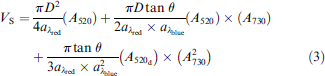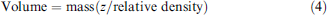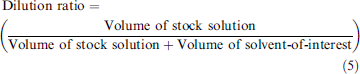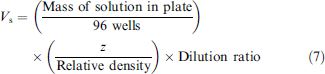Abstract
Multichannel volume dispensing devices, such as automated liquid handlers, are widely used in drug discovery assays and other high-throughput screening processes. The performance of these systems is heavily based on the ability to deliver proper volumes of specific reagents. For instance, because concentrations of species within an assay are volume dependent, assay integrity and the subsequent interpretation of assay results are directly tied to liquid-handler performance. When liquid handlers are used to aspirate/dispense aqueous-based reagents, there are many accepted methodologies used to calibrate/verify the system's ability to properly perform within a user's tolerance window. In other situations, however, liquid handlers are used to dispense complex or nonaqueous reagents, such as dimethyl sulfoxide (DMSO), serum, and aqueous-based mixtures with detergents, for which there may not be standard, or simple, methodologies to verify liquid-handler performance. Discussed herein is the recent research on broadening existing methods for accurately assessing liquid-handler performance when dispensing complex or nonaqueous reagents. Accurate and reliable adjustment of liquidhandler protocols for varied reagent types could have farreaching adoption in all scientific communities. Using this methodology, target aliquots of multiple solutions were dispensed with a calibrated syringe and subsequently validated via dual-dye ratiometric photometry. Various custom sample solutions were prepared to incorporate (a) 75–90% DMSO (v/v), (b) 20% glycerol (v/v), and (c) 50% ethanol (v/v). (JALA 2006;11:172-80)
Keywords
Introduction
Methods of determining the amount of liquid volume dispensed from a liquid-handling device are well documented and include gravimetric, fluorometric, and photometric approaches.1–6 None of these methods, however, is particularly well suited for calibrating liquid-handling devices to precisely and accurately dispense all liquid types. In particular, some methods are not optimally designed for calibrating liquid handlers to dispense liquids that are nonaqueous or complex. As discussed herein, the term complex is used to refer to any liquid type (aqueous, organic, etc.) that has one or more components or additives, such as other liquid types, salts, sugars, detergents, surfactants, and proteins. Although liquid-handling instruments may be used to dispense a wide array of reagent types, it is commonly known that method parameters, such as aspirate/dispense rates or dialed-in target volume, can vary significantly between different solvent types resulting in different end volumes. Thus, a methodology used for aqueous solutions may dispense a significantly different volume if used for organic solvents. To accommodate varying solvent types, some liquid-handler software packages incorporate computational algorithms, commonly known as “user-defined liquid classes”, which provide users with the ability to adjust dispense methods to compensate for potential solvent-dependent performance differences. However, unless an accurate measurement of the liquid-handler performance is determined for each method, each solvent, and each target volume, a potential false-sense of performance could ultimately result in downstream troubleshooting and economic loss. This article discusses a new methodology for preparing customized sample solutions for assessing a liquid-handler's performance as measured using the Artel Multichannel Verification System (MVS, Westbrook, ME). 6
Quantifying Dispensed Volumes with the MVS
The MVS is a complete system based on dual-dye ratiometric photometry. Because this system has recently been described in detail, 6 only a brief overview is provided here. The MVS system is composed of various components including aqueous-based dye solutions, dimensionally characterized microtiter plates, and a microtiter plate reader. The operating principles of the MVS involve dispensing the target volume of a sample solution containing both red and blue dyes into the wells of a characterized microtiter plate. This dispense is carried out by the liquid-handling device that is being tested. A diluent, which contains the same concentration of blue dye as in the sample solution, is also dispensed into each well to a defined total working volume (either before or after the sample dispense), and does not have to be carried out by the liquid handler being tested. The dispensed solutions are then mixed and the absorbance values of the two dyes are measured (at 520 and 730 nm) and used to determine the volume of sample solution that was delivered.
Because the MVS operates by the principles of absorbance photometry, it is ultimately governed by the well-known Beer–Lambert law, shown in Eq. (1), which relates the measured absorbance (A) of a solution at a given wavelength (λ) to the molar absorptivity (ε) of the solution at wavelength λ, the concentration (C) of dye in the solution, and the pathlength of light (l) through the solution:
When using the Beer–Lambert law for measurements made with a vertical beam spectrophotometer, for example, a microtiter plate reader, it becomes convenient to combine the concentration (C) and the molar absorptivity (ε) into a new term called the absorbance per pathlength (aλ), as defined by Eq. (2):
The MVS system calculates the volume of sample solution dispensed into the wells of a microtiter plate by incorporating a known absorbance per pathlength (aλ) for each dye, red (aλred) and blue (aλblue), in the sample and diluent solutions with the known dimensions of the microtiter plate wells (bottom diameter D, side-wall taper angle θ), and the measured absorbance for each dye (A520, A730). For a microtiter plate with truncated cone-shaped wells, for example, round-well plate, the sample volume is determined using Eq. (3):
Although a more detailed and complete discussion of the dual-dye MVS method for the evaluation of automated liquid-handling devices is presented in references 6 and 7, a few important items should be noted. First, it is important to note that aqueous-based volume-verification measurements performed with the MVS are traceable to the National Institutes of Standards and Technology, and photometry is an approved method by the International Organization of Standardization (ISO 8655-7: 2005) for volume verifications. Second, the initial MVS system allowed for assessing liquidhandler performance only for aqueous-based solvents. The work reported herein describes efforts to expand the use of MVS to other commonly used solvents. The new method described below is a process for preparing a custom sample solution using a solvent-of-interest. By accurately combining a solvent-of-interest and a concentrated dye stock solution with known aλred, a custom sample solution is prepared that both mimics the solvent-of-interest and can be measured with the MVS. Target aliquots of the custom sample solution can be aspirated, dispensed, and quantified to correlate the liquid-handler's performance to the original solvent-of-interest. Figure 1 is a flowchart showing an overview of the methodology described in this article.
A flowchart describing the preparation of custom sample solutions, which are evaluated using a dual-dye photometric approach.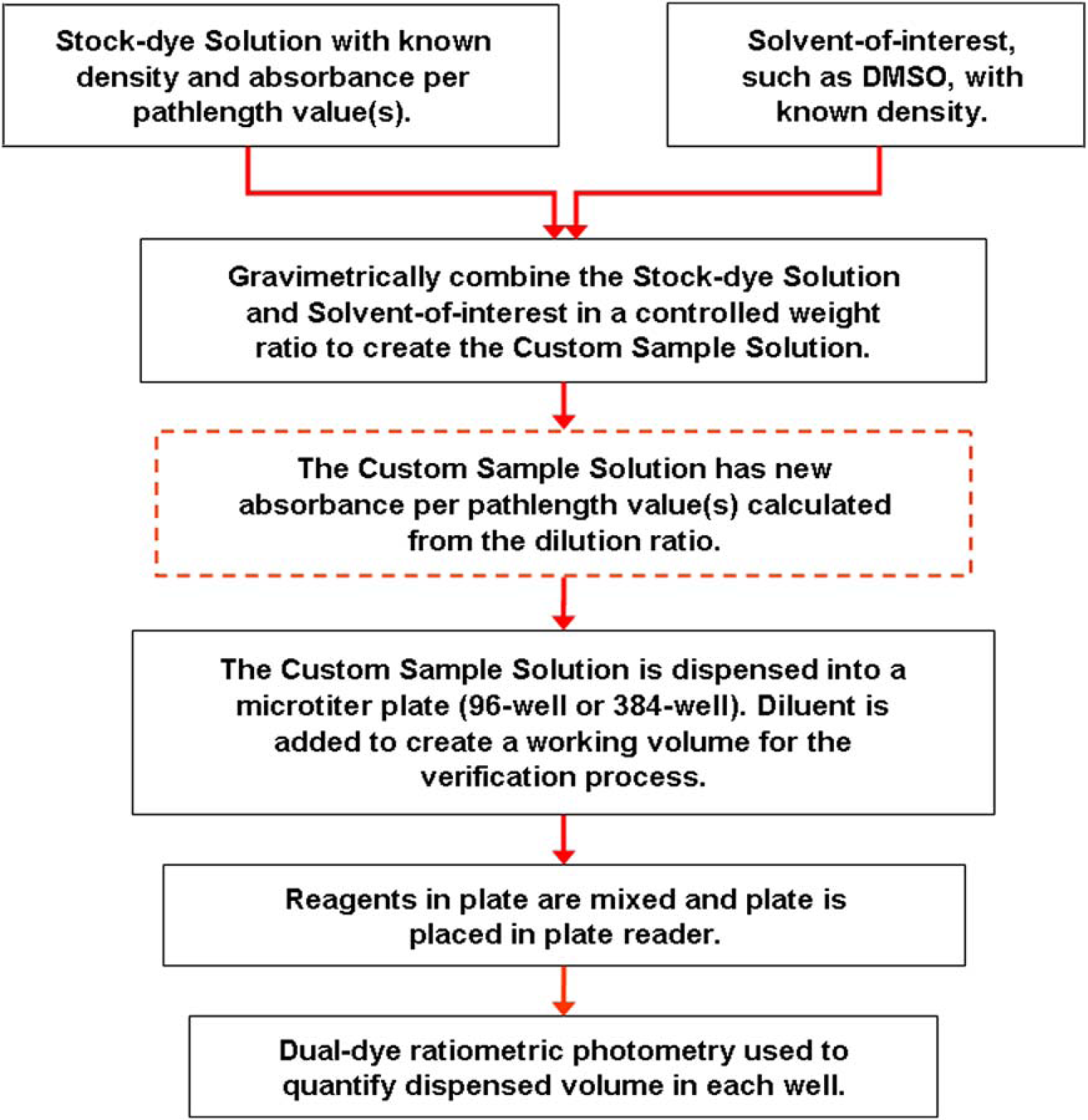
Stock Solutions Used to Prepare Custom Sample Solutions
Stock solutions, which can also be referred to as stock-dye solutions, are similar to MVS sample solutions in that they consist of well-defined composition. Because these stock-dye solutions are defined by aλred values, they can be gravimetrically combined with a solvent-of-interest, such as dimethyl sulfoxide (DMSO), to create a custom sample solution. The custom sample solution is prepared in a controlled, stepwise manner so that the dilution ratio is used to determine the new aλred value. In other words, a very controlled amount of dye is added to the solvent-of-interest so that the custom sample solution can be quantified using dual-dye ratiometric photometry.
Density Considerations and Dilution Factors
The custom sample solution is made by mixing a stock-dye solution with a solvent-of-interest by using a gravimetric procedure. By determining the weight of the solvent-of-interest and the weight of the stock-dye solution, and incorporating the relative density of each, the volume contribution for each liquid component can be determined using Eq. (4)
Eq. (4) is a more accurate version of the relationship between volume, mass, and density, where better accuracy of the volume determination is achieved by using the z-correction factor (z), which accounts for fluctuations in relative densities for each solution as a function of atmospheric conditions (buoyancy factor of air and density changes as a function of atmospheric temperature, pressure, and humidity). Once each solution is weighed and the volume contributions for each liquid component are determined in Eq. (4), the values can be substituted into Eq. (5) to determine the volume-based dilution ratio
Accurately knowing the dilution ratio allows for an accurate calculation of the new absorbance per pathlength values for the custom sample solution. The dilution ratio is used to calculate a revised absorbance per pathlength for each dye through Eq. (6):
where the new absorbance per pathlength (a′λ) is determined as the product of the stock-dye solution's absorbance per pathlength and the dilution ratio.
This new, or revised, absorbance per pathlength is substituted into the MVS volume calculations. The method described above shows how the MVS can be used to measure custom sample solutions. The MVS performance is based on measuring absorbance values for each custom solution between approximately 0.4 and 2.4 absorbance units, which ultimately dictates the total concentration of dyes allowed in the custom sample solutions. In other words, solutions with a relatively large aλred value, and therefore a higher concentration of red dye, can be used for measuring smaller volumes of sample. For the purpose of this article, the custom sample solutions were prepared to be in the same working volume range as the commercially available MVS sample solutions. For instance, in a 96-well plate, a custom sample solution prepared with DMSO might work for target volumes between 0.4 and 4.3 μL, which for the sake of this article, are referred to as group D sample solutions. A second DMSO sample solution might cover 1.8–11.0 μL (group C), and a third might work between 9.1 and 49.9 μL (group B).
Materials and Methods
All purchased materials were used as received. DMSO (99.9% spectroscopic grade; 1.1 g/mL), glycerol (Biochemika Ultra; 1.262 g/mL), and ethanol (ACS reagent, absolute; 0.7892 g/mL) were acquired from Sigma–Aldrich. The MVS and all MVS-related materials were from Artel (Westbrook, ME). A Sartorius R160D analytical balance was used for all gravimetric determinations, except for the gravimetric comparison using a syringe (see below). All organic solvents were used with caution, and protective gear and hoods were used where applicable. All solutions were allowed to equilibrate to room temperature prior to being used.
Stock Solutions 1 and 2
Stock solution 1 was prepared to have an absorbance of approximately 740 at 520 nm in a 1-cm pathlength cuvette. Stock solution 2 was prepared to have a target absorbance of approximately 72 in a 1-cm pathlength cell at a wavelength of 520 nm. The exact absorbance per pathlength values for each stock solution used for each experiment are defined below. The density values for stock solutions 1 and 2 are 1.0077 and 1.0059 g/mL, respectively, as determined by the relative density of each solution versus deionized water. All relative density measurements were made with a pycnometer (Kimble-Kontes, Vineland, NJ).
Preparing Custom Sample Solutions
All solution components were weighed on the analytical balance. Following each component addition, the balance was allowed to reach equilibrium, the information was collected, and the balance was tared before the next component was added. In all cases, a clean, amber glass bottle and cap were used to prepare and store a custom sample solution. Once all components were added, the bottle was mixed by inversion at least 20 times. The custom sample solutions were designed and prepared to incorporate similar absorbance per pathlength values as the commercially available, aqueous-based MVS sample solutions.
DMSO-Based Sample Solutions
One group of custom sample solutions (group D) were prepared by mixing 7.0 g of stock solution 1 (aλred = 737:832) with 22.96 g of pure DMSO to make a 25% stock solution 1 in DMSO (v/v). This mixture consisted of 75% DMSO (v/v) and, as calculated by the MVS software, these solutions covered the approximate volume range of 0.4–4.3 μL in a 96-well plate. A second group of DMSO sample solutions (group C) were prepared by mixing 3.0 g of stock solution 1 (aλred = 737:832) in 29.837 g of pure DMSO to make a 9.9% stock solution 1 in DMSO (v/v). Each of these mixtures consisted of 90.1% DMSO (v/v) and covered the approximate volume range of 1.8–11 μL in a 96-well plate. A third group of DMSO sample solutions (group B) were prepared by mixing 6.0 g of stock solution 2 (aλred = 72:7378) in 25.92 g of pure DMSO to make a 20.2% of stock solution 2 in DMSO (v/v). These mixtures consisted of 78.8% DMSO (v/v) and covered the approximate volume range of 9.1–49.9 μL in a 96-well plate. The density of one DMSO sample solution (group C) was determined with a pycnometer to be 1.1029 g/mL.
Gravimetric Dilutions with DMSO-Based Sample Solutions
Three different DMSO-based sample solutions were prepared to include one solution in each group (D, C, B). Each of the three DMSO-based sample solutions was then used to prepare multiple (five to six) large-volume batches via a controlled dilution method. For instance, the group D sample solution was gravimetrically diluted with aqueous diluent to create six different 60-mL batches, where each batch consisted of a different target volume. Each of these large-volume dilutions was then used to fill every well in a 96-well plate to a total volume of 200 μL, and the weight of the dispensed solution was collected for the filled plate (minus the tare weight of the empty plate). This process was similar for the group C and group B sample solutions. The ratio of DMSO-based sample solution to diluent is based on the 200-μL working volume for the wells of a 96-well plate. For example, for a 5-μL target volume, the ratio would equal 5/200 of DMSO-based sample solution to total solution and the target sample volume (Vs) would be calculated as:
Into each well of a 96-well microtiter plate, 200 μL of the dilution sample was dispensed using an eight-channel Rainin handheld electronic pipette. Before using the MVS for the volume-verification measurement, each filled plate was weighed using the analytical balance.
Stability of DMSO-Based Sample Solutions
Two different DMSO-based sample solutions (group C) were prepared and kept in the dark at room temperature until use. The two solutions were named DMSO Solutions C1 and C2 and prepared with stock solution 1 (aλred = 737:832). DMSO Solution C1 was prepared with 5.1628 g of stock solution 1 and 51.481 g DMSO (Fig. 2 represents the software's screen shot used when preparing DMSO Solution C1). DMSO Solution C2 was prepared with 5.2749 g of stock solution 1 and 58.0378 g of DMSO. On each of the nine testing days during the 41-day period, a 3-mL aliquot of the solution was first transferred to a glass vial, which was sealed with a crimp-top cap. Using a calibrated syringe, each solution was aspirated from the glass vial and dispensed in replicates of eight. Following the target volume aliquot, 192 μL of diluent was simultaneously added to each well using an eight-channel Rainin handheld electronic pipette. During the 41-day testing period, three different calibrated syringes (see reference 6 for more detail on these specialized syringes), each with a slightly different mean volume, were used. A 7.910-μL syringe was used for the first six data points, a 7.990-μL syringe for the seventh data point, and a 7.980-μL syringe for the last two data points. Significant work has been carried out internally to establish these specialized syringes as volume transfer standards for aqueous solutions. Because these syringes can reproducibly dispense a calibrated volume (Table 1), and because each syringe had a different calibration value, the relative inaccuracy to the calibrated syringe value was used as an unbiased comparator to monitor potential performance degradation of each solution (Fig. 4).
An image of the MVS software's graphical user interface used when preparing a custom sample solution. This particular screen shot was used when preparing DMSO Solution C1. Based on the absorbance per pathlength of the stock solution and the weights and densities of both liquid components, the software calculates a usable volume range for the specific custom sample solution for a 96-well plate and a 384-well plate. The custom sample solution is also known as an alternative solution. Multiple DMSO-based sample solutions were prepared to have a specific, gravimetrically determined target volume (x-axis, see text). By using gravimetry as a standard, these target volumes, which covered 0.5–44.7 μL, were then used to validate the performance of the MVS for measuring the DMSO-based sample solutions. All relative inaccuracy performance data are within ±2.5%. The error bars represent the CV% of the measurement for each target volume. The figure legend is as follows for each of the DMSO-based sample solutions: Two DMSO-based sample solutions were prepared and periodically evaluated with an 8-μL calibrated syringe over a 41-day period. As shown by relative inaccuracy data, the stability in performance did not degrade during the testing period for either solution. All relative inaccuracy performance data are within ±0.7%. The error bars represent the CV% in each measurement. The figure legend is as follows for the two DMSO-based sample solutions: 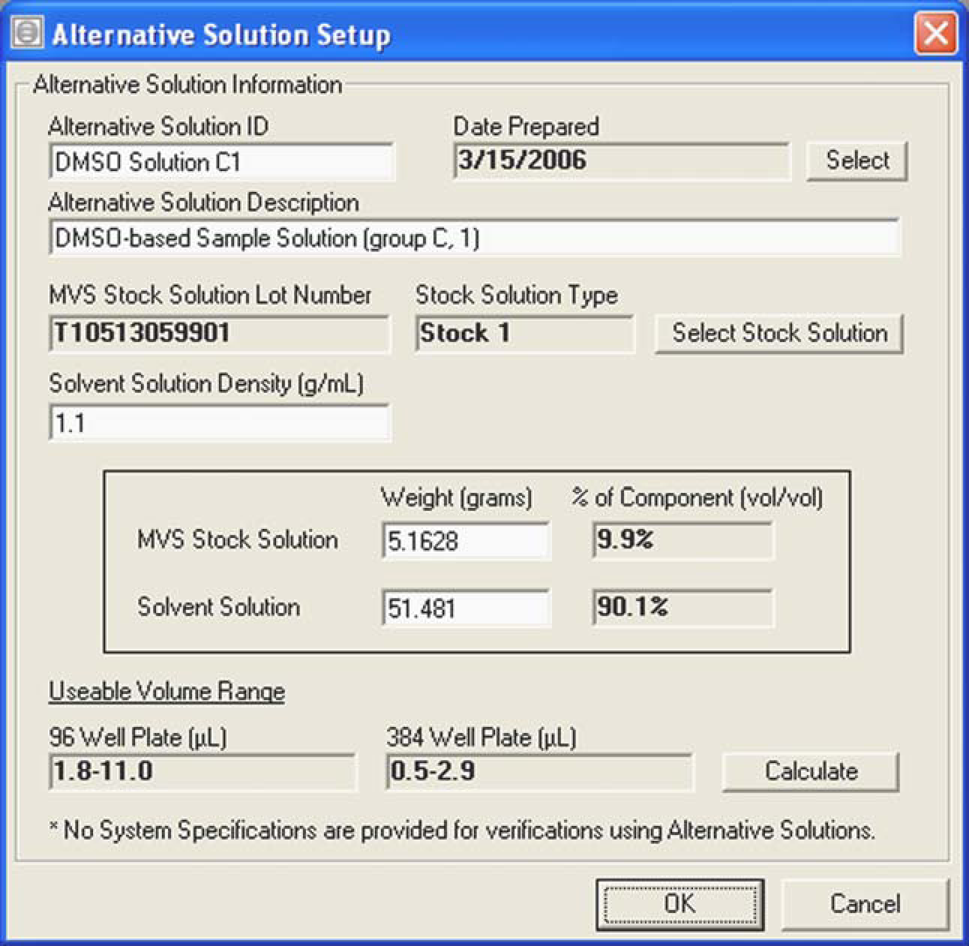

 group B,
group B,  group C, and
group C, and  group D.
group D.
 DMSO Solution C1 and
DMSO Solution C1 and  DMSO Solution C2.
DMSO Solution C2.
Gravimetrically measured performance of a syringe used as a volume transfer standard for three different solution types
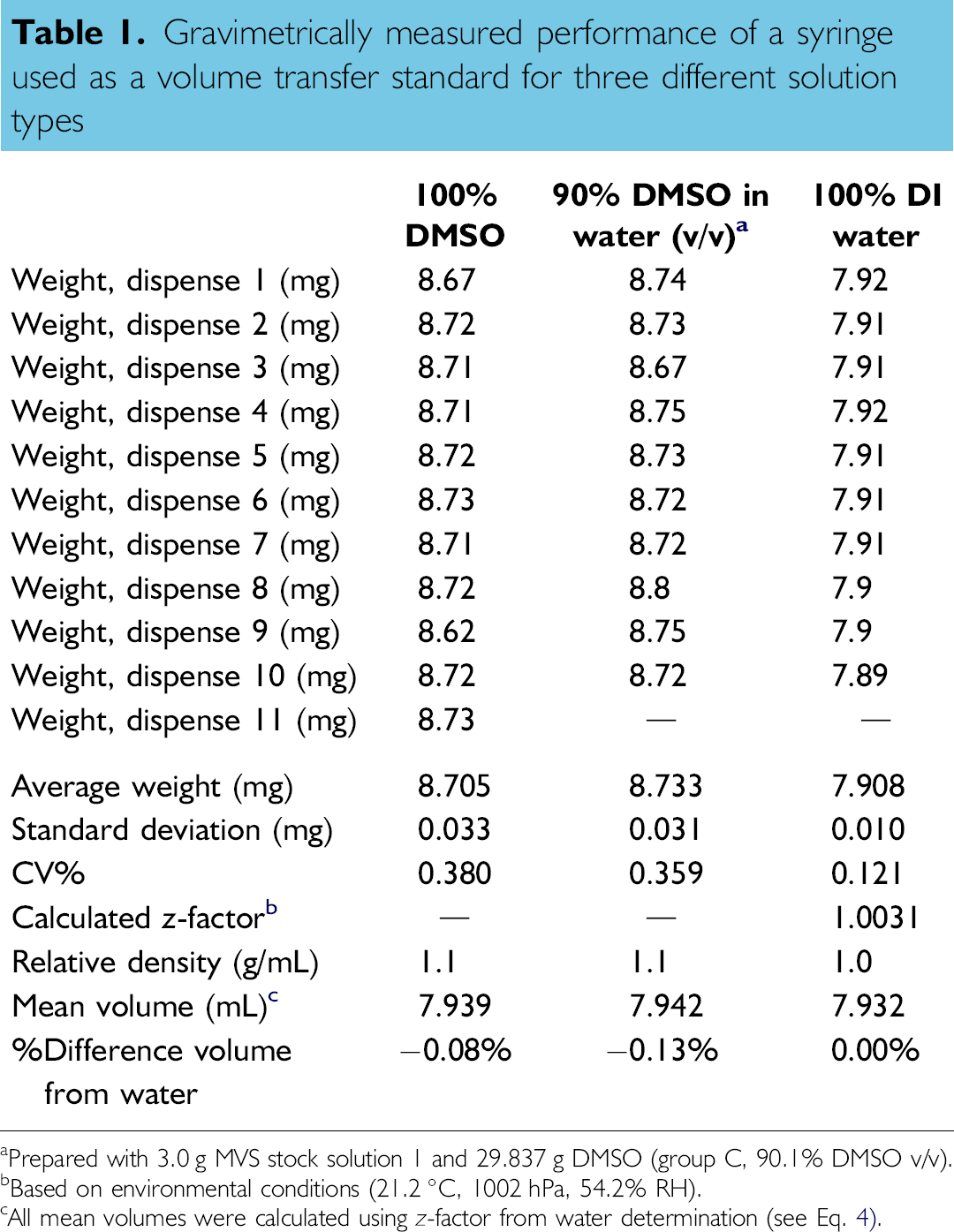
Prepared with 3.0 g MVS stock solution 1 and 29.837 g DMSO (group C, 90.1% DMSO v/v).
Based on environmental conditions (21.2 °C, 1002 hPa, 54.2% RH).
All mean volumes were calculated using z-factor from water determination (see Eq. 4).
Other Custom Sample Solutions
Two different (non-DMSO) custom sample solutions were also briefly evaluated. As opposed to the process of combining only two liquid components, that is, DMSO and stock solution, the following example used three components and each one was weighed separately, taking into account individual density value to determine the total volume contribution from each component. Once prepared, these solutions were periodically evaluated by dispensing with an 8-μL calibrated syringe during the 28-day testing period. A sample solution of 20% glycerol in water (v/v) was prepared with 3.5189 g stock solution 1 (aλred = 742:0406), 8.6293 g glycerol, and 25.0169 g deionized water. The resulting solution contained 9.879% stock solution 1 (v/v) and 19.344% glycerol (v/v). A sample solution of ~50% ethanol in water (v/v) was prepared with 4.8085 g stock solution 1 (aλred = 742:0406), 18.8037 g ethanol, and 20.0765 g deionized water. The resulting solution contained 9.803% stock solution 1 (v/v) and 48.95% ethanol (v/v). As stated in the above section, two different calibrated syringes were used to dispense the other custom solutions. The two syringes used had slightly different calibrated values. A 7.910-μL syringe was used for the first two data points and a 7.980-μL for the last three data points (Fig. 5).
Two other custom sample solutions were also prepared and evaluated with an 8-μL calibrated syringe. The two custom sample solutions were comprised of 20% glycerol in water (v/v) and 50% ethanol in water (v/v). As shown by the relative inaccuracy, the performance did not degrade during the 28-day testing period. All relative inaccuracy performance data are within ±2.2%. The error bars represent the CV% in the measurement. The figure legend is as follows for the two custom sample solutions: 
 50% ethanol in water (v/v) and
50% ethanol in water (v/v) and  20% glycerol in water (v/v).
20% glycerol in water (v/v).
Calibrated Syringes as Volume Transfer Standards
Gravimetrically calibrated 8-μL Hamilton syringes with Chaney adapters were used as volume transfer standards. Each syringe was used to aspirate samples from a vial filled with custom sample solution followed by a dispense into a 96-well microtiter plate. It is important to note that the attached Chaney adapters were modified from the commercially available version by mechanically securing the collarplunger assembly as well as by stabilizing the plunger stop-arm. Significant internal testing using these customized Chaney adapters has shown that the modified syringes have excellent reproducibility over multiple operators, multiple locations, and multiple detection systems (internally collected data, as well as data from Fig. 4 in reference 6). All syringes are gravimetrically calibrated in an A2LA 17025 accredited test laboratory before being used as volume transfer standards. Because the reproducibility of the syringes has been proven, they can be used as volume transfer standards thereby removing device variability from the test procedure. One of the syringes was used to gravimetrically compare three solution types: pure DMSO, 90% DMSO in water (v/v), and DI water (100% aqueous). Each solution was placed in a glass vial and sealed with a septum crimp-top cap. The syringe was used to dispense 10–11 replicates of each solution onto a Mettler Toledo MT5 balance, which has incorporated draft shields and a humidified evaporation trap. Between additions of water, the evaporation was measured to be 0.002 mg after a 20 s period. For the 100% DMSO solution and 90% DMSO in water solution, the humidified evaporation trap was removed, and a lid was added to the draft shield chamber. Both DMSO and the DMSO-based custom solution gained weight between dispenses with values at 0.025 and 0.044 mg, respectively, over a 20 s period. These values were each used to adjust the individual dispense volumes for each solution, that is, the corresponding weight change due to evaporation or absorption of atmospheric water was adjusted for each sample. Table 1 reflects the corrected values for each dispense. Also, during this comparison, the temperature, pressure, and relative humidity, which were 21.2 °C, 1002 hPa, and 54.2% RH, respectively, were used to calculate a z-factor and accurately adjust the gravimetric results for atmospheric-dependent density changes of each sample.
Volume Verification of the Custom Sample Solution
Aside from the gravimetric dilution study, an aliquot of each custom sample solution was dispensed into individual wells of a microtiter plate using one of the calibrated syringes. To this aliquot, the working volume was adjusted to 200 μL in each well of the 96-well plate using a handheld, multichannel pipette. Although 384-well plates can be used, only 96-well plates were used in this study. For example, if 8 μL of custom sample solution was added to a well in a 96-well plate, 192 μL of diluent was added to create a working volume of 200 μL. The custom sample solution could be added before or after the addition of diluent. Evaporation and/or contamination of the sample is minimized by the use of microtiter plate covers, which are placed onto the plates between sample transfers. Also, the target sample solutions used for syringe work are contained in glass vials sealed to minimize evaporation. Once the working volume was achieved in each well, a plate-shaker device was used to mix the solutions, and each plate was placed into the MVS for volume determination measurements. Prior to making any absorbance measurements, information such as the solvent-of-interest's density value and the weights of the solvent-of-interest and stock solution (g) were entered into the MVS software. Some of the information, such as the stock solution's absorbance per pathlength and density values, are embedded in barcodes and passed to the software through a bar code reader. Once the information was entered into the software, the usable volume range for each solution for two plate types, 96-well or 384-well, was computed. Figure 2 shows the graphical user interface used when preparing a custom sample solution.
One item of note should be made with respect to evaporation. As opposed to gravimetric testing, evaporation of delivered sample volumes is not a large concern for MVS measurements (within reason). For the MVS approach, the sample and diluent reagents have “known” dye concentrations from which all calculations are based. In a 96-well plate filled with 200 μL of aqueous solution per well, a typical evaporation over a 15-min period is ~0.5–1 μL per well. Let us assume a 0.5 μL test sample is dispensed into a dry well of a microtiter plate and allowed to completely evaporate. When the diluent is added to the well, the dye residue is readily reconstituted into the diluent, and the total volume is lowered by 0.5 μL than expected. Thus, the error in the results is −0.25%, which is acceptable in almost all testing scenarios.
Results and Discussion
The majority of the research and evaluation for preparing custom sample solutions has been focused on the organic solvent DMSO. Because most compound management groups store their compound libraries in DMSO or a DMSO-based solution, verifying a liquid-handler's performance for delivering DMSO is of extreme importance, especially to the compound library screening community. The method of preparing a DMSO-like sample solution to check a liquid-handler's performance is critical for the subsequent interpretation of experimental results, that is, if the dispense volume of DMSO is inaccurate, there is a likely chance that the perceived concentrations of species in solution of the DMSO-based assay (targets, antibodies, antigens, etc.) will also be inaccurate, potentially leading to false-positive or false-negative interpretations.
Three DMSO-based sample solutions, one in each group (D, C, B), were prepared and subsequently diluted with aqueous diluent to create 17 batch volumes of 60 mL. By way of gravimetric dilution, each batch volume prepared was equal to a specific target volume. For instance, one batch volume of a DMSO-based sample solution had a gravimetrically determined target volume of 4.178 μL and another had a target volume of 7.353 μL. Each of the 17 solutions was dispensed into an entire microtiter plate, for example, 17 plates were prepared and tested. Each of these plates produced a data point for comparing the MVS output results versus the gravimetrically determined target volume. The large-volume dilution method is an accurate way of comparing gravimetric and photometric methods, especially at low-volume levels, and proves that DMSO-based sample solutions can be used to assess liquid-handler performance for DMSO. Figure 3 shows the relative inaccuracy % for each of the three groups of DMSO-based sample solutions, which covered a total volume range of 0.5–44.7 μL. All of the relative inaccuracy data correlating gravimetry to the MVS for these DMSO-based sample solutions are within ±2.5%.
It should be reiterated that each of the three groups of solutions is prepared with different chemistries and therefore, each group does not necessarily follow a linear response with the gravimetrically determined target volume (Fig. 3). The different MVS stock solutions are used to offer more, or less, dye for each different preparation (see Materials and Methods). The slight trend for the group B solutions (10.847–44.7 μL) might be the result of the varying ratio between two dye types. As a reminder, the MVS method is based on a sample solution with a red dye and a blue dye, as well as a diluent with blue dye at the same concentration as in the sample solution. The current approach deviates from the MVS method in that the concentration of blue dye present in the custom sample solution can be significantly lower than that in the diluent. This does not pose an issue for small test volumes (< 10 μL) as the bulk of the total test volume is made up of diluent. In the 10–50 μL range, however, more blue dye comes from stock solution 2 and less comes from the diluent. Because the second dye is first being diluted in the solvent (DMSO) and less blue dye comes from the diluent, this seems to be causing a downward trend as the target volume increases. Due to this fact, the current form of this test method does not allow for testing volumes above 50 μL.
Two DMSO-based sample solutions were also used to monitor solution stability over time. Using a calibrated 8-μL Hamilton syringe with modified Chaney adapter, the solutions were tested in replicates of eight on each day of testing. Figure 4 shows the stability in the performance of both solutions. As measured by relative inaccuracy, which shows all data to be within ±0.7%, the performance did not degrade during the 41-day testing period. It is important to note that the calibrated syringes can be used to dispense multiple types of solutions. Table 1 shows a gravimetrically determined comparison for replicate dispenses on a balance for three different solution types. Regardless of the solution type used, the calculated mean volumes for the other two solution types are within 0.13% of water and therefore, the syringes can be used as transfer standards for these various solution types.
The discussed methodology allows for volume-verifying a solution with similar flow characteristics and physical properties as the original solvent-of-interest. For instance, the group C DMSO-based sample solutions are 90% DMSO (v/v) and the measured density value of 1.1029 g/mL is near identical to that of pure DMSO (1.1 g/mL). These prepared sample solutions also show reasonable stability over time, which could mean that the methodology becomes a little easier to implement because the sample solutions have to be prepared less often for routine liquid-handler performance checks.
In addition to the DMSO-based sample solutions, two other custom sample solutions were briefly evaluated. A 20% glycerol in water (v/v) and a 50% ethanol in water (v/v) were individually prepared as custom sample solutions and dispensed using one of the 8-μL calibrated syringes. Figure 5 shows the stability in the measurement over the 28-day period and all relative inaccuracy data are within ±2.2%. These results indicate that it might be possible for individual users to use this methodology to assess device performance for specific or proprietary solutions for critical volume transfers at assay-specific volumes.
Conclusions
The results presented herein indicate the ability to use a custom sample solution with the MVS to understand liquid-handler “behavior”. This methodology also allows programming and troubleshooting of automated aspirate/dispense method parameters for specific test liquids at critical target volumes. The flow characteristics of the resulting sample solution correspond to the flow characteristics of the solvent-of-interest, such as DMSO. The method is robust, rapid, and easy to use in the laboratory by in-house personnel so that the volumetric performance of the device can be verified frequently and with minimal device downtime. Although preparing a custom sample solution for volume verifications could conceivably work with any liquid type, the approach does have some foreseeable limitations, that is, although this method conceivably works for any solution type, the current model may not allow for verifying a solution made from a solvent, which is highly immiscible with water. The method does not permit the testing of 100% organic solvent because the aqueous-based dye stock component must be added to, and thereby diluting, the test liquid. The aqueous-based stock-dye solution should also be miscible with the solvent-of-interest, and the absorbance characteristics of the dyes should not be significantly changed or altered. When diluted in the solvent-of-interest, the dyes in the stock-dye solutions must be relatively stable and should not precipitate. Also, the density value of the solvent-of-interest must be known or measured, and this solution should not contain components in solution that significantly absorb light in the dye's spectral window. It is important to note that it may be necessary to conduct trial-and-error evaluations of mix ratios for compatibility of the solvent-of-interest with a selected stock solution for a desired volume range.
The methods discussed are focused toward quality control for liquid delivery, for all reagents and all assays. One of the main goals of this study is to emphasize the importance of liquid delivery for the proper interpretation of experimental results. A viable approach has been shown to enable the quantification of small volumes of varied solution types. An important advantage of this method is that it can be potentially used for various reagents and reagent mixtures, such as 90% DMSO in water (v/v), 50% ethanol in water (v/v), or 20% glycerol in water (v/v). It is thought that, if implemented correctly, this method can be an advantage over other volume-verification methods because it is a complete system with characterized components, methods, and reagents. The user, therefore, need not worry about preparing stock solutions, accurately weighing powder on a balance, filtering impurities/nonsolubles, measuring the quality or shelf-life of the dye stock, preparing solutions for “standard curve generation”, or using a calibrated pipettor to dispense the standard solution. The user also does not have to worry about using a proper five or six place balance with environmental controls, draft shields, and evaporation traps to weigh low-volume targets. This method is also rapid because there is a simultaneous measurement for both accuracy and precision data for each independent pipettor channel. It must be pointed out, however, that although this system does have inherent advantages, other volume-verification methods such as single-dye approaches or gravimetry, may be just as reliable if they are correctly implemented.