Abstract
GRAVI, presented here in its automated version, is a new bench-top sized immunoassay platform combining the advantages of microfluidics with those of simplified robotics. Characterized by dramatically reduced time to result (< 10 min) and significantly decreased sample/reagent consumption, the cost-efficient biosensor instrumentation allows performing multimenu analysis with minimal laboratory infrastructure.
Coupled to a robotic liquid handler, the system dispenses samples and reagents from conventional plates or tubes into microchannels of a microchip (GRAVI-Chip), in which assays are processed and results readout. As in conventional 96-well microtiter plates, the microchannels have a standard spacing of 9 mm to facilitate automation.
With solely gravity and capillary force-driven fluidics within the microchannels, liquids are free to flow while magnetic beads, functionalized with the antibody of choice, are trapped nearby incorporated electrodes by virtue of a magnet array. Following assay performance and electrochemical signal detection in the parallel microchannels, chips are regenerated by magnet release and rinsing of beads out from the microchannels.
Applicability of the presented immunoassay platform, delivering 100 results per hour, is exemplified here with results from the validation of an immunoglobulin assay for antibody quantification in mammalian cell cultures. Adapted to run on the GRAVI platform, this competitive assay covers a dynamic range of two orders of magnitude.
Introduction
Over the past few years, DiagnoSwiss has developed polymer devices with the advantages of microfluidics for bioanalytical applications, including enzyme-linked immunosorbent assays (ELISAs) 1 and nanoelectrospray sampling. 2,3 Laser photoablation and plasma etching processes were used for the purposes of prototyping and production of microchips, respectively. In their current version, these microchip consumables harbor eight parallel microchannels, with integrated electrodes for electrochemical signal readout 4 or derivatization of elements in situ. 5,6
Frédéric Reymond, Ph.D.

Microfluidics, applied to ELISA testing, very significantly speeds up analysis and reduces sample consumption. The applicability of our microfluidics-based system for performing immunoassays was demonstrated in previous reports. The various sandwich or competitive ELISA reagents were successively flowed through the microchannels, to iteratively build the labeled immune complex, which was quantified by chrono-amperometric readout. 7 As described in experiments for the detection of vitamin B9 in milk powder, 8 devices such as peristaltic pumps and syringes were interfaced with our microfluidic biosensors to force liquid flows and control microfluidics within the microchannels.
Extensive characterization of the microchip fluidics 9 has meanwhile allowed us to develop a platform in which liquid flows are solely driven by gravity and capillary forces. The simple fact of placing microchips on tilted holders allowed eliminating maintenance-intensive pumps and valves.
In the mentioned reports, the capture antibody was bound to the inner surface of the microchannels, and reagents were sequentially pumped through the microchannels. Formation of immune complexes naturally depends on the affinity and avidity of antibody for its antigen. In addition, reaction kinetics in this configuration were limited as a consequence of analyte flowing by, rather than diffusing to the immobilized antibody. 1,9 To overcome these limitations without compromising the advantages of microfluidics (low sample and reagent consumption, and short time to results), our system was evolved to run assays with paramagnetic nanoparticles. Capture antibody is immobilized on nanoparticles, which can be preincubated with sample, in tube. After incubation, the mixture is flowed through the microchannels, and the paramagnetic nanoparticles are trapped nearby the electrodes by virtue of a magnet array. The biological reactions (occasionally requiring longer incubation times) can thus be freely adjusted in function of the assay. Reactions leading to formation of the immune complex can be performed in tube or in the microchip, while washing steps and detection of the enzymatic reaction take place in the microchannels. In this manner, the microchip merely serves as physical biosensor, in which tests with a variety of microbeads can be performed in successive runs. The flexibility to consecutively use microbeads functionalized with different capturing moieties translates into a full random-access solution. 10
Here, we present a novel microfluidic biosensor platform dubbed GRAVI, the dimensions of which are compatible with standard microtiter format (i.e., 9-mm microchannel spacing, as in a 96-well microtiter plate). A robotized version of the platform (dubbed GRAVI-Lab and illustrated in more detail below) represents an all-in-one ELISA platform for liquid handling, incubation, washing, and readout.
There are different examples of microfluidic systems with automated instrumentation. Caliper Technology, for instance, built the EZ-Reader, 11 which is an elegant platform using glass electrophoresis chips to separate preloaded samples (as in the so-called BioAnalyser 12 ) and which loads reagents from a microtiter plate. Another example is Gyros, who developed a pipette-head aspirating samples from a microtiter plate, and which repositions the probes for liquid transfer to disk wells in circular configuration. 13 In the field of mass spectrometry analysis, Advion has developed a system where samples from a microplate can be introduced in a reader, which sequentially transfers the samples to a microfabricated silicon nozzle for dispensing by electrospray ionization. 14,15
Various other (chromatography-like) systems have been evolved, either for clipping microfluidic channels or chambers onto the column (e.g., the label-free systems of Biacore, 16 SensiQ, 17,18 and Qsense 19,20 ), or in which the microchip serves as full alternative to chromatography (such as the HPLC Chip of Agilent Technologies 21 ). The requirement to feed chips, however, has made the development of specific liquid handlers or robots necessary in all these automated microfluidic systems. Such systems thus represent quite complex solutions, hardly suitable for incorporation of microfluidic chip technology in conventional laboratory instrumentation.
Infrastructure requirements are regarded as an important hurdle to the wide acceptance of microfluidic systems. According to A. Chow “one bottleneck of microfluidics remains to be the efficiency in interfacing the macroscopic world to the microfluidic chip, including introducing sample into the chip and getting processed material out from the chip.” 22
In contrast to other systems, GRAVI-Lab is designed for integration in most any eight-channel liquid handler, with minimal need for customization. Simple liquid-handling robots are thus readily upgradable into fully automated ELISA platforms for swift analysis with least sample and reagent consumption.
Material and Methods
Electrochemical Microchips
GRAVI-Chip, shown in Figure 1, is a polyimide foil-based microchip with a linear array of eight independent microchannels. Gold microelectrodes, incorporated in the microchannels, allow for electrochemical signal readout. The electrodes are connected to metallic tracks, conducting the electrical current to the external reader. In its present version, the chip microchannels are 1.5 cm in length, and have a cross-section of 90 × 200 μm 2 , thus leading to microchannels with a total internal volume of approximately 300 nL. With 30-μL reservoirs at the microchannel inlets and outlets, and given a microchannel spacing of 9 mm (i.e., the 96-well microtiter plate standard), the system is fully compatible with conventional pipetting devices. The GRAVI-Lab platform (see below) can harbor two GRAVI-Chip units, for running 16 assays in parallel. The actually commercialized version of GRAVI-Chip is suitable for up to 100 detections per channel over a 30-day period.
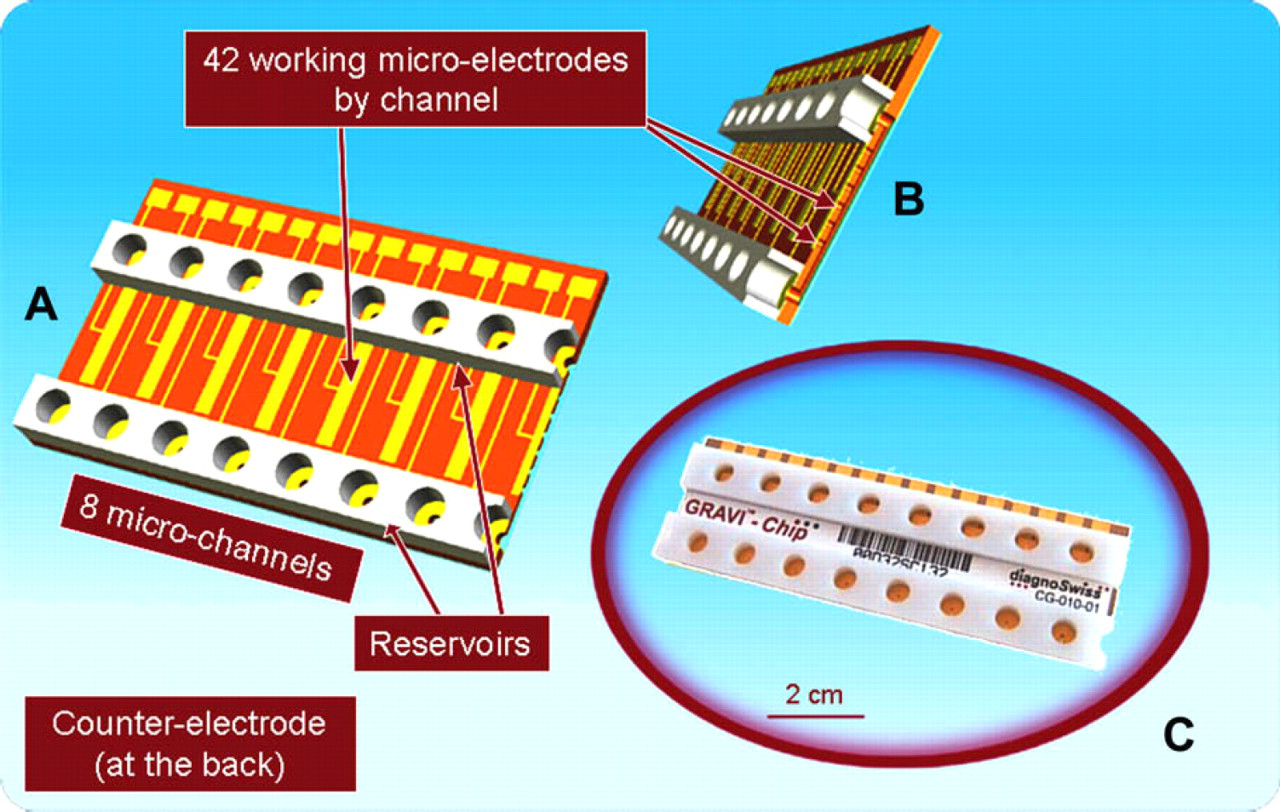
Views of the GRAVI-Chip showing (A) upper side of the eight-microchannel chip with its solution reservoirs and electrical network; (B) cross-section of a microchannel with its array of electrodes integrated in recesses; (C) picture of a GRAVI-Chip consumable.
Integrated Hardware
Features of the GRAVI-Lab instrument are shown in Figure 2. Microchip holders are fixed to a tilted surface (for gravity-driven fluidics) and incorporate a heating block (37 °C). Electrical contacts connect the microchannel electrodes to the potentiostat for electrochemical readout. The magnet array can be opened or closed by a simple robot movement. The GRAVI-Lab platform is also equipped with relay cards, for the control of independent shakers (or other outputs), which can be snapped on to the working space of the liquid handler. The GRAVI-Lab instrument, mounted on a compatible plate support, is directly snapped onto a Tecan MSP 9250 robot (see Fig. 3). The washing station is positioned to allow adding a maximum number of reagent and sample plates on the robotic station.
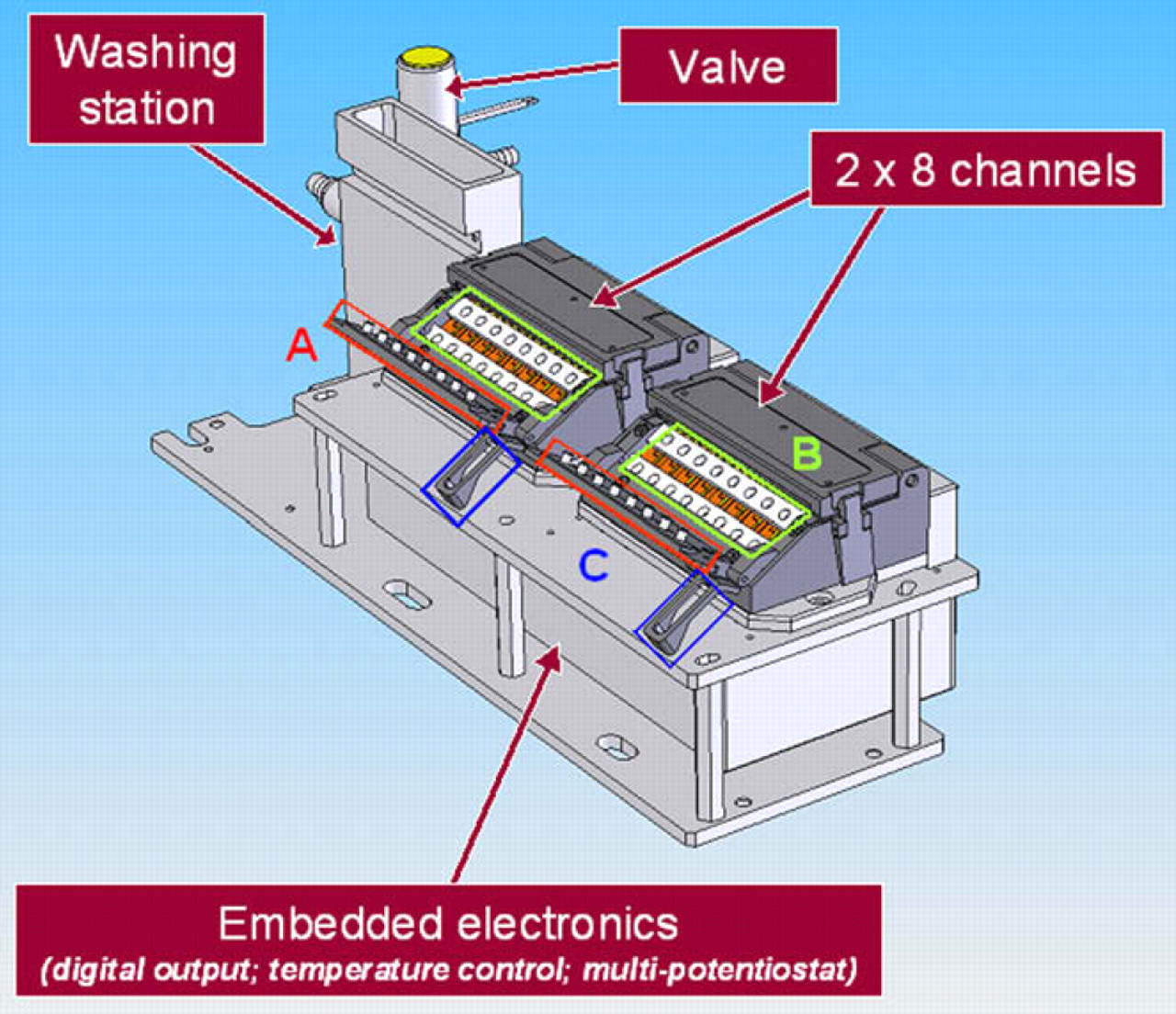
Schematic illustration of the GRAVI-Lab apparatus showing (A) magnet array; (B) GRAVI-Chip units (tilted for gravity-driven fluidics within the device); (C) magnet lid (for robotic opening and closure of the magnet array).

Picture of the GRAVI-Lab coupled to an MSP 9250 robot from Tecan Cavro: (A) GRAVI-Lab unit; (B) washing station; (C) reagent plate; (D) stock plate of microbeads and secondary antibody; (E) mixing plate; (F) sample plates (stock plate and mixing plate are supported on computer-driven shakers).
Software: for Platform Control and for Result Display
GRAVI-Soft is a master software controlling and synchronizing all actions performed by the robotized GRAVI-Lab, including robot movement and liquid-transfer functions. In the presented configuration, one GRAVI-Lab, six microtiter plates, and one washing station are embarked on the working space of the MSP 9250 liquid handler. Each of the microtiter plate positions can be equipped with plate shakers, piloted individually by GRAVI-Soft (e.g., to warrant optimal mix of bead-containing solutions and halting of the shaker for probe access).
GRAVI-Viewer is the complementary software, which allows programming assay protocols and exploiting experimental results. Preprogrammed protocols can be uploaded and adapted with GRAVI-Viewer, in which every GRAVI-Lab action is defined. Exploitation of results is possible at any time with GRAVI-Viewer, because there is no interference with GRAVI-Soft. To facilitate result interpretation, raw data can also be transformed into color intensities, which are displayed as a conventional microtiter plate image (see ref. 23).
Reagents
The GRAVI platform has been evolved to work with super paramagnetic microbeads (of 1 μm diameter or less, to prevent sedimentation or clogging). Suitable microbeads can be obtained from different sources (e.g., Dynabeads My-One from Invitrogen Dynal, Norway or Estapore from Merck, France). Microbeads are either precoupled (with a specific affinity reagent) or coated with different molecules for passive or active coupling of antibodies or antigens. Signal generation is enzyme mediated, by hydrolysis of substrate into an electrochemically active product within the microchannels. The enzyme can, for example, be coupled to a secondary antibody as used in a typical ELISA. Alkaline phosphatase works with p-aminophenyl phosphate (PAPP), and the generated chrono-amperometric signal is easily detected by the electrodes built in the microchannels. Virtually any affinity reagent can be used to establish most any immunoassay on the open GRAVI-Lab platform.
Principle of Elisa Processing in GRAVI-Lab
The GRAVI-Lab ELISA test presented here is a one-step assay (samples and enzyme conjugate are incubated with functionalized magnetic nanoparticles, before the mix is dispensed into the microchip). The assay principle is summarized in Figure 4, and key steps are explained in the following.
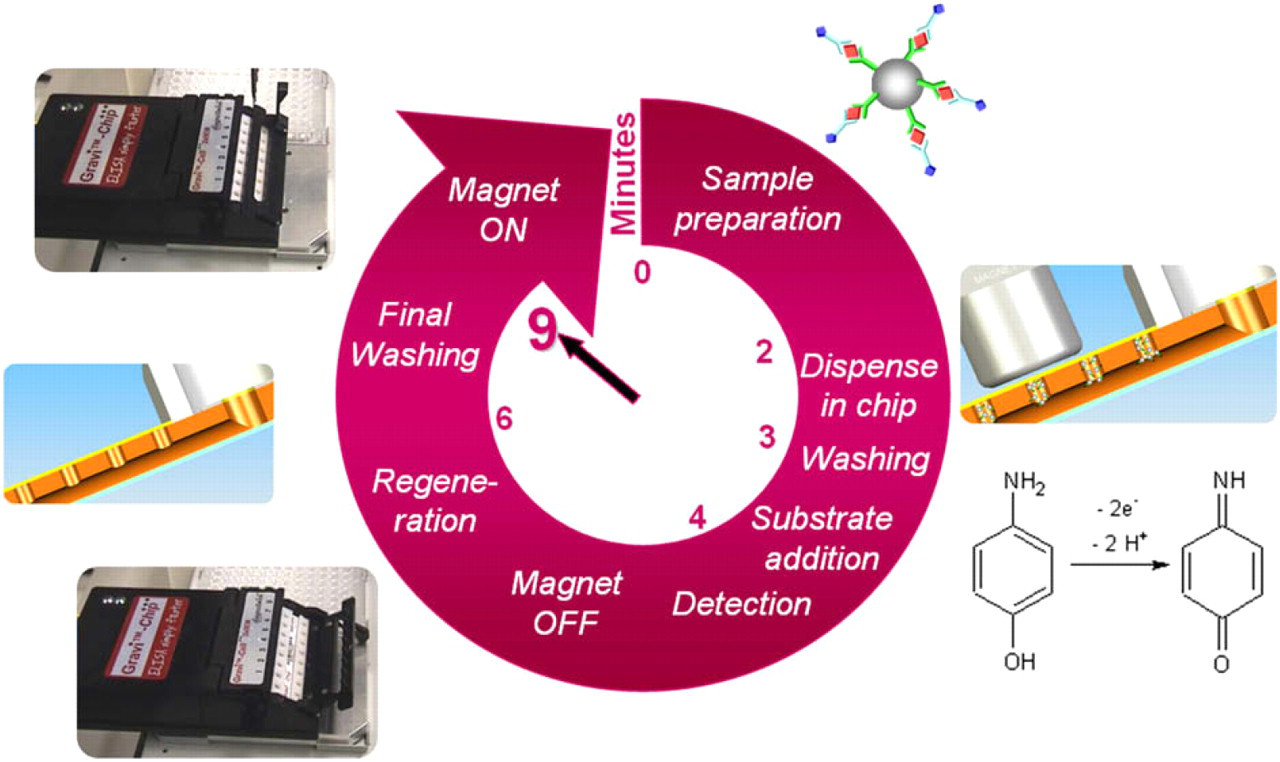
Cycle of a bead-based immunoassay with robotized GRAVI instrumentation.
Chip Priming
Before starting a first assay, the robot dispenses buffer (Tween 20, Sigma Aldrich) in the bottom GRAVI-Chip reservoirs. The buffer (heated to 37 °C in the chip) fills up the microchannels by capillarity within a few seconds. During “capillary fill,” the robot flips the magnet array onto the microchip, in view of trapping paramagnetic nanoparticles nearby the electrodes within the microchannels.
Sample Preparation
GRAVI-Soft instructs the robot to mix 10-μL coated beads from the stock plate, with 5–10-μL sample into wells of a reaction plate (see Fig. 3). The reaction plate is left to incubate (from a few minutes to 1 h, depending on the assay) with the shaker turned on. In one-step immunoassay protocols, conjugate is directly added to the mixture in the reaction plate (to allow formation of the immune complex as needed for the assay, whether competitive or in sandwich format). In two-step protocols, only beads and sample are preincubated (and conjugate is added at a later stage).
Dispensing and Washing Step
Upon incubation in the reaction plate (time enough to reach equilibrium or to obtain a sufficient signal), the liquid handler aspirates 10-μL incubation mix, which it dispenses in the top reservoirs of the microchip. The solution flows freely through the microchannels, while beads are trapped nearby the electrodes by virtue of the magnet array flipped onto GRAVI-Chip (see Fig. 2). Within a few minutes (time during which the robot prepares reagent/sample mixes for next assay series), the solution has entirely passed.
The robot subsequently dispenses 20-μL washing buffer into the upper chip reservoirs, to evacuate unbound enzyme and contaminants. Upon flow of approximately half the volume of washing buffer, the robot aspirates excess liquid from the upper reservoirs. This washing procedure is repeated three times, if desired with signal readout at each round (as blank control).
Substrate Addition and Electrochemical Detection
For enzyme-mediated generation of electrochemical signal, corresponding substrate is dispensed in the top reservoirs of the microchip. Substrate rapidly replaces the washing buffer, and the enzymatic reaction (e.g., hydrolysis of PAPP into electrochemically active p-aminophenol) occurs at the bead surface. Electrochemical signal, generated by application of a 300 mV potential versus Ag/AgCl, is measured over a time span of 0.5 s and repeated up to ten times in each of the microchannels. Signal development is immediate, and the measured electrical current is directly linked to the concentration of immune complex, and thus the amount of enzyme within the microchannels. Indeed, the evolution of current over time follows Michaelis-Menten kinetics, 7,8 and the concentration of the target analyte is given by the current slope at origin in the individual microchannels.
For signal detection, the flow of enzyme substrate is halted to allow for the enzymatic reaction product to accumulate in the microchannels. To stop the flow, the robot simply removes excess substrate solution from the top reservoirs. As a consequence, with gravity and capillary forces in equilibrium, “stop-flow” conditions are immediately reached.
Chip Regeneration
Once the assay concluded and signal readout, the magnet array is flipped away from the microchip by the robot (see Fig. 2) by a simple motion of the liquid-handler probes. To efficiently denature and evacuate beads and reagents from the microchannels, the robot dispenses a dimethyl sulfoxide-containing (Sigma Aldrich, Germany) regeneration solution into the upper chip reservoirs. After a flow of 30 s, the upper and the lower reservoirs are emptied and rinsed twice with the washing buffer. This procedure eliminates contaminants from the upper reservoirs and the microchannels, as confirmed by values (within experimental error) obtained from repeated measurements before and after regeneration (results not shown).
Finally, the magnet array is replaced in capture position by the robot, and the next series of samples (premixed in the reaction plate) can be injected.
Qualification of the GRAVI-Lab
To determine the intrinsic performances of the GRAVI-Lab platform, qualification experiments were conducted with a one-step alkaline phosphatase (ALP) assay. Microbeads (1 μm diameter) coated with streptavidin (Dynabeads My-One Streptavidin T1, Invitrogen Dynal, Norway) were used as solid support, and biotinylated ALP (Vector Laboratories Inc., USA) as enzyme label.
The sample mix, consisting of 0.05-mg/mL microbeads and 31.25-ng/mL biotinylated ALP, was prepared by mixing of 1-mL streptavidin-coated microbeads at 0.1 mg/mL in washing buffer (i.e., 100-mM Tris [hydroxymethyl aminomethane], 100-mM sodium chloride with 1% bovine serum albumin, and 0.1% Tween 20 [Sigma Aldrich, USA] at pH 9), with 1-mL biotinylated ALP solution at 61.5 ng/mL. After 10-min incubation time, 140 μL of this mixture was deposited in wells of a microtiter plate, which was shaken on the robot (see Fig. 2). For assay qualification, a protocol was created in GRAVI-Soft so as to perform a series of ten successive assays and microchip regenerations.
According to this protocol (repeated in loop), the robot proceeded to close the magnetic lid, pipette 10 μL reaction mixture in the top chip reservoirs, allow solution flow into the microchannels for 80 s, add 10-μL substrate buffer (i.e., 5-mM PAPP in 100-mM 2-methylaminoethanol and 2-mM magnesium chloride [Sigma Aldrich] at pH 9.8), let flow for 15 s, and remove excess substrate from the top chip reservoirs. After electrochemical detection by amperometric measurement at 300 mV versus Ag/AgCl, the robot performed a chip regeneration as described in the previous section.
Figure 5 shows example results for such successive assays, with the intra- and interchannel variability of the platform. The coefficient of variation was less than 5% for each channel (3.5% in average), and the interchannel variability was under 10%. These qualification experiments demonstrate that each channel can be efficiently calibrated.
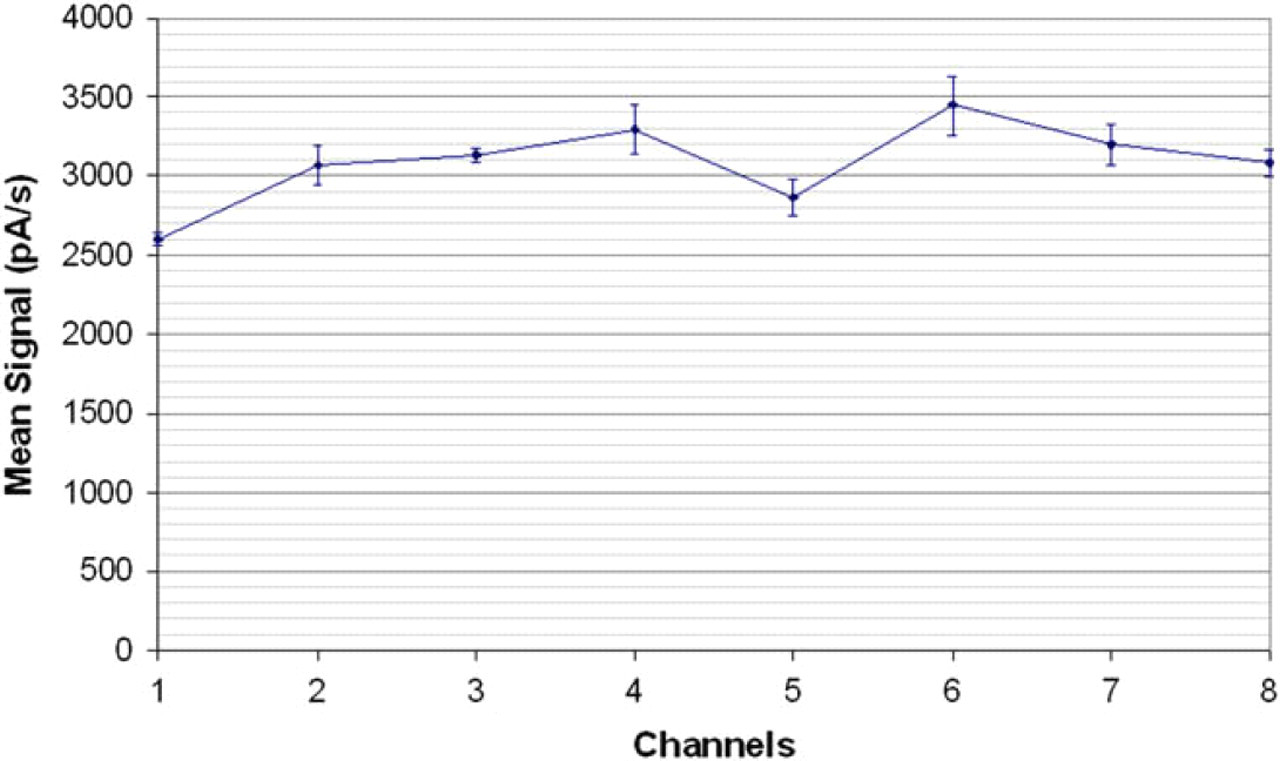
GRAVI-Lab qualification curve, showing signal averages and standard deviations obtained from ten successive determinations of biotinylated alkaline phosphatase (31.25 ng/mL) coupled to streptavidin-coated microbeads (0.05 mg/mL) in the eight different channels of a chip.
Example Application: Therapeutic Antibody Monitoring
For the production of therapeutic antibody, ELISA testing is an ideal procedure to identify the appropriate clone, to select optimal production conditions, as well as to check integrity and reactivity of the product, during and after processing. Nevertheless, developers often only quantify the product, before tests to verify affinity of the antibody for its therapeutic target are actually performed.
The aim of the present example was to develop an assay kit for capturing human antibody, based on the affinity of protein A for the constant (Fc) region of an antibody. In recombinant protein production, downstream processing strategies are often based on the capture of the Fc region by lengthy preparative chromatography. It is therefore of interest to develop a rapid means of testing.
For this purpose, a competitive ELISA was optimized on the GRAVI-Lab platform. Paramagnetic beads of 1 μm diameter and coated with protein A (New England Biolabs, USA) were mixed with the supernatant of mammalian cell cultures containing immunoglobulin G1 (IgG1) antibody, and with a fixed concentration of ALP-labeled antigoat IgG (produced in rabbit: Sigma Aldrich) serving as competition partner (see Fig. 6). In the presence of a large concentration of recombinant antibody, the bead-coupled protein A is saturated by nonlabeled antibody, thus resulting in a weak signal. The signal, however, rises with decreasing recombinant antibody concentration (because a larger amount of protein A binding sites are occupied by the more abundant, ALP-labeled, IgG).
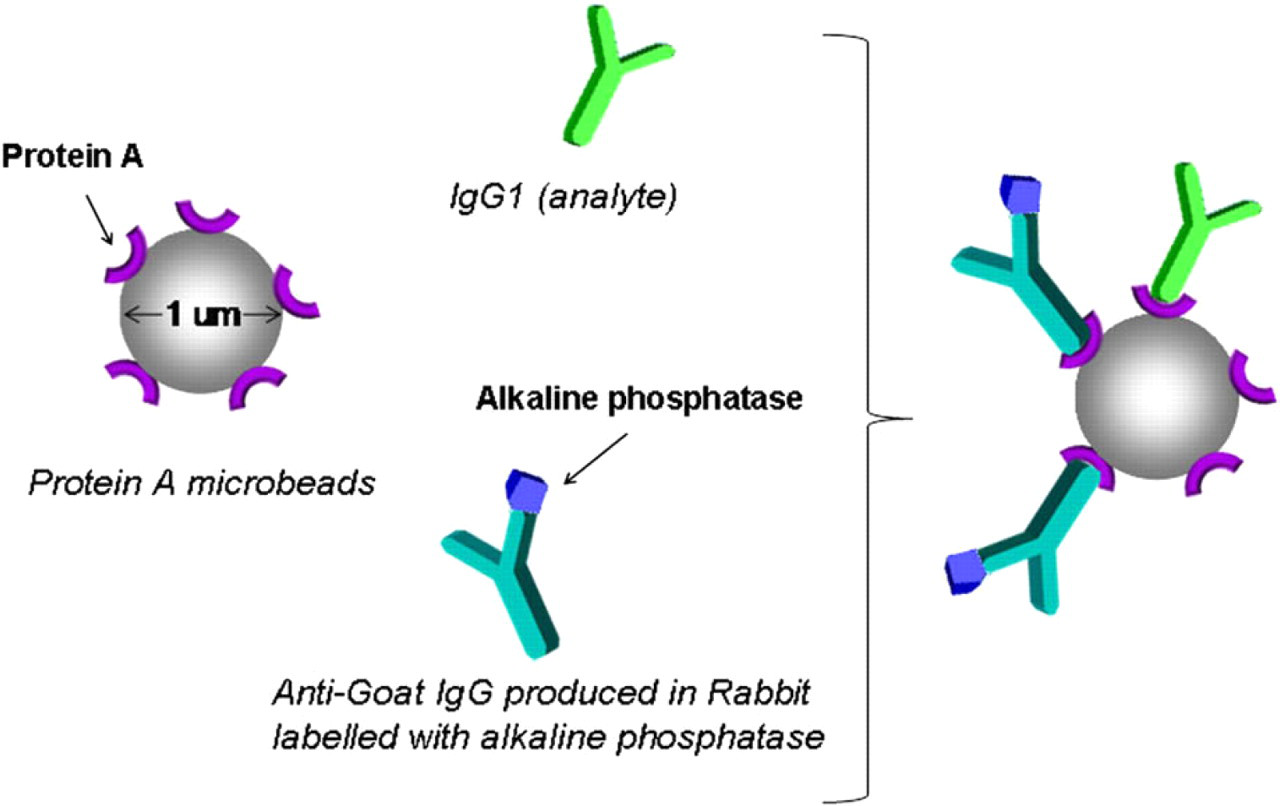
Competitive enzyme-linked immunosorbent assay principle for therapeutic antibody detection (mixing of protein A-coated beads with immunoglobulin G sample and alkaline phosphatase-labeled conjugate to form the affinity complex; after 2-min incubation, the mixture is dispensed into the GRAVI-Chip).
In this competitive immunoassay, 2-μL IgG solution was mixed with 10-μL protein A-coated bead solution in the reaction plate, to which 10-μL solution of enzyme-labeled secondary antibody was added. For the different bead and conjugate concentrations tested, see below. The mixtures were incubated for 2 min, before being dispensed into the GRAVI-Chip. Upon mixture flow, the microchannels were washed with 20-μL washing buffer, which was let flow though the microchannels for 30 s.
After the addition of 20-μL PAPP substrate buffer, the amperometric detection was performed ten times in each of the 16 microchannels, and the Michaelis-Menten kinetics was hence followed over 1 min. Counting in the time required for the robot to pipette the various solutions and wash the pipetting heads between each action, the complete assay duration after this protocol was 9 min, including the chip regeneration time.
Assay Optimization
To determine the most appropriate conditions for analyzing IgG in cell-culture samples on GRAVI-Lab, we fine tuned the concentrations of protein A beads and of secondary antibody. A stock solution of recombinant antibody (at a concentration of 400 μg/mL) (graciously provided by Selexis, Switzerland) was used for calibration. Eight calibrators were prepared by serial dilutions (1:2) of the stock solution with washing buffer, yielding calibration solutions of different IgG1 concentrations (decreasing from 400 to 3.1 μg/mL).
In the first round of experiments, the optimal bead concentration was determined by variation of this parameter: from 0.5 to 0 mg/mL, while maintaining a fixed concentration of ALP-labeled IgG (stock solution diluted 1:4000). As shown in Figure 7, there is a clear dependence between signal and bead concentration. The signal-to-noise ratio was good (>10) for bead concentrations greater than 0.02 mg/mL. To ensure signal detection and minimize consumption, a bead concentration of 0.05 mg/mL was chosen for the further test optimizations.
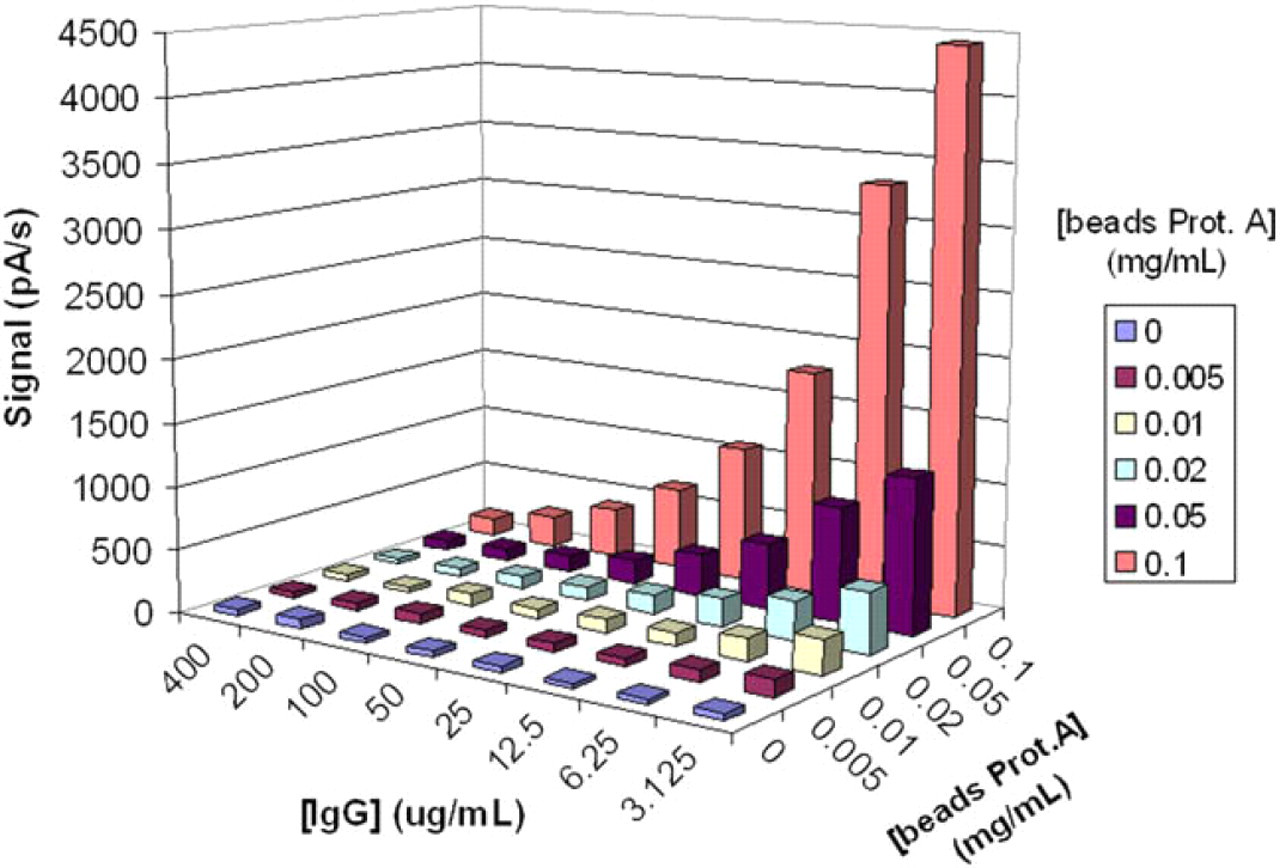
Three-dimensional representation of the current signals for the competitive immunoassay of human immunoglobulin G (IgG) in GRAVI-Lab, as a function of IgG concentrations (varying from 400 to 3.1 μg/mL) and of protein A-coated bead concentrations (ranging from 0.1 to 0 mg/mL).
In the second experiment round, the conjugate variable was optimized by testing different concentrations thereof (dilution factor ranging from 1:2000 to 1:15,000), while maintaining a constant bead concentration (0.05 mg/mL). As shown in Figure 8, competition was insufficient at lower enzyme concentrations, which did not allow discrimination of higher IgG concentrations. Conversely, highest conjugate concentrations did not allow for detection of low IgG concentrations (because competition cannot take place in these conditions, and beads are therefore saturated by enzyme conjugate). A conjugate dilution of 1:4000 was deemed best for efficient competition with recombinant antibody.
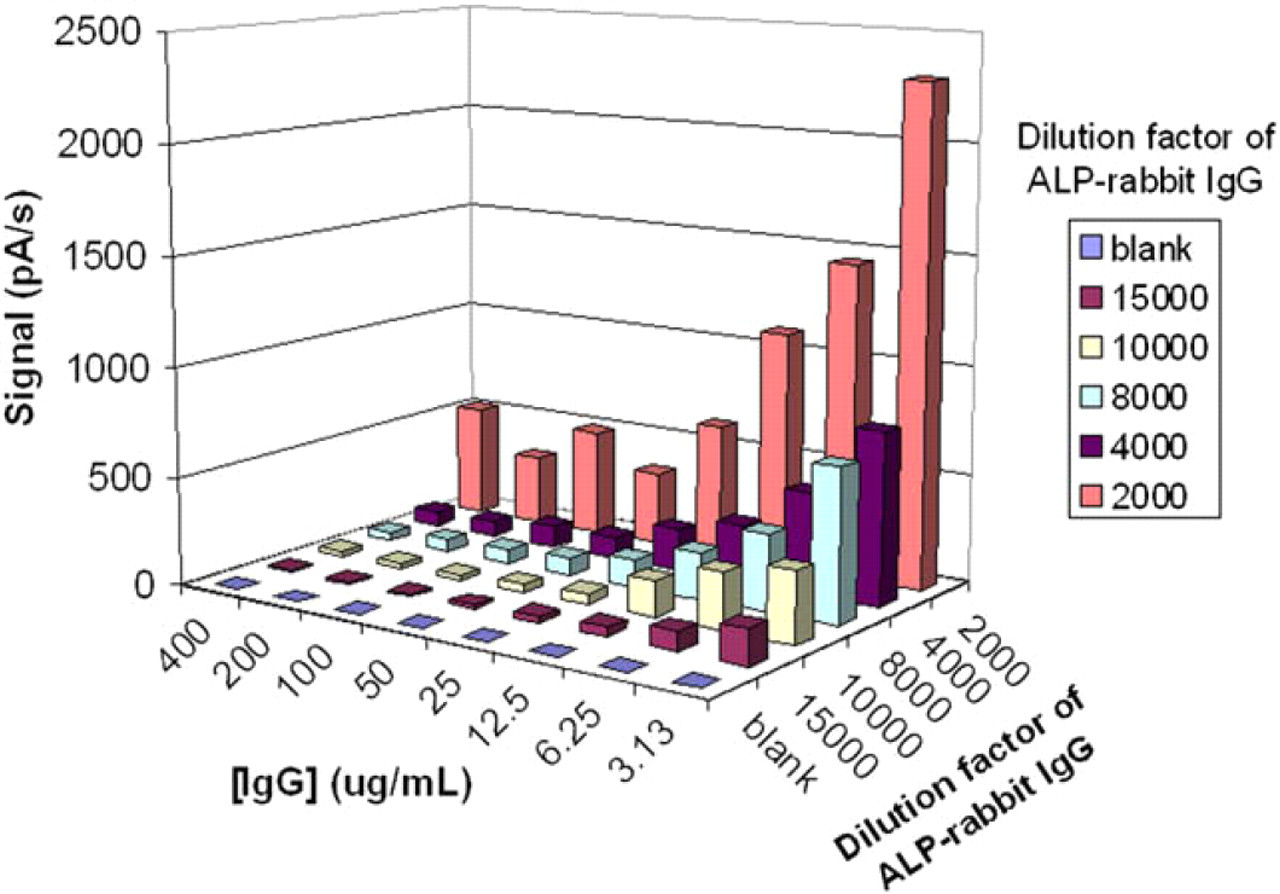
Three-dimensional representation of current signals in a competitive human immunoglobulin G (IgG) immunoassay on GRAVI-Lab. Signals vary in function of IgG concentrations (ranging from 400 to 3.1 μg/mL) and of alkaline phosphatase-labeled rabbit IgG dilution factor (from 1:2000 down to 1:15,000, plus blank).
Detection of Recombinant Antibody
To assess GRAVI platform performance in real conditions, the variable parameters were set in function of the findings mentioned above (dilution factor of 1:4000 for the conjugate concentration, and protein A beads at a concentration of 0.05 mg/mL).
The eight calibration solutions of IgG1 samples prepared above were analyzed following the protocol outlined above. A typical calibration curve example, showing the signal average over eight experiments at each concentration, is presented in Figure 9. The results reveal the typical sigmoïdal curve of a competitive ELISA, with a dynamic range covering two orders of magnitude.
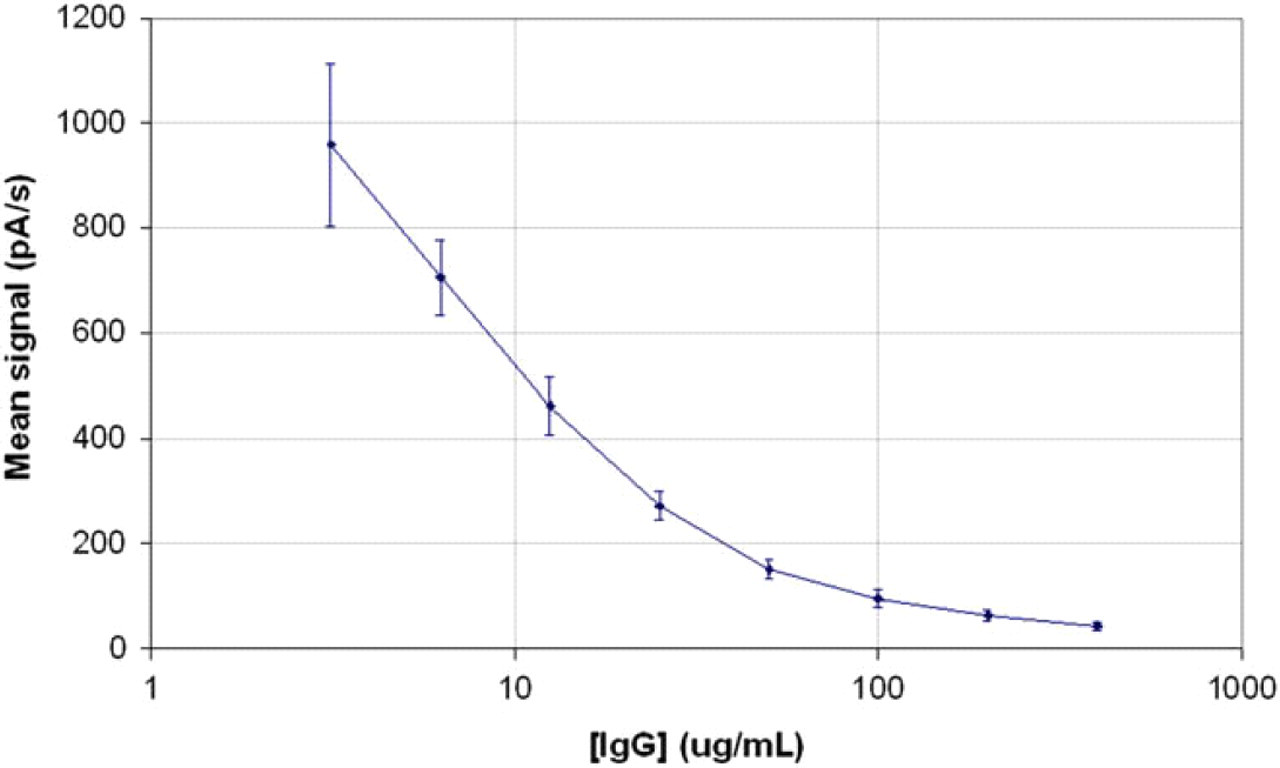
Example of calibration curve for the detection of human immunoglobulin G (IgG) by competitive enzyme-linked immunosorbent assay in GRAVI-Lab, using protein A-coated beads at a concentration of 0.05 mg/mL and an alkaline phosphatase-labeled rabbit IgG conjugate dilution factor of 1:4000.
As indicated by the error bars in Figure 9, the coefficient of variation ranges from 10% to 15%. Due to robot pipetting errors during sample preparation steps, the standard deviation is larger in this example than was in the ALP experiment described previously. Optimization of robot parameters and incubation times is on going, in view of decreasing variation coefficients for the performance of assays with the automated GRAVI-Lab, in fully unattended mode.
Following the outlined protocol, GRAVI-Lab processes 96 samples in 55 min without the necessity of manual intervention during the assay procedure. Including 3-min time for the chip regeneration, total duration for processing 16 assays was roughly 9 min.
Conclusion
The so-to-say maintenance-free GRAVI concept (with no pumps, tubes, nor valves) is an innovative solution to combine the speed and economics of microfluidic systems, with standard robotics. Because compatible with conventional 96-well plate format, GRAVI-Lab represents a flexible and convenient solution replacing sample preparation, incubation, washing, and detection stations: all in one.
Here, the GRAVI system was coupled to a robotic liquid-handling station, which allows performing up to 4 × 96 tests unattended and yields data at a pace of 16 results every 9 min.
As demonstrated by quantification of IgG in mammalian cell-culture aliquots, GRAVI performs competitively (dynamic range of greater than two orders of magnitude in the present competitive assay), with the additional advantages of minimal time to results and extremely low sample and reagent consumption.
With microchips suited for multiple regenerations, the GRAVI technology is a time- and cost-efficient ELISA platform with which to enhance open robotic stations. GRAVI is ideally positioned where fast results are needed, where tests are performed on scarce and precious specimens, and where automation is required to reduce hands-on time for immunoassay processing.
Acknowledgments
The authors would like to thank the European Commission for financial support of part of this work (NeuroTAS project of the 6th EU Research program, grant number NMP4-CT-2003-505311).
