Abstract
The importance of drug delivery has increased over the past decades, and significant advances have been made in the development of novel technologies. This review focuses on the use of different polymer drug delivery systems and their advancement toward clinical applications.
Keywords
The area of drug delivery systems has gained increasing attention in recent years because of limitations of conventional drugs. For example, the severe toxic side effects of anticancer drugs on healthy tissues may cause physicians to reduce the administered dose, delay treatment, or even discontinue the course of chemotherapy. Drugs that are widespread in the body can affect normal organs, resulting in dose-limiting side effects (e.g., cardiac toxicity of doxorubicin) and restrict the amount of drug that can be administered. Low concentration of drugs in the target tissues will lead to the suboptimal therapeutic effects and require more frequent administration.1–3 Recent advancements in proteomics and genomics have led to the discovery of a large number of potentially clinically useful biologic medications. However, these biologics can also have shortcomings, including being large-molecular-weight proteins or polypeptides susceptible to proteolytic degradation, hydrolysis, chemical modification, and denaturation. These properties often result in loss of activity and/or poor bioavailability of biologic drugs upon systemic administration.4,5 Many useful drugs are hydrophobic in nature and are difficult to solubilize in an aqueous environment, thus making it difficult to achieve a convenient pharmaceutical format for administration using conventional delivery strategies. 2 To address these and other challenges, novel drug delivery technologies have been developed for more convenient, controlled, and targeted delivery.
Drug delivery systems can be internal or external devices or materials that facilitate the administration of medications to a patient. Internally administered drug delivery systems can be composed of a variety of materials that are functionalized with therapeutics, ranging from metalloids to polymers to lipids.1,2,6 The advantages of these systems include the following: (1) lowering the adverse side effects of administered drugs by extending their release at a lower systemic dosage or targeting their delivery to the desired cells, (2) improving the bioavailability of peptides and proteins by protecting them against different degradation and inactivation processes in the body, (3) enhancing the solubility of poorly soluble drugs, (4) reducing the cost of treatment by increasing the drug efficacy and lowering the drug dosage for certain therapeutic effects, (5) improving patient compliance by reducing the frequency of administration and the chances of missing or erring in a dose.1–3,5,6 On the commercial side, more and more pharmaceutical companies have realized the advantages that new drug delivery technologies bring to enhance the value of their current products, and they are adopting these technologies to reformulate their off-patent drugs to fight off generic competition. In addition, numerous potentially effective therapeutic agents developed by large pharmaceutical companies have been tabled because of severe toxicity during preclinical or clinical trials. The use of novel drug delivery technologies provides the potential that these drugs can be reformulated, and the substantial funds that were spent in the research and development of these therapeutics can be recovered.
Polymer-based drug delivery systems can be broadly classified into three types: polymer-drug conjugate systems, reservoir-based systems, and monolithic matrix systems. In polymer-drug conjugate systems, drugs are delivered in the form of covalent conjugates with water-soluble and biodegradable polymers. This has the potential to enhance the solubility of poorly soluble drugs and the bioavailability of rapidly degraded therapeutics. 5 In reservoir-based systems, the drug reservoir is enclosed within polymer coatings. The drug releases through the rate-controlling porous polymeric membrane. Monolithic matrix systems are similar to reservoir-based systems, but in this case, the drug is dispersed or dissolved within a rate-controlling polymer matrix. In both reservoir-based and monolithic matrix systems, drugs are noncovalently contained or embedded within the polymer matrices.3,5 This review focus on the reservoir-based, controlled-release polymer drug delivery system.
Reservoir-Based System
The reservoir-based system is one of the most common controlled drug delivery systems to date. In these systems, a drug core is surrounded by a polymer film, and the drug release rate is controlled by the properties of the polymer (e.g., polymer composition and molecular weight), the thickness of the coating, and the physicochemical properties of the enclosed drug, such as solubility, drug particle size, and molecular weight.3,7 Reservoir-based systems are most beneficial for one of the following two applications: (1) a mid-/long-term administration of a medication that is localized to a specific region (i.e., organ, body cavity, etc.). This is usually done if the area being targeted is difficult to reach via systemic administration (i.e., eye, ear) and/or the drugs administered are toxic and may require a long-term course of dosing (i.e., cancer treatments). (2) A drug depot for long-term systemic administration. This is generally administered as an intramuscular or subcutaneous injection or implantation. Based on the morphology, the reservoir-based system can be classified as the following categories ( Fig. 1 ).
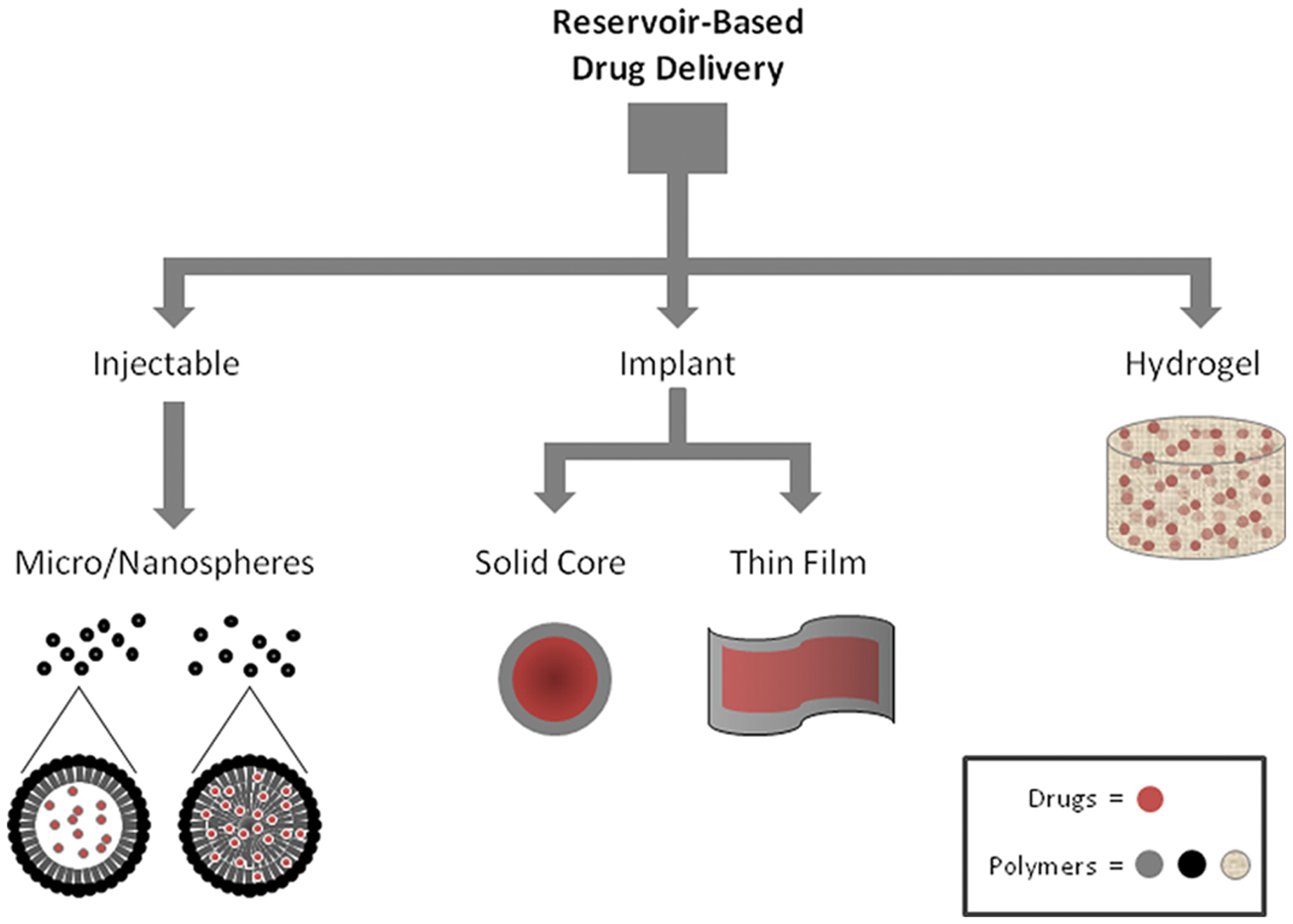
Classes of reservoir-based polymer drug delivery systems.
Medical Devices Coupled With Drug Delivery Systems
An important influence in the development of stand-alone reservoir-based delivery systems is the implanted medical device coupled with extended drug delivery. The implantation surgery can be invasive and sometimes cause inflammation or infection. The coupled delivery of active therapeutics with medical devices can reduce or eliminate the trauma associated with the implantation. In addition, medical devices can serve as a vehicle to locally deliver certain drugs to the implantation site directly, enabling in situ therapeutic treatment for some diseases. This review focuses on stand-alone drug delivery systems. However, the development of drug delivery coatings is important to touch upon as advances in this field have had significant impacts on the development of stand-alone systems.
Polymers used in the medical device drug delivery systems are required to be biostable, nonbiodegradable, biocompatible, and stable in contact with metals. In the early 1980s, polyurethane was used by Medtronic Corporation as an insulator for pacemaker leads. However, the degradation of this polymer in the body and the associated inflammatory and fibrotic reaction have limited its continued usage in the development of novel medical devices using microfilamentous scaffolds. Poly(styrene-block-isobutylene-block-styrene; SIBS) was developed to minimize these deficiencies. SIBS is a biostable thermoplastic elastomer, which shows physical properties overlapping silicone and polyurethane. SIBS was first introduced in the medical industry by Boston Scientific Corporation (BSC) in 2002. BSC introduced a drug-eluting TAXUS® coronary stent to reduce the incidence of coronary bypass procedures and associated morbidities. 8 Various other polymeric materials have also been used with implanted medical devices for drug delivery, with stents being a common area of research. These polymers include a PEVA, PBMA, and PBMA combination, phosphorylcholine, different fluoropolymers, and Parylene C.9–13
Microspheres and Nanospheres Particles for Injection
Microspheres and nanospheres are some of the most desirable types of parenteral delivery systems due to the small sizes of these particles. Microspheres and nanospheres are small drug particles coated with different polymers, which can be administered by a routine injection using narrow-gauge needles.1,7 They can also be administered orally. Upon administration, drugs are released in a rate-controlled manner from the polymer by diffusion or by degradation of the polymer matrix. The drug release rate for such systems is controlled by the polymer molecular weight and composition, particle size, and morphology. The size of the injected microspheres and nanospheres can be a limitation to their route of administration. For example, for intravenous administration, the administered particles must be smaller than the diameter of the capillaries (5–10 µm) in order to avoid being caught in the body’s circulatory system. 14 Particles greater than this size are useful for intramuscular, subcutaneous, or cavity injection.
Microspheres and nanospheres can be prepared either via the polymerization of monomers or from linear polymers. The polymerization of monomers can be achieved by various techniques, such as emulsion, dispersion, and suspension. Emulsion is typically used to form uniform particles at a nanometer scale (10 to 1000 nm). The technique typically involves the dispersion of a hydrocarbon monomer and a water-soluble initiator in water. A surfactant is employed for the formation of uniform micelles, and polymerization takes place inside these micelles. Dispersion polymerization results in particle size at the micrometer scale (0.5 to 10 µm). The technique involves dissolving the monomer, initiator, and stabilizer (an organic polymer consisting of hydrophobic and hydrophilic parts) in an organic medium, and polymerization takes place inside the monomer droplets. The polymer beads precipitate out from the organic solvent. Suspension polymerization is usually employed to produce micron-sized particles (50–500 µm). In suspension polymerization, the monomer is dispersed in a water phase with a stabilizer and an initiator, which is soluble in the monomer phase. Polymerization occurs within the monomer phase. In addition to the polymerization techniques mentioned above, a significant amount of work has also been conducted on dispersion polymerization in supercritical CO2, which eliminates the usage of toxic solvents and may prove to be beneficial to medical applications. 7 Therapeutics are incorporated into the microspheres or nanospheres either via encapsulation in the polymer sphere or by dispersion within the polymer sphere matrix. The form of drug incorporation depends on the chemical properties of the drug and polymers used and the method used to produce the microspheres or nanospheres.
Additional common microsphere and nanosphere preparation techniques include solvent evaporation and spray drying. These methods are useful for polymers that cannot be made by the emulsion process, such as polylactic acid (PLA) and polyglycolide (PGA). They are also beneficial for some naturally occurring polymers (e.g., chitin, chitosan, and cellulose). The solvent evaporation technique (or double emulsion technique) can be briefly described as follows: the drug is dissolved in water and then dispersed in an organic solvent (usually dichloromethane [DCM]) containing the polymer, forming the first emulsion. The first emulsion is then dispersed in a stabilized aqueous medium (usually polyvinyl alcohol) to form the second emulsion. Microspheres or nanospheres are formed as DCM evaporates and the polymer hardens, trapping the drug inside the particles. 15
The first Food and Drug Administration (FDA)–approved controlled-release system was Lupron Depot, injectable microspheres composed of lactic acid–glycolic acid copolymer and leuprolide acetate. This system was introduced in 1989 to treat prostate cancer, and the drug release lasted for 30 days. 3 Other microsphere or nanosphere drug delivery systems subsequently approved by the FDA include AmBisome (liposomal amphotericin B for fungal infection treatment), 16 Adagen (PEG-adenosine deaminase for severe combined immunodeficiency disease), 17 Doxil/Caelyx (Stealth PEG-stabilized liposomal doxorubicin for refractory ovarian and breast cancer), 18 DepoCyt (liposomal cytosine arabinoside for meningitis), 19 ONTAK (denileukin diftitox for cutaneous T-cell lymphoma), 20 Mylotarg (gemtuzumab ozogamicin for CD33+ relapsed acute myeloid leukemia), 21 PEG-Intron (PEG-interferon α-2b for hepatitis C), 22 and Neulasta (pegfilgrastim for reduction of febrile neutropenia associated with chemotherapy). 23
Hydrogel Systems
Hydrogels are three-dimensional, cross-linked networks of water-soluble polymers. Historically, there has been some debate in the field as to whether hydrogels are a separate class of drug delivery from monolithic matrices. For the purposes of this review, the two are considered separate entities. Hydrogels are highly absorbent (up to 99.9% water), and they can be made from natural or synthetic polymers. The use of biodegradable hydrogels as carriers for controlled drug delivery has sparked particular interest because of their biocompatibility and inertness toward various drugs, especially proteins. Furthermore, hydrogels can be formulated into a variety of physical forms, such as slabs, microparticles and nanoparticles, and films. The highly porous nature of hydrogels makes the drug release rate significantly depend on the diffusion coefficient of the drug molecules through the gel network. The porosity of hydrogels can be easily tuned by controlling the degree of cross-linking, which will affect the release rate of the entrapped drug particles.5,24,25
Hydrogels can be prepared by both physical and chemical cross-linking. Physical cross-linking can be triggered by the change of pH, temperature, and ionic strength and a variety of physicochemical interactions (e.g., hydrophobic interaction, charge condensation, hydrogen bonding, stereocomplexation, and supramolecular chemistry). Chemical cross-linking involves adding additional cross-linking entities to covalently bond to hydrogels, leading to the chemical modification of the system. Small-molecule and polymer-polymer cross-linking are the two major cross-linking strategies in this category. Hoare and Kohane have done a thorough review on these techniques in their recent publication. 24
Despite the various advantages of using hydrogel systems for drug delivery, there are limitations as well. The ability of hydrogels to rapidly swell with water can lead to fast release of the loaded drug and rapid degradation of the polymer. Hydrophilic drugs delivered through hydrogel systems typically have a release period of hours to days, which is much shorter than those delivered by microspheres or nanospheres based on more hydrophobic polymers. To extend the effectiveness of hydrogels for drug delivery, several strategies have been developed, such as enhancing drug-hydrogel bonding by physical interactions (ionic interaction or copolymerization), introducing covalent bonds between drugs and hydrogels, and modifying the microstructure of hydrogels (e.g., increasing the percentage of cross-linking monomer incorporated in the gel, forming a second hydrogel network within a prepolymerized hydrogel, generating a reduced-permeability layer at the hydrogel surface, and coformulating particulate systems into hydrogel matrix).24,25
Hydrogel-based drug delivery systems have been developed for many applications. For example, controlled release of bone morphogenetic protein-2 by biodegradable hydrogels has shown enhancement of ectopic bone formation. 26 Solid lipid nanoparticles containing ibuprofen have been incorporated into dextran hydrogels and are shown to be suitable for oral formulations. 27 Multiple injectable hydrogels have been developed for the treatment of ear disorders. These include insulin-like growth factor delivered by gelatin hydrogels, which demonstrate hearing improvement in patients with sudden sensorineural hearing loss, 28 and dexamethasone-containing poloxamer hydrogels.29–31
Implants
In comparison with other reservoir-based drug delivery systems, implants are less common. However, multiple variations of these extended drug release implants have been used extensively in the ophthalmology arena. One main reason for this is that the intraocular structures are easily accessible and are confined and isolated from the circulation by the inner and outer blood-retinal barriers. In the most common incarnation of this implant drug delivery system, a core of solid drug is surrounded by a permeable polymeric membrane whose thickness and permeability control the release rate of the drug into the body. The implants can be modulated into different shapes, such as rods, films, plugs, pellets, and discs. The release kinetics of this system suggests that if the drug concentration within the reservoir is constant (partially dictated by the solubility of the drug), the driving force of the drug release is constant diffusion through the polymer coating, and zero-order release kinetics can be achieved. The drug release rate is determined by factors such as the release area, the thickness of the polymeric membrane, the implant form, and the drug solubility. 32
Depending on the polymer used, the implant systems can be classified into nonbiodegradable and biodegradable implants (further expanded upon below). The polymers most employed in the nonbiodegradable implants include silicone, polyvinyl alcohol (PVA), and ethylene vinyl acetate (EVA). PVA is easily permeable for a variety of lipophilic drugs, whereas EVA is impermeable to most drugs and is used as a membrane around the drug core to reduce the release area of an implant. Silicone can be tailored to be both a permeable or impermeable layer depending on the thickness and grade of silicone used. Polymers used for biodegradable systems can be either naturally occurring polymers (e.g., bovine serum albumin, human serum albumin, collagen, and gelatin) or synthetic polymers, such as PLA, PGA and polylactic-co-glycolic acid (PLGA) copolymer. With biodegradable implants, drug release is thought to occur during polymer degradation. As such, there is an initial drug-burst release usually associated with this type of system. This can be a major disadvantage when constant release kinetics are desired for the duration of therapeutic administration.4,5,32
Currently, there are only three implants for the eye that have been approved by the FDA: Retisert, Vitrasert, 33 and Ozurdex. 34 Retisert, marketed by Bausch & Lomb, is an intraocular implant that contains fluocinolone acetonide (FA), to treat noninfectious uveitis. Retisert is composed of an FA tablet containing microcrystalline cellulose, magnesium stearate, and PVA. Vitrasert, also marketed by Bausch & Lomb, is a controlled-release intraocular implant that contains 4.5 mg of ganciclovir surrounded by PVA/EVA. Vitrasert is used to treat cytomegalovirus (CMV) retinitis, the major ocular infection in AIDS patients. 33 Ozurdex, developed by Allergan Inc. (Irvine, CA), is a biodegradable intravitreal implant that provides sustained delivery of 700 µg dexamethasone to the retina and vitreous for the treatment of macular edema and noninfectious posterior uveitis. 34
Various additional technologies (both polymer and non–polymer based) for implantable reservoir-based drug delivery have been developed to expand use beyond ocular applications. These include the development of polymer beads and films for implantation. For example, Biocompatibles, a UK-based company, has developed a pipeline of biodegradable therapeutic-releasing beads for various applications. Multiple products have been approved for use outside of the United States and have progressed to clinical trials in the United States. These drug-loaded biocompatible beads are primarily hydrogels composed of either PVA or sulphonate-modified N-Fil.
The development of films for transdermal drug delivery and drug-eluting coatings for medical devices has led researchers to adapt these technologies for independent implantable drug delivery systems. This has led to the creation of polymer-based films for drug delivery. Some early applications for thin film drug delivery have also been in the ocular arena,32,35 but development has expanded to the treatment of inflammation and cancer. These films have some advantages over traditional implanted reservoir systems in that they possess a flexible footprint that allows for increased versatility for applications in the body (i.e., contour to different cavities, organs, etc.). There are some limitations as well, including a reduced drug load as compared with some other reservoir-based delivery systems. However, because of the platform nature of this system and the films’ flexible physical properties, these films fill a unique niche for drug delivery technologies. Parylene, a polymer that has been commonly used for decades to coat implanted medical devices, has been used both as a flexible backbone of these films and as a controlled release coating over deposited drugs. 13 These films have been developed further to incorporate additional advanced materials (i.e., block co-polymers, nanodiamonds) to facilitate drug loading and release.36,37 Various additional nanoporous polymeric materials have been studied for their controlled release potential, but because their physical properties, most would have applications as drug delivery coatings for implanted medical devices. Additional work would need to be performed with these materials to develop a stand-alone drug delivery system.
The major drawbacks for the implant drug delivery systems are the need for a surgical implantation and the potential requirement to remove the implant after it is empty. Recent research has been focusing on miniaturization of the implants (allowing for direct injection of the implant without a complicated surgery) and adopting biodegradable polymers to the system (eliminating the need for implant removal). In addition, thermo- and light-sensitive polymers are being incorporated into implantable drug delivery systems to achieve localized and targeted delivery.1,4
Polymers for Reservoir-Based Controlled Release Systems
Polymers used in the drug delivery systems can be classified into the following categories: diffusion controlled (nonbiodegradable), chemically controlled (biodegradable), and externally triggered systems (smart polymers responded to pH, temperature, etc.).
Nonbiodegradable Polymers
The polymers that are commonly used in diffusion-controlled systems are usually nonbiodegradable. In these systems, because the polymers are not biodegradable, there is usually no initial burst release, and the release kinetics are determined by the thickness and permeability of the polymer, the release area, and the solubility of the drug.32,33 The nonbiodegradable polymers that have been used most are silicone, cross-linked PVA, and EVA. These polymers have been approved by the FDA to be safe for the body. EVA, impermeable to many drugs, is often used as a membrane surrounding the drug core to reduce the release area (so as to reduce the drug release rate). PVA, on the other hand, is permeable to various lipophilic drugs, so it is often used as a controlled elution membrane in the release area. The thickness of the PVA layer can be tuned to achieve desired release kinetics. Silicone can be used as an impermeable or permeable material, depending on the thickness and grade used. Several implants using nonbiodegradable polymers have been developed to treat eye diseases such as CMV retinitis, uveitis, and diabetic retinopathy.32,33 Additional nonbiodegradable polymers currently under development in drug delivery systems include various block co-polymers (vesicles and coatings) and parylene (films and coatings). 36
Biodegradable Polymers
Biodegradable polymers find widespread use in the drug delivery industry. There are two types of biodegradable polymers: natural polymers and synthetic polymers. Collagen and gelatin are the two natural biodegradable polymers that have been deployed most for drug delivery systems. There are numerous advantages of collagen, including its biocompatibility, nontoxicity, and the ease of isolation and purification of large quantities. However, collagen is known to cause immunogenic responses in some patients, thus limiting its utility. A variant, atelocollagen, which is prepared by removing the telopeptide from collagen, has been used to decrease the potential immunogenicity. Other disadvantages of collagen include its poor mechanical strength and the difficulty of developing reproducible release rates.5,38 Gelatin is a thermoreversible polymer. It is widely used for drug delivery because of its easy availability, low antigen profile, and low binding to drug molecules. However, in preparing the drug delivery system, gelatin is cross-linked with glutaraldehyde, which can bind to and inactivate some protein drugs.4,5
Synthetic biodegradable polymers include PLA, PGA, PLGA, polycaprolactone, polyparadioxane, polyphosphoesters, polyanhydride, and polyphosphazenes. Among these, PLA, PGA, and their copolymer PLGA are the most well-defined and used polymers in drug delivery. 39 Their precursors, lactic acid and glycolic acid, come from corn starch or cane sugar, which provide a great advantage over conventional polymers synthesized from oil. These polymers are well suited for use in drug delivery because they are biocompatible with living tissue, and their degradation products, lactic and glycolic acids, are biocompatible and are easily eliminated from the body. PLA, PGA, and PLGA have been used to make microspheres for vitreoretinal and inner-ear drug delivery.33,40 They have also been used for peptide drug delivery systems because of their ability to shield the drug from enzymatic attack. Lupron Depot, Zoladex, and Decapeptyl are examples of FDA-approved peptide drug delivery systems using PLGA. 5
Platform technologies for the delivery of therapeutics packaged in biodegradable polymer matrices have been developed by multiple companies, including PR Pharmaceutical (using PLGA) and StarPharma (using dendrimers). Both systems use polymers to encapsulate therapeutics into microparticles or nanoparticles to increase the serum persistence of the drug as it is slowly released from the biodegradable packaging. This allows for a decrease in the toxic side effects of drug administration and increases patient compliance. The microparticles/nanoparticles can either be injected or mixed in a gel-like substance for topical administration, depending on the desired location of activity.
The drug release rate in a biodegradable system is generally thought to be controlled by the degradation of the polymer. There are two types of degradation: surface and bulk degradations. In surface degradation, the polymer matrix is progressively removed from the surface, whereas the polymer volume fraction remains fairly unchanged. In bulk degradation, no significant change occurs in the size of the polymeric system until it is fully degraded, whereas the fraction of polymer remaining in the system decreases over time. Both forms of degradation often result in autoacceleration (i.e., the degradation product[s] further catalyze the degradation process). Therefore, there is always an initial burst release and a final burst release of drug associated with these biodegradable drug delivery systems. Continuing attempts are being made to minimize the burst effects and achieve a pseudo–zero-order kinetic of drug release.4,32
Smart Polymers
Smart polymers, or environmental responsive polymers, are macromolecules that display physicochemical change in response to environmental stimuli, such as change in temperature, pH, ionic strength, redox potential, biochemical agents, and ultrasound.4,41 The interests in smart polymers was sparked by the advantages these polymers could bring to drug delivery, including ease of application, localized delivery of drugs with site-specific action, prolonged delivery period, and decreased systemic drug dosage to minimize the associated side effects.
Many polymers show abrupt changes in their solubility upon temperature change, and this property can be deployed to develop polymer solutions that undergo sol-gel transition near their lower critical solution temperature. In these delivery systems, a liquid polymer/drug solution is injected into the target site at ambient temperature. As the body temperature warms up the solution, a polymer gel is formed, entrapping the drug in the matrix. Extended-release formulation is achieved by the diffusion of the drug from the polymeric gel. One disadvantage of this system is a high initial burst release due to the shrinkage in volume, which expels a large amount of drug. Some polymers that show thermo-sensitivity are poly (N-isopropylacrylamide; PNIPAAM), poly (ethylene oxide)-poly (propylene oxide)-poly (ethylene oxide) triblock copolymers (PEO-PPO-PEO), and poly (ethylene glycol)-poly (lactic acid)-poly (ethylene glycol) triblocks (PEG-PLA-PEG). 42 Bovine serum albumin (BSA) has been shown to release gradually from a system made of temperature-responsive chitosan grafted with PEG (PEG-g-chitosan). 43 Release profiles demonstrated an initial burst release of BSA in the first 5 h, followed by a sustained, diffusion-driven release until ~70 h. OncoGel, which is composed of paclitaxel and a PLGA-PEG-PLGA thermo-sensitive polymer system (ReGel), is used for injection in tumors and release of paclitaxel over a period of 6 wk. 35 OncoGel injection physically targets paclitaxel to the tumor site while limiting its circulation, resulting in an acceptable safety profile.
Because physiological pH varies systematically in the body, there has been a tremendous amount of effort to develop polymers capable of responding to pH changes. In addition to normal tissue pH variation, diseased and inflamed tissues show different pH profiles from healthy tissue. It is also well known that cancerous tumors produce acidic conditions in the extracellular milieu (pH ~6.5). 44 A pH-responsive polymeric system exhibiting sol-gel transition over physiologically compatible pH ranges has been developed. The polymers used in this system include acrylic acid and methacrylic acid. 42 In this system, the polymer matrix swells when the ambient pH reaches 8.5, releasing the entrapped drug from the system. Bae and colleagues45,46 have developed pH-sensitive polymeric systems to deliver the anticancer agent doxorubicin to the target tumor cells.45,46 Additional systems have been investigated using polymers that are sensitive to more than one stimulus. For example, PLA/PLGA-PEG diblock or PLA/PLGA-PEG/PLA/PLGA triblock pH/thermo-sensitive biodegradable hydrogels have been developed as a sustained delivery system of biological agents. In vitro studies of a PLGA-PEG-PLGA system show sustained release of Fc-leptin over a 7- to 10-d period. 42
Future Trends and Commercialization Aspects
Research on polymeric extended drug delivery has shown great success in the past few decades in mediating safe and effective delivery of therapeutics to treat a wide variety of medical conditions. However, several challenges remain. The advanced treatment of diseases requires the precise delivery of a regulated dose of medicine for the desired time period. In addition, this administration must be done in a highly regulated and site-specific manner to achieve therapeutically relevant concentrations in the cells. Future therapeutic delivery systems should also be able to recognize key indicators of a particular disease and monitor the release rate of the drug accordingly. These programmable drug delivery systems offer an opportunity to create fully autonomous systems, which increases the precision of drug payload. Classical chemical engineering principles and control theories should be incorporated into designing such systems. One of the major shortcomings for many microsphere/microvesicle delivery systems can be their limited drug-loading capacity. The continued development of nanotechnology as applied to the drug delivery field strives to overcome this limitation and may able to achieve a much higher drug:polymer ratio. In addition, micronized solid drug core implants can be adapted for injection. This can further increase the drug-loading capacity over a microsphere/microvesicle delivery system.
Ultimately, the end goal of any drug delivery system is the production of a useful clinical tool. As such, a number of requirements need to be addressed when determining the feasibility of translation of these technologies from the laboratory to the clinic, and there are many potential pitfalls in this process. First and foremost is the scalability of manufacturing. Even the most useful drug delivery system will never be commercialized and used by physicians if the processes for production are not amendable to scaling up. The development of high-throughput, highly reproducible, and high-yield processes for production is vital for the successful translation of drug delivery technologies. The use of self-assembly processes for the production of polymers, matrices, and films can facilitate the commercialization of drug delivery systems. In addition, scalable spin, spray, and vapor deposition techniques are processes that are currently under further development.
There is often a separation between the investigators who are developing drug delivery technologies in an academic setting and the processes that are required for translation into a commercial world. For many investigators, their focus is on technical design and intellectual pursuit, and they have no entrepreneurial interests. Although tremendous advances are made by these investigators in the drug delivery arena, these advances may not be harnessed for clinical applications. Other investigators make the leap into the commercial world and start small businesses for the commercialization of their technologies. These companies represent some of the best opportunities for further development and commercialization of promising drug delivery systems.
One interesting aspect to consider when investigating the translation of drug delivery technologies to the clinic is the funding process for small businesses. The traditional funding route for a start-up company is multiple rounds of financing via angel and venture capital investment groups. In this case, investors are looking for a product, a large market, and a potential for significant market share by the developed product. A successful company will progress to a point where they can commercialize the product or be acquired by a larger company, accompanied by a significant return on investment for the angel and venture groups.
Some of the most valuable drug delivery technologies are platforms for which there are many potential applications and markets. When progressing through a traditional fundraising pathway, companies are often forced to choose a "lead" product for continued development. Although this can work, a company with useful technology may go out of business if this lead product is not successful or deemed desirable. One of the most unfortunate consequences of this is that a platform technology with huge potential may be abandoned.
To circumvent this, small companies need to be creative with their research and development strategy and diversify their product development. One resource to aid in this process is the Federal small business grant program. Various departments of the US government have allocated funds for the support of small businesses, including significant amounts for biotechnology. Many universities are actively fostering collaborations between their academic laboratories and small businesses. Additionally, large pharmaceutical companies have also allocated funds to support the development of promising biotechnologies from universities and small businesses. These resources can be harnessed to aid small businesses in the development of multiple products and technologies for potential commercialization and translation to the clinic.
Footnotes
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The authors received no financial support for the research, authorship, and/or publication of this article.
