Abstract
Drug delivery is still a challenging mission in therapeutic treatment. Research on biomedical micro-electromechanical systems (BioMEMS) has led to a diverse range of microsystems for curative applications. This paper introduces miniaturized controlled valves and drug reservoirs for drug delivery systems. Detailed microfabrication processes, optimized package, and optical/electrochemical detection of the proposed device are described. The release mechanism of the device is controlled by a bilayer actuator valve, which consists of a conductive polymer polypyrrole (PPy) film and a thin metal gold (Au) layer. The PPy layer is electrochemically polymerized on the Au layer. Therefore, further miniaturization of the device is possible through microfabrication of the Au layer. A polydimethylsiloxane (PDMS) package is also introduced to prevent the flap from being blocked by the surrounding tissue of the human body. In addition, a parylene coating is applied to minimize the permeability of PDMS. The release process is then verified by an optical and electrochemical detection system.
Introduction
Currently, the most common techniques used to deliver drugs include pills, ointments, and intravenous solutions. Using these drug-delivery methods, a patient is administered with a high medication dose, which is quickly diluted and loses its efficiency. When drug concentration drops below the therapeutic level, the administration of a new dose is required to sustain the drug effect (see Fig. 1a). To improve the controlled release techniques, innovative drug delivery technologies 1 –3 have become extremely important in the pharmaceutical development. Among these, one popular technique is characterized by using micro-electromechanical system (MEMS) technologies, in which microneedles 4 –6 and implantable microchips 7 are used. The microneedles are used to deliver tiny amounts of drug through transcutaneous process to improve the compliancy of patients, whereas the implantable microchip helps to release the medicine through microvalves by controlling the dissolving of the covered material on the valves. Drugs can also be inserted into pumps 8 , 9 or biodegradable microspheres, 10 , 11 which degrade slowly over time, to control the release of the drugs. The pumps or biodegradable microspheres are placed directly into the treated areas of the body, such as eyes or under the skin, where the drugs can have greater effects. These types of delivery systems allow drugs to be delivered for longer periods of time. The above drug delivery systems can improve the safety and effectiveness of drugs, and in some cases even allow for new types of treatment. However, these systems cannot respond to the change of physiological environment. To address this problem, we introduced a responsive drug delivery system 12 consisting of biosensors, drug reservoirs, control circuitry, and microbatteries. When the reading of biosensor indicates the concentration of therapeutic agent is outside the normal range, the control circuit will activate the drug-release valves and the drug administration can take place automatically. With the responsive drug delivery system, drug levels in patients can stay within the window of optimal efficiency (see Fig. lb) for an extended period of time. To control the drug release, a reliable valve is crucial for a successful responsive drug delivery device. Electroactive polymer actuators made of polymer polypyrrole (PPy) for biomedical applications have been demonstrated in the literature. 13 , 14 PPy/metal-layered structures can serve as actuator valves to open and close the reservoirs. 15
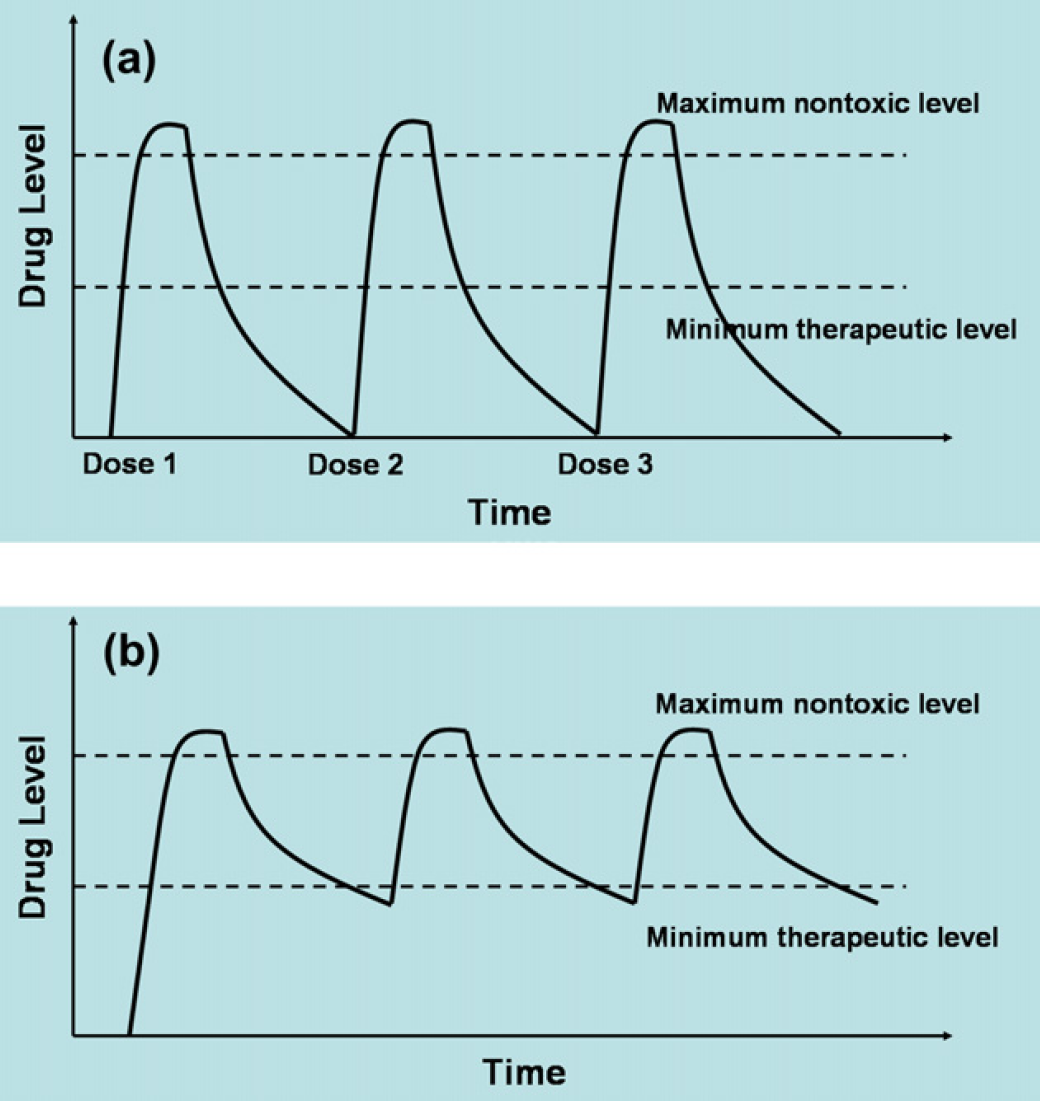
(a) Traditional drug delivery. (b) Responsive drug delivery.
In our demonstration, the advanced release system has been miniaturized into an implantable size of 1 cm × 1 cm with the integrated counter electrodes, working electrodes, and protective reservoirs. By changing the size of the drug reservoir, the amount of medication can be controlled.
Experimental
Fabrication
Four-inch single crystal silicon wafer covered with a thin silicon dioxide is used as a substrate material. RCA (five parts of deionized (DI) water, one part of NH4OH, and one part of H2O2) cleaning procedure is applied before metal deposition process. The metal layer for the flap leads is gold (Au). To enhance the adhesion of Au, a thin chromium layer is deposited before forming a thicker film of Au by vapor deposition. All the depositions are achieved by using e-beam evaporation. The Au layer is then patterned by using a photolithographic process. A thin layer of positive photoresist (Shipley 1827) is deposited using spin coating, followed by a soft bake on a hot plate. The photoresist is then exposed under UV illumination through a patterned iron oxide mask in a Karl Suss MA 6 mask aligner and subsequently developed in MF 319 developer. Au etchant and chromium etchant are subsequently used to remove the undesired metal coverage. Finally, the remaining photoresist is stripped from the substrate by using acetone.
The drug-release opening is covered by the Au/PPy bilayer flap. To enable flap operation, a thin layer of polyimide is used to prevent the metal layer of the flap from sticking to the substrate. Diluted polyimide is spun-coated on the surface, which is then baked on the hotplate. Positive photoresist (Shipley 1827) is applied on top of the polyimide thin film and patterned using the MA6 aligner. The polyimide layer is patterned simultaneously with the positive photoresist in the MF 319 developer. The remaining photoresist is stripped by acetone. A second layer of Au is deposited using e-beam evaporation and then patterned to form the Au layer of the flap. In the next step, the Si wafer is etched through to form the drug-release aperture. A layer of positive photoresist (AZ 4620) is spin-coated onto the backside of the substrate. Using backside alignment, the openings on the backside of the substrate are aligned exactly underneath the flap. The wafer is flipped over (face down) and attached to the holding chuck. The etching is performed in a deep reactive-ion etching (DRIE) machine to etch through the 500-μm-thick Si wafer. The polyimide layer is also etched out during the DRIE etching.
After the microfabrication process as described above, a PPy layer is electrochemically deposited on the working electrodes. Sodium dodecylbenzesulfonic salt (NaDBS, Al-drich) and pyrrole (98% Aldrich) are mixed in DI water with concentrations of 0.1 M each to polymerize pyrrole on the working electrode. The electrochemical polymerization is carried out by applying a fixed potential of 0.5 V (vs. Ag/AgCl reference electrode) on the working electrode. Figure 2 shows the composition of the drug-release device.
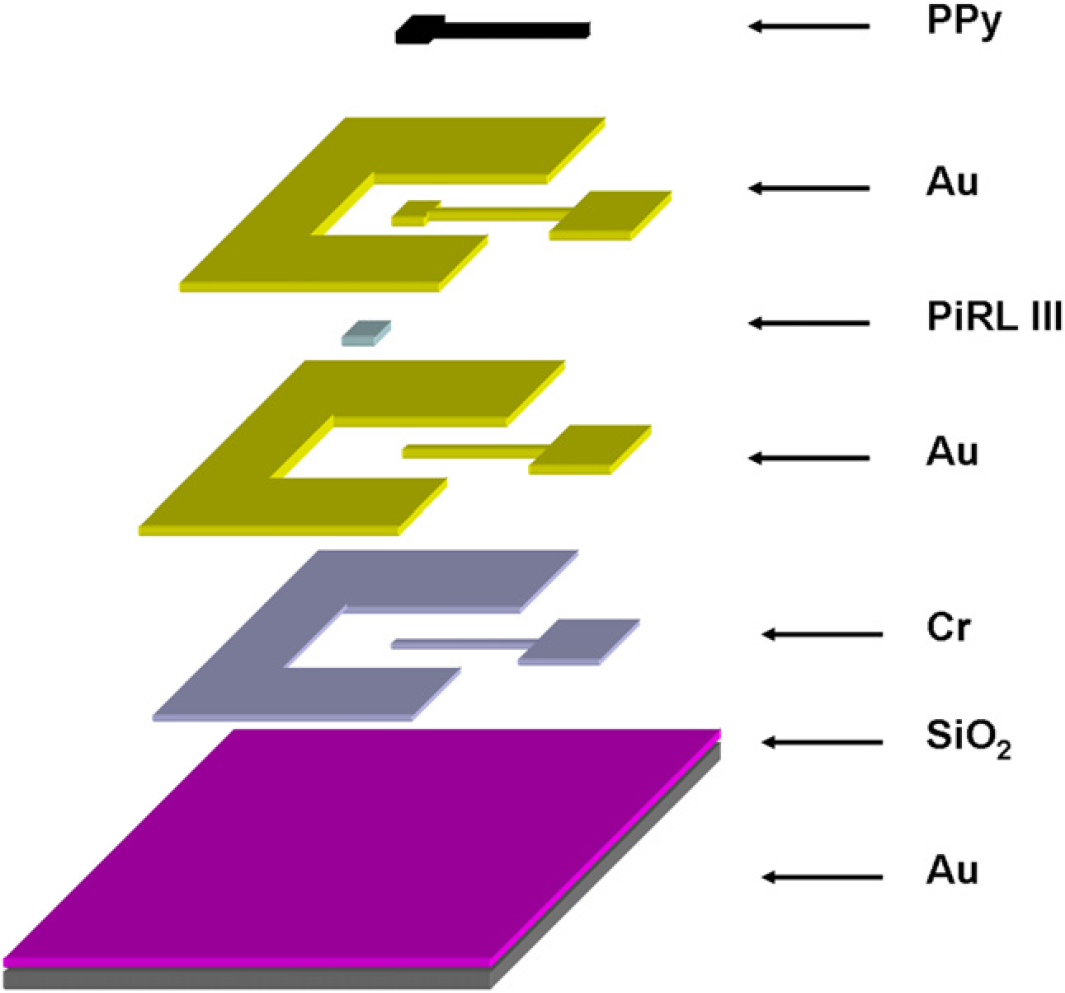
Composition of drug-release device.
The drug storage reservoir/protective enclosure is made from Dow Corning (Midland, MI) PDMS using a molding process. The base (Sylguard 184 silicone elastomer) and the curing agent (silicone resin solution) are thoroughly mixed in a weight proportion of 10:1. Leveler is used to achieve a uniform thickness of PDMS structure. The wafer is cured for 10 min at 90 °C to polymerize the PDMS film. The resulting PDMS structure is carefully peeled off from the mold. To reduce the permeability of PDMS, a thin layer of parylene is deposited on the PDMS reservoir. 16 Double side adhesive film is cut and used to seal the PDMS reservoir onto the silicon substrate.
Design of Electrochemical Detection
The electrochemical detection is performed using a test platform that includes a polymethylmethacrylate (PMMA) substrate with a channel (6.5 cm long, 2 mm wide, and 3 mm deep) connecting two reservoirs (2 mm deep and 5 mm in diameter). The channel is filled with DI water, and the two electrodes are positioned in the reservoirs as illustrated in Figure 3. The drug-release device is put on top of the PMMA substrate with its release aperture aligned with the channel, and the PDMS reservoir is filled with 3 M NaCl solution. To prove the release process, a constant 1 V potential is applied to the electrodes, and current changes versus time are recorded.
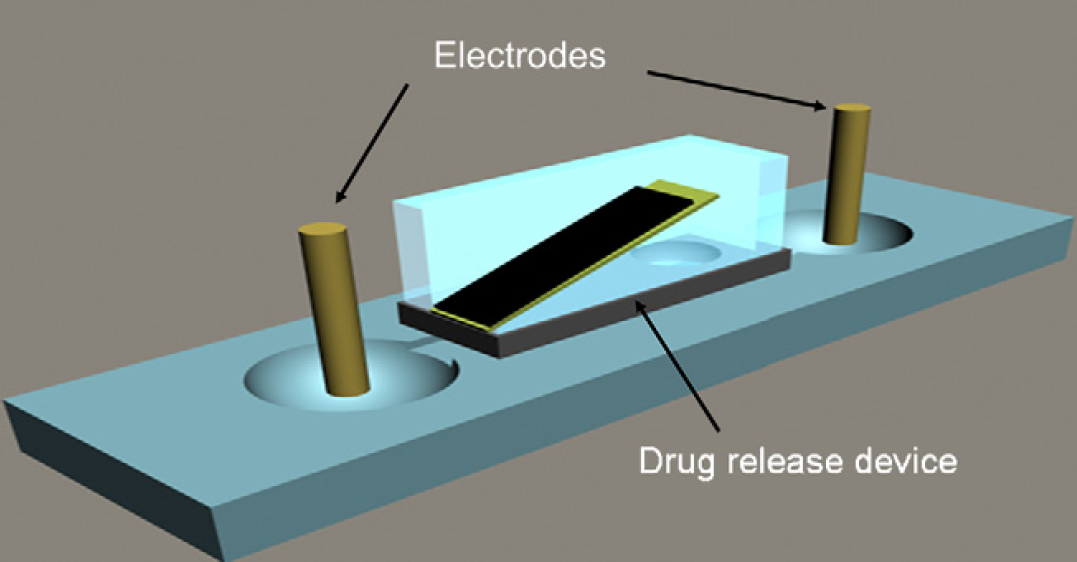
Design of electrochemical detection system.
Results and Discussions
Fabrication and Package
Figure 4a shows the microfabricated drug-release device, the adhesive film, and the molded PDMS reservoir. The size of the release hole is 200 μm in diameter and the width of each working electrode is 100 μm. The dimension of the counter electrode is much larger than the working electrode to ensure the reaction on counter electrode not to restrict the electrochemical deposition rate on the working electrode. An adhesive layer is cut to seal the PDMS cap and the Si substrate. The dimension of the molded PDMS cap is 5 mm × 5 mm × 0.6 mm (depth). The complete package is composed of the Si wafer with valve, the adhesive layer, and the PDMS cap (Fig. 4b).

(a) Drug release device, adhesive layer, and PDMS reservoir before assembling. (b) Packaged drug delivery system.
Parylene Coating
The special coating system, SCS/PDS 2010 is used to process parylene coating. The PDMS cap is placed in the coating chamber and 5 g of parylene C is stored in the thermal column. After 2 h of evaporation, a 1.2-μm-thin layer of parylene is coated on the PDMS surface. A 0.1-g phosphate-buffered saline (PBS) solution is then stored in the parylenecoated PDMS chamber against a piece of Si wafer and sealed with the waterproof epoxy. The same amount of PBS solution is stored in an uncoated PDMS chamber as a control experiment. The result (see table 1) shows that the vapor evaporation rate decreases from 8% per day to 1% per day. To further reduce the permeability of PDMS, a thicker layer of parylene coating may be applied.
Permeability of PDMS before/after parylene coating

Controlled Valve
The operation of the valve opening and closing is performed in the 0.1 M PBS (pH = 7.4) solution which has a similar ionic strength, pH value, and ion concentration to body fluids. An optical microscope and a charge-coupled device (CCD) camera are used to observe the actuating process from the top view. In Figure 5a, the valve is in the closed state with no potential applied. To break the adhesion of the flap and the surface, a higher initial potential (1.2 V) must be applied. Then a lower potential (0.8 V) can be used to control the actuator (Fig. 5b).

Bilayer flap (a) at 0 V and (b) when 1.2 V potential applied.
Electrochemical Detection
To quantify the performance of the PPy valve, we choose an electrochemical detection method. A constant potential (1 V) is applied to the electrodes which are positioned at the two ends of the channel. The ionic concentration of solution increases after the valve is opened to release NaCl solution (3 M) into the channel. The conductivity change of the solution can be quantified by the current measurements. As shown in Figure 6, the flap is in the neutral (closed) state between 0 and the 30th second. A constant potential (1.2 V) is applied to the flap at the 40th second. After 10 s, a current increase is observed which demonstrates the release of NaCl solution from the reservoir. At the 70th second, a negative potential (–1.2 V) is applied to reverse flap deformation and to close the valve. The current level reaches a plateau after the valve is closed and remains constant for the duration of the experiment. This result indicates that the above procedure can be used to quantify the amount of solution released from the reservoir by the changes in current measurements.
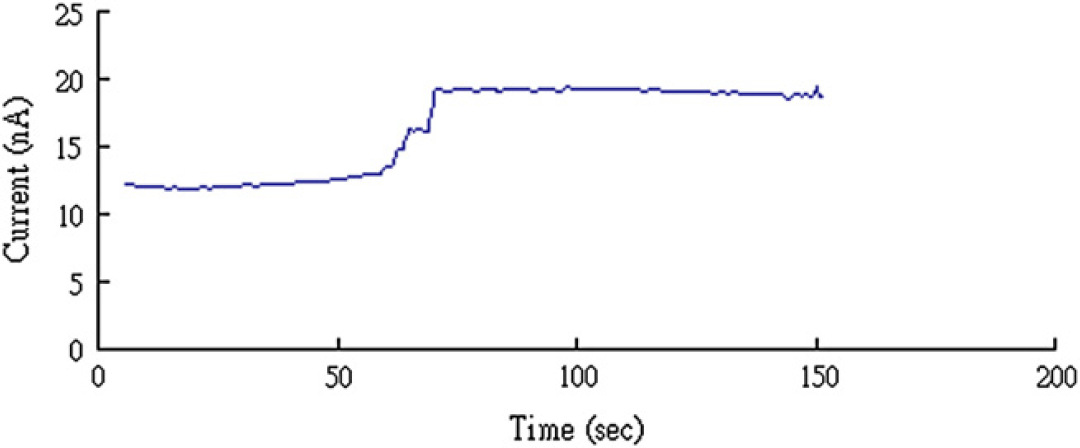
Electrochemical detection: Current increase when the NaCl solution release through the opened valve. Current reaches plateau after valve is closed.
Conclusion
The applications of microtechnology in the biomedical area play an important role in developing new therapeutic methods. A microfabricated drug-release device has numerous potential advantages over traditional therapeutic methods. This research presents a packaged device with implantable dimension. By using optical observation and electrochemical detection, we prove that the valve can be opened and closed with a small bias. Because the actuator may be blocked by the surrounding tissue, a scalable PDMS cap that serves as both a reservoir and a flap protection is developed. The permeability of this PDMS cap is significantly reduced after the parylene coating according to the experiment results. As the prototype has been successfully demonstrated in vitro with similar environment in the human body, it is promising that this release system can be an implantable device. In vivo animal tests are planned to perform in the near future.
Acknowledgments
The authors would like to thank Dr. Lawrence Kulinsky for helpful discussions. The device fabrication was done in INRF (Integrated Nanosystems Research Facility) at University of California Irvine.
