Abstract
This work aims at a new class of methods for electric field-mediated separation of biomolecules. In contrast to standard techniques, electrophoretic transport was not performed in a single-phase homogenous fluid, but in a system of stratified layers. The large surface-to-volume ratio characteristic for microfluidic systems allows the interactions of biomolecules with liquid/liquid interfaces to be examined and the suitability of corresponding effects for applications in the field of biomolecular separations and enrichment to be assessed. To perform these types of studies, a micro flow cell was fabricated into which two coflowing immiscible liquid phases can be introduced. Subsequently, the electrophoretic transport of biomolecules driven by an electric field perpendicular to the channel was examined.
To investigate the transport phenomena related to electrophoresis in stratified two-phase systems, aqueous solutions consisting of polyethylene glycol and dextran were prepared, which allowed a stable interface to develop. Transport within one phase and an enrichment of proteins at the phase boundary has been established. In addition, other kinds of fluid combinations such as water and propylene carbonate have been examined, also supporting the enrichment of proteins at the phase boundary.

Introduction
Following the work of Albertsson, 1 aqueous two-phase systems (ATPSs) are finding ongoing and increasing applications in extractive separations and high-sensitivity biomolecular assays. ATPSs are formed by mixing one or more polymers or surfactants with water and can be used to selectively partition and purify target proteins or other types of sensitive biomaterials such as cell organelles or amino acids. 2 –5 One well-studied ATPS consists of aqueous solutions of polyethylene glycol (PEG) and dextran. 2, 6
Due to the high water content of both phases (70–90% w/w), the main advantages of ATPSs are high biocompatibility and low interfacial tension minimizing degradation of biomolecules. Experimental investigations have suggested that multiple factors influence protein partitioning, including the type of the ATPS and their compositions: the protein sizes and conformations as well as the buffer type, concentration, and pH value. 7, 8
Depending on the pH value of the surrounding solution proteins acquire different charges and thus different electrophoretic mobilities, which can be exploited either for electrophoretic separations or isoelectric focusing. 9 Normally, both of these separation approaches are based on a continuous buffer solution in a single-phase medium without any interfacial boundaries.
In this study, the main idea is to use two different properties of biomolecules: their physico-chemical affinities to the fluid phases of an ATPS and their electrophoretic mobilities when an external electric field is applied. The focus is on examining the interaction of biomolecules with fluidic interfaces and on establishing novel schemes for biomolecular separation and sample enrichment. Instead of a macroscopic system, which was used by Clark and colleagues, 10 – 12 Levine and Bier, 13 and Levine et al, 14 a microfluidic system was chosen for that purpose because of the inherent short diffusion lengths and large surface-to-volume ratios. In particular, the study of transport of proteins across the phase boundary can favorably be performed in transparent, polymeric microchannels.
Bi or multilaminated ATPSs based on PEG/dextran were formed inside microchannels with an electric field applied perpendicular to the main flow direction. By that the transport of different types of proteins from the preferred phase into the nonpreferred phase and vice versa was examined.
An additional two-phase system studied consisted of an aqueous buffer and the organic solvent propylene carbonate (PC). By adding quarternary ammonium salts to the system, PC can be made electrically conductive. Due the fact that proteins are not dissolvable in PC the applied electric field can be used for protein concentration at the phase boundary.
Experimental Details
Materials
All proteins, dextran of 500.000 Da average molecular weight (D 5251), PEG of 8.000 Da average molecular weight (P 2139), and PC were obtained from Sigma-Aldrich (Schnelldorf, Germany). Water was deionized and further purified using a Millipore (Schwalbach, Germany) Milli-Q water treatment unit. The proteins were labeled with fluorescence markers using the Alexa Fluor 488 Protein Labeling Kit (A-10235, Molecular Probes/Invitrogen, Karlsruhe, Germany).
The experiments with the ATPS were performed with labeled proteins dissolved in either the PEG- or the dextranrich phase (in the following also termed “PEG phase” and “dextran phase”). The two-phase system was prepared by dissolving 3.8 wt% PEG and 5.5 wt% dextran in a buffer solution (potassium phosphate, 5 mM) with different pH values (pH 6, pH 7, and pH 8). After stirring and settling overnight, the phases separated with a sharp stable phase boundary developed between them. The upper (less dense) PEG-rich phase (PEG phase) consisted of 5.7 wt% PEG, 1.0 wt% dextran, and 88.6 wt% of buffer solution. The lower (denser) dextran-rich phase (dextran phase) consisted of 1.9 wt% PEG, 9.5 wt% dextran, and 93.3 wt% of buffer solution. 10 Finally, the two phases were separated by a pipette and stored in different vessels. The gel enabling a fluidic decoupling of the microchannel from the buffer reservoirs consisted of 1.5 wt% agarose (Sigma-Aldrich, Schnelldorf, Germany) and is based on the same buffer as used for preparing the PEG/dextran two-phase system.
In the case of the water/PC two-phase system, bovine serum albumin (BSA) was dissolved in a buffer solution (potassium phosphate, 5 mM) and tetrabutylammoniumhydrogensulfate (Sigma-Aldrich, Schnelldorf, Germany) was added to PC to a final concentration of 1 M.
Microdevice
The polymethylmethacrylate (PMMA) microdevice was fabricated using a CNC micro milling machine and was either temporarily sealed by adhesive tape or permanently sealed by attaching a PMMA foil using a solvent-assisted bonding process. The microchannel design is shown in Figure 1. The chip consists of three inlets and one outlet. The width, depth, and length of the separation channel are 800 μm, 120 μm, and 30 mm, respectively. The channel is located between “comb structures” which are filled with agarose gel separating it from the buffer reservoirs on the backside of the chip where electrolysis takes place when an electric field is applied. The gel matrix enables a fluidic decoupling between the microchannel and the buffer reservoirs and serves as an ion conductor through which the electric field is guided. The channel comprises 40 gel bridges on each side, each having a cross section of 350 μm × 100 μm and a distance of 300 μm between each other.
After the channel structure is sealed, the agarose gel solution is heated up to 100 °C and filled into the comb structures from the backside of the chip. Driven by capillary forces the gel solution flows toward the separation channel and stops at the openings to the channel, which act as flow barriers for hydrophilic solutions. The comb structures are connected to reservoirs on the backside of the chip, which, after the agarose solution has gelled, are filled with the same buffer as used for preparing the two-phase system, cf. Figure 1.
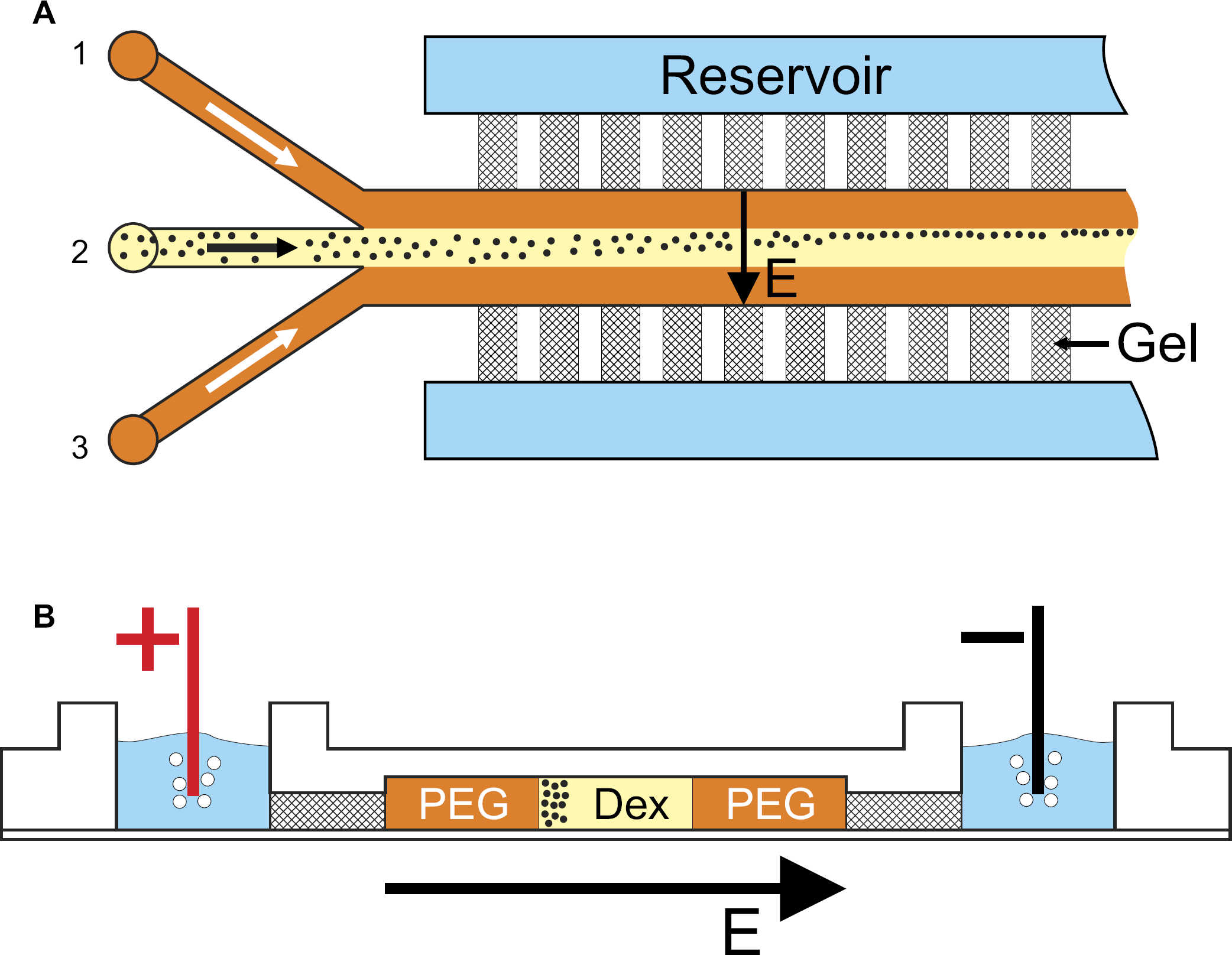
Setup of the microfluidic chip. Proteins are dissolved in their preferred phase, here dextran phase, and injected into the microchannel. The fluidic lamella is located between two PEG phase lamellae injected through two side channels. The transverse electric field allows driving the molecules selectively to one of the two phases, or enables accumulation at the phase boundary. The gel bridges act as part of the channel walls and electrically connect the separation channel with the reservoirs on the backside of the chip. Channel dimension: 800 × 120 μm (w × d). Flow rates: dextran 0.02 mL/h, PEG 0.1 mL/h.
Experimental Procedure
The PEG and dextran phases are coinjected into the microfluidic structure using syringe pumps (KD Scientific Inc., Holliston, MA, USA) at different flow rates. The proteins are dissolved in either the dextran phase or the PEG phase and are injected through inlet 2, cf. Figure 1. The corresponding liquid phase is flanked by the opposite phase fed to inlets 1 and 3 such that a system of three coflowing liquid lamellae is formed. Owing to the different viscosities of the two phases the flow rates have to be adjusted appropriately to achieve uniform lamella widths. For the experiments reported in this article, the flow rates were chosen to 0.1 mL/h and 0.02 mL/h for the PEG and dextran phase, respectively. The corresponding residence times of proteins within the electric field are long enough to influence their distribution across the channel significantly.
In the case of the PC-water system, two inlets were used and thus only two lamellae were formed within the main channel. The flow rates were chosen to 0.3 mL/h and 0.4 mL/h for PC and water phase, respectively.
The protein distributions for a subsequent evaluation are recorded by mapping the fluorescence intensity using a fluorescence microscope (CKX 41, Olympus, Hamburg, Germany) with a CCD camera attached to it (Kappa DX 30 C, Gleichen, Germany). All these images were taken at a position 30 mm downstream of the confluence of the feed streams, close to the exit of the channel. The time spans between the images were 3 min giving the system enough time to respond to a change in electric field.
Results
Owing to the small aspect ratio of the microchannel and a low (PEG/dextran) or moderate (water/PC) interfacial tension, a stable two-phase flow consisting of three parallel lamellae in the case of PEG/dextran and two parallel lamellae in the case of water/PC is possible. Undulations or instabilities of the phase boundary of a PEG/dextran system were only observed at comparatively high voltages (> 15 V). 15
Eletrophoretic Transport of Proteins in PEG/Dextran ATPS
If the proteins are dissolved in the preferred dextran phase and no electric field is applied, the proteins hardly penetrate the phase boundary. In this case, the phase boundary appears to impede transport. Only if the electric field exceeds a specific threshold value the proteins increasingly overcome that barrier. In the case of BSA, the threshold voltage is approximately 3 VDC and appears to be independent of the pH value, as shown previously. 15
If the proteins are dissolved in the nonpreferred PEG phase, they easily diffuse into the dextran phase after the two phases get in contact. An additional electric field leads to an enhanced and directed transport into the preferred dextran phase. This additional electrophoretic migration across the phase boundary starts immediately when the E-field is switched on and leads to a rapid decrease of fluorescence intensity within the PEG lamella, also at low voltages, cf. Figure 2.
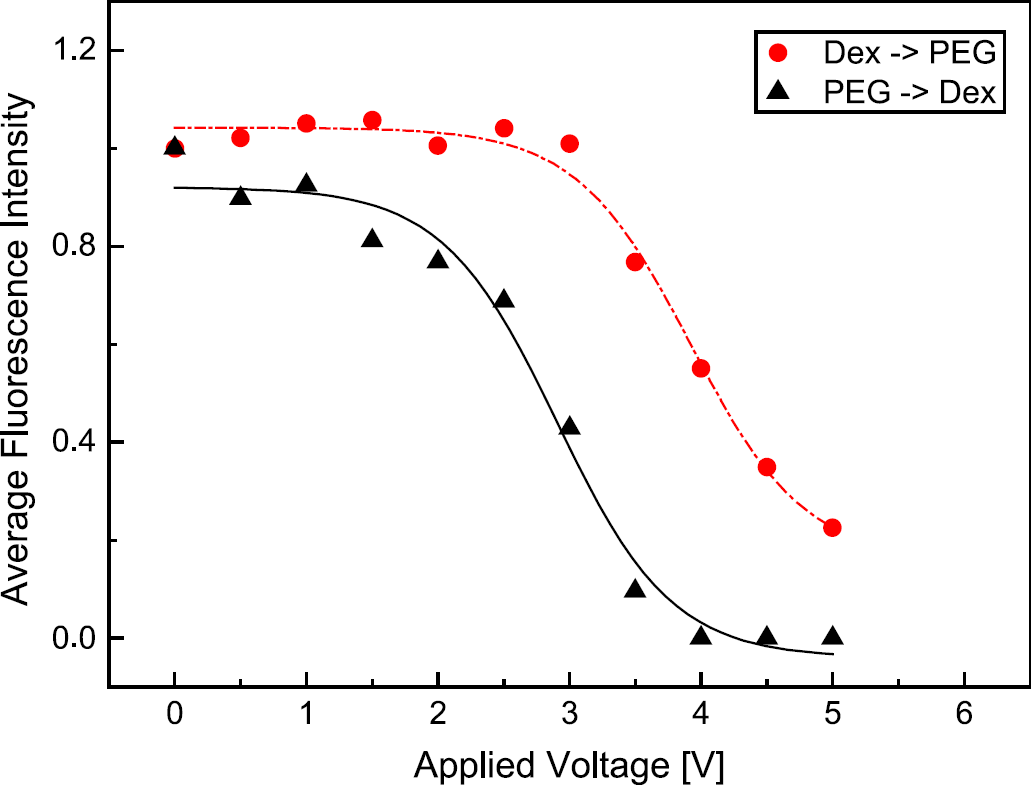
Comparison of fluorescence intensity of BSA within the lamella into which BSA is injected. PEG→ Dex: BSA is injected in the nonpreferred PEG phase. By applying an electric field the proteins rapidly leave the phase and penetrate the phase boundary without any hindrance. Dex→ PEG: BSA is injected in the preferred dextran phase, the phase boundary impedes protein transport. Only when a voltage of about 3.0 VDC is reached, the BSA molecules increasingly penetrate the phase boundary. Buffer: potassium phosphate, 5 mM, pH 7.
This resistance-like behavior and enrichment of proteins at the phase boundary has also been discussed by Theos and Clark, 12 and Levine and Bier 13, 14 and takes place only in buffer systems that provide a significant double layer potential due to inhomogeneous distribution of ions, such as a phosphate buffer. 16 –20 Double layer formation at liquid-liquid interfaces is a well-investigated phenomenon and has recently been illuminated in more detail based on molecular-dynamics simulations (see [21,22] and references therein).
It is straightforward to use the setup described above for protein separation, which, in its most general form, is based on exploiting different protein properties, such as protein size, its hydrophobicity, or its isoelectric point. To examine if there is any dependency on the type of protein, the experiments on electrophoretic transport in PEG/dextran systems were performed with different proteins. In addition to bovine serum albumin (BSA, 66 kDa), protein samples of beta-galactosidase (116 kDa) and carbonic anhydrase (29 kDa) were examined.
Similar to the observed behavior of BSA, the other protein types also display a high affinity to the dextran phase. This means that if the proteins are injected into this phase they hardly leave it by diffusion. After applying an electric field all tested protein species show a movement and redistribution within the dextran phase, but still no increased movement across the phase boundary itself as long as the field strength stays below a certain threshold value. For a specific voltage range the average fluorescence intensity (averaged over a dextran lamella) stays at the same level until the proteins start to leave the preferred phase and penetrate into the PEG phase. This finally leads to a decrease in fluorescence intensity. When increasing the electric field strength it is self-evident that all proteins will start to leave the dextran phase sooner or later. Remarkably, however, in the case of the PEG/dextran system, all protein species start to overcome the phase boundary within nearly the same voltage range of about 2.5–3 V DC. Only in the downward slope there seems to be some difference between the different protein types, cf. Figure 3, an issue that is being clarified in ongoing experiments aiming at determining E-field-dependent partitioning coefficients.
It seems that the observed behavior has to be attributed to a quite universal mechanism dominating the transfer of molecules between the phases. Because all of the proteins studied have a higher physico-chemical affinity to the dextran phase than for the PEG phase, this difference in chemical potentials itself impedes transport by diffusion or electromigration. However, the systematics of protein partition coefficients in ATPSs 23 suggests that the height of the chemical potential barrier is different for the different proteins studied here. Therefore, it is speculated that the transport resistance due to the different affinities to the phases is not the dominant effect. The most natural explanation for the “universal” shape of the fluorescence intensity curves is the formation of an electric double layer at the phase boundary. As discussed by several authors, 16 20 the ions of the buffer solution will usually have different affinities to the two different aqueous phases. Consequently, they partition between the phases, resulting in charge separation and the formation of an electric double layer at the phase boundary. In this double layer, an electric field could be formed in such a way that it opposes the transport of proteins from the dextran to the PEG phase. Because the electric double layer potential difference is the same for all protein species, the corresponding transport resistance would be the same, thus offering a possible explanation for the observed “universality” of the curves in Figure 3.
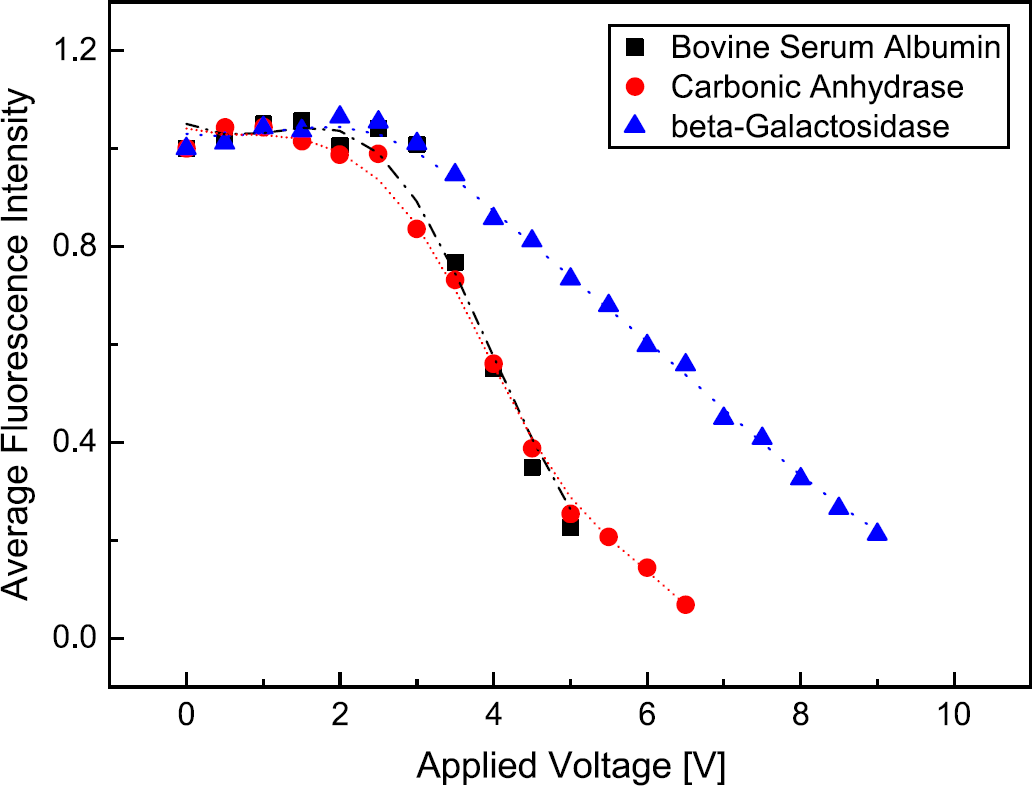
Comparison of the fluorescence intensity of different proteins, which are injected in the preferred dextran phase. A notable transport of proteins into the PEG phase is observed only at a voltage of about 3 VDC.
Eletrophoretic Transport of Proteins in Water/PC Two-Phase Systems
Owing to the fact that BSA is not readily dissolvable in organic solvents such as PC, this system was used for examining the concentration of proteins at the phase boundary itself. To obtain a significant electric field within the aqueous phase the organic phase has to be conductive. Otherwise, most of the electric potential drop will occur in the latter and almost nothing will remain for the transport of the proteins within the aqueous fluid lamella. For that purpose tetrabutylammoniumhydrogensulfate was added up to a concentration of 1 M, which gives a conductivity of 3.8 mS within the PC phase.
In this case, the microfluidic system comprises two parallel lamellae and again an electric field is applied perpendicular to the flow direction. At a vanishing electric field the BSA molecules show a uniform distribution within the water lamella. Upon applying a voltage of 15 VDC, a concentration of BSA is observed directly at the phase boundary between PC and the aqueous phase. The width of the band of concentrated proteins is about 60 μm, cf. Figure 4. A voltage increase up to 20 V DC leads to a narrowing of the band down to approximately 20 μm and to an increase of the fluorescence intensity of 35%. However, this value is not fully representative because in some parts of the concentrated band the maximum fluorescence intensity which can be recorded by the fluorescence camera is already reached. A further increase of the electric field strength does not lead to a higher concentration of BSA molecules at the phase boundary, but to a deformation of the boundary itself. As the camera only captures 2D distributions this deformation manifests itself as a visible band widening. In all cases and within the examined voltage range, no penetration of BSA into the PC phase was observed and only small amounts of denatured and deposited BSA got visible after several minutes of flow.

Bilaminated PC (top)-water (bottom) system. When an electric field is applied, the proteins accumulate at the phase boundary. An increase of the field strength leads to an increase of protein concentration. (1): 0 VDC, (2): 15 V DC, (3): 20 VDC.
Conclusion and Outlook
Experiments on the electrophoretic transport of BSA molecules in binary or tertiary lamella arrangements of immiscible phases have been carried out for two liquid-liquid systems. Both fluid combinations showed novel phenomena that, to our best knowledge, have not yet been observed before. For the system with aqueous solutions of PEG/dextran, an E-field can be used to drive the sample molecules perpendicular to the phase boundary, thus enabling a much faster transport than purely by diffusion. In this system, based on a potassium phosphate buffer, the boundary between the two phases impedes protein transport. BSA molecules initially in the PEG phase penetrate the phase boundary without any hindrance. If BSA is dissolved in the dextran phase and an electric field is applied perpendicular to the liquid lamellae, a negligible transport across the phase boundary is observed until a certain field strength is reached. Only after exceeding this threshold voltage the proteins start to leave the preferred phase and the average fluorescence intensity within the dextran lamella decreases. With respect to the “breakthrough voltage” of this “resistance”, no significant differences between different protein species could be detected.
One future aim is to determine the partition coefficients of different proteins at non-zero electric field, thus generalizing previous studies at vanishing E-fields. Also, alternative ATPSs based on other polymer types or surfactants will be the focus of future studies. With the developed microfluidic system it should be possible to examine whether or not there is a combination of parameters that allows for protein separations not directly based on size or isoelectric point, but on more general criteria.
The water/PC system having been examined as an alternative to the PEG/dextran system opens up possibilities for the enrichment of biomolecules or other charged particles by electrophoresis combined with a two-phase flow. This version could be used for a continuous concentration of different biomolecules such as DNA or proteins. Proteins can be moved toward the phase boundary within the aqueous lamella. This leads to a strong protein concentration at the interface within a band of approximately 60 μm and less width. For both types of two-phase systems the final goal of our work is to use all these effects for novel microsystem-based separation and enrichment techniques for biomolecules.
Acknowledgment
This work is supported by the German Research Foundation, priority program 1164.
