Abstract
Automation systems are well established in industries and life science laboratories, especially in bioscreening and high-throughput applications. An increasing demand of automation solutions can be seen in the field of analytical measurement in chemical synthesis, quality control, and medical and pharmaceutical fields, as well as research and development. In this study, an automation solution was developed and optimized for the investigation of new biliary endoprostheses (stents), which should reduce clogging after implantation in the human body. The material inside the stents (incrustations) has to be controlled regularly and under identical conditions. The elemental composition is one criterion to be monitored in stent development. The manual procedure was transferred to an automated process including sample preparation, elemental analysis using inductively coupled plasma mass spectrometry (ICP-MS), and data evaluation. Due to safety issues, microwave-assisted acid digestion was executed outside of the automation system. The performance of the automated process was determined and validated. The measurement results and the processing times were compared for both the manual and the automated procedure. Finally, real samples of stent incrustations and pig bile were analyzed using the automation system.
Keywords
Introduction
The automation of life science processes requires the development of suitable robotic concepts to support a complete automation. 1 Highly developed industrial automation solutions are mainly designed and realized as proprietary systems for specific applications and are used for some years without any major system modifications. Two general concepts for robot application can be found in classical industry automation: as transport systems or for direct task execution. 1 Manual processes are transformed to automated processes using the robot. The human arm and hand should be replaced by the robot arm with an end effector to reproduce human skills. Application areas are the automotive industry, precision engineering, the food industry, heavy-duty mechanical engineering, electrical engineering, and medicine. 2 In such automation systems, robots are used for single tasks, such as welding, painting, screwing, and polishing. 2
Since the beginning of life science automation, several robotic concepts have been developed to support a complete automation.1,3 The aim of automating life science processes is the design and realization of systems that ensure the typical range of functions required for biological, chemical, biochemical, and analytical processes. According to the included functionalities, different specific applications can be performed on these automation systems. The automation concepts in life sciences can be classified according to the type of usage of the robot: central system integrator, flexible robot, and integrated robotics. 1 In the first concept, the robot is used as a system integrator and acts only as a transportation element for material transfer between several substations to realize essential tasks, such as dispensing, aliquoting, heating, shaking, and measuring. This concept is widely used in life science processes.1,3–9 It ensures a relative high flexibility and is mostly realized by a hierarchical system design. The task-performing devices have to be adapted to the robot handling to enable an automated delivery and pickup of the samples by the robot.
In this study, an existing automation system was further enhanced to extend the applicative scope.4,5,10 The system follows the concept of the central system integrator. 1 The locally decentralized and applicative open system design 1 enables flexible use in a multitude of completely different applications in both structural analysis 4 and elemental analysis.5,11 –13 The flexibility was reached by suitable and easily changeable adaptions according to the application processes to be executed. The new process involves the sample preparation of solid stent incrustation samples, including microwave digestion for mineralization, dilution of the sample solutions, analysis using inductively coupled plasma mass spectrometry (ICP-MS), and evaluation of the measurement results.
Background and Applicative Scope
Various diseases of the gall bladder and the biliary tract require an endoscopic implantation of a biliary endoprosthesis (stent).14 –21 This form of therapy results in low complications22,23 but has a major problem: inside the stents, deposits may accumulate to a complete closure, which requires the stent replacement.15,24 –26 The reasons for closure are still not completely clear. 15 The clogging occurs mainly in stents made of plastic; biofilms of bacteria and fungi are involved in sludge formation.17,27,28 As a contribution to develop new stents with no or low clogging, and therefore to improve this form of therapy, an analysis procedure to determine the elemental composition of the incrustations was developed. In a first study, a manual miniaturized method for the determination of calcium and trace metals in the stent incrustations using ICP-MS was validated. 29 An automated method to ensure reliable and comparable results, as well as to increase the sample throughput, was developed and validated. The measurement results and the processing times are compared for both the manual and the automated procedure. In addition, real samples of both incrustations of biliary endoprostheses and pig bile were analyzed.
Aims of Automation
Nearly all subprocesses of this application can be fully automated. With respect to additional costs for special automated devices, some subprocesses, such as crushing, drying, fine milling, and weighting, are currently performed manually. The microwave digestion procedure can also be integrated into the automation system. A microwave digestion device requires an additional exhausting system to prevent damage to the sensitive mechanical robot parts due to highly toxic and corrosive vapors generated. For this reason, the microwave device was not integrated into the automation system presented. Thus, additional transportation steps between these two stations are required, which can be performed by human operators or mobile robots.
In the first subprocesses, the incrustations were removed from the stents, dried at room temperature to constant weight, finely ground using a mortar, and stored in sample containers. Self-made reference material for method and system validation can directly be used; no further treatment is required. 29 The following steps include weighing, adding digestion acid (nitric acid, HNO3), predigestion for 20 min, final dilution, and analysis. The analysis is performed using ICP-MS, which also requires an additional exhausting system. In this study, an external ICP-MS device in another room was used, but integration into an automation system is also possible. 1 Figure 1 gives an overview of the general process workflow.
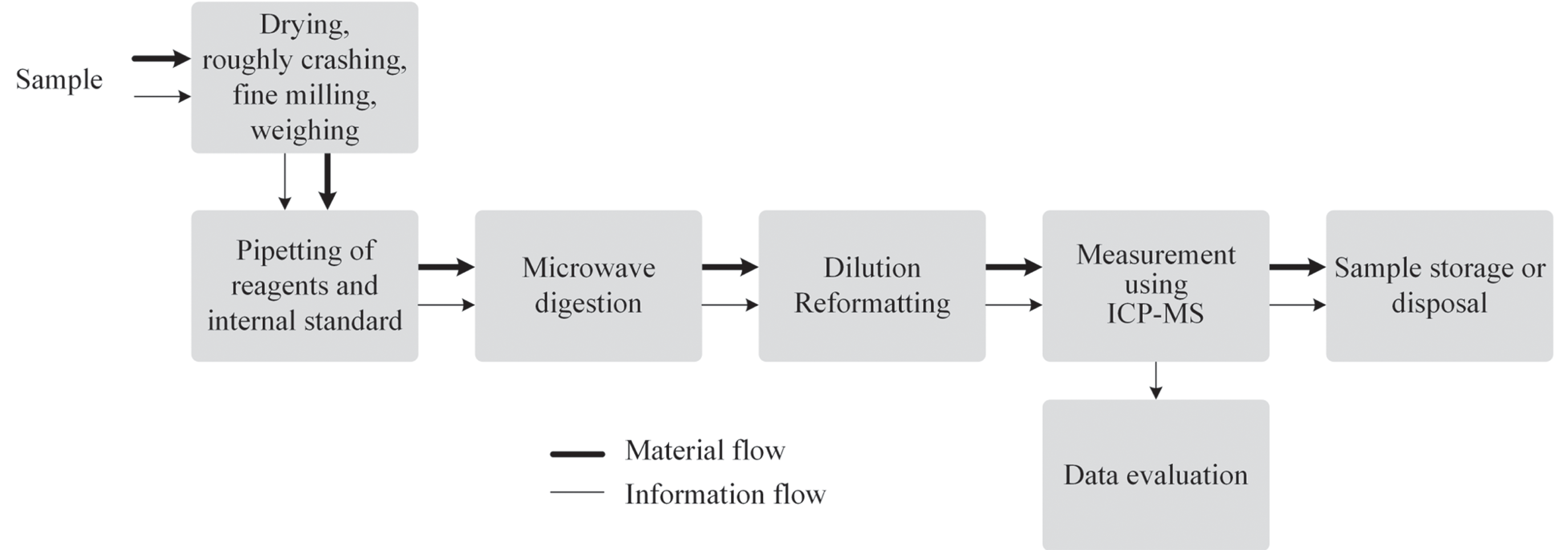
General process workflow for the determination of calcium and trace metals in stent incrustation samples using ICP-MS.
For the automated procedure, several subprocesses can be defined. The subprocess of liquid handling includes pipetting of the standard solutions, the addition of the internal standard to the digestion acid, and the addition of the prepared digestion acid to the solid sample. The subprocesses of dilution and reformatting involve the final dilution (1:12.5) of the digested sample solution to decrease the acid concentration and the transfer of the sample solutions from the microwave digestion vessels (vol. 10 mL) in vials, which can be handled by the autosampler of the ICP-MS (vol. 8 mL). The subprocess of analysis using ICP-MS involves a multielement analysis method for simultaneous determination of calcium and several trace metals. The subprocess of transport includes the initial transport to supply the weighed samples prepared in microwave digestion vessels, digestion acid, ultrapure water, and labware. Further transportation tasks between several stations of the automation system are realized by the central system integrator. The final transportation step is needed for storage and/or disposal of samples, chemicals, and used labware.
One aim of automating the sample preparation of incrustations in biliary stents and bile was to increase the operating safety for the laboratory staff. The human operator is protected from potential hazards, such as concentrated acid, toxic digestion solutions, and the formation of harmful nitrous gases. Furthermore, in manual operation the human operator has to work with his arms and hands under the exhaust system but with his head outside. For pipetting multiple samples (in detail, pipetting concentrated acid to solid samples and dilution of steaming acid digestions), partially ergonomically unfavorable postures are required. An automation of these processes prevents overexertion, stress, or diseases of the musculoskeletal system.
Another aim for automation is an effective time use of laboratory processes. The automated pipetting procedure needs more time than manual pipetting caused by using two liquid handlers and several transportation steps. But in this time, the laboratory assistant is free and can perform other processes. Thus, a higher number of samples can be processed in parallel, which increases the performance and throughput of a laboratory. In our thinking, the benefits of automation not only apply for a specific process, but also can have an impact on the overall performance of laboratories.
Furthermore, the automation should reduce pipetting errors, the unintentional contamination of samples and solutions, and mistakes caused by accidental interchanging of the sample vessels. In the method validation, such major human errors are not included since they are not related to the method itself. The reasons for such mistakes are very individual: no optimal daily form or fitness, bad concentration, stress, overexertion, private issues, and so forth. In the case of such errors, the measurement series have to be repeated. But repeating a measurement is not always possible, especially by using real human samples with a very low sample amount. Automation prevents such human-caused deviations and saves time and sample material.
System Concept and System Design
The recent automation system was designed and realized for the automated sample preparation in the field of analytical measurement: in elemental analysis, such as the determination of mercury in wood material, as well as of calcium and phosphor in bones, and in structural analysis.1,4,5,10 –12 The system was adapted and redesigned to the requirements of the determination of calcium and heavy metals in biliary endoprostheses. Therefore, new types of labware were integrated, different liquid handling methods were created and evaluated, and new process control methods were generated.
The automation system consists of the central system integrator and the functional subunits of sample storage, liquid handling, sample treatment, and analytics. Figure 2 shows the complete system as a three-dimensional computer-aided design (3D CAD) drawing.
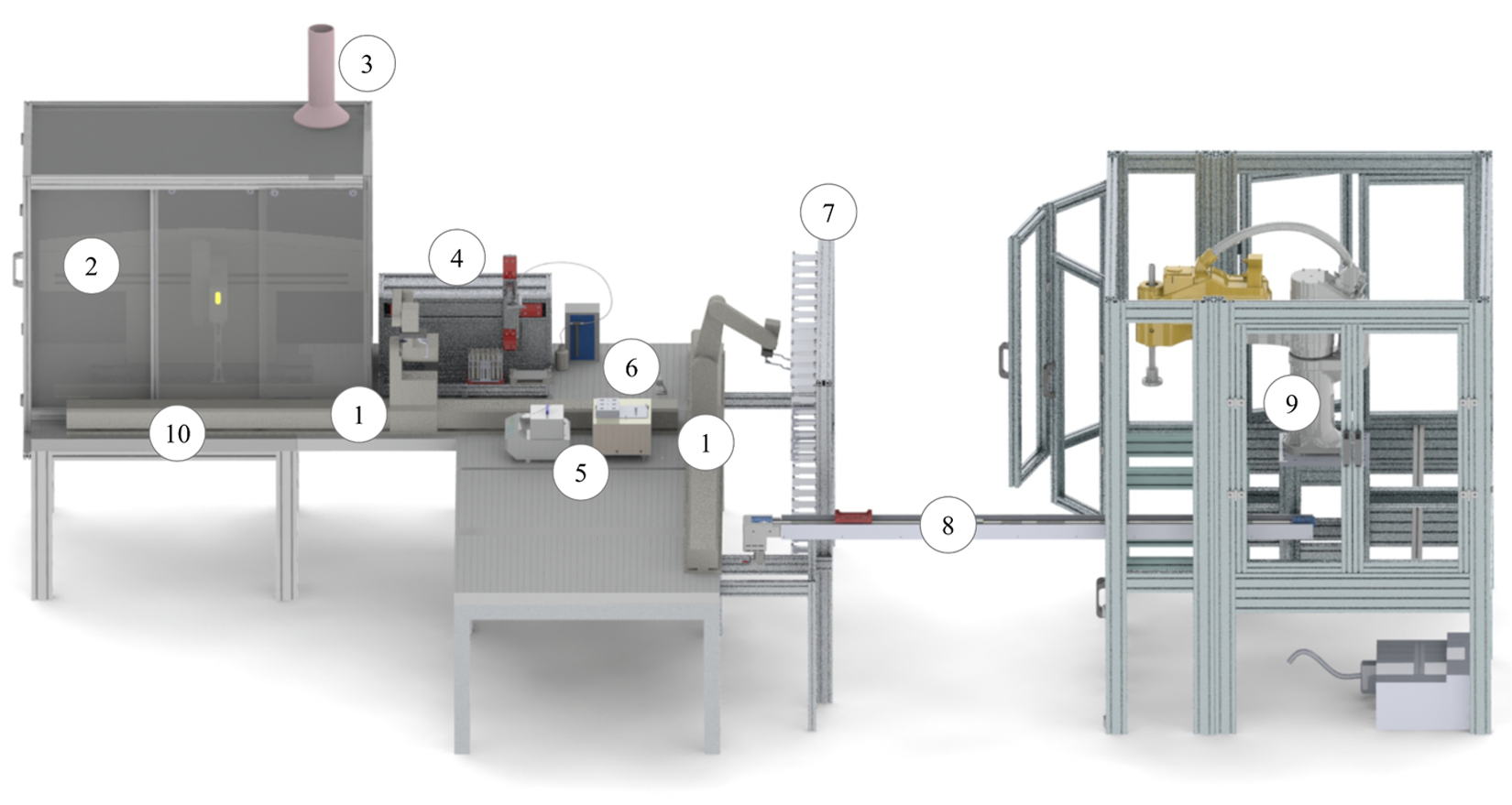
3D CAD drawing of the complete automation system: (1) central system integrator with two ORCA laboratory robots moving on orthogonal linear rails (Beckman Coulter, Krefeld, Germany); (2) liquid handler Biomek 2000 with housing (Beckman Coulter); (3) exhaust ventilation; (4) single-vial liquid handler (celisca, Rostock, Germany); (5) thermomixer Comfort (Eppendorf, Hamburg, Germany) and thermomixer MKR23 (HLC BioTech, Bovenden, Germany); (6) labware regrip station; (7) storage system for labware, reagents, and samples; (8) linear rail with shuttle; (9) SCARA robot TS60 (Stäubli, Bayreuth, Germany) with balance and crimp station; and (10) sample transfer station.
The central system integrator connects the different functional subunits and transports all required materials. Due to the high number of subunits, the workspace was extended using two ORCA laboratory robots (Beckman Coulter) moving on two orthogonal rails. The robots are connected using a regrip station. This allows a bilateral exchange of labware and a regripping in different orientations of the labware. Using intelligent scheduling, the sample throughput can be significantly increased using two ORCA robots.
The sample storage functional unit involves activities and devices related to the safe storage of samples and chemicals. In elemental analysis, all samples and solutions are exclusively arranged in perfluoroalkoxy (PFA) copolymer, polypropylene (PP), polyethylene (PE), or polytetrafluoroethylene (PTFE) vessels. The volumes range from 10 to 100 mL. These single vessels were arranged on special racks in standardized microplate format, which allows easy handling by the robots and positioning at the defined positions (automated labware positioner [ALP]).1,4,5,11 All racks are equipped with PTFE covers to ensure a tight sealing of the single containers. The presented concept comprises the idea of capping the whole rack in microplate format, including the implemented vessels, with just one cover. Irrespective of the vessel diameter, the gripper tools of both the central system integrator and the liquid handler are capable of gripping this cover in one single step for simultaneous opening and covering of up to 24 vessels. One exception is the screwing of caps on the digestion vessels to perform the microwave digestion, which can be performed manually or automated using a capping station. A storage system with 196 positions for sample containers in microplate format with different heights is integrated on the deck to provide chemicals, samples, and labware. A manual or automated feeding of the storage system is possible.
The liquid handling functional unit involves two different liquid handlers. The preparation of calibration standards, the addition of the internal standard and digestion acid, and the final dilution are performed on a liquid handling system, Biomek 2000 (Beckman Coulter). The liquid handler is equipped with a single pipetting tool to enable the processing of single vials. Changeable tips are used to prevent cross-contamination. The pipetting tips have a special filter inside to avoid the raising of corrosive vapors into the pipette cannulas to prevent damage to the sensitive mechanical liquid handler parts. Biomek 2000 can be operated in a typical volume range from 1 to 1000 µL.
An additional single-vial liquid handler was integrated for handling larger volumes (celisca) ( Fig. 3 ).1,4,5,11 The horizontal and vertical needle movements are realized with two linear rails and carriages driven by stepper motors over drive belts. A pneumatic carriage was used to realize the third degree of freedom (orthogonal to horizontal linear rail). One rack with single vessels and the related cover can be placed on predefined positions. The dosage is realized with a ML511C dispenser (Hamilton, Bonaduz, Switzerland) and a 1010TLL syringe (vol. max. 10 mL). The single-vial liquid handler is used for the dosage of water in the application presented. A passive wash station is integrated to avoid cross-contamination, whereby the needle is rinsed from the outside with a solvent collected in a waste container.1,4,5,11 For processing racks with different numbers of single vessels (e.g., 2 × 3 and 4 × 6), the single-vial liquid handler was optimized using a special rack adapter.
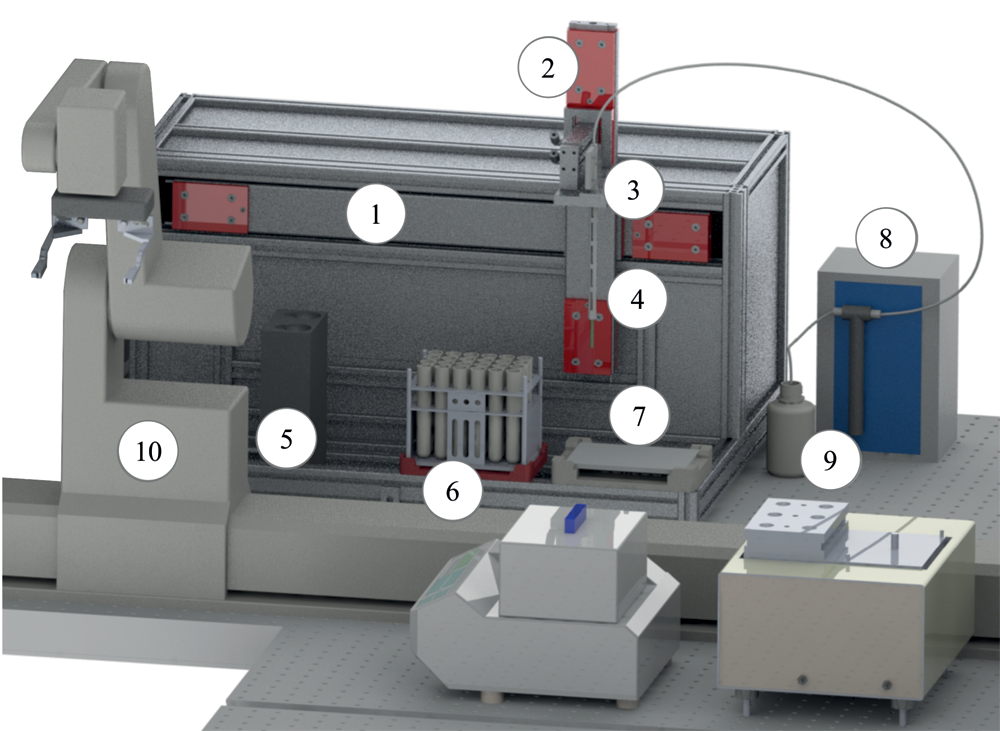
3D CAD drawing of the single-vial liquid handler (celisca): (1,2) horizontal and vertical linear rails, (3) pneumatic carriage, (4) needle with needle guide, (5) wash station, (6) ALP for positioning tube racks, (7) ALP for positioning racks covers, (8) dispenser ML511C (Hamilton), (9) system liquid, and (10) ORCA laboratory robot (Beckman Coulter).
The sample treatment functional unit involves the sample preparation using a microwave-assisted acid digestion to convert the solid samples into a liquid form. This is performed using the microwave device Mars5 (CEM, Kamp-Lintfort, Germany). The device used in this study is located outside of the automation system under a separate laboratory hood due to safety reasons.
The analytics functional unit includes an ICP-MS 7700x (Agilent Technologies, Waldbronn, Germany) and an autosampler ASX-500 (Cetac, Neuss, Germany) for automated sample supply.
Materials and Methods
The manual steps mentioned above were transferred into an automated procedure based on the manual procedure, which is described in detail in a previous publication. 29 The most important materials and instrument parameters used in the automated procedure are briefly described in the following paragraphs.
Chemicals and Reagents
The ICP standard solutions were purchased from Merck (Darmstadt, Germany): multielement standard IV with 23 elements and single-element standards of calcium (Ca), lutetium (Lu), and rhenium (Re), each with a concentration of 1000 mg/L. Lutetium and rhenium were used as internal standards (ISTD1 and ISTD2) for error correction in the sample introduction and for correction of vaporizing effects during the microwave digestion of the solid samples. 12 Nitric acid Suprapur (65%) and hydrochloric acid Suprapur (30%), purchased from Merck, and ultrapure water were used to prepare the microwave digestion, calibration solutions, diluted samples, and rinsing solutions. The methods were validated using self-made reference material containing calcium oxalate, cholesterol, ICP multielement standard IV, and potato starch. 29 To reduce inhomogeneities of this material, it should be new prepared prior to the validation experiments and mixed carefully before weighting.
Preparation of Samples and Standard Solutions
The internal standard rhenium (ISTD2) was mixed with nitric acid (65%) to a final concentration of 1 mg/L. A volume of 1 mL of this acid solution was added to 30 mg sample. The predigestion was performed for 20 min at room temperature with open vessels. The microwave device Mars5 and associated digestion vessels Xpress (vol. 10 mL) from CEM were used in the microwave digestion procedure, which was performed with the following parameters: temperature–time ramp for 20 min to 150 °C (302 °F) and 20 min hold time. After the digestion, the vessels were cooled to room temperature and then uncapped. The dilution was done with 5.75 mL ultrapure water and 0.5 mL digestion solution in a plastic tube (vol. 15 mL). The calibration standards were prepared in nitric acid (8%, v/v) with five concentration levels: calcium with 0.101, 0.505, 1.010, 5.050, and 10.100 mg/L, and all other elements with 1, 5, 10, 50, and 100 µg/L. All solutions were exclusively arranged and stored in PE or PP vessels.
Measurement Parameters
The determination of calcium and metal content was realized using an ICP-MS 7700x (Agilent Technologies) under the following conditions: 1550 W radio-frequency power, 10 mm sample depth, 0.65 L/min carrier gas flow, 0.40 L/min dilution gas flow, 0.10 rps nebulizer pump speed, and 13 °C (55.4 °F) spray chamber temperature. Argon was used as plasma, carrier, and dilution gas. The sample introduction was automatically performed using the autosampler ASX-500 from Cetac (Omaha, Nebraska). The rinse times between two samples were 55 s at 0.3 rps of the nebulizer pump (water with 5% nitric acid and 1% hydrochloric acid), followed by 45 s at 0.4 rps (water with 8% nitric acid).
Results and Discussion
Realized Process
The sample preparation performed using the automation system is divided in two subprocesses: the predigestion and the dilution. In the beginning, the required solutions and labware were manually provided to the storage system. Concentrated acid and digestion solutions were directly provided on the liquid handler Biomek 2000 under the housing to avoid hazardous vapors in the nonhoused area of the automation system. The two ORCA laboratory robots performed the transport steps to the required stations. In the predigestion process, 1 mL of the nitric acid with internal standard was added to the samples in the microwave vessels. After resting for 20 min with open vessels (predigestion), the microwave digestion was performed. Then the vessels were cooled down to room temperature, decapped, arranged in racks, and provided to Biomek 2000. The dilution was performed using Biomek 2000 and the single-vial liquid handler. For the dilution, two different workflows were tested. The diluted sample solutions were transported to the autosampler of the ICP-MS, and analysis was performed. The used labware was transported back to the storage system. Figure 4 shows the complete process (using dilution method 2) with all the required process steps.
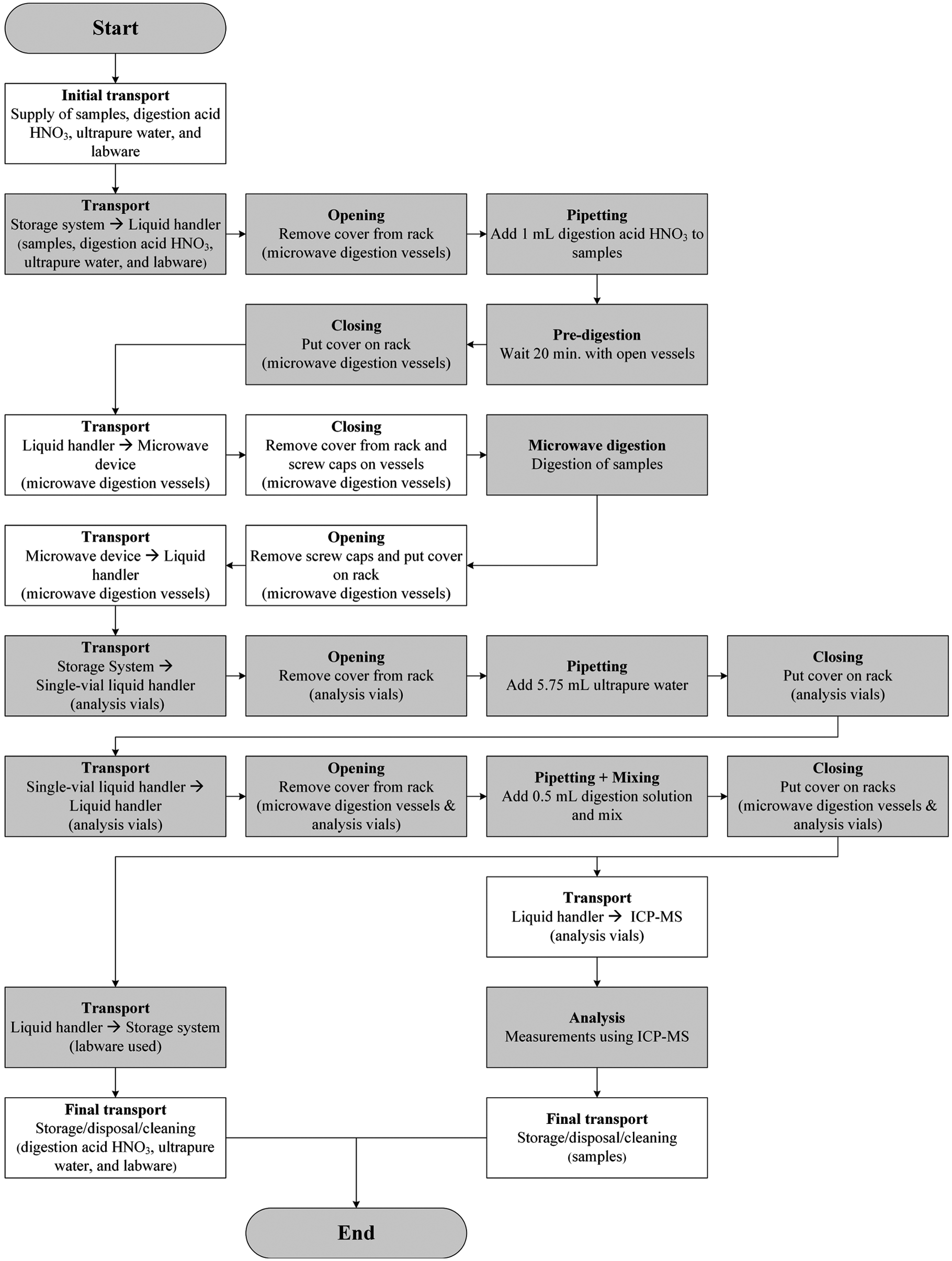
Flowchart to visualize the general workflow of the complete process, including sample preparation and analysis (boxes with white background, manual processing; boxes with grey background, automated processing).
Evaluation of Dilution Methods
There are some challenges to meet in automated dilution of microwave digestion solutions provided in low-volume vessels (10 mL). The microwave digestion is usually performed using magnetic stirrers to mix the solution during the digestion. If no magnetic stirrer is required, boiling stones must be added to prevent boiling retardation. Magnetic stirrers or boiling stones remain in the digested solution during the dilution process in manual procedures. The laboratory staff aspirates the sample solution with the pipette while pushing the stirrer/stones aside. In automated procedures, the liquid handler is not able to push the stirrer/stones aside since their exact positions are unknown. This results in a collision of the pipetting tip and the stirrer/stones. Three PTFE agitator balls with a 6 mm diameter (Omnilab, Bremen, Germany) are alternatively used to avoid collisions, which are arranged on the vessel bottom to form a small hole in their middle. Figure 5 shows the different options. No collisions were observed in process runs using agitator balls, but occasionally, a ball was ejected from the vessel containing the digestion solution with concentrated acid. A new rack cover was designed with holes for the pipetting tips ( Fig. 6 ). The cover remains on the rack during the pipetting procedure to ensure safe processing. Furthermore, this cover reduces the evaporation of highly corrosive acid vapors.
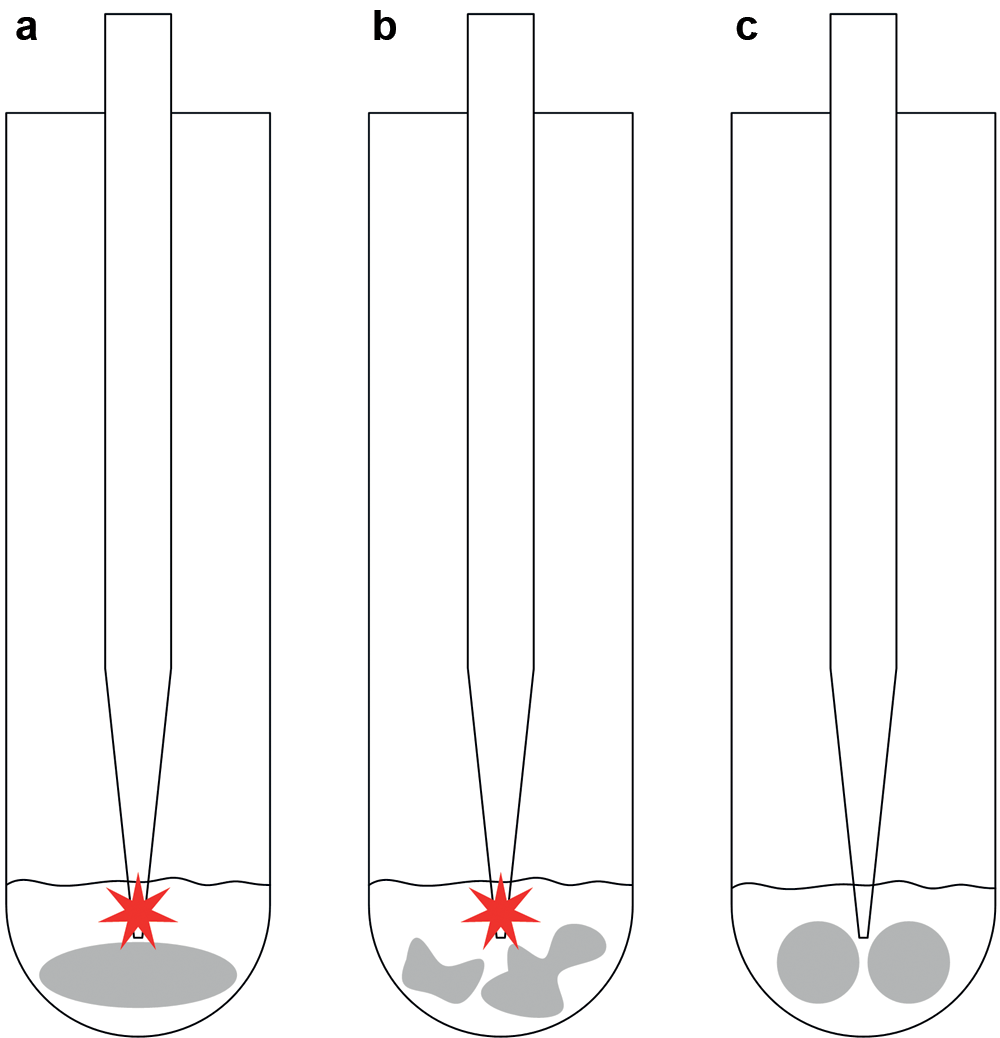
Realization of collision-free automated solution aspiration. (

Pipetting process using protection covers to prevent ejecting agitator balls.
During the dilution step, a reformatting of the sample vessels was performed. The sample solutions were transferred from the microwave digestion vessels into water-filled PP tubes (vol. 15 mL), which can be handled by the mass spectrometer’s autosampler. A maximum of 24 tubes can be arranged on one rack. To perform the dilution, two methods can be applied: using only the liquid handler Biomek 2000 (method 1) and using both the single-vial liquid handler and the liquid handler Biomek 2000 (method 2). Figure 7 shows the general workflows of the two dilution methods.
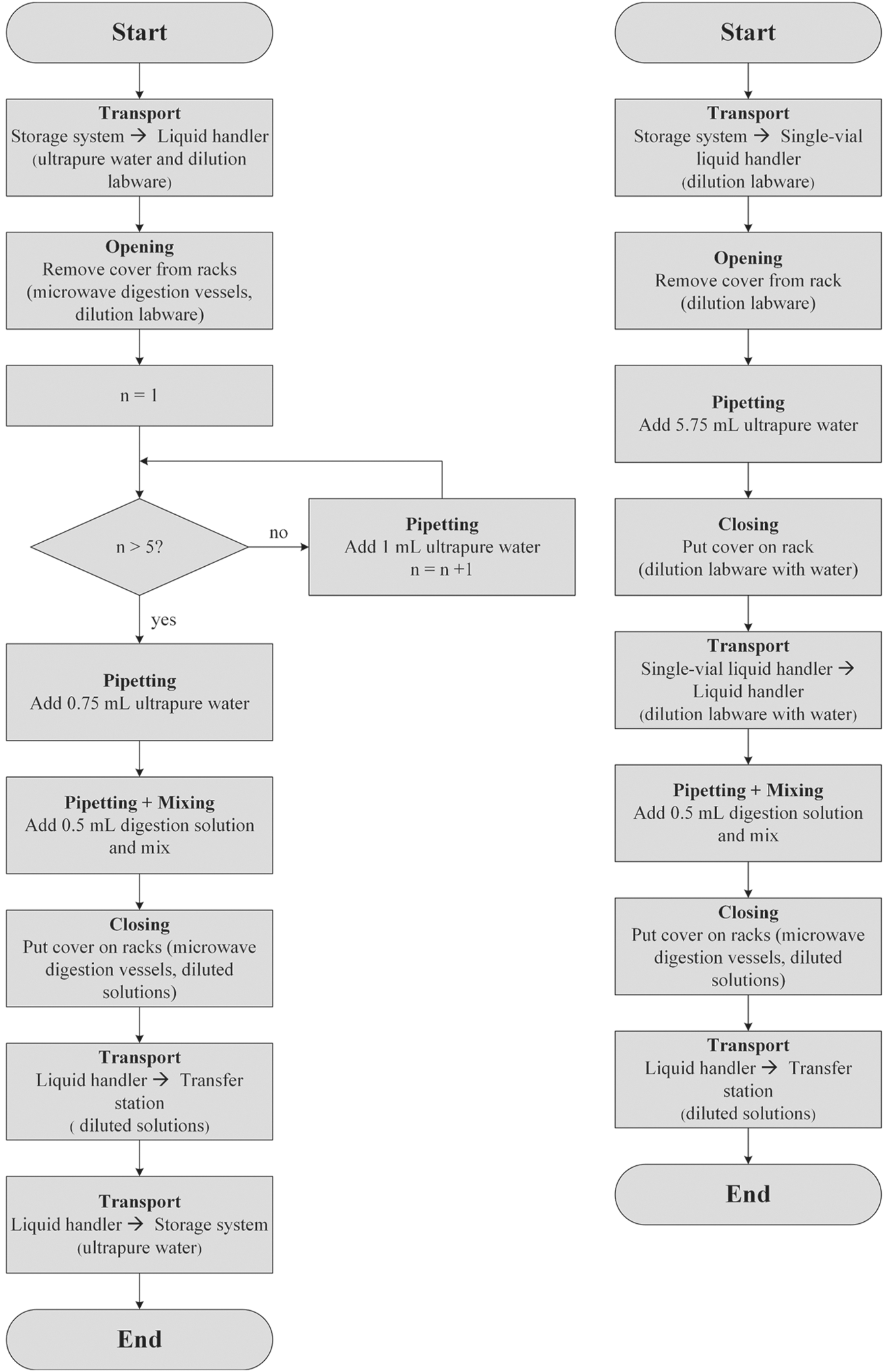
Flowchart to visualize the general workflow of two different dilution methods. Left: Dilution method 1 using Biomek 2000 (Beckman Coulter). Right: Dilution method 2 using both the single-vial liquid handler (celisca) and Biomek 2000.
In the first method, 24 empty PP tubes arranged on one rack were provided to the liquid handler Biomek 2000 for filling with 5.75 mL of ultrapure water. Six pipetting steps were required due to the liquid handler’s maximum volume of 1 mL (5 × 1 mL and 1 × 0.75 mL). Subsequently, a 0.5 mL sample solution was added. Only one device was used for the dilution step, which decreased the required transportation. A disadvantage was the number of required pipetting steps due to the requested volume. This required more pipetting time and reduced the precision.
In the second method, 24 empty PP tubes arranged on one rack were provided to the single-vial liquid handler for filling with 5.75 mL of ultrapure water in one step per vessel. Subsequently, the rack was transported to Biomek 2000 for pipetting of 0.5 mL of the sample. This method required an additional transportation step since two different liquid handling devices were used.
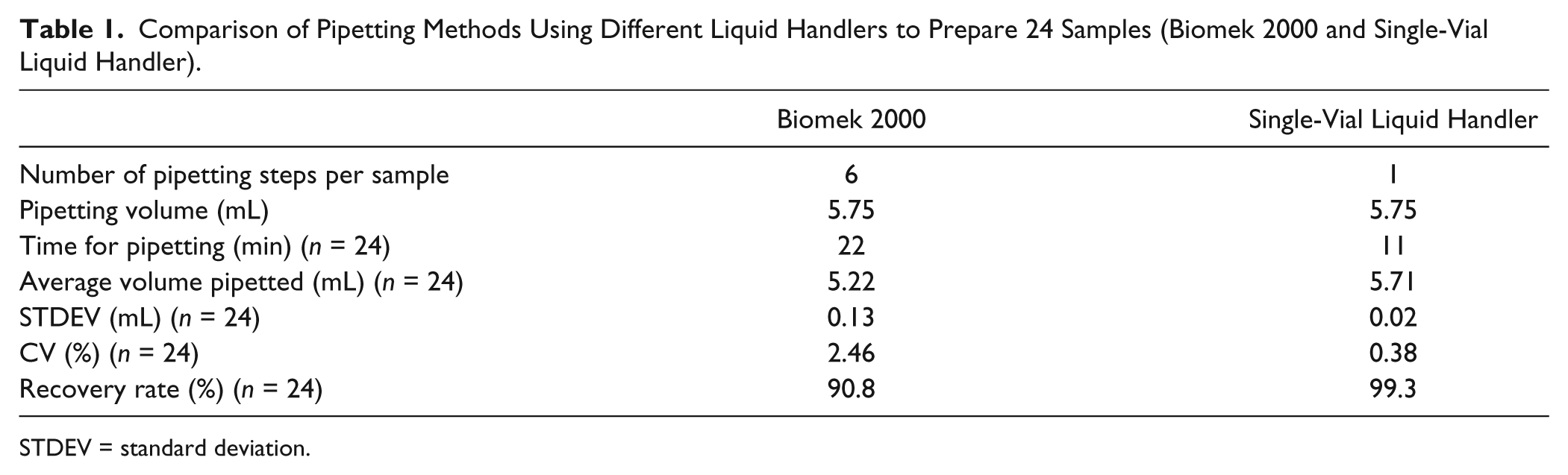
The pipetting of the sample solution was similar in the two dilution methods and was performed using Biomek 2000. To compare the performance of these methods, the different pipetting procedures were executed and the achieved results compared ( Table 1 ). Using the one-step pipetting procedure with the single-vial liquid handler, the pipetting time and the coefficient of variation (CV) were decreased and the recovery rate increased. The total time of the dilution process was reduced from approximately 42 min to approximately 28 min. For these reasons, the final automation process uses the second dilution method.
Comparison of Pipetting Methods Using Different Liquid Handlers to Prepare 24 Samples (Biomek 2000 and Single-Vial Liquid Handler).
STDEV = standard deviation.
Automation Control
The scheduling software SAMI Workstation Ex 4.0 (Beckman Coulter) was used to control the automation system. The workflow is programmed by generating a SAMI method. This includes the definition of the labware, the starting and final labware positions, and programming of the substations. For the sample preparation, two SAMI methods have been created: for the addition of digestion solution ( Fig. 8 ) and for the dilution of the samples after the microwave digestion. In the two methods, up to 24 microwave digestion vessels arranged on four racks can be handled in one run.
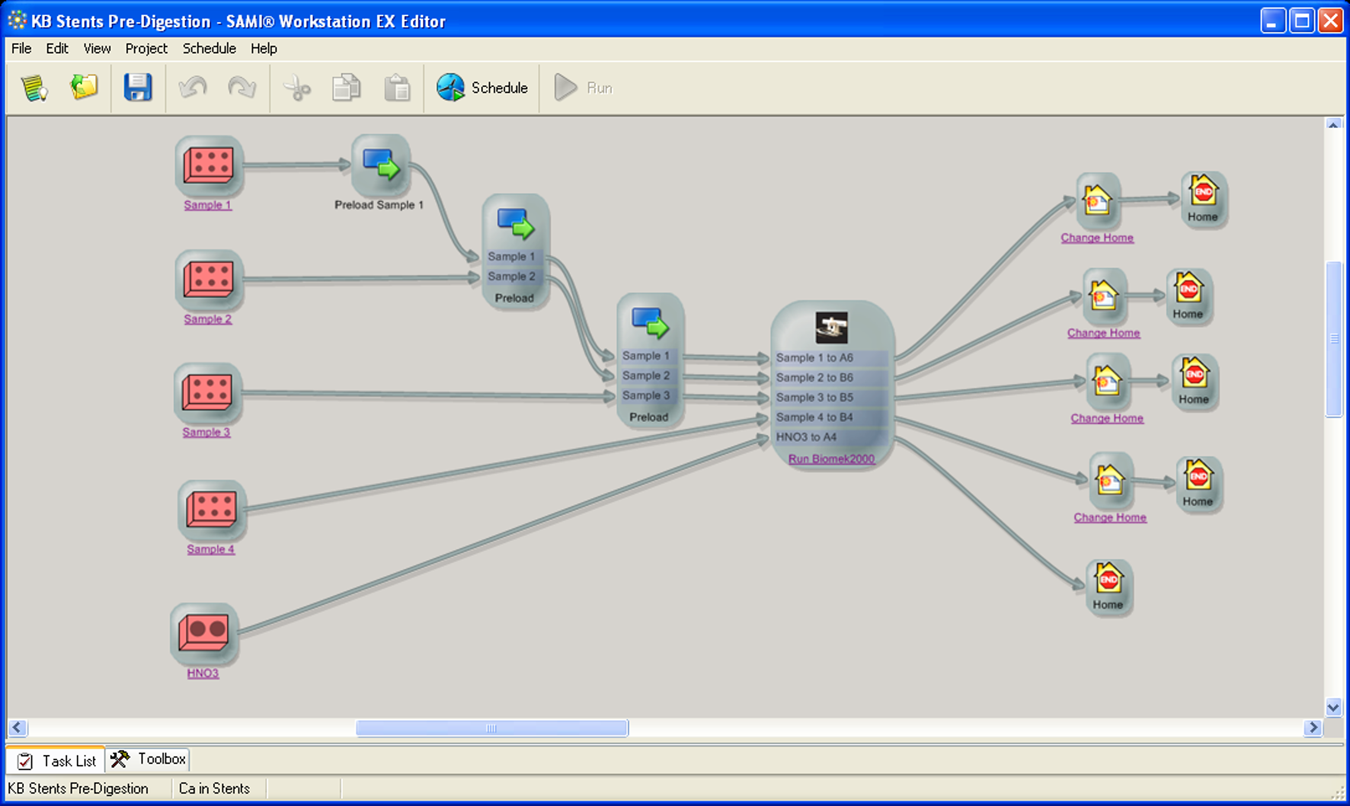
Screenshot of SAMI editor with method for pipetting the digestion acid prior to the microwave digestion.
The SAMI scheduling algorithm determines the optimal workflow to process four racks with a maximum of 24 vessels in one run (also called family). The two ORCA laboratory robots work in parallel to transport the vessels from the storage system to the liquid handler ( Fig. 9 ). The number of families can be increased to perform multiple runs and is only limited by the storage system providing the required samples, chemicals, and labware.

Realized automation system in action: (1) central system integrator with two ORCA laboratory robots moving on orthogonal robot rails (Beckman Coulter); (2) liquid handler Biomek 2000 with housing (Beckman Coulter); (3) exhaust ventilation; (4) single-vial liquid handler (celisca); (5) thermomixer MKR23 (HLC BioTech); (6) regrip station; (7) storage system for labware, reagents, and samples; and (8) sample transfer station.
The single-vial liquid handler was integrated into the automation system and in the SAMI software to realize pipetting or liquid dispensing steps for larger volumes up to 10 mL. The configuration of the liquid handling processes is realized with the device software, which provides two dialogues: “action config” (also called editor) and “configuration config.” In the action config dialogue, the operator initializes all system parts (e.g., XYZ portal, pumps, and valves) and the park position, and creates the liquid handling methods. A typical transfer process includes an aspirating and a dispensing step, but in the method presented in this study, only dispensing of the system medium (ultrapure water) is used. The position of the needle head, the transfer volume, and the dispensing rate can be defined. Using the dialogue configuration config, the parameters of the liquid handler parts are saved as system configuration and calculation basis. The height of the washing blocks and racks, the needle length, and the dimensions of the sample tubes are important parameters for positioning the XYZ portal. 1 Figure 10 shows the configuration of the single-vial liquid handler for dispensing 5.75 mL of ultrapure water in the dilution process after the microwave digestion.
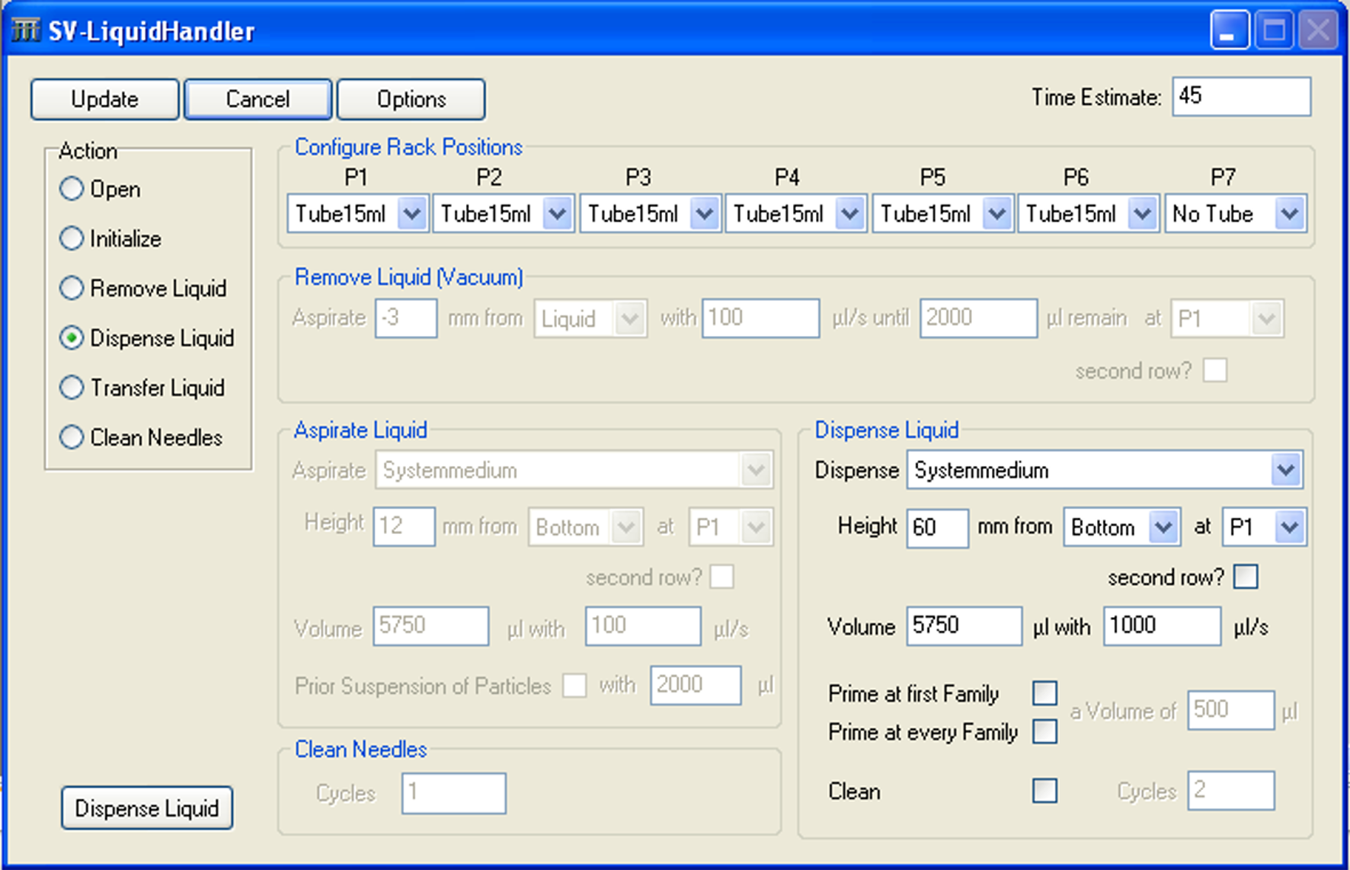
Screenshot of single-vial liquid handler editor with method for the dilution of the samples.
System and Method Validation
The automated sample preparation of the self-made reference material was validated with the following parameters: repeatability, recovery rate, within-laboratory precision, method stability, measurement precision, and the limits of detection (LOD) and quantification (LOQ). Finally, the performances of the manual 29 and automated procedures were compared.
The measurement data were acquired and preprocessed using the instrument software MassHunter 4.2 Workstation Software for ICP-MS version C.01.02 (Agilent Technologies). The calculation of the statistical validation parameters and the final data evaluation were processed using the software Analytical Data Evaluation (ADE).30 –33
The repeatability and the recovery rate were determined by preparing and measuring 22 samples of reference material on 1 day. The results achieved followed a normal distribution, which was verified by the
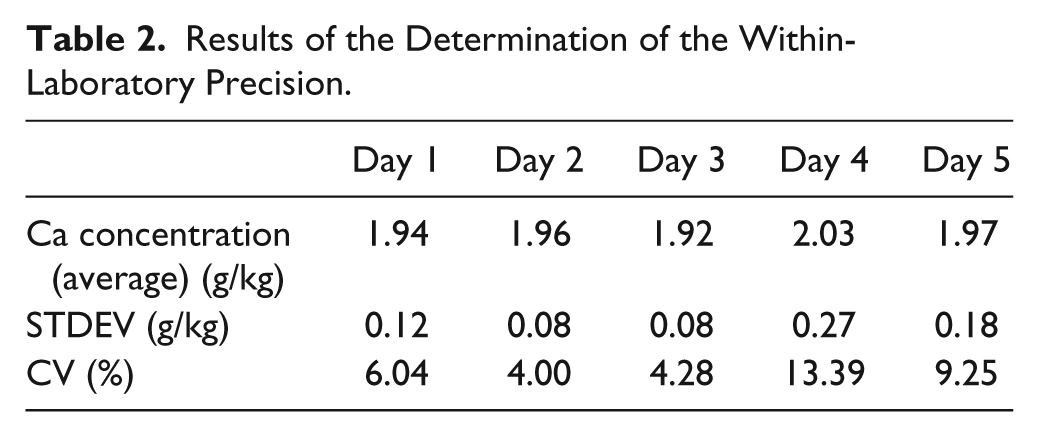
The within-laboratory precision was determined with 11 samples prepared and measured on 5 consecutive days. The daily mean values ranged from 1.92 to 2.03 g/kg, and the CV values from 4.00% to 13.39% ( Table 2 ).
Results of the Determination of the Within-Laboratory Precision.
The measurement precision was determined using one sample measured 10 times. The mean value was 2.00 g/kg, and the CV value was 0.73%. Ten blank samples were prepared and measured for the determination of the LOD and LOQ. In the measurement solutions, the values were 0.11 mg/L (LOD) and 0.26 mg/L (LOQ). In the solid samples, the values were 44.61 mg/kg (LOD) and 108.53 mg/kg (LOQ). These values were calculated according to the general definitions.34 –36 The results achieved are summarized in Figure 11 .
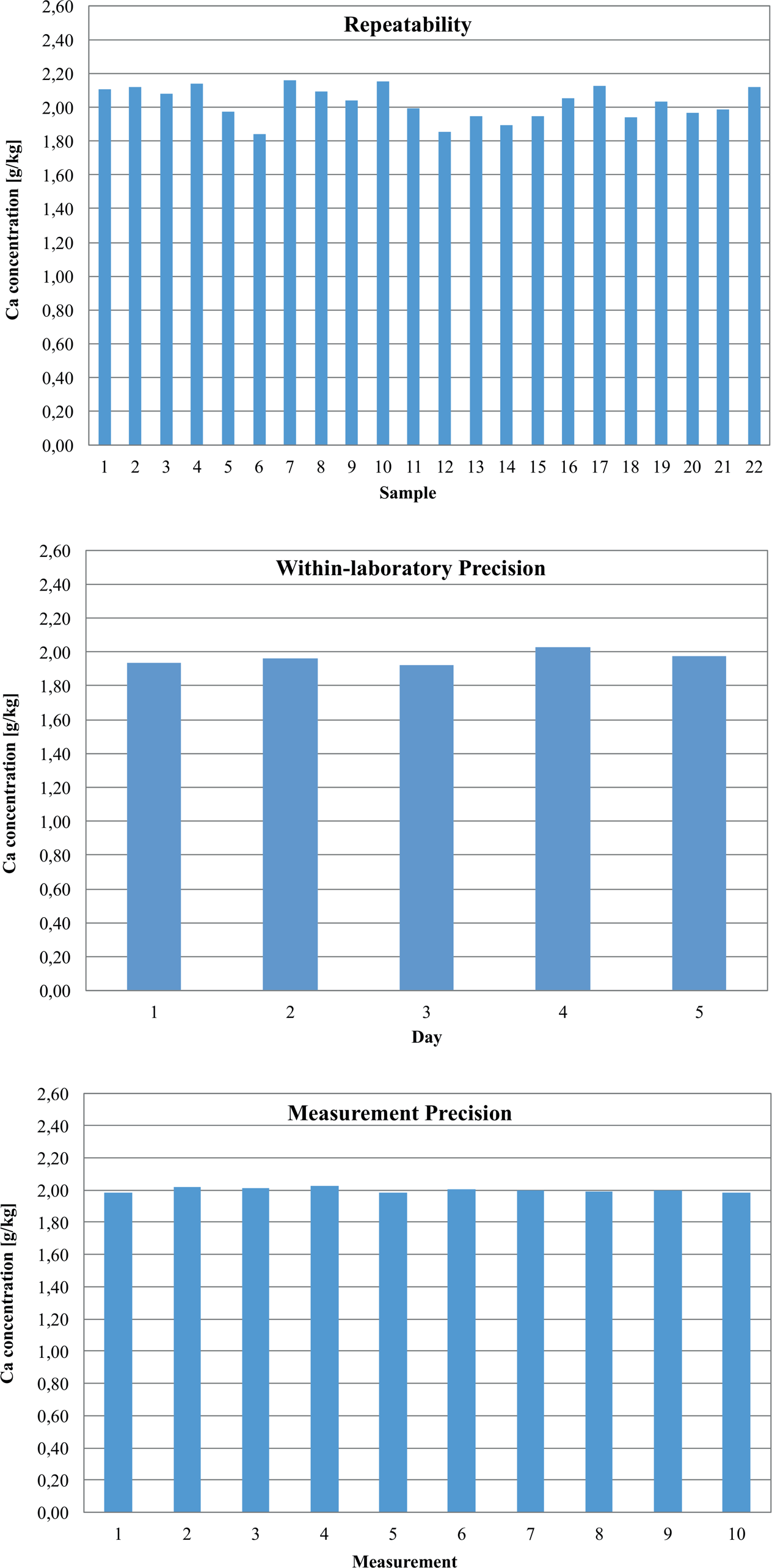
Results of the determination of repeatability, within-laboratory precision, and measurement precision using automated sample preparation.
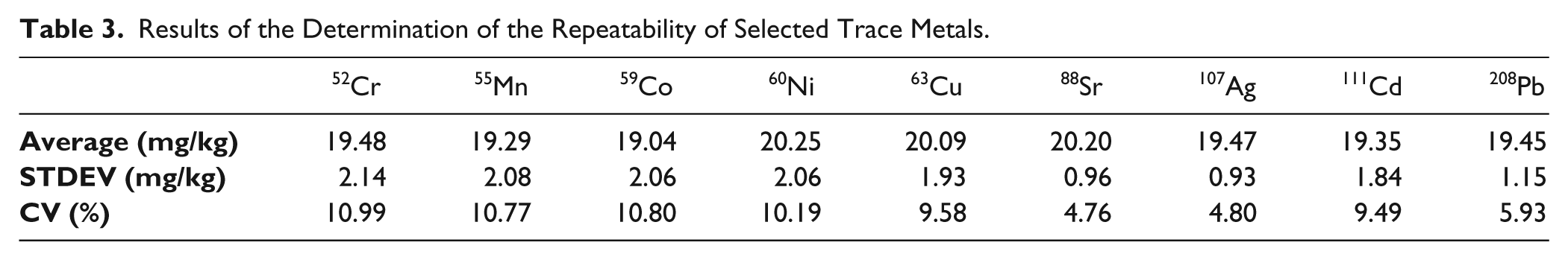
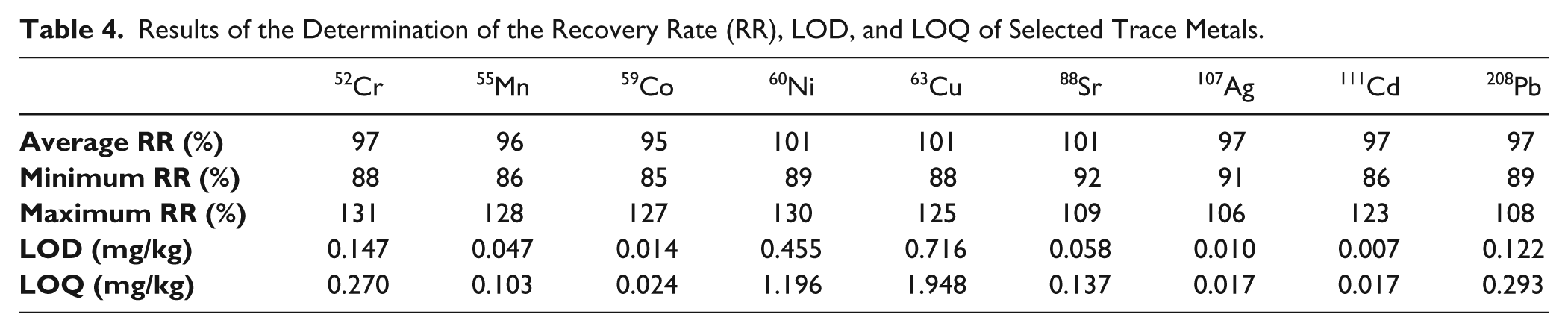
In addition, the results for selected trace metals were evaluated. Table 3 gives an overview of the results of the determination of the repeatability of selected trace metals. Table 4 shows the recovery rates, as well as the LOD and LOQ values.
Results of the Determination of the Repeatability of Selected Trace Metals.
Results of the Determination of the Recovery Rate (RR), LOD, and LOQ of Selected Trace Metals.
Comparison of Manual and Automated Methods
Qualitative and Quantitative Comparison
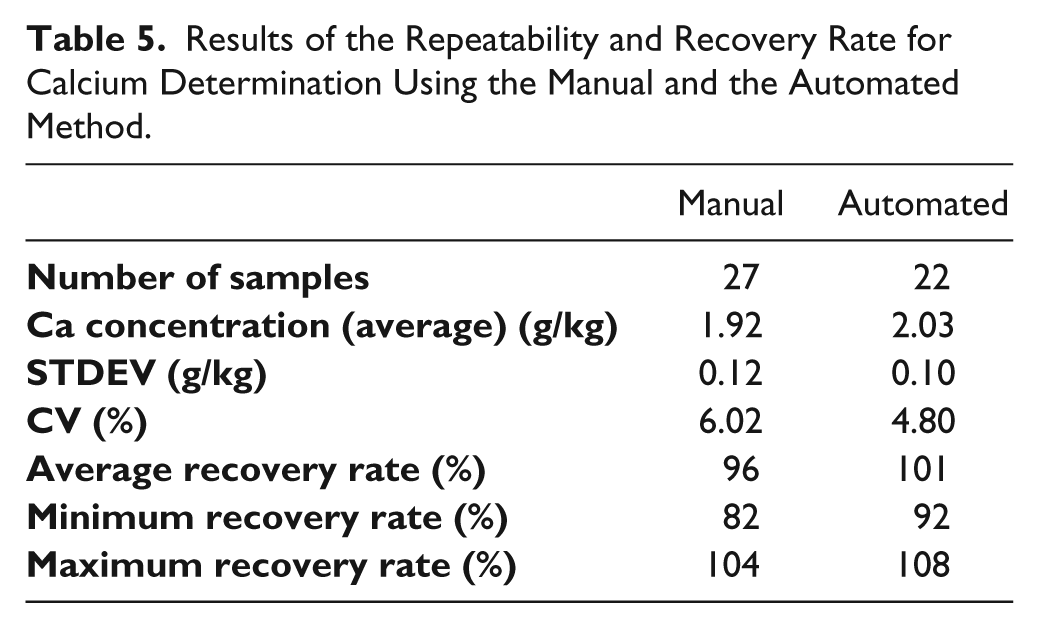
The automated sample preparation method developed is suitable for the determination of calcium and heavy metals in biliary endoprostheses and is in accordance with the results of the previously developed manual method. 29 The results of the repeatability and recovery rate for calcium determination using the manual and the automated method are shown in Table 5 . A comparison of the two methods using a box-whisker plot is shown in Figure 12 .
Results of the Repeatability and Recovery Rate for Calcium Determination Using the Manual and the Automated Method.
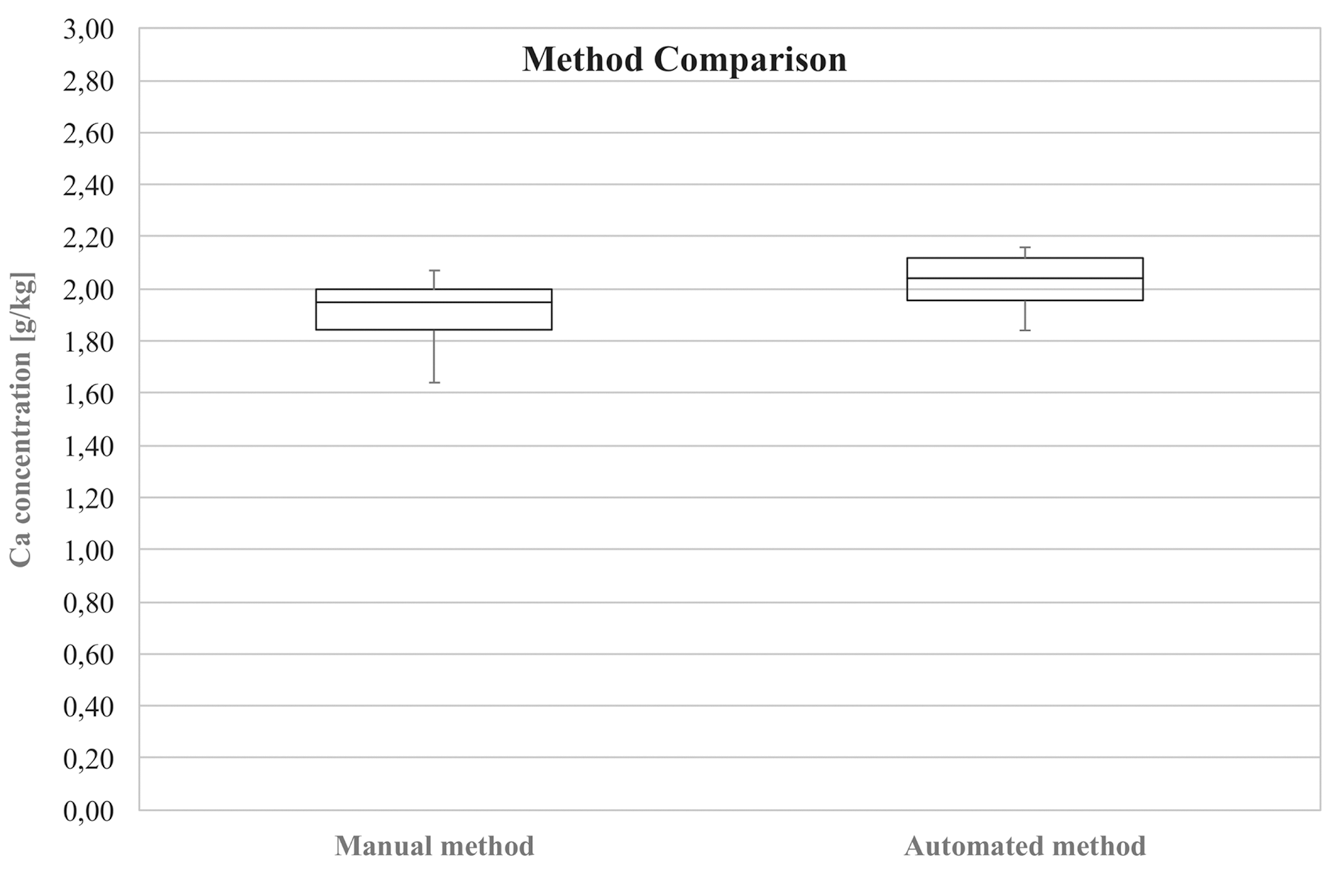
Comparison of the manual and the automated method for the determination of calcium using a box-whisker plot.
Process Comparison
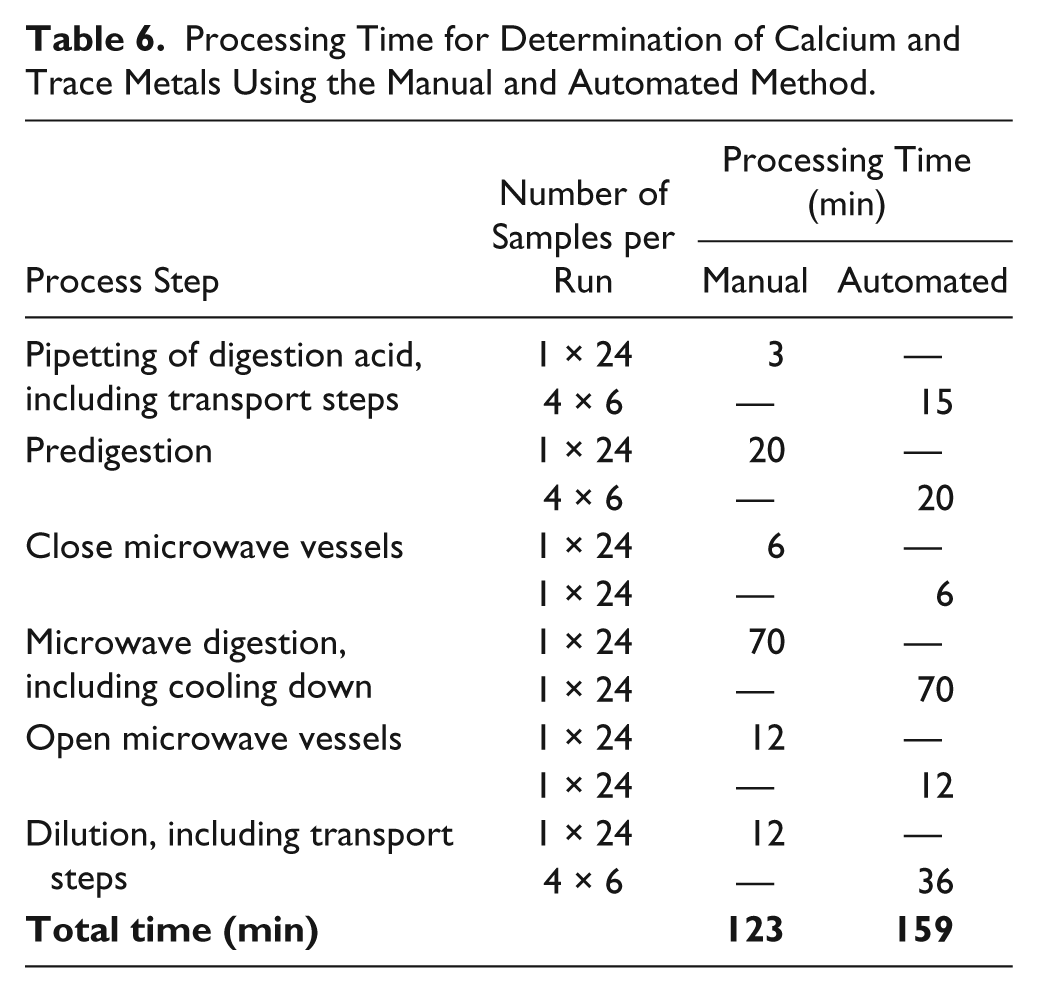
The manual and automated methods were further compared related to their process workflow and processing times. In the manual procedure, all 24 samples can be prepared in one run. A maximum of 40 samples can be processed in one microwave run (full turntable). In the automated method, the sample vessels are arranged on special racks for robot-based handling. In one process run (also called “in one family”), a maximum of four racks, each with six samples, can be handled due to the limited space on the deck of Biomek 2000. Depending on the method, positions for one rack with acid (adding of acid prior to microwave digestion) and one rack with water vessels and its cover (dilution after microwave digestion) are further required. In comparison with manual processing, the automated processing is more time-consuming. The reasons are processing steps such as placing and removing the covers on/from the racks and transportation steps between the several stations (storage system, Biomek 2000, and single-vial liquid handler). Table 6 gives an overview of the processing times of the manual and the automated procedure.
Processing Time for Determination of Calcium and Trace Metals Using the Manual and Automated Method.
Real Samples
Stent Incrustations
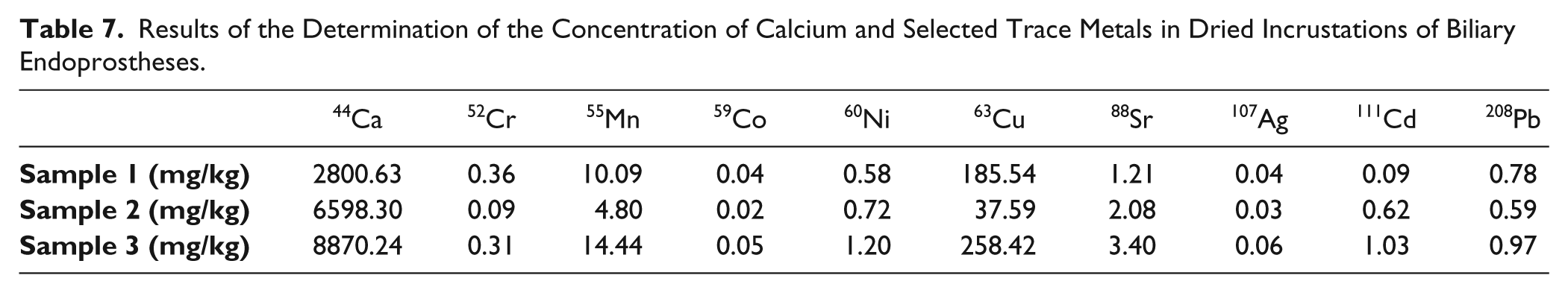
Three human incrustations of clogged biliary endoprostheses were analyzed with the method presented. After removal from the patient, the complete stents were fixed in a solution of 0.9% sodium chloride and 2.5% glutardialdehyde. Prior to the analysis, the incrustations were removed from the stent and prepared as described in the Materials and Methods section. The concentrations of calcium depend on the sample and range from 6.6 to 28.8 g/kg in dried material. An additional dilution (factor 5 or 10) is required for large concentrations to reach the working range of the measurement instrument (ICP-MS). Table 7 gives an overview of the concentrations of calcium and selected trace metals in three incrustation samples.
Results of the Determination of the Concentration of Calcium and Selected Trace Metals in Dried Incrustations of Biliary Endoprostheses.
Pig Bile
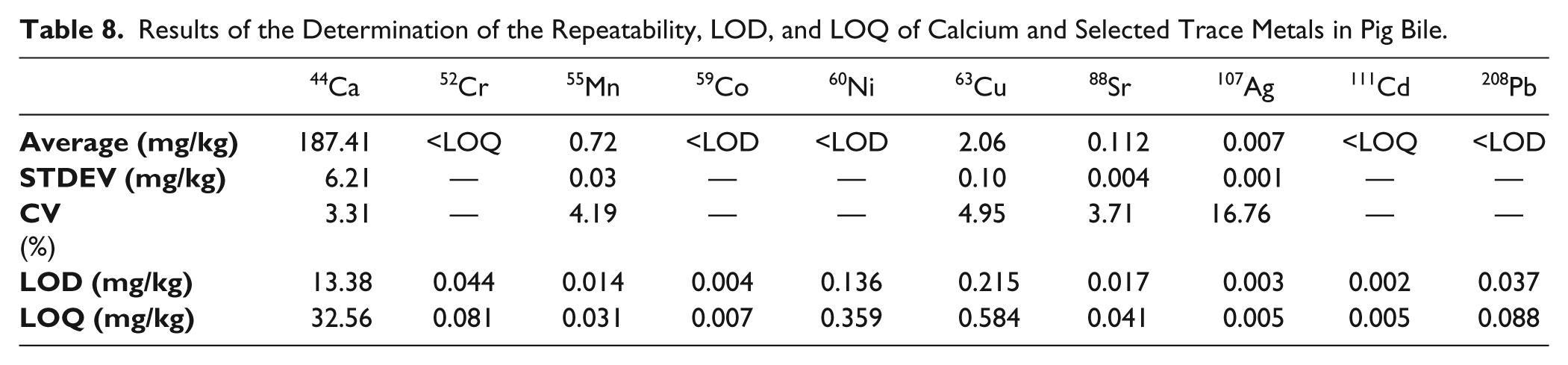
In addition, the method was applied to a further matrix. Not only solid stent incrustations can be analyzed, but also it is possible to determine calcium and trace metals in bile. The sample was pooled and contained equal parts of bile from pigs of different ages. The pooled sample was fixed in the ratio of 1:10 (v/v) with glutaraldehyde (25%). The sample preparation is similar to the preparation of stent incrustations. A volume of 100 µL of pig bile was pipetted and weighted in the microwave vessel. A set of 23 single samples of pig bile was used to evaluate the repeatability. With 10 blank samples, the LOD and LOQ values were determined. Table 8 shows the results of the determination of calcium and selected trace metals in pig bile.
Results of the Determination of the Repeatability, LOD, and LOQ of Calcium and Selected Trace Metals in Pig Bile.
Summary and Outlook
The manual method for elemental analysis using ICP-MS of incrustations of biliary endoprostheses was successfully transferred to an automation system. It enables the determination of calcium and trace metals in both solid matrices, such as dried incrustations, and liquid matrices, such as bile. The automation concept of the developed system uses two ORCA laboratory robots as central system integrators to realize large transportation distances on a very flexible automation platform. The difference of total processing time of the two methods is 36 min, resulting from additional transportation steps required in the automation system. The validation parameters of the manual and automated procedures are similar. The method and the automation system presented enable a precise and fast analysis of medical samples. This should be a contribution to the determination of biliary endoprostheses with no or lower clogging behavior.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors thank the German Federal Ministry of Education andResearch for supporting the funding program KMU-innovativ: Medizintechnik, contract number 13GW0082D.
