Abstract
We have developed a fully automated laboratory robotic system (FA-LAS) capable of conducting most of routine experiments. Main objectives for developing this novel robotic system are to streamline the analytical tasks by automating sample preparations and analytical procedures, and to ensure the safety of analysts by assigning risk-involving procedures, such as handling of highly active substances.
FA-LAS integrates numbers of unique functional devices and is highly competent for analyses of various research and development products.
Comparative evaluations of FA-LAS with conventional manual preparation for drug substances and pharmaceutical products' real quantitative high performance liquid chromatography (HPLC) tests fulfilled this study's targeted criteria, which are 100 ± 5% for accuracy and coefficient of variation (%CV) ≤ 3% for repeatability for both HPLC analyses of drug substances and pharmaceutical products. In addition, several advantages over conventional manual methods were confirmed, such as reducing “working hour” for analysts, eliminating “working-hour” restriction, and decreasing drastically lag time for each prepared sample toward HPLC analysis.
FA-LAS's unique features will work as a bridge between the sample management system and automatic documentation system, and it will reduce drastically in human resources, budgets, and time frame for development of pharmaceutical products.
Introduction
The development of pharmaceutical products requires tremendous amounts of routine analyses to guarantee the quality of pharmaceutical products. To date, various types of laboratory automation systems have been developed to help expedite analyses, but no single system has been capable of conducting fully automated analyses that include sample preparations for pharmaceutical products. Therefore, a fully automated laboratory robotic system (FA-LAS) capable of conducting most routine experiments has been developed.
The FA-LAS was developed primarily to streamline analytical tasks by automating sample preparations and analytical procedures, and to ensure the safety of analysts by assigning risky procedures to the system, such as handling of highly active substances.
The FA-LAS integrates a number of unique functional devices and is highly competent for the analysis of various research and development products. Devices include a volume adjuster using volumetric flasks, a decrimper capable of removing rubber stoppers altogether, a multivalve high-throughput autosampler for high performance liquid chromatography (HPLC) analyses, and a novel powder dispenser. The newly developed powder dispenser offers highly accurate and precise performance with almost all types of powders, including sticky, clumped, or ultra-fine powders.
Also, a multifunctional handling arm and varieties of unique devices allow the handling of any sample form (liquid, powder, tablet, capsule, and lyophilized samples) in any type of container (syringe, crimp-top vials, and screw-cap vials/tubes).
FA-LAS is designed to save time for analysts by automating experimental preparations, enhance analytical task efficiencies by handling routine experiment work for analysts during working hours or off-hours, eliminate analyst-to-analyst technological transfer involving manual operations, and improve safety for analysts by preventing exposure risks to highly active substances, etc.
Although routine analyses are “time/money/effort consuming” in the drug development process, sample management, documentation, and checking the data and document can be even more “time/money/effort consuming.”
A sample management system capable of fully automated sample storage and sample retrieval, including entering and dispatching management of stored sample, is already available from several life sciences supply companies; and an automatic documentation system able to create text is being developed.
In the meantime, FA-LAS's unique features can work as a bridge between a sample management system and an automatic documentation system, and it can contribute to achieving “total automation” from sample management through documentation.
In this article, we introduce details of the system's components and the performance advantages of FA-LAS over conventional manual operations. Also, to confirm the capability of FA-LAS for development studies of pharmaceutical products, comparative evaluations of FA-LAS with conventional manual preparation are reported as part of Performance Qualification studies for actual operation.
Methods and Materials
Overview and System Design of FA-LAS
Overview and system design of FA-LAS are shown in Figure 1. Figure 1A-C illustrates how FA-LAS is composed of a main unit, which assumes sample preparation of various sample forms in various containers, and an HPLC unit, which can assume up to four different HPLC analyses of prepared samples. The system design of FA-LAS is shown as a schematic image (Fig. 1D), because the main unit is hermetically closable, and inside conditions for temperature and humidity are controlled constantly using the temperature- and humidity-adjusting device (4–3 in Fig. 1D). This function enables reliable experimental results for temperature and/or humidity sensitive experiments, and it enables the handling of deliquescent powders, which typically are almost impossible to handle in humid conditions.

Overview and system design of fully automated laboratory robotic system (FA-LAS). (A) Main unit. (B) HPLC unit. (C) Inside view of main unit. (D) System design of FA-LAS.
System Components of FA-LAS
System components of FA-LAS are listed by function in Table 1, which also demonstrates how FA-LAS integrates different functions with unique functional devices (Figs. 2–8).
System components of FA-LAS


Tumbling device.

Decrimping device.

Capping/decapping device.

Devices for adjusting volume. (A) Prefilling of volumetric flask by volumetric flask declining/rotating device and two-channel pipetting nozzle. (B) Monitor image of automatic volume adjusting device. The mechanism of volume adjusting is based on three-point (center and both sides) recognition for liquid surface and gauge line of volumetric flask with high-speed CCD camera, and distance moving by each droplet solution is optically detected to predict end points.

Multifunctional handling arm. The arm consists of three hands (hand I–III). Hand I: Pipetting nozzle, pH meter, volumetric pipette, and extension arm for handling microplate and the HPLC sample tube rack. Hand II: Volumetric flask. Hand III: Crimp-top vial, screw-cap vial/tube, and volumetric flask.

Multipurpose four-channel dispenser. The arm consists of four dispensing probes (probe I–IV). Probe I: Liquid-sampling probe. High-volume (up to 1200 μL) disposable tip and piercing tip. Probe II: Liquid-sampling probe. Low-volume (up to 250 μL) disposable tip. Probe III: Gas blow (Air, N2, Ar). Probe IV: Powder dispensing nozzle and specific vacuuming nozzle.

Two-channel pipetting nozzles. Nozzle I (N2 substient): Inner nozzle for liquid pipetting and outer nozzle for N2 gas infusion. Nozzle II (alcohol rinsing): Inner nozzle for dispensing alcohol and outer nozzle for aspirating alcohol. Nozzle III (prefilling volumetric flask): Inner nozzle for liquid pipetting and outer nozzle for N2 gas infusion.
Multifunctional Feature of FA-LAS
A list of functional devices for different sample types and different containers is presented in Table 2 along with information about functions and quantitative capabilities. Multifunctional capability is one of the most important features of FA-LAS, and these varieties of unique functional devices permit most routine analyses using any sample form in any type of container.
List of functional devices by their specificity for each sample form and container

Comparative Evaluation of FA-LAS with Conventional Manual Preparation
Comparative evaluations of FA-LAS and conventional manual preparation are tested as part of Performance Qualification studies. HPLC analyses, which are real quantitative tests for the drug substances and pharmaceutical products, are carried out by FA-LAS. In these evaluation studies, concentrations obtained from peak areas are subjects for judgment, and the criteria for these evaluation studies are set to 100 ± 5% for accuracy and coefficient of variation (%CV) ≤ 3% for repeatability.
Method for Drug Substances
A system flowchart and time schedule for HPLC analysis of drug substance samples are shown in Figure 9A as a schematic image.

System flowchart and time schedule for HPLC analyses of drug substance and pharmaceutical product. (A) Drug substance. (B) Pharmaceutical product.
Briefly,
Drug substance sample solutions in screw-cap tubes were incubated at 37 °C for 3 h. After homogenizing the sample solution in a screw-cap tube by five times of tumbling manipulation with the tumbling device, 400 μL of each sample was diluted to 20 mL with mobile phase of HPLC analysis (50 mmol/L Sodium Phosphate pH 7.0, 300 mmol/L NaCl, and 0.05% Sodium Azide) in a 20 mL volumetric flask. The diluted sample solution was mixed by stirring with a magnetic stirrer. Two hundred microliters of the diluted sample solutions and a standard solution were dispensed into HPLC sample tubesand placed in the HPLC unit. Sixty microliters of each sample solution and standard solution were analyzed by the HPLC device.
Method for Pharmaceutical Products
A system flowchart and time schedule for HPLC analysis of pharmaceutical product samples are shown in Figure 9B as a schematic image.
Briefly,
Pharmaceutical product sample solution in crimp-top vials was homogenized by five times of tumbling manipulation with the tumbling device. After removing an aluminum lid and rubber stopper of the crimp-top vial by the decrimping device, 1 mL of each of sample was diluted to 20 mL with the mobile phase of the HPLC analysis in a 20 mL volumetric flask. The diluted sample solution was mixed by stirring with a magnetic stirrer. 200 μL of the diluted sample solution and a standard solution were dispensed into HPLC sample tubes, and placed in the HPLC unit. 60 μL of each sample solution and standard solution were analyzed by the HPLC device.
In addition, the prepared samples were analyzed by the validated HPLC system (2695 Separation Module, Waters) to evaluate accuracy and repeatability of sample preparation steps by FA-LAS and its HPLC unit.
Software of FA-LAS
FA-LAS is controlled by two separate software programs: LTRoboticSystem and LTAutoSamplerSystem, which are written using Microsoft VisualBasic. NET 2003, and developed by INTRON (Fig. 10A, B). LTRoboticSystem controls the operation of each device in the main unit using RS232C, and LTAutoSamplerSystem controls operation of each device in the HPLC unit using RS232C. The LTRoboticSystem and LTAutoSamplerSystem are connected by RS232C.

Fully automated laboratory robotic system software top-level screen images. (A) LTRoboticSystem. (B) LTAutoSamplerSystem.
Results
Comparative evaluations of FA-LAS with conventional manual preparation for the HPLC analyses of drug substances and pharmaceutical products as part of Performance Qualification studies.
Overall Accuracy and Repeatability
Method for Drug Substance As shown in Table 3, accuracy and repeatability for drug substance's HPLC analysis were 99.4 ± 1.4% and 1.4% ( Accuracy and repeatability for comparative evaluation studies of fully automated laboratory robotic system (FA-LAS) with conventional manual preparation
%CV: Coefficient of variation
Method for Pharmaceutical Product As shown in Table 3, accuracy and repeatability for the pharmaceutical product's HPLC analysis were 101.9 ± 3.2% and 3.2% (
To evaluate the accuracy and repeatability of the sample preparation step of FA-LAS and its HPLC unit, prepared samples of pharmaceutical products were analyzed by a validated HPLC system, and the result of this evaluation study was compared with the overall accuracy and repeatability of pharmaceutical products. As shown in Table 3, accuracy and repeatability for this study was 102.3 ± 0.4% and 0.4% ( These results suggest the defectiveness of the HPLC unit in FA-LAS. After conducting extensive investigations, the cause of the decreased accuracy and repeatability for these comparative evaluation studies turns out to be the newly developed parallel HPLC autosampler. Upgrading of the parallel HPLC autosampler is currently in progress.

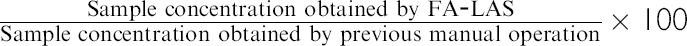
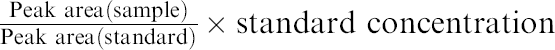
Advantages of FA-LAS
Method for Drug Substance As shown in Table 4, “working hours” for analysts and the total time required for the HPLC analysis of drug substance was reduced more than 4 h when the FA-LAS method was compared with the conventional manual method. Comparison of “working hour for analyst” between FA-LAS and manual operation FA-LAS = fully automated laboratory robotic system.

Method for pharmaceutical product
As shown in Table 4, “working hours” for analysts and the total time required for the HPLC analysis of pharmaceutical product was reduced more than 2 h when the FA-LAS method was compared with the conventional manual method.
In the FA-LAS method, preparation time for additional samples is easily absorbed by the HPLC analysis time, so the effect of increasing the number of samples is insignificant. Therefore, it is obvious that the differences between conventional manual methods and the FA-LAS method increase as the number of samples increases.
Method for Drug Substance As shown in Table 5, time lag in the HPLC analysis was reduced more than 10 h when the FA-LAS method was compared with the conventional manual method. Comparison of “time lag” for each prepared sample toward the HPLC analysis FA-LAS = fully automated laboratory robotic system. This time lag is possible to be shortened down to less than 10 min.

Method for Pharmaceutical Product
As shown in Table 5, time lag in the HPLC analysis was reduced more than 20 h when the FA-LAS method was compared with the conventional manual method.
In FA-LAS method, waiting time for each sample in the HPLC analysis is easily controllable regardless of the number of samples. Therefore, it is obvious that the differences between conventional manual methods and the FA-LAS method increase when the number of samples increases. Moreover, if the time lag is increased even more, the stability of sample solutions in autosampler of HPLC system must be confirmed.
Conclusion
Comparative evaluations of FA-LAS with conventional manual preparation for HPLC analyses of drug substances and pharmaceutical products confirm that FA-LAS is capable of conducting fully automated HPLC analyses, including sample preparations. Because the newly developed parallel HPLC autosampler is unfinished, overall accuracy and repeatability is just about enough to fulfill the criteria for these evaluation studies. Upgrading of the parallel HPLC auto-sampler is currently in progress, however, accuracy and repeatability of sample preparation steps are comparable to the manual preparation conducted by most of the workers and researcher in our department. This precise sample preparation capability of the FA-LAS improves robustness of most analyses requiring sample preparation, and eliminates analyst-to-analyst technological transfer.
In addition, other than expected advantages, such as reducing “working hours” for analysts, eliminating “working-hour” restrictions, and cutting down total analysis time, the evaluation studies of fully automated HPLC analyses by FA-LAS demonstrate an important advantage over manual preparation by drastically reducing lag time for each prepared sample in the HPLC analysis. This additional advantage eliminates concerns about storage time of prepared samples in poorly temperature-controlled devices like most of HPLC autosamplers. The parallel HPLC autosampler in FA-LAS is designed to have better temperature-control capability (4 °C∼ room temperature) than most commercial autosamplers.
In addition to the HPLC analyses of drug substances and pharmaceutical products, many other analysis evaluation studies are planned, including HPLC analysis of powder and tablet samples, peptide mapping, sugar chain analysis, and a sample preparation of water content assay for freezedried products.
Actual operation of FA-LAS is starting with fully automated stability assays based on the HPLC analyses of drug substances and pharmaceutical products evaluated in this study.
After beginning fully automated actual operation of FA-LAS, bridging the study of the sample management system and automatic documentation system using FA-LAS is planned for the development of a “total automation system.” In the development process of pharmaceutical products, there are strict rules for handling drug samples and standard substances and their records, such as log data for storage temperatures, sample and standards use records, and entering and dispatching records. These troublesome duties along with test results documentation will be swept away with a commercially available sample management system combined with an additional weighing system.
A “total automation system” composed of a sample management system, FA-LAS, and automatic documentation system will lead to drastic changes in human resources, budgets, and time frames for the development of pharmaceutical products.
Acknowledgment
We gratefully acknowledge Intron Co., LTD. for its support in the development and construction of FA-LAS.
