzers used were Gamry Reference 600 and HP 4194A, wherein the measurements were taken by connecting to the electrodes through the external pins.
Samples
CRP and MPO are cardiovascular biomarkers and detection of such multiple biomarkers mark the physiological condition of a myocardial infarction. 15 Patients with CRP levels less than 1 μg/mL in their blood have very low risk of a heart attack, whereas patients with more than 30 μg/mL face a high risk. 16
The CRP and MPO pure protein samples and their antibodies were prepared using standard serial dilution techniques after preparing a standard stock solution of high concentration. CRP and MPO were preserved with 0.1% sodium azide. A 1-mg/mL stock solution was prepared followed by serial dilutions up to 100 pg/mL.
Human serum (U.S. Biological, Swampscott, MA) was diluted to create a sample that has 4 μg/mL of CRP. Known concentrations of CRP were also spiked with human serum followed by centrifuging for 3 min at (1100 ×g) for them to mix well.
Experimental Protocol
There are four steps associated with the single capture immunoassay before protein binding in preparing the device for protein detection using this mechanism: (1) establishing control and background compensation, (2) chemical functionalization, (3) antibody saturation, and (4) blocking. After these steps, the protein sample is injected into the system for characterization.
The first step is to identify background effects. Open and short circuit measurements were noted for appropriate electrical compensation. Then, an impedance curve was measured with the ionic buffer 1x PBS on and between the electrodes that acted as the first reference. This first reference curve gave the baseline measurements and compensated for all the constant electrical elements discussed before.
Standard biochemical functionalization was used to immobilize the antibodies onto the IrOx sensing sites using a biotin/streptavidin linker. The nanowires were first coated with 4 μg/mL of streptavidin and allowed to incubate for 15 min. Such high concentration was used to ensure saturation of streptavidin on nanowires. Streptavidin strongly binds to the nanowires through strong physical adsorption. Meanwhile, antibodies were conjugated with 4 μg/mL of biotin to form biotinylated antibodies and allowed to incubate for 15 min. After incubation, the nanowires were washed with the 1× PBS to remove all unbound streptavidin molecules. Now the biotinylated antibodies were injected onto the streptavidin-coated nanowires till saturation. Biotin binds very tightly to the tetrameric protein streptavidin with a dissociation constant Kd in the order of 10−15 mol/L that is the strongest known protein-ligand interaction, approaching the covalent bond in strength. Because of this strong biotinavidin linking, the antibodies stayed strongly crosslinked to the nanowires.
The next step in the detection methodology was to saturate the nanowires with monoclonal antibodies. Starting from a lower concentration, increasing concentrations of antibodies were biotinylated and added to the nanowires and the impedance increase was monitored while incubation (15 min). At a particular concentration, there was no increase in impedance and it reached saturation. This concentration is the saturation concentration of antibodies that corresponds to the state where almost all possible sites on the nanowires were occupied by the antibodies. Hence, this saturation concentration is used throughout to increase sensitivity of detection by maximizing probability of protein binding and reduce the number of unoccupied sites for nonspecific binding. To further the above, a blocking step was performed, where a 3% bovine serum albumin/PBS buffer was injected onto the sensing sites to block all remaining unoccupied sites on the nanowires to eliminate nonspecific binding of antigens. The impedance measured after this step provides the final reference for the measurements.
Now, the device was ready for protein detection in real-time. The change in impedance measured from this final reference was completely attributed to protein binding and hence used for the characterization and quantification of the proteins. This change in impedance denoted the change in Cdl induced by protein binding. A range of concentrations of pure and serum samples were tested in real-time, where the impedance at 1 kHz was noted continuously at regular intervals with protein addition. The following section discusses the results and the performance metrics achieved by the device toward detecting two proteins CRP and MPO.
Results and Discussion
This section shows the results from initial control experiments, dose response attained for two proteins CRP and MPO in pure and serum samples, and their respective cross-reactivity analyses. Results attained from antibody saturation show the enhanced surface area offered by the nanowires along with the high physical adsorption rate of IrOx making it ideal for protein detection. A DC bias of 200 mV was applied between the WE and counter electrode during all experiments. The frequency of detection is 1 kHz, as noted in the previous section.
Starting from a lower concentration (500 ng/mL), increasing concentration of the monoclonal antibodies was added to the nanowires in a stepwise manner. At 10 μg/mL for anti-CRP and 8 μg/mL for anti-MPO, impedance reached saturation and hence marked the saturation concentration of the respective antibodies.
Protein Dose Response in Pure and Serum Samples
The concept of “real-time” detection is the ability to monitor the protein concentration in a sample right after the sample has been injected into the device for measurement. Monitoring a steady increase in impedance corresponds to the minimum concentration of that protein present in that sample in real-time. It also reaches endpoint within 4–5 min, enabling a possibility of quicker diagnosis and treatment. After antibody saturation, the protein sample was injected into the system and the respective change in impedance for different doses of the pure samples of CRP and MPO was observed in Figure 3A and B.
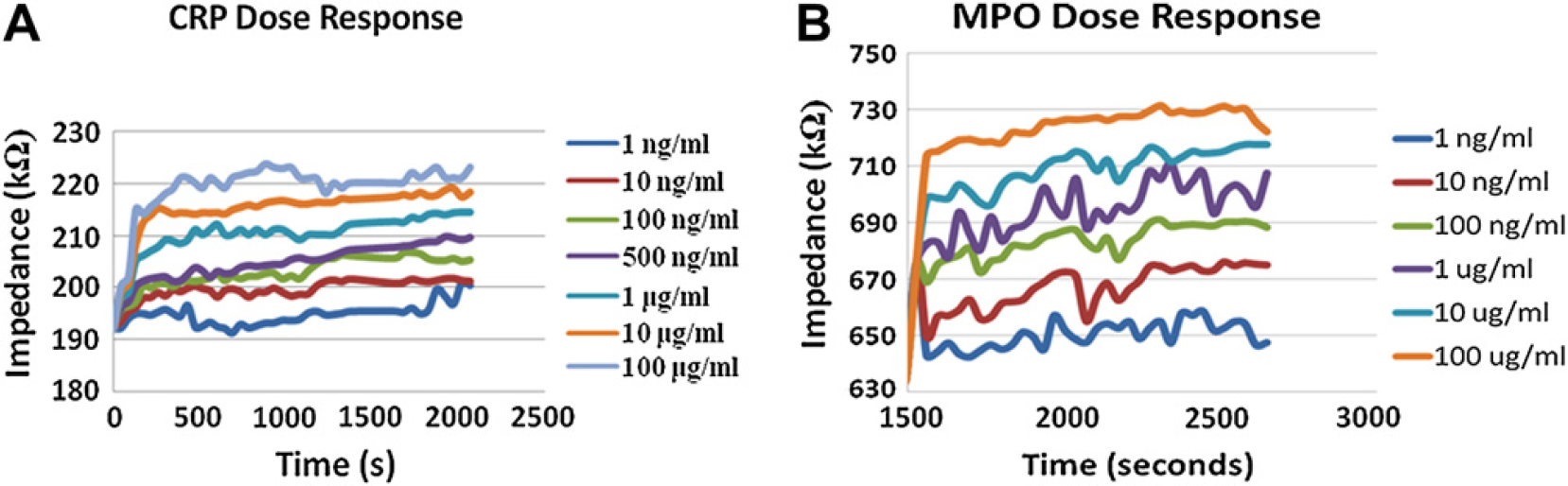
(A) Impedance dose response for CRP after anti-CRP saturation (error margin [EM] ∼ 1–3%). (B) Impedance dose response for protein MPO after anti-MPO saturation (EM ∼ 1–4%).
As seen from Figure 3A and B, there is an increase in impedance with protein binding. This in turn means that there is an increase in capacitive reactance and hence decrease in the double layer capacitance. With antigens binding to its respective antibodies, the impedance increases and finally attains saturation within few minutes, hence marking a constant value at the end. This value is used for characterization and quantification of proteins. It is also observed that the percentage change in impedance increases with the increase in concentration of the antigens, hence showing high sensitivity toward pure sample protein detection. The lower limit of detection was found to be 1 ng/mL for CRP and 500 pg/mL for MPO. The linear dynamic range of detection extends from 1 ng/mL to 100 μg/mL (5 orders of magnitude), which is much higher than ELISA (1–2 orders).
To test the efficiency of the device toward detecting proteins in complex, physiologically relevant samples, CRP was spiked in human serum. Human serum consists of a mixture of competing proteins and it was diluted to a concentration where CRP was 4 μg/mL. Pure 1 μg/mL of CRP (50% v/v) was spiked with the diluted serum stock solution and tested. Figure 4 shows the dose response for this analysis and it shows the higher change in impedance with spiked CRP serum showing its selective binding to CRP in the presence of several competing proteins. Protein CRP at concentrations (> 1 μg/mL) could be detected up to 85% accuracy when present in human serum. Hence, this accuracy in detection confirms that the potential of operating this device is in “real-life” conditions to detect proteins in clinically relevant concentrations.
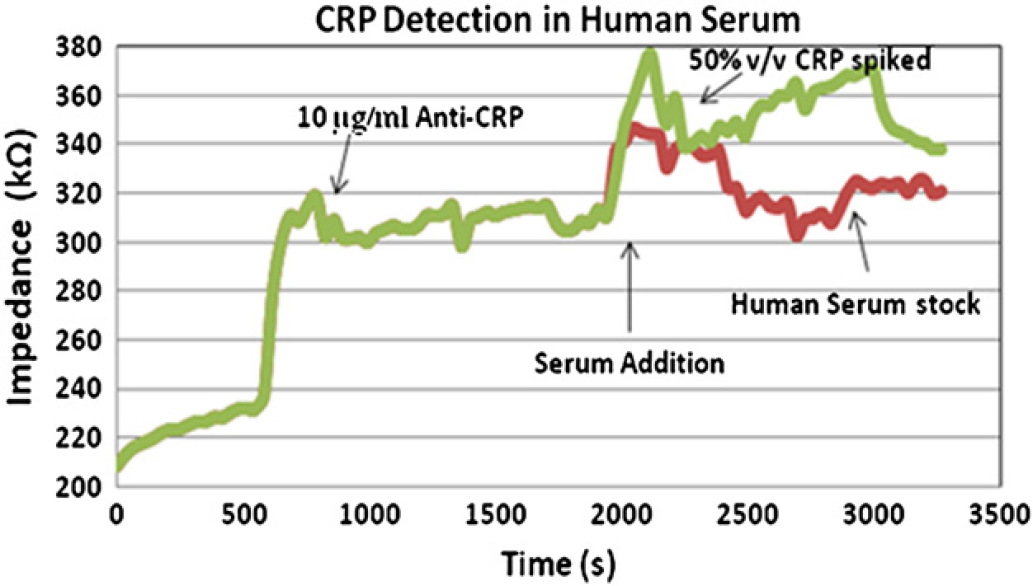
CRP detection in serum sample—human serum with spiked serum containing 50% v/v of CRP (error margin ∼ 1–3%).
Figure 5 shows the dynamic range of detection for proteins CRP and MPO in pure samples and CRP in human serum sample wherein the percentage change in impedance is plotted with increase in concentration of the protein. A linear trend is observed within the 1 ng/mL–100 μg/mL range for both the proteins.
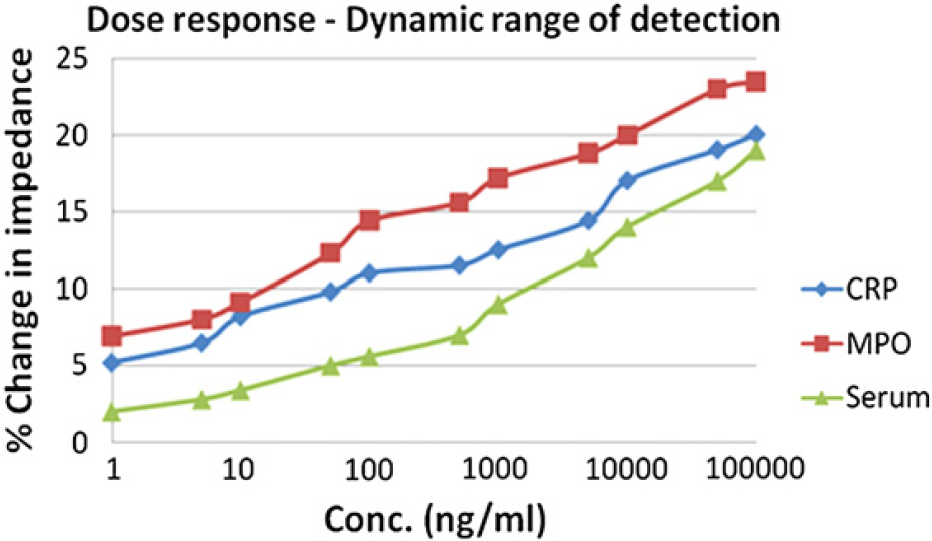
Dynamic range of detection showing percentage change in impedance over concentration.
Selectivity Analysis
Two sets of cross-reactivity analysis experiments were performed: (1) proteins CRP and MPO were introduced to anti-CRP saturated nanowires (Fig. 6A) and (2) proteins CRP and MPO introduced to anti-MPO saturated nanowires (Fig. 6B). A standard 10-μg/mL concentration of CRP and MPO were used for both the analyses. The amount of cross-reactivity was found to be less than 10% of that of selective binding of proteins. The increase in impedance is significantly higher when the corresponding protein was added to its antibody suggesting high selectivity.
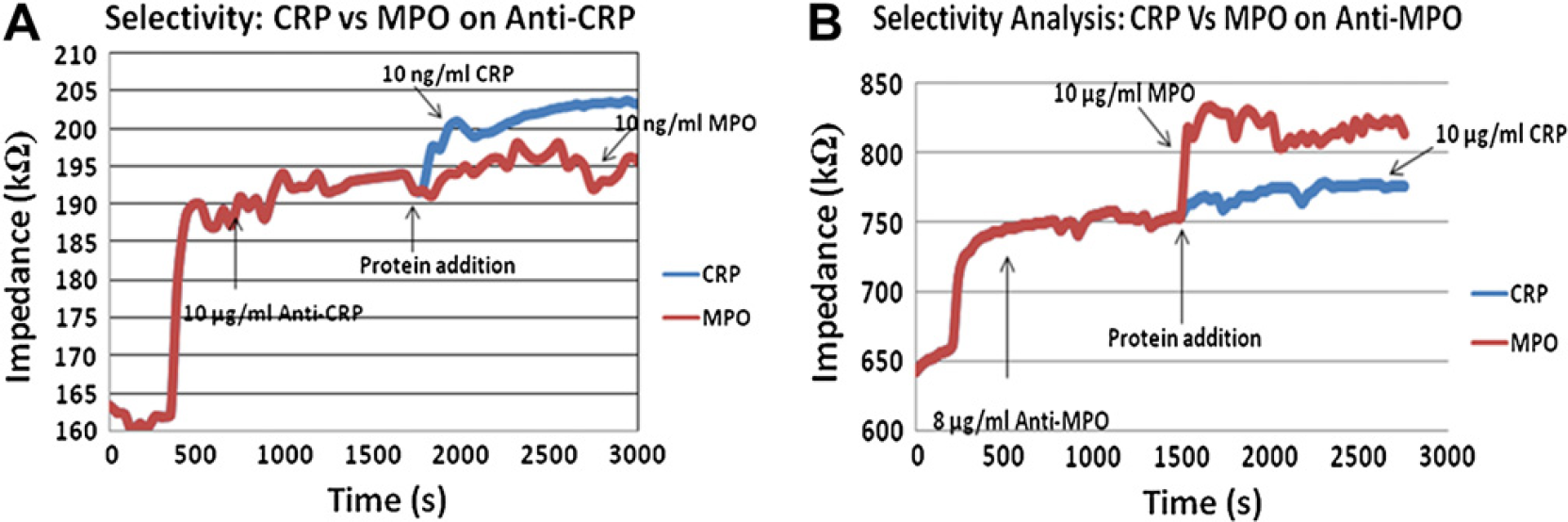
(A) Selectivity analysis of proteins CRP and MPO on anti-CRP (error margin [EM] ∼ 1–3%). (B) Selectivity analysis of proteins CRP and MPO on anti-MPO (EM ∼ 1–4%).
Figure 6A and B show that increase in impedance when a protein binds to its corresponding antibody is much higher than otherwise. This signifies the amount of selectivity achieved by this device. There is some amount of nonspecific binding observed (<8%) but its corresponding increase in impedance is lesser than the increase induced at the lowest limit of detection (1 ng/mL). Hence, the nonspecificity factor is negligible at this point.
Conclusions
The prototype that was designed, fabricated, and evaluated was an IrOx vertical nanowire array platform also known as the IrOx nanomonitor. We have demonstrated the capacity of IrOx nanowires toward high sensitivity real-time protein detection. Nanowires have much larger surface area for biomolecule interaction than thin films, hence offering significantly higher sites for protein interaction. We believe that because of the increased surface area of interaction they help confine the protein molecules, hence potentially retaining protein efficacy for longer periods by maintaining their active conformation. IrOx has much higher electrical sensitivity at metal/liquid interfaces than many metals because of its high sensitivity to surface charge variations. These factors make IrOx nanowires ideal for electrical biomolecule sensing and protein biomarker profiling.
Selective detection is achieved by the incorporation of specific antibodies on the nanowires. Because of this specific binding of the proteins on the nanowire/liquid interface, perturbations in the electrical double layer are monitored and quantified for the specific protein. Detection time is in few minutes and being a “point-of-care” device, the infrastructure cost for this device is very low. Hence, along with being highly portable, this nanomonitor offers highly improved performance metrics over competing technologies like ELISA.
“Real-time” monitoring is definitely advantageous over endpoint monitoring as it gives time-continuous data enabling quicker diagnosis and treatment. Concentration of protein biomarkers effectively determine the physiological state of a disease and IrOx nanomonitors have successfully demonstrated their ability toward effective real-time protein detection. They could also potentially be used as a rapid label-free screen for disease biomarkers and can be developed as a lateral flow immunoassay methodology because of the rapid charge transduction through IrOx nanowires. This rapid charge transduction produces distinct capacitance changes associated with the protein binding event and is the basis for the protein detection profile.
There are a few limitations in the current IrOx nanomonitor system—nonspecific binding and cross-reactivity as observed in serum samples. As can be seen from the selectivity graphs, we see a small impedance change (∼5%) with the presence of CRP on antibodies of MPO. This is because of the cross-reactivity happening between competing proteins. It also includes contributions from background effects, environmental effects, and external circuitry. This nonspecific binding has constrained the detection limit to higher pg/mL. Higher sensitivities could be achieved by reducing or optimizing this phenomenon through customized protein-specific biochemical functionalization of the nanowire surfaces for detecting specific proteins, improved noise reduction, and throughput enhancement from multiple site detection. Having measurements at multiple lower frequencies and building a suitable averaging system will make the detection more accurate by reducing system noise. System noise could be further reduced by building much better passivation layer to isolate the sensing sites from the environment. Hence with better isolation, it would be possible to measure at even lower frequencies with negligible noise, therefore, leading to much higher sensitivity in detection.
For this device to serve to be suitable for clinical diagnostics applications, it needs to simultaneously detect multiple protein biomarkers rapidly from the human blood samples without ambiguity at clinically relevant concentrations, while being very easy to handle, inexpensive, and highly portable. The current article is a demonstration of the potential of the IrOx nanowire-based nanomonitors for real-time and rapid protein detection with performance metrics comparable with other standard immunoassay techniques such as ELISA. Future research is directed toward achieving multiplexed detection of many protein biomarkers with lower limits of detection and with lower cross-reactivity.
