Abstract
Methods for the rapid determination of the enantiomeric excess (ee%) of organic substrates, especially for HTS, are often the “bottleneck” in a process. For this purpose, a new process of entirely automated sample preparation and the determination of ee% using electrospray ionization-mass spectrometry (ESI-MS) has been developed. Various substrates and new auxiliaries were explored to enhance the methodical scope. In combination with a very versatile liquid-handling system (HTS-PAL) and a comprehensive processing equipment, a multitude of standardized reaction vessels can be managed with the presented system. As an example of use, the ee% determination of I-phenylethanole via ESI-MS is compared to state-of-the-art GC analysis. In addition, a HTS suitable data processing network was constructed that allows postrun data manipulation and the automated data transfer to analysis and visualization templates with a maximum amount of automation.
Introduction
The basic necessity for high-throughput screening (HTS) systems to analyze large quantities of samples is a crucial problem when investigating enantioselective reactions or transformations with the need to determine the enantiomeric excess (ee%) of the desired product. With conventional analysis via CE, GC, or HPLC it is often difficult to fulfill these requirements. 1 Electrospray ionization-mass spectrometry (ESI-MS) is a suitable method for HTS applications, and a multitude of different techniques are estabilished.2-10 In general, MS is not a competent technique to directly analyze chirality but is an attractive option to be applied in high-throughput application techniques. 11 That MS enables the detection and specification of masses of desired targets in a parallel manner without interference, creates the basis for a fast determination of ee% via parallel kinetic resolution with mass-tagged auxiliaries.12-14 To take full advantage of this methodology for a high-throughput capable system, it is necessary to combine the latter with a very versatile and easily adaptable sample processing system. 15 Furthermore, to avoid the cause of a new bottleneck, all measurement data should be processed with a minimum of manual labor.
Experimental Details
Experimental
All reactions were carried out in 96-well microtiter plates (MTP) (200 μL) or screw cap vials (2 mL) under standard reaction conditions. Starting materials and solvents were used as received from commercial suppliers. Proline derivatives
Analysis Parameters
ESI-MS experiments were performed on Agilent 1946 VL (single quadrupole) MS with ChemStation Software Rev. 10.02 (Agilent, Waldbronn, Germany). ESI conditions (positive ion mode) are as follows: nebulizer and drying gas, purified nitrogen; nebulizer pressure, 30 psig; drying gas flow, 10 L/min; drying gas temperature, 250 °C; and capillary voltage, 4 kV. For a multipurpose analysis, all product ions can be detected in SCAN mode (50-1000 m/z), or in selected ion mode for an enhanced sensitivity. Eluent (MeOH) was provided by Agilent 1100 Series HPLC binary pump with a flow rate of 0.5 mL/min.
Determination Principle
ee% determination of a substrate with ESI-MS is based on a parallel kinetic resolution with pseudoenantiomeric, mass-tagged, chiral derivatization reagents. The mass of such an auxiliary is directly related with its absolute configuration. In general, under suitable reaction conditions, each enantio-mer of a desired chiral substrate will react with both pseudoenantiomeric auxiliaries. However, each set of reactions, depending on matched or mismatched case, has a different reaction rate (see Figs. 1 and 2).
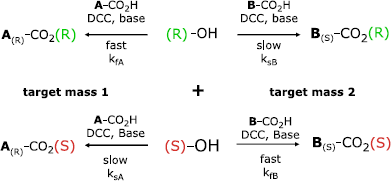
The relative rates of the competing fast and slow reactions of pseudoenantiomeric, mass-tagged, R- and S-auxiliaries with the substrate characterize the kinetic resolution selectivity.
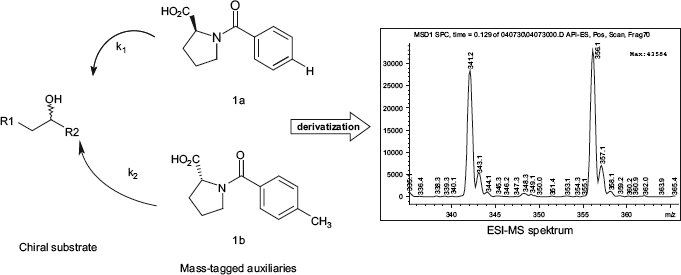
General scheme for the reaction of chiral alcohols with chiral, mass-tagged acids in the presence of DCC and a base.
This kinetic resolution selectivity, defined as the relative rates of the competing fast and slow reactions of the mass-tagged enantiopure R- and S-auxiliaries with the substrate, was recorded in a defined calibration procedure. With this calibration at hand, the analysis of an authentic sample, derivatized under identical reaction conditions, yields the enantiomeric excess. All measured calibrations consist of three or five different ee% values, normally 100 ee% S-enantiomer, 50 ee% S-enantiomer, racemate, 50 ee% R-enantiomer, and 100 ee% R-enantiomer. A smaller subdivision of the measurement range is not essential.
Automation
The embedded liquid-handling system for diluting, dosing, injecting, and transferring of the samples is based on a HTS-PAL from CTC Analytics AG (Switzerland), which allows temperature-controlled storage, a fast injection cycle time, barcode reader, and can be adapted to any major LC-MS system (see Fig. 3). A self-made conversion of an agitator accessory into a temperature-controlled well-plate shaker enables a well defined and thus reproducible sample processing. Sample processing was executed as described above. After injection by means of the HTS-PAL injection valve, the sample is transferred via peek capillary into the spray chamber of the connected MS. Raw data were analyzed with the ChemStation software and transmitted to the analysis and visualization program.
CTC setup for automated derivatization.
Results and Discussion
New Chiral Auxiliaries
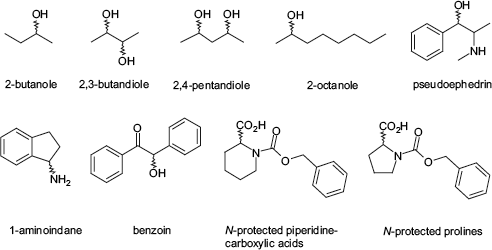
For the expansion of the applicable derivatization reagents, new commercially available chiral auxiliaries were introduced. Namely two N-protected amino acid derivatives, N-Z-proline and N-Z-piperidine-2-caboxylic acid, and two alcohols, 2-heptanole and 2-octanole, were examined in detail (see Fig. 4).
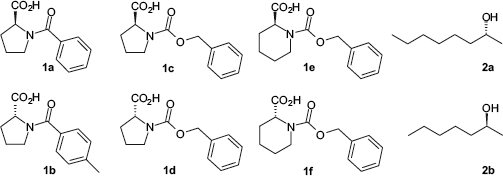
Set of chiral, mass-tagged auxiliaries for the parallel kinetic resolution.
A derivatization of amino acid derivatives with the selected enantiopure 2-alcohols
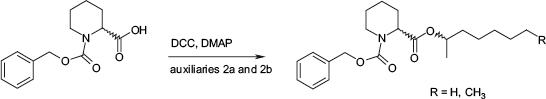
Reaction scheme for the derivatization with alcohols
On the other hand, the N-protected amino acid derivatives
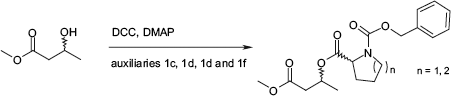
Reactions of pseudoenantiomeric N-benzoyloxycarbonyl amino acid auxiliaries with l-phenylethanole.
Figure 7 documents the measurement results of the consistency check for all possible combinations of the enantiopure derivatization reagents.
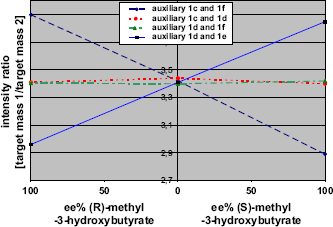
Selectivity calibration curve for methyl-3-hydroxybuty-rate with derivatization reagents
The compliance of the four major demands for a successful and reliable ee% determination with the presented methodology is clearly evident:
a reliable calibration curve; matching of all curves at the same y-axis value; no selectivity for the auxiliary combinations with identi cal absolute configuration; and auxiliary combinations with reverse set of absolute con figuration leading to the reverse selectivity.
On the basis of the shown data, the implementation of the new pseudoenantiomeric auxiliary combinations is indemnified.
New Representative Substrates
To expand the applicability of the ee% determination via ESI-MS, a number of new substrates were tested.
For all substrates shown in Figure 8, calibrations were recorded and the applicability and reliability for an automatized derivatization with pseudoenantiomeric, mass-tagged auxiliaries was proved. As mentioned before, some of the substrates themselves can be used as chiral auxiliaries.
New substrates for the ee% determination using ESI-MS.
Automated Processing
The analytical process can be divided into three parts regarding automation capabilities: sample preparation, measurement, and data processing.
The sample preparation for the derivatization reactions is exceptionally straightforward. With the HTS-PAL system, samples can be fed in any 96-well MTP type or other vessel with normal injection facility. The remote control CTC software Cycle Composer allows a very flexible programming of sample lists, and methods and macros for liquid-handling sequences.
A multitude of different trays and stacks, dilutors, solvent reservoirs, and wash stations can be joined. Because of the use of well-prepared stock solutions and the inherent stability of the method toward small deviations from optimum reaction mixture consistence, the use of a time-consuming highly precise liquid-handling system is not decisive. Nevertheless, the use of syringes required washing cycles between each dosing step. Through this, liquid-handling procedures were slightly prolonged.
All LC-MS system parameters were controlled by the ChemStation software, and the required CTC Cycle Composer software can be fully integrated in the LC-MS data acquisition systems.
An HTS suitable data processing was achieved by a self-made macro for ChemStation applications. After raw data acquisition, the offline postrun macro performs the extraction of the required MS data and generates an excel data file on the basis of a preselected analysis and visualization template.
The Applied System
To demonstrate the applicability of the whole automated system, the ee% determination of 1-phenylethanole with the auxiliaries
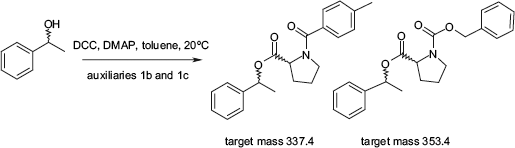
Derivatization reaction of l-phenylethanole with pseudoenantiomeric auxiliaries
A reaction time of 1 h was found to be sufficient for the completion of the desired derivatization reaction. Together with a sample processing time of 3 min and an estimated measurement time of 1 min for each sample, the overall measurement time for 96 samples amounts approximately to 8 h.
In comparison, the analysis via GC took 14 h with an included triflouroacetic acid derivatization step, or 28 h for the analysis of the unprotected substrate. Therewith, the average measurement duration can be shortened by a factor of two to four times.
The comparison between Figures 10 and 11 shows the exceptional conformity between ESI-MS and GC analyses. GC analysis is commonly known for its accuracy and low relative standard deviation concerning ee% determination. The GC measurements performed resulted in deviations lower than 2% for the ee% value correlated with the theoretical value as well as the RSD within a series of measurements. For the ESI-MS measurements, the RSD within a series was < 3% and the deviations for the correlation with the theoretical ee% values were lower than 3%, except for two series with 6% and 8% deviation, respectively.
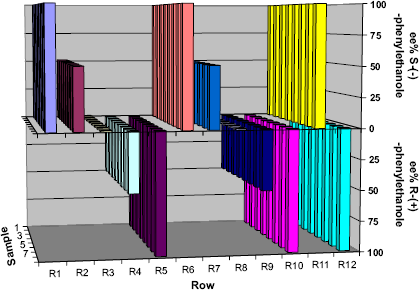
ee% determination of underivatized I-phenylethanole via GC.
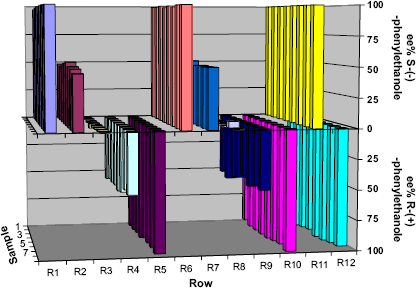
ee% determination via ESI-MS after parallel kinetic resolution with pseudoenantiomeric auxiliaries
All obtained measurement data were processed offline via ChemStation macro and directly transferred into customized analysis and visualization templates. The time effort is in the range of several minutes and can be neglected.
Conclusions
For this methodology, a large variety of acids, alcohols, amides, amines, and amino acid derivatives are applicable and could be used as substrate or auxiliary. In combination with standardized reaction vessels and a very versatile and precise sample preparation system (HTS-PAL), the measurement duration can be averagely shortened by a factor of 2-10. Furthermore, because of the automated preparation, the measurement error can be reduced as well. Further developments to shorten sample preparation times are still under progress.
Footnotes
Acknowledgment
The project was supported by the Federal Ministry of Education and Research and the Ministry of Commerce Mecklenburg-Vorpommern.
