Abstract

Keywords
Overview
Advances in microelectromechanical systems (MEMS) have allowed the microfabrication of polymeric substrates and the development of a novel class of controlled delivery devices. In contrast to the traditional microsphere delivery vehicle, micro-fabricated devices may be specifically designed flat and thin to maximize contact area with the intestinal lining. This flat design may also minimize the side areas that are exposed to constant flow. The devices can be microfabricated to incorporate single or multiple drug reservoirs to provide unidirectional release for any number of drugs/biomolecules of interest. Furthermore, specific areas of the device can be targeted for surface modification. These specifically tailored three-dimensional physical and chemical features, together, provide a micromachined vehicle with the capacity to target cells, promote unidirectional controlled release, and enhance permeation across the intestinal epithelial barrier (Figure 1).
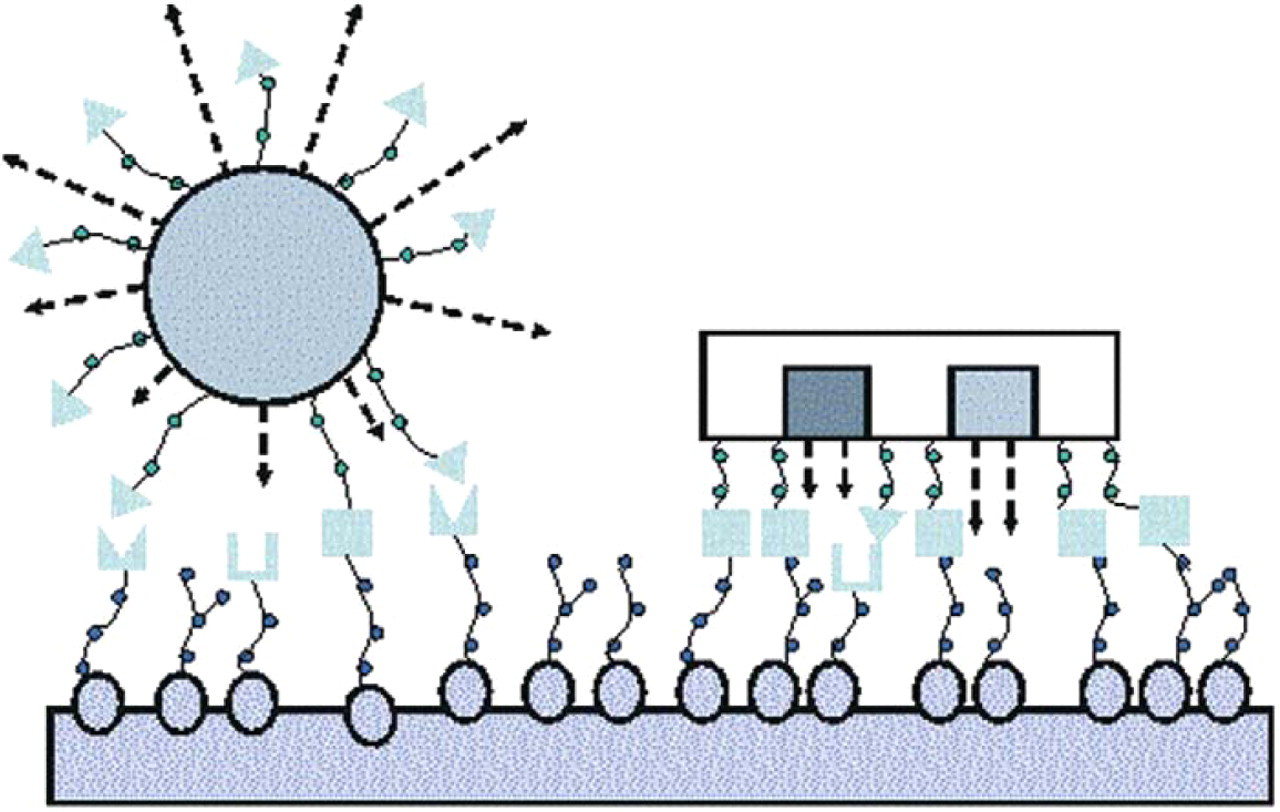
Schematic of microsphere adhesion compared to microdevice adhesion.
Introduction
Many macromolecular drugs are poorly available when administered orally. The human gastrointestinal (GI) tract has a naturally low level of permeability since it is designed to resist absorption of peptides, proteins and large molecules until they are broken down into smaller molecules. Several approaches are currently being developed to enhance the oral delivery of macromolecular drugs including protective coatings to protect peptides during trans-port, 1,2 and bioadhesive agents to induce contact of the peptide to the intestinal wall. 3,–5 In addition, the use of permeation enhancers and protease inhibitors to augment uptake and prevent degradation are being explored. 6,7 Although all these approaches have been shown to improve upon the oral bio-availability of large molecules, none of these approaches offers a complete solution for adequate and safe oral administration. Therefore it is important to explore alternative approaches, perhaps using technologies borrowed from other disciplines, such as MEMS.
Experimental methods
Microfabrication
Poly(methyl methacrylate) (PMMA) microdevices were fabricated on 2-inch (111) silicon wafers (Virginia Semiconductor, Fredericksburg, PA) using PMMA (950,000 MW, suspended in 11% anisole solvent, MicroChem, Newton, MA) and standard microfabrication methods (Figure 2, Table 1).
Microfabrication process for constructing PMMA microdevices
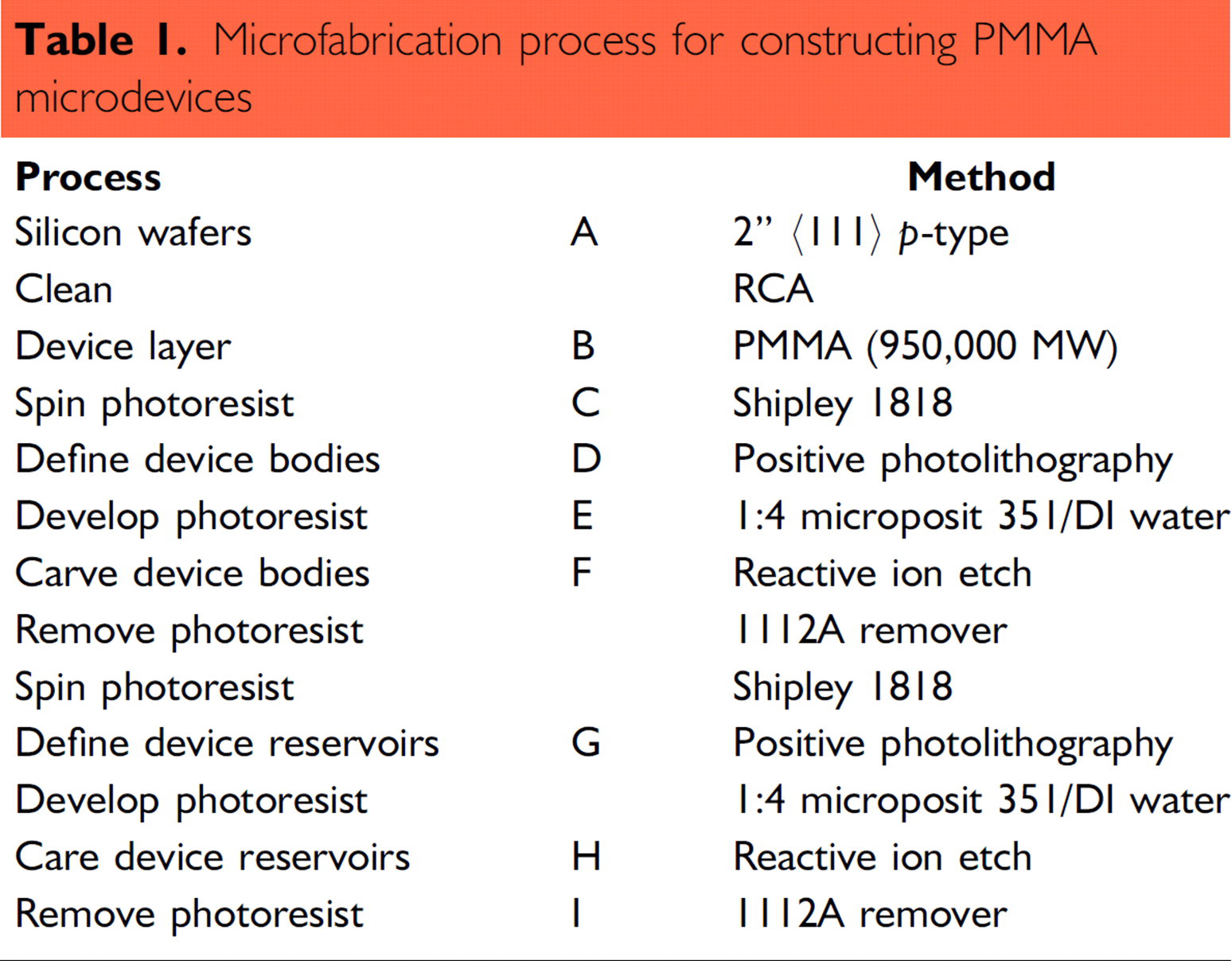
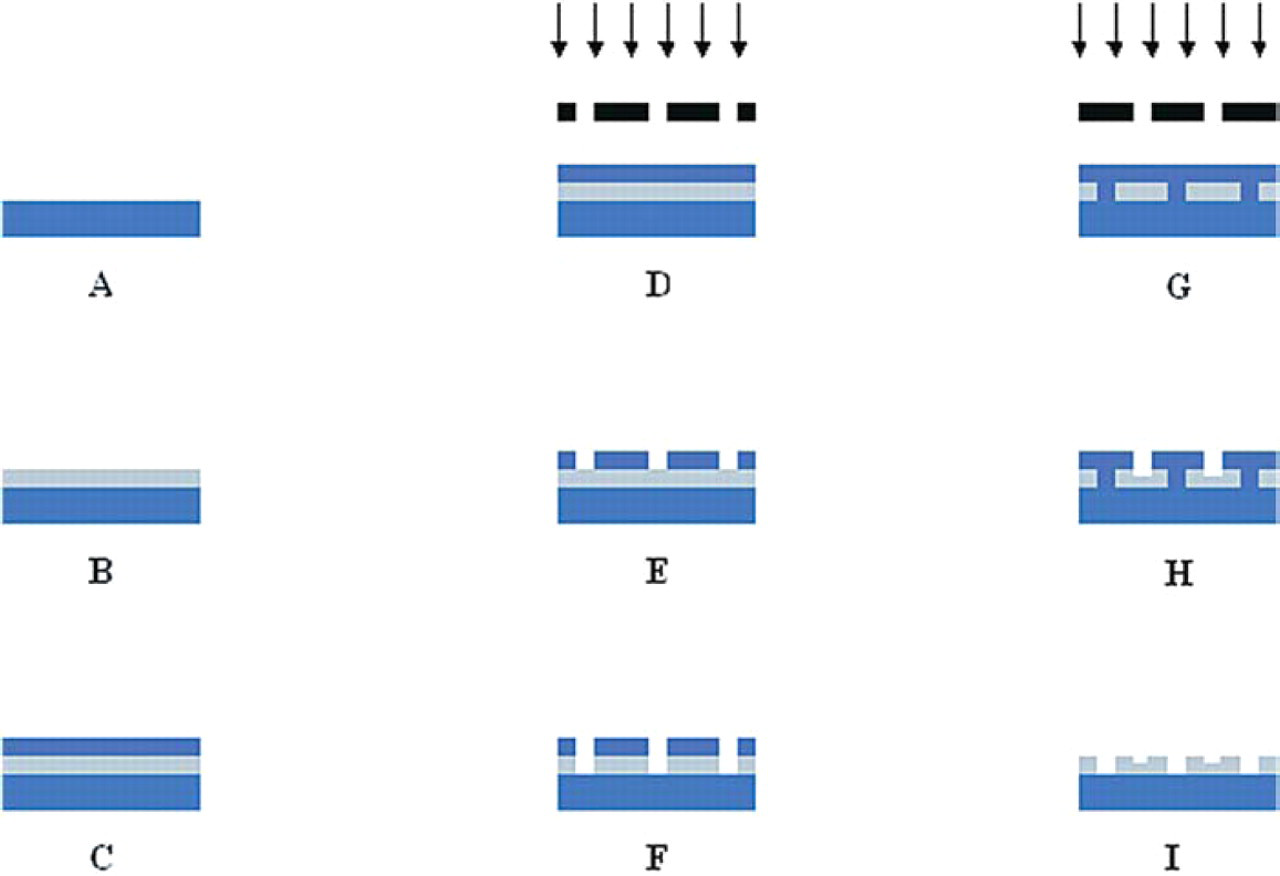
Microfabrication process. Corresponds to Table 1.
Surface Chemistry
PMMA was rendered cytoadhesive to human colorectal carcinoma (Caco-2) cells by attachment of lectins, a group of molecules able to recognize surface glycoconjugate structures of intestinal cells. The surface modification process consists of (A) aminolysis with N-lithioethylenediamine, 8,9 (B) avidin immobilization using a hydroxy-succinimide catalyzed carbodiimide reagent, and (C) lectin attachment utilizing avidinbiotin attraction and interaction 10 (Figure 3).
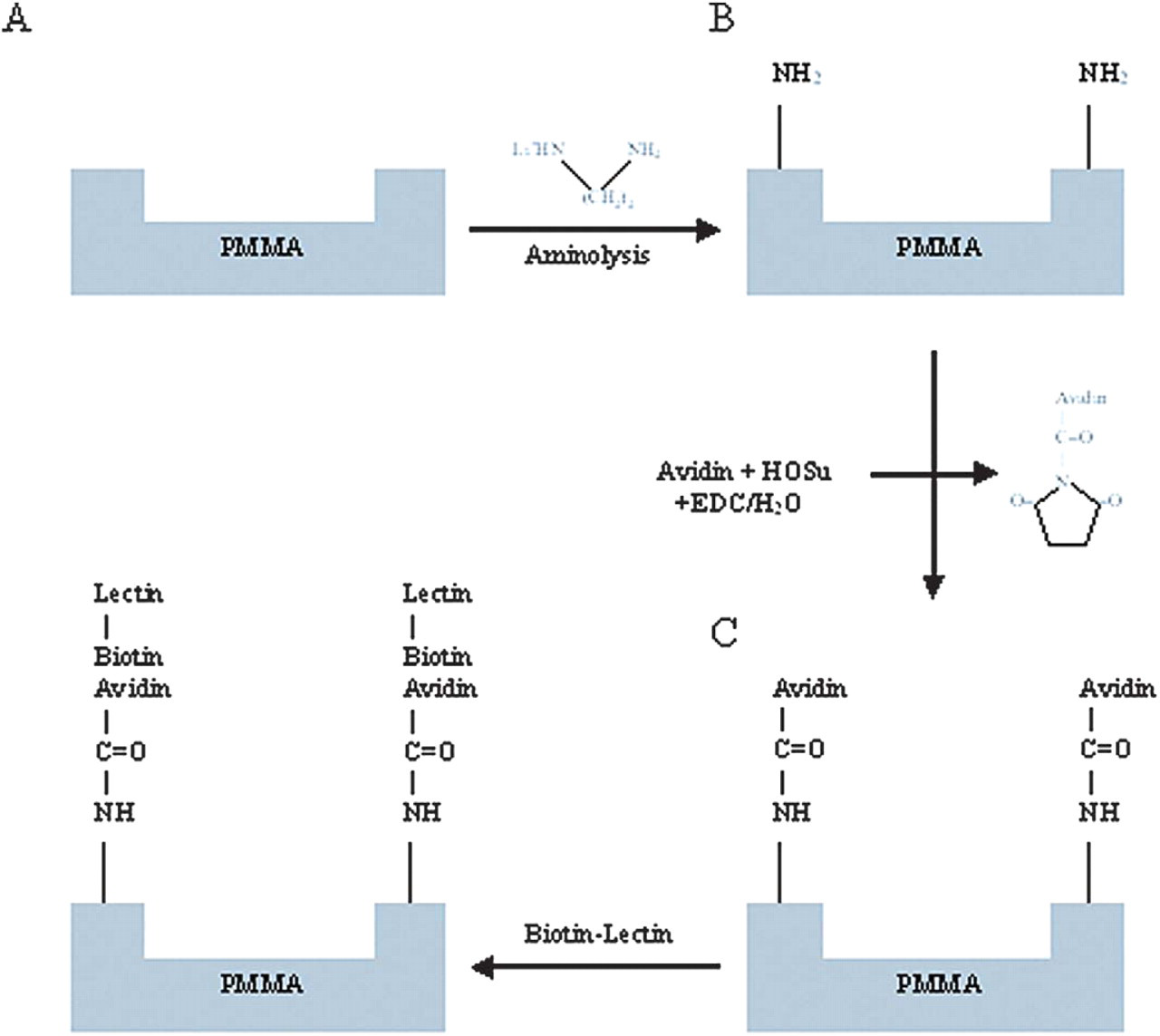
Surface modification process. (A) Aminolysis with N-lithioethylenediamine; (B) Avidin immobilization using a hydroxy-succinimide catalyzed carbodiimide reagent; and (C) Lectin attachment utilizing avidin-biotin attraction and interaction.
In Vitro Characterization
The human colorectal carcinoma cell line, Caco-2, was used to study the affect of attaching tomato lectin (Caco-2 cell specific) to the microdevice. Cytodhesion studies were performed by determining the percent of unmodified/lectin-modified microdevices able to attach to the Caco-2 mono-layer over time. Stability of the interaction between the microdevice and cells were compared to those of PMMA microspheres. Transport of molecules across the Caco-2 monolayer pre- and post- application of the microdevices was studied in order to observe the affect of microdevice interaction on monolayer permeability.
Results
The microdevices were successfully constructed from PMMA by applying a series of standard microfabrication processes including photolithography and reactive ion etching. Though it is possible to produce a multitude of devices with various dimensions and shapes, the prototype device was maintained at 150 μm × 150 μm × 5 μm with 80 μm × 80 μm × 2 μm reservoirs centered in the particle body (Figure 4). Cytoadhesive properties of the PMMA microdevices were evaluated by characterizing their interactions with Caco-2 monolayers (Figure 5). It was shown that specific binding of the microdevices to Caco-2 cells may be mediated by the type of lectin which is attached. After 15 120 minute incubation periods, PMMA microdevices conjugated with tomato lectin (specific to Caco-2 cells) showed a two- to six-fold increase in Caco-2 cell recognition over peanut lectin-conjugated (un-specific to Caco-2 cells) and unmodified microdevices (Figure 6). The stability of the microdevice-cell interaction was observed under static conditions. The PMMA lectin-modified microdevices were relatively stable; approximately 96% of the originally bound microdevices remained bound. In contrast, only 25% of the originally bound lecitin-modified microspheres remained bound (Figure 7). The potential of PMMA microdevices to open Caco-2 tight junctions was assessed by observing changes in the flux of phenol red across a cell monolayer when grown on a transwell permeable support. The cells reached confluency at day 4 post-seeding, and achieved complete formation of tight junction and inhibition of cell proliferation at day 8. After 21 days post-seeding, Caco-2 monolayers were unexposed (control) or exposed to approximately 400 PMMA microdevices or 400 tomato-lectin modified microdevices. The change in permeability was plotted as the apparent permeability after exposure to the devices divided by the apparent permeability at day 21. An initial increase in permeability occurred in those monolayers exposed to the PMMA devices (approximately three-fold over the control) suggesting some effect on the epithelial integrity. However, after 49 hours, permeability was recovered suggesting that no irreversible damage was induced. This may suggest a time frame in which drugs delivered from the microdevice may experience enhanced permeability across the epithelial layer due to intrinsic properties of the PMMA (data not shown).

SEM micrograph of an array of PMMA microdevices fabricated on a silicon wafer. 150x.

SEM micrograph of PMMA microdevices bound to Caco-2 monolayer. 200x.
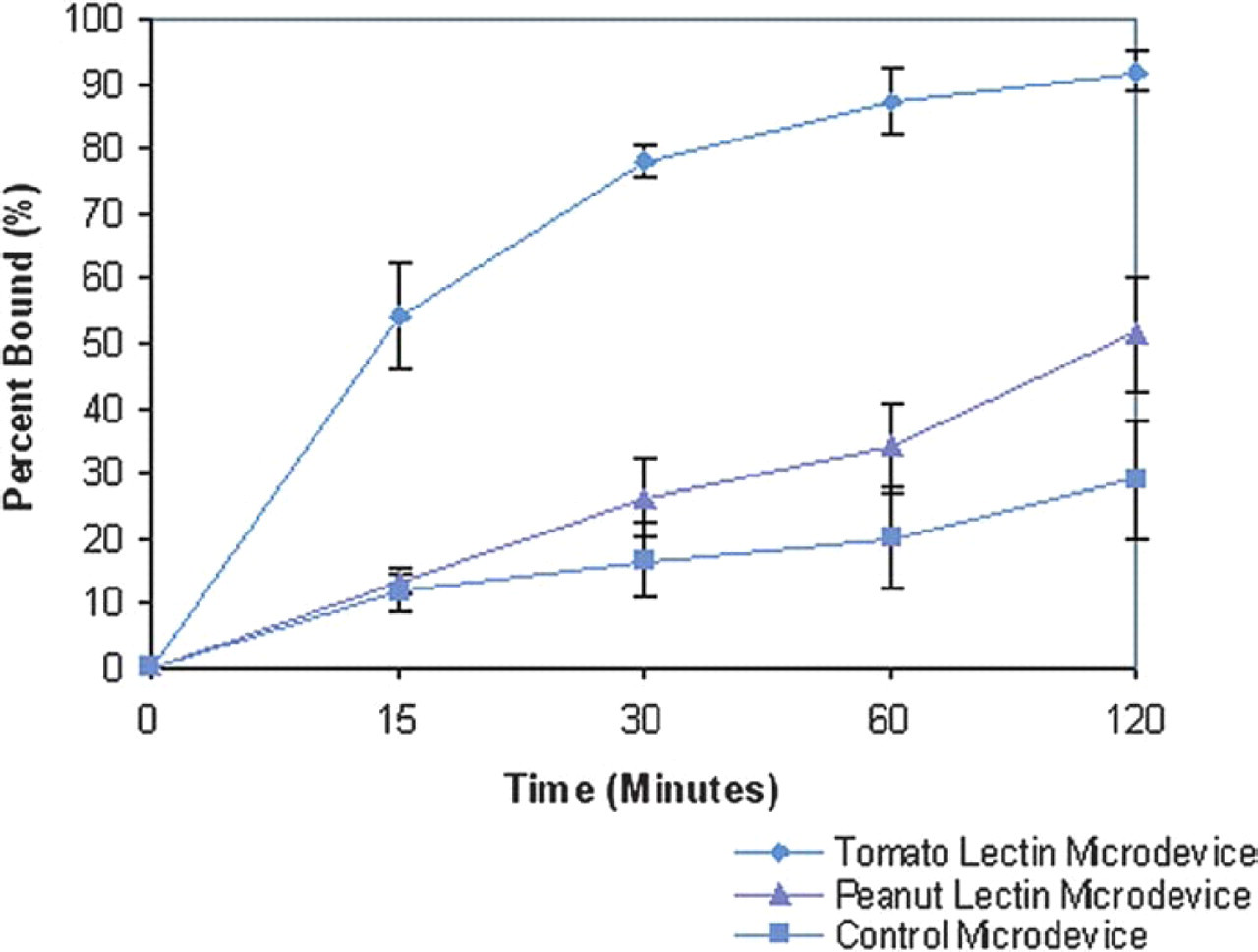
Microdevice adhesion to Caco-2 monolayer over a two-hour period.
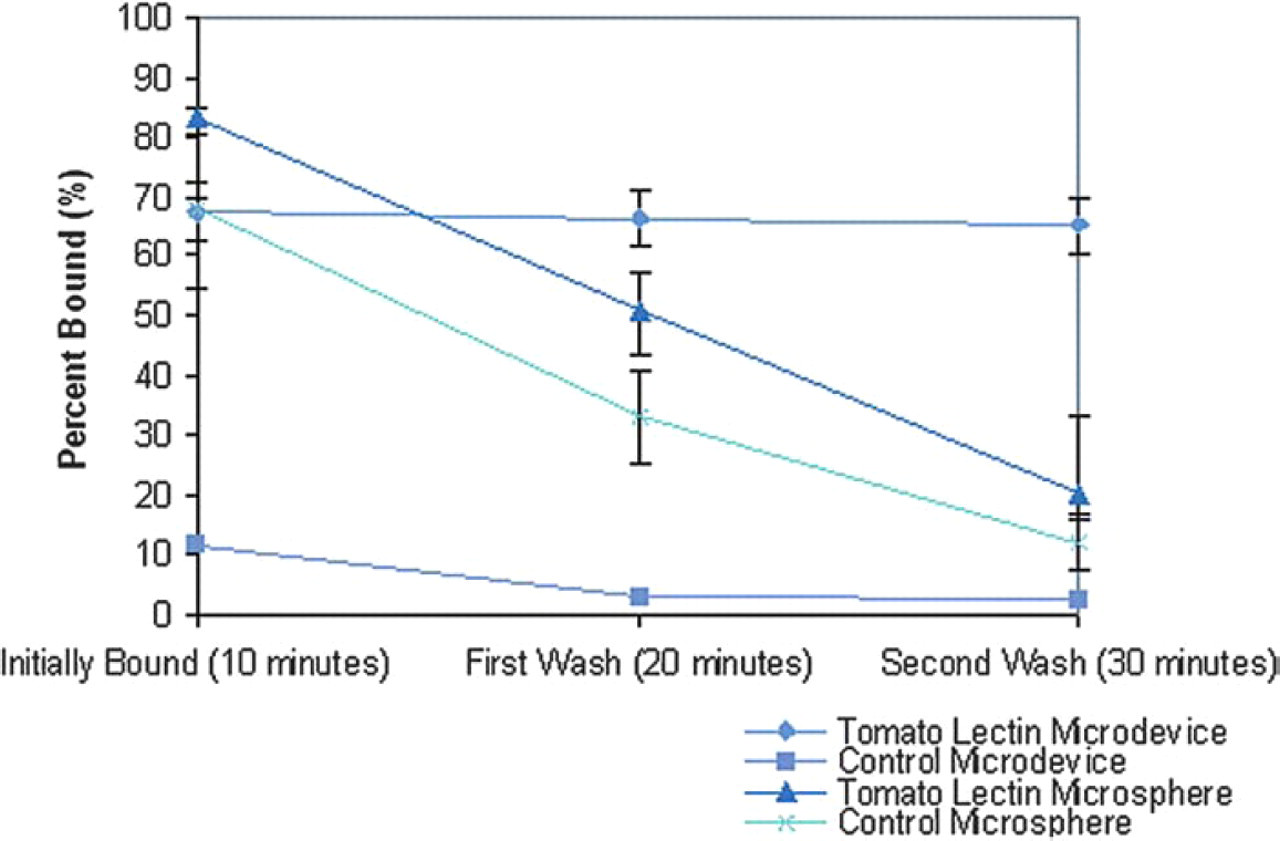
Stability of PMMA microdevice and PMMA micro-sphere adhesion to Caco-2 monolayer.
Conclusions
This research has shown that the proposed PMMA micro-devices can be precisely manufactured utilizing traditional microfabrication techniques, chemically modified on a single side to incorporate cell-targeting molecules, induce an initial increase in permeation across Caco-2 cell monolayers, and have improved cytoadhesion to Caco-2 cells over control devices. These microdevices remain anchored to Caco-2 cells in comparison to the unstable attachment of PMMA microspheres and do not cause any irreversible damage to epithelial integrity or cell structure. In conclusion, these findings demonstrate the advantages of using micromachined, asymmetrically cytoadhesive PMMA microdevices for applications in intelligent oral drug delivery.
Acknowledgments
This project is funded by the National Aeronautics and Space Administration (NASA) Graduate Student Research Program, the Center for the Integration of Medicine and Technology (CIMIT), and the National Science Foundation (NSF) Career Award.
Special thanks to Michael Lubeley and Anlee Krupp.
