Abstract
Low-volume liquid handling capabilities in bioanalytical workflows can dramatically improve sample processing efficiency and reduce reagent costs, yet many commercial nanoliter liquid handlers cost tens of thousands of dollars or more. We have successfully adapted a low-cost and open-source commercial pipetting robot, the Opentrons OT-1, to accurately aspirate and dispense nanoliter volumes. Based on fluorescence measurements, the modified OT-1 was able to reproducibly transfer 50 nL of water with less than 3% measurement error and 5% coefficient of variation (CV). For 15 nL transfers, the volume measurements indicated less than 4% error and 4% CV. We applied this platform to the preparation of low-nanogram proteomic samples for liquid chromatography–mass spectrometry analysis, demonstrating that the modified OT-1 is an effective platform for nanoliter liquid handling. At a total materials cost of less than $6000, including the commercial liquid handler and all modifications, this system is also far less expensive than other platforms with similar capabilities, placing automated nanoliter handling within reach of a far broader scientific community.
Introduction
Ongoing improvements to bioanalytical instrumentation are leading to ever-decreasing detection limits and sample input requirements that power advances in high-throughput screening, single-cell analyses, and the mapping of biochemical expression across tissues with high spatial resolution.1–3 However, to fully realize the benefits of these analytical developments, concomitant improvements in up-front sample handling approaches are required. For example, reducing sample amounts without also paring preparation volumes can lead to an increased portion of the sample being lost to the surfaces of containers (e.g., well plates) due to nonspecific adsorption.4–7 Similarly, the low resulting sample concentrations may experience decreased reaction rates for enzymatic and chemical reactions during processing. 8 Reducing sample-processing volumes can alleviate these issues and also save costs when precious reagents are used. To address this need, an impressive array of automated liquid-handling systems capable of nanoliter dispensing have emerged in recent years.9–12 Some systems, such as the Beckman Coulter Echo series acoustic dispensers, provide high throughput and eliminate the possibility of cross-contamination through contactless dispensing.13,14 However, these commercial systems often cost tens of thousands of dollars to >$100,000, putting them out of reach of many laboratories. In addition, many solutions exist for nanoliter dispensing, but few can retrieve nanoliter volumes for transfer to nonoptical detection platforms such as liquid chromatography–mass spectrometry (LC-MS).
We recently developed a workflow termed nanoPOTS (Nanodroplet Processing in One pot for Trace Samples), 15 which miniaturizes sample preparation volumes to the nanoliter scale for highly reproducible, quantitative, and in-depth proteome profiling at16–20 and near15,21–23 the single-cell level. A home-built robotic liquid handler combines a three-axis translation stage, high-resolution optics, and an integrated syringe pump. The system can dispense and withdraw liquids with subnanoliter volume control, submicrometer positional accuracy, and cross-contamination of 0.003% or less. 24 Instead of interfacing with conventional well plates or vials, samples are prepared on glass slides that have been photolithographically patterned with hydrophilic spots. Samples and reagents are retained by a hydrophobic boundary that surrounds each nanowell, and the potentially adsorptive surface to which a sample is exposed is reduced to <1 mm2. 15 As a result of the reduced adsorptive losses and enhanced reaction kinetics afforded by the miniaturized sample processing, in-depth and label-free proteomic analysis of single mammalian cells is now feasible.16–20 Moreover, a larger number of proteins can now be identified and quantified from trace samples in a label-free workflow than was previously possible for samples that contain 500 times more total protein. 25 However, as with commercial nanoliter liquid handlers, this custom robotic platform is costly to construct at >$50,000 and in addition requires substantial engineering expertise. These obstacles significantly limit the dissemination of nanoPOTS and related technologies to other laboratories.
To address the high cost of automated liquid handling, the startup company Opentrons (Brooklyn, NY) recently introduced the OT-1 and subsequently the OT-2 systems. 26 These are low-cost, open-source solutions for automated pipetting but are limited to microliter volumes and therefore insufficient for applications such as processing trace samples for proteome profiling. Here, we have sought to dramatically reduce the cost and complexity of nanoliter pipetting by modifying the low-cost commercial OT-1 platform. Using three-dimensional (3D)–printed parts, we modified the OT-1 to accommodate a 10 µL syringe in place of the stock pipette, thus extending the pipetting capabilities to the nanoliter range. The built-in stepper motor that serves as the “thumb” to actuate the pipette piston on the standard OT-1 now serves as a syringe pump to dispense and withdraw liquids using the syringe. This modification enables reproducible pipetting of volumes as small as 15 nL. The modified platform also has sufficient positional accuracy to reproducibly address microfabricated nanowells in a nanoPOTS chip and can be used to prepare low-nanogram samples for proteomic analyses similar to the in-house-built robot. The modified OT-1 is thus an effective platform for nanoliter liquid handling and, at a total materials cost of less than $6000, is substantially less expensive than commercial platforms while offering improved precision in the nanoliter range.
Materials and Methods
Reagents and Chemicals
Deionized water was purified with a Millipore Milli-Q RG water purification system (Darmstadt, Germany). Dithiothreitol and iodoacetamide were purchased from Thermo Fisher Scientific (Waltham, MA) and freshly prepared in 50 mM ammonium bicarbonate buffer each day before use. MS-grade trypsin and Lys-C were products of Promega (Madison, WI). Unless otherwise mentioned, all other reagents were obtained from Sigma-Aldrich (St. Louis, MO).
Modification of the Opentrons OT-1 Liquid-Handling System
An Opentrons OT-1 liquid-handling system was modified for nanoliter pipetting, as shown in
Figure 1
. The pipettes that were included with the OT-1 system were removed, and a 10 µL Gastight syringe (Hamilton, Reno, NV) was mounted in place of one of those pipettes using 3D-printed adapters (

Opentrons OT-1 platform before and after modification. (
The Opentrons API was replaced with a home-built LabView program (National Instruments), which was used to synchronously control the movement of the 3D stages and the liquid dispensing of the syringe. The LabView program offers thorough user control over settings and actions available to the robot.
Measurement of Liquid-Handling Accuracy and Precision
Characterization of pipetting performance was primarily accomplished by aspirating a plug of liquid into a 200 µm i.d. fused silica capillary (

Dispensing 50 nL volumes into 1-mm-diameter nanowells. (
A fluorescence-based method served as an orthogonal means to characterize liquid-handling performance. To generate a calibration curve, sodium fluorescein standard solutions were created by diluting 10 mM stock solutions to 2 to 10 µM with 50 mM sodium borate buffer (pH 9.3). A handheld pipette was used to transfer five replicate 10-µL aliquots of each solution to a 384-well plate (Corning 3544, Corning, NY). For each test volume, 10 wells were first filled with 10 µL of buffer. The modified OT-1 was programmed to dispense fluorescein into the buffer-filled wells to produce final concentrations of 6 µM fluorescein in each test well. Fluorescence readings were taken using a Biotek Synergy H4 Hybrid Microplate Reader (Winooski, VT). The excitation and emission wavelengths were set to 485/13.5 and 528/17.0, and sensitivity was set to 120. Accuracy for each dispensed volume was determined by comparing fluorescence intensity from the test solutions with the calibration curve; reproducibility was determined from the fluorescence intensity of replicate measurements.
Confidence intervals for the volume measurements were computed by bootstrap resampling in R using the boot and boot.ci functions (boot package v1.3-20 with the following settings: first-order normal approximation, ordinary simulation, 1000 replicates, confidence level = 0.95).
Cell Culture and Proteomic Sample Preparation
HeLa cells (ATCC, Manassas, VA) were grown in Dulbecco’s Modified Eagle’s Medium (Mediatech, Inc., Manassas, VA) supplemented with 10% fetal bovine serum and 1× penicillin/streptomycin at 37 °C with 5% CO2. Cells were prepared as described previously 15 and counted using a TC20 Automated Cell Counter (Bio Rad Laboratories, Hercules, CA). Dispensing and sample collection were performed using the OT-1 system according to the previously published nanoPOTS protocol, 15 which consists of a single step for cell lysis, protein extraction, and reduction followed by dispensing and incubation steps for alkylation, Lys-C digestion, trypsin digestion, and acid quenching. This was designated as the standard protocol and has been detailed elsewhere. 19 A simplified protocol was then adapted from the standard protocol by removing the alkylation and Lys-C digestion steps and reducing trypsin digestion from overnight to 4 h for an overall sample preparation time of ~6 h instead of ~18 h.
LC-MS Setup
The LC-MS analyses were similar to those of previous studies.15,16 Briefly, the solid-phase extraction (SPE) precolumn and LC column were slurry packed with 3 µm C18 packing material (300-Å pore size, Phenomenex, Torrance, CA). The SPE column was prepared from a 5-cm-long fused silica capillary (75 µm i.d., 360 µm o.d., Polymicro Technologies). The LC column was prepared from a 50-cm-long Self-Pack PicoFrit column with an i.d. of 30 µm and a tip diameter of 10 µm (New Objective). The sample storage capillary was connected to the SPE column with a Valco Nanovolume union (Houston, TX) having a 100 µm bore. The sample was loaded and desalted in the SPE precolumn by infusing Mobile Phase A (0.1% formic acid in water) at a flow rate of ~320 nL/min by connecting to the split-flow line of the LC pump (see below) for 10 min. The SPE precolumn was then connected to the head of the LC column using Valco Nanovolume unions. The LC separation flow rate was ~50 nL/min, which was split from 350 nL/min with an UltiMate 3000 RSLCnano pump (Thermo Fisher). A linear 95-min gradient of 5% to 25% Mobile Phase B (0.1% formic acid in acetonitrile) was used for separation. The LC column was washed by ramping Mobile Phase B to 90% over 5 min and holding at 90% for another 5 min. The column was then reequilibrated with Mobile Phase A for 20 min prior to the subsequent analysis.
A Q Exactive HF-X mass spectrometer (Thermo Fisher) was employed for all data collection. An electrospray voltage of 1.9 kV was applied at the source, the ion transfer tube was set at 250 °C for desolvation, and the ion funnel radio frequency level was set at 45. A full MS scan range of 375 to 1800 m/z and Orbitrap resolution of 120,000 (at m/z 200) were used for all samples. The automatic gain control (AGC) target and maximum injection time were set at 1E6 and 50 ms. Data-dependent acquisition mode was used to trigger precursor isolation and sequencing. Precursor ions of +2 to +6 charge states were isolated with an m/z window of 2 and fragmented by higher energy collision-induced dissociation with a normalized collision energy of 29%. The signal intensity threshold was set at 8000. To minimize repeated sequencing, dynamic exclusion with a duration of 30 s and mass tolerance of ±10 ppm was used. MS/MS scans were performed in the Orbitrap on the 10 most intense peaks. The AGC target was 1E5, and MS2 resolution was 60,000 with a maximum injection time of 240 ms. MS data analysis was performed using MaxQuant as described previously. 19
Results and Discussion
Commercial nanoliter liquid-handling systems are prohibitively expensive for many laboratories, and few provide both dispensing and aspiration capabilities with high precision in the nanoliter range. We sought to modify the low-cost Opentrons OT-1 automated liquid-handling platform for nanoliter pipetting and demonstrate its suitability for preparation of trace proteomic samples. There are, of course, many additional applications that should likewise benefit from similar nanopipetting capabilities. Specific design requirements that we pursued in the current study were (1) sufficient positional accuracy to reproducibly address 1-mm-diameter nanowells, (2) precise dispensing and aspiration of nanoliter volumes within a humidified environment to minimize evaporation, and (3) a total materials cost of <$10,000. Each of these objectives has been achieved as described below.
Key modifications of the OT-1 robot included construction of a chamber around the entire platform, as shown in
Nanoliter pipetting may require more accurate positioning of the dispensing tip than a typical liquid handler interfacing with, for example, 96-well plates. To be compatible with the nanoPOTS workflow, the OT-1 needs to dispense 50 nL of liquid consistently and accurately into 1-mm-diameter nanowells. This requires sub-millimeter lateral positioning accuracy across an array of wells patterned on a glass slide, as well as sub-100-µm accuracy in the vertical direction to ensure that the pipetted liquid contacts the nanowell surface rather than wicking up the exterior wall of the dispensing tip. We calibrated the capillary tip to the nanowell chip surface by manually aligning the tip to fiducial marks on the chip to define the plane of the chip, 15 after which liquid handling and tip positioning were controlled in the software. After calibration, we programmed the OT-1 to dispense 50 nL of water (tinted with red food dye for easier visualization) into eighteen 1.0-mm-diameter wells. As shown in Figure 2 , the capillary tip successfully interfaced with each nanowell across the array, indicating that the positional accuracy of the OT-1 was sufficient to accommodate the nanoPOTS workflow.
We first evaluated the liquid-handling capabilities of the modified OT-1 by aspirating programmed volumes of 50 nL of water into a glass capillary and measuring the length of the resulting plugs under a microscope, as described in the Materials and Methods section. The resulting observed volumes averaged 49.9 nL with a 95% confidence interval (CI) of 49.39 to 50.40 nL (
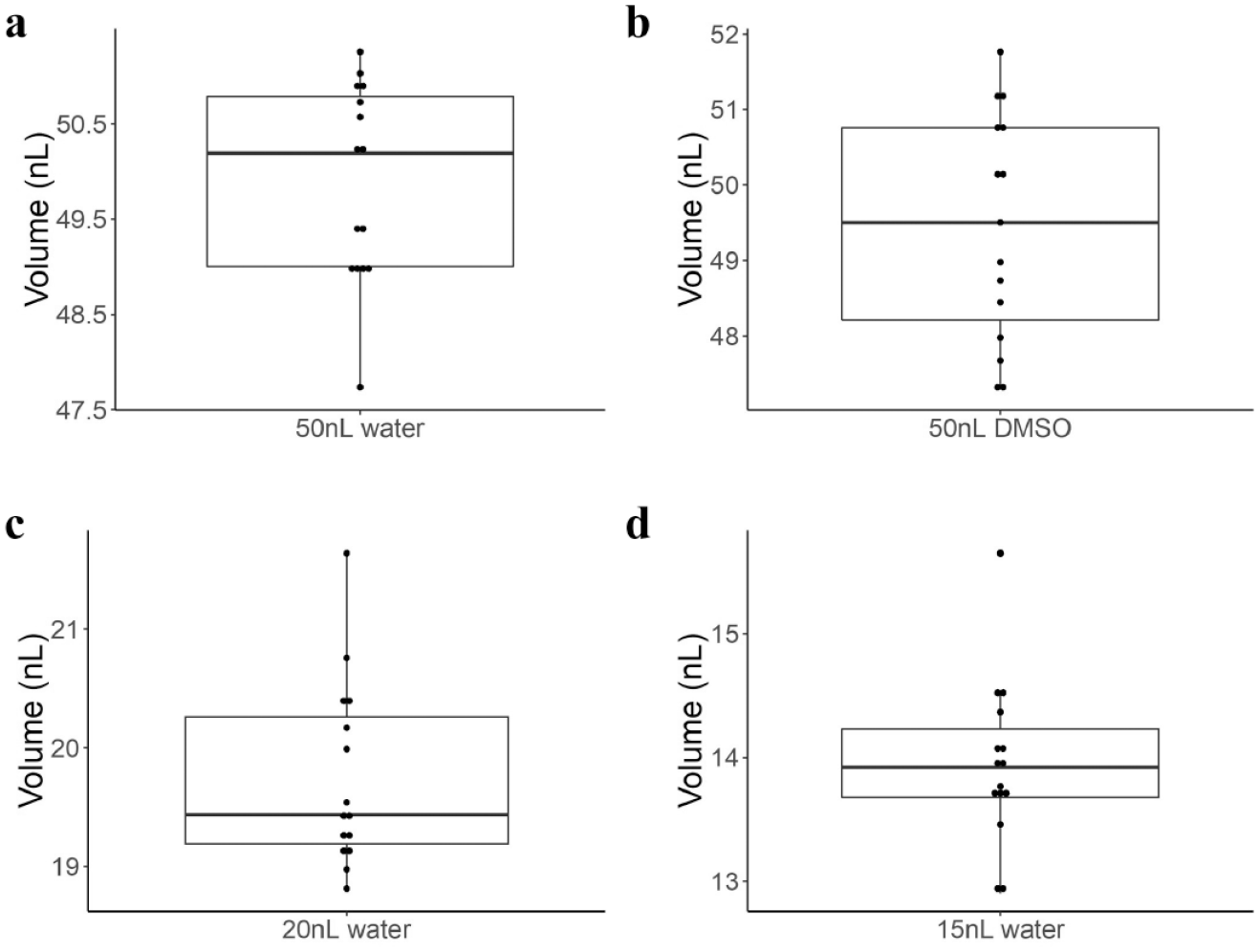
Evaluation of nanoliter pipetting accuracy for programmed volumes of (
However, as the error at 15 nL could potentially be an artifact of our limited ability to accurately measure shorter droplet plug lengths, we also evaluated the pipetting performance by dispensing programmed volumes of sodium fluorescein of known concentration into 10 µL buffer solutions. We programed the modified OT-1 system to add either 50, 20, or 15 nL of fluorescein solutions at concentrations of 1.4, 3.4, and 3.8 mM, respectively, to wells containing 10 µL of buffer to produce final concentrations of ~6.0 µM. The well plate was centrifuged, and fluorescence readings were made using a plate reader. Using this method, average values of 51.4 nL (95% CI: 50.9 to 51.9 nL), 20.0 nL (95% CI: 19.9 to 20.2 nL), 15.5 nL (95% CI: 15.4 to 15.6 nL) were observed for the 50, 20, and 15 nL trials, respectively. These correspond to measurement errors of 2.8% for 50 nL, 0% for 20 nL, and 3.4% for 15 nL volumes. The calculated CVs were 5.2% (95% CI: 4.6 to 5.9%), 3.1% (95% CI: 2.7 to 3.5%), and 4.4% (95% CI: 3.0 to 5.9%), respectively, for 100 replicate measurements of each volume. These orthogonal measurements provide further confirmation of the accuracy and precision of the modified OT-1 system. The pipetting precision of the modified OT-1 compares favorably with commercial offerings. For example, SPT Labtech (Melbourn, UK) reports a CV of 7.5% for 50 nL volumes for its low-volume Mosquito X1 and Mosquito LV products, 29 and the Tecan Freedom EVO (Männedorf, Switzerland) liquid-handling workstation reports a 5.2% CV for 100 nL, 30 which was the smallest volume reported. The Beckman Coulter Echo 650 Series reports an impressive 2.5 nL transfer capability with a <10% deviation from target volume and a CV of <8%. 31
The 50 nL pipetting steps currently employed for preparation of many trace proteomic samples in the nanoPOTS method is well within the capabilities of the platform. Images captured on a microscope of liquid dispensed in 50 nL increments into nanowells (

Comparison of pipetted volumes using the in-house-built nanoliter pipettor (Custom) and the modified Opentrons liquid handler (OT-1). Water was added to wells using each dispensing system in 50 nL increments.
For many applications, throughput can be a primary concern when selecting an automated liquid-handling system. The OT-1 is customizable, with motor speeds, fluid delivery rates, and plate/reservoir locations all adjustable by the user. This yields a large parameter space that can suit a wide variety of applications. In this work, we set the speed of the translation stages to 1000 mm/min and the fluid delivery rate to 50 nL/s. With these settings, we found that we could accurately aspirate a liquid and deliver nanoliter volumes to 10 wells in succession in a standard 384-well plate with a total time of approximately 3 min. Dispensing an aliquot of liquid and moving to the next well took ~11 seconds. The translation stages can be set to at least 4000 mm/min, and the fluid delivery rate can be set to 300 nL/s for potentially higher throughput, but pipetting performance was not determined at these higher speeds. The relatively low throughput of the platform as used here may be limiting for some uses but is not a concern for applications such as nanoPOTS, in which each downstream analysis takes hours.
The nanowell chips typically used for nanoPOTS maintain 4.5 mm on-center spacing (matching a 384-well plate) for compatibility with cell isolation platforms such as laser-capture microdissection.
22
However, other applications will benefit from higher-density well formats, and we sought to characterize the modified OT-1 for such use cases. An array of nanowells having a diameter of 1.2 mm with an interwell spacing of 0.5 mm were constructed by laser cutting adhesive tape on a glass slide. The modified OT-1 dispensed 100 nL volumes of DMSO into each well as shown in
We used the modified OT-1 robot to prepare trace proteomic samples on two glass nanowell chips: one using the standard protocol and the other using a simplified protocol (see the Materials and Methods section). Each proteomic sample comprised 68 to 72 HeLa cells. Three samples from each chip were selected for LC-MS analysis, and the raw data were analyzed as described in the Materials and Methods section. As shown in Figure 5 , the average number of identified protein groups from samples prepared using the standard and simplified protocols was 1952 and 2305, respectively, and the average number of identified peptides was 10,457 and 13,324, respectively, using standard 1% false-discovery rate filtering criteria and MS/MS identifications only (i.e., no MS1-level feature matching). The proteome coverage achieved is similar to that of previous studies, and any difference is attributed to the different MS instrumentation rather than nanoliter liquid handling. This preliminary comparison between the standard and simplified nanoPOTS protocols showed the methods to be comparable and may indicate that the added steps of alkylation and Lys-C can be eliminated and that trypsin digestion time can be significantly reduced for similar samples. Overall, the proteomics results show that the functionality of low-cost commercially available liquid-handling robots such as the OT-1 can be expanded with simple modifications to accommodate the nanoPOTS workflow.
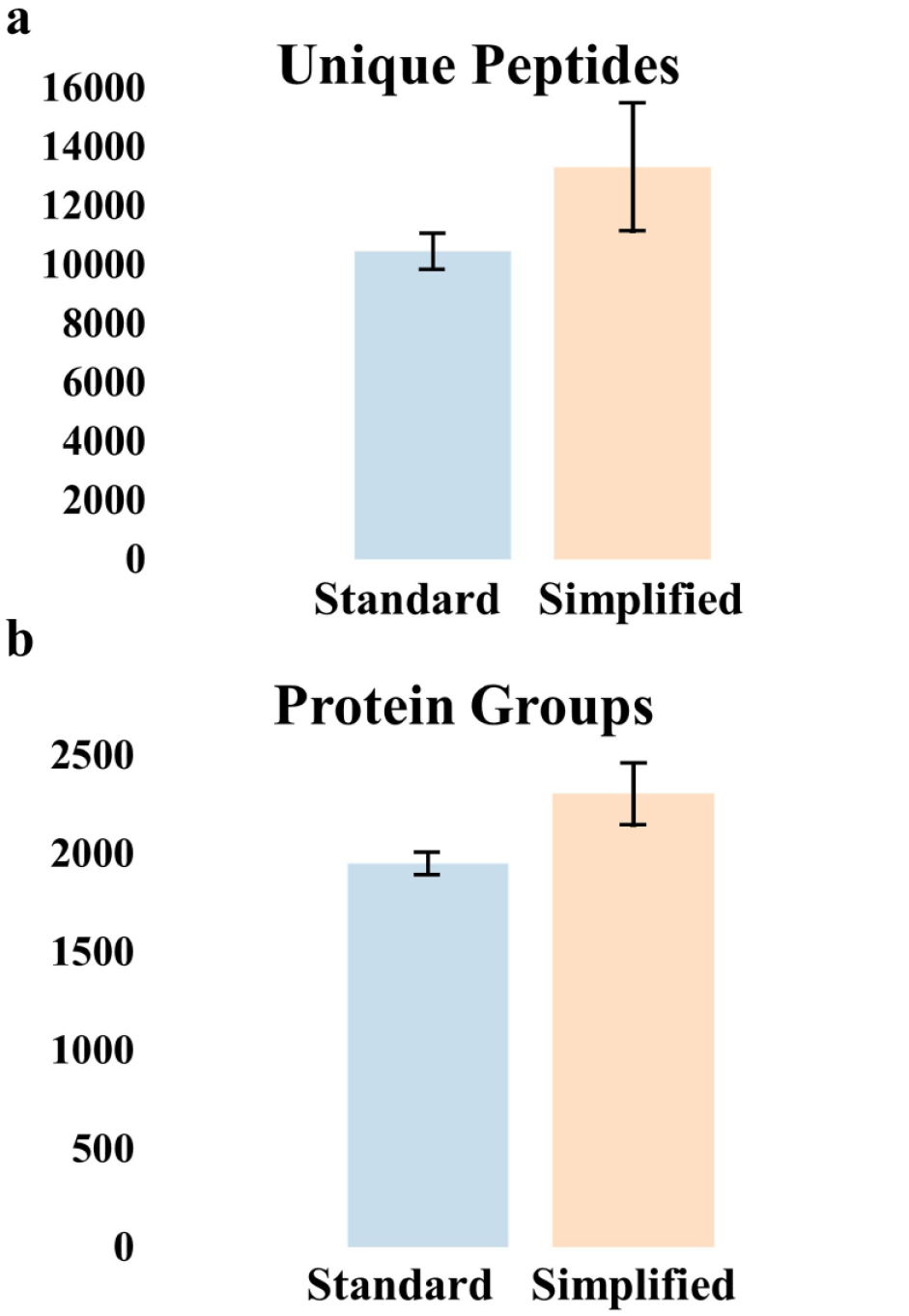
Number of identified peptides (
Conclusion
We have demonstrated that with straightforward modification, a low-cost commercial liquid-handling platform (Opentrons OT-1) can be used for highly accurate pipetting of nanoliter volumes. Given the greatly reduced cost of the modified platform relative to commercial offerings, this should put nanopipetting in reach of many laboratories that have been limited by budget restrictions and by the complexity of building comparable systems in house. This makes the preparation and analysis of trace biological samples far more economical and accessible.
Supplemental Material
sj-pdf-1-jla-10.1177_2472630320973591 – Supplemental material for Adapting a Low-Cost and Open-Source Commercial Pipetting Robot for Nanoliter Liquid Handling
Supplemental material, sj-pdf-1-jla-10.1177_2472630320973591 for Adapting a Low-Cost and Open-Source Commercial Pipetting Robot for Nanoliter Liquid Handling by E. Enoch A. W. Councill, Nathanial B. Axtell, Thy Truong, Yiran Liang, Adam L. Aposhian, Kei G. I. Webber, Ying Zhu, Yongzheng Cong, Richard H. Carson and Ryan T. Kelly in SLAS Technology
Footnotes
Acknowledgements
The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Supplemental material is available online with this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Research reported in this publication was supported by the National Institutes of Health under award numbers R33CA225248 and R01GM138931.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
