Abstract
The red blood cells (RBCs) from patients with such diseases as pulmonary hypertension (PH) and diabetes (Type I and II) have a unique physical property, namely, the deformability of their RBCs are different from those of healthy patients. For example, patients with PH and diabetes have RBCs whose deformability is less than that of healthy patients. Unfortunately for these patients, there have been very few reports relating these somewhat abnormal RBC deformabilities to a cause/effect of the aforementioned diseases. Our group believes there is a link between the physical properties of these RBCs, the chemical properties they induce, and the resultant physiological consequences of the induced chemical properties. However, characterization or verification of the physical, chemical, and physiological processes is difficult because current analytical tools do not enable all three processes to be monitored simultaneously. Here, the ability of microchip technology to monitor physical, chemical, and physiological events that may occur in each of the diseases will be discussed. These events, which are physically stimulated on-chip, are monitored both optically (chemiluminescence) and electrochemically (amperometry), and when taken collectively, become excellent in vitro models of important in vivo processes.
Introduction
Of all the automated processes, the human body and its associated physiology is one of the most well-regulated and homeostatic processes. Unfortunately, due to its complexity, the processes occurring in the human body are constantly under investigation. Moreover, attempts at altering or maintaining the appropriate physiological processes (e.g., through pharmaceutical drugs) are generally studied in conditions that do not truly mimic those found in vivo. That is, most studies involving a new drug candidate will probably occur in an in vitro system prior to animal or human subject experiments. However, such in vitro studies typically do not involve all of the conditions that would be encountered in vivo and rightly so; attempting to mimic the bloodstream, tissues, organs, etc., in a single in vitro environment is a near impossible task.
There are numerous reasons why an in vivo environment using in vitro methods is difficult. First, in vivo processes often involve the bloodstream, which is a dynamic tissue; not only in regards to its constituency, but also in the fact that it is a flowing, moving stream. Moreover, there are other types of cells other than those found in the blood that are major determinants of physiological processes. If those cells are to be incorporated into an in vitro mimic of in vivo events, proper care and maintenance of such cells will be of utmost importance and require a great deal of attention.
As daunting as it may seem, strides are already being taken towards the development of an in vitro model (on a microfluidic platform) of such complex in vivo systems as the circulatory system, 1 –3 the central nervous system, 4 and to some extent, the interface of these two important systems. One area that is providing a platform for the development of such in vivo mimics is microfluidics employing lab-on-a-chip technology. 5 –8 A microfluidic platform enables many of the properties required for the development of such in vivo mimics. For example, the continuous flow property of microfluidics allows for continuous flow of blood, or portions of blood, through channels patterned on the device. In addition, the planar platform of microfluidic devices possesses the same dimensionality of standard cell culture flasks, albeit on a much smaller scale.
Here, based on recent developments in the field of microfluidics, an attempt is made to predict the future role of automation in the field of microfluidics and how microfluidics is poised to play a major role in biomedical research. Indeed, there are many areas in which automation is already an important part of microfluidics. In order to maintain some type of focus throughout this article, the topics of automation and microfluidics will be confined to their potential in biological analyses, specifically the biological systems mentioned above (the microcirculation).
Microfluidics and the Microcirculation
One of the main roles of erythrocytes in vivo is to deliver oxygen to tissues and organs. However, a new role for the red blood cell (RBC) has been recently proposed. 9 Namely, when subjected to mechanical deformation, as would be the case in resistance vessels (arterioles and capillaries having diameters between 25–100 μM and 10–25 μM, respectively) in vivo, RBCs release micromolar amounts of adenosine triphosphate (ATP). This ATP has been shown to stimulate nitric oxide (NO) production in endothelial cells that line the inside of these resistance vessels. The importance of this NO production is that NO is one of the most potent vasodilators found in vivo, and, as such, is one of the only gaseous phase signaling molecules. 10 –12 Moreover, through its ability to stimulate NO production, RBC-derived ATP thus becomes a determinant of vascular tone and mediator of blood pressure. 13 –15
Of course, to further understand the mechanism describing the role of the RBC in controlling vascular caliber shown in Figure 1, it would be ideal to place various probes or sensors into the intact circulation and quantitatively measure ATP and NO. However, such in vivo measurements would be very difficult due to the complexity of the measurement environment and the positioning of the probe. 16 Therefore, our group has recently taken the opposite approach; that is, rather than taking the probe to the circulation, it may be more appropriate to bring the circulation to the probe.
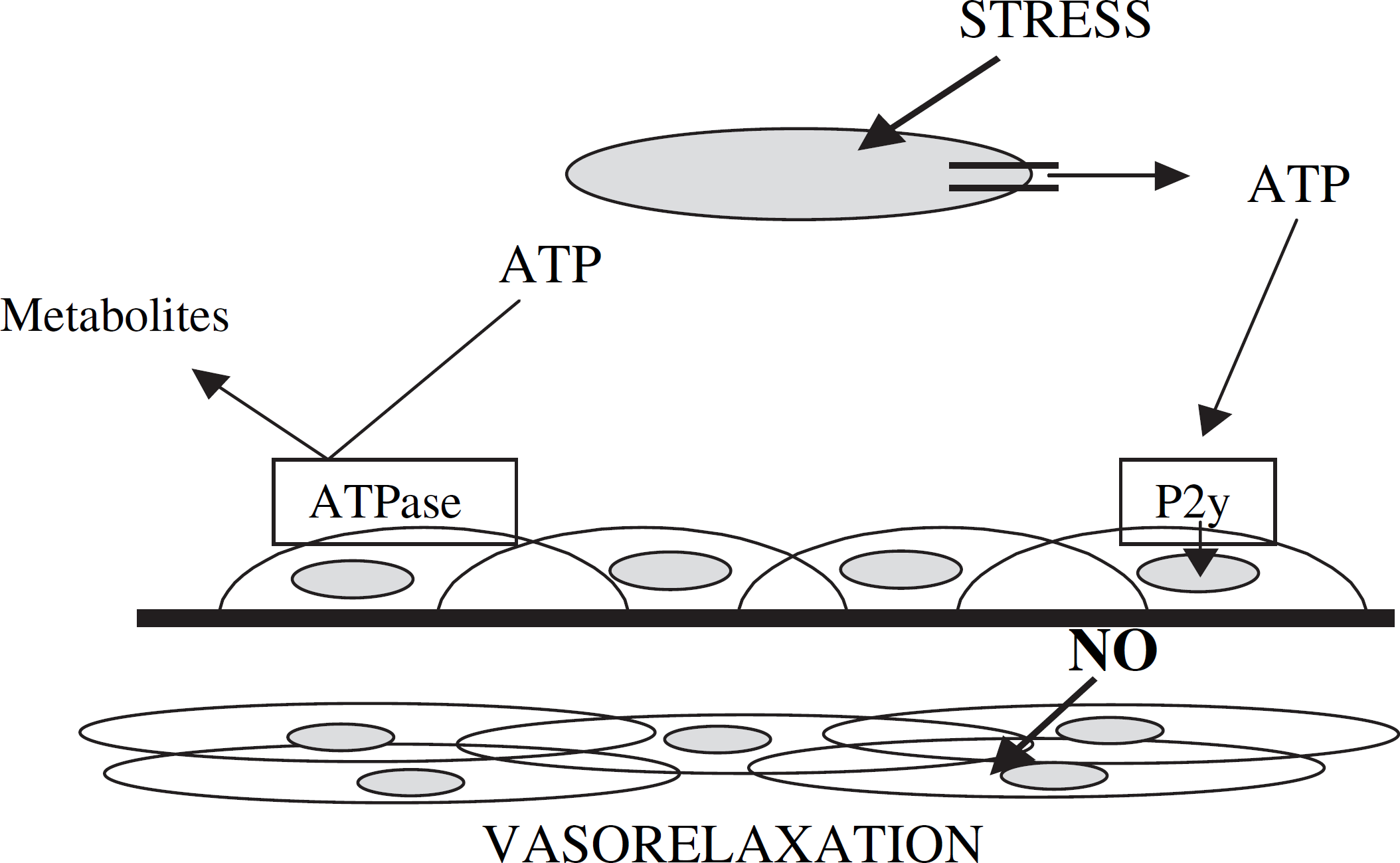
Mechanism for control of vascular caliber by RBC-derived ATP. As the RBC traverses resistance vessels in vivo, it is subjected to mechanical deformation. The deformation-induced release of ATP stimulates NO production in endothelial cells that line the circulatory vessel. In this construct, the RBC thus becomes a determinant of vascular diameter.
As an initial attempt, microfluidic devices have been patterned with channels whose diameters approximate those of resistance vessels found in vivo. 1 Specifically, microchips were fabricated using standard lithographic technology and those methods reported in the literature. 17,18 Masters were made on silicon wafers using an SU-8 photoresist and photolithographic procedures employing masks in the form of negative films. All flow designs were made using negative transparencies that were originally drawn in Freehand. The masters were cast against a mixture of poly(dimethylsiloxane) (PDMS) and cross-linking agent and allowed to cure for ∼2 h at 75°C. After completely curing, the chip was removed from the wafer and reversibly sealed over a glass plate that had access holes for introducing hydrodynamically pumped reagents and analytes.
Such devices having patterned channels of micrometer dimensions provide an excellent mimic of the microcirculation. To date, our group has employed such devices to mechanically deform RBCs as they traverse these channels. 1 Upon deformation, as mentioned above, RBCs release ATP in micromolar amounts. This ATP has been determined using a chemiluminescence assay for ATP. Due to the transparent nature of the PDMS microfluidic device, the luminescence produced during the assay can be determined by positioning the device over a photomultiplier tube. The measured light is directly proportional to the amount of ATP present in the sample (i.e., the ATP released from the RBCs).
Determination of the RBC-derived ATP is only half of the mechanism shown in Figure 1. That is, once released from the RBC, the ATP also is a stimulus for the production of endothelium-derived NO. In order to completely elucidate the exact mechanism, or to at least determine the role of RBC-derived ATP in the control of vascular caliber, NO must be quantitatively determined. The challenge here lies in the fact that the NO produced in vivo is produced by endothelial cells. Therefore, to truly learn more about the vasodilation mechanism through the production of NO, a system is required that contains endothelial cells.
Previously, our group demonstrated that an endothelium could be created on the inside wall of microbore tubing whose diameters approximated those found in resistance vessels in vivo. 19 However, the success rate for production of such endothelial cell-containing tubes was approximately 25%, because the tubes would often become brittle and break due to the coatings (fibronectin) on the inside of the tubing. Moreover, due to the cylindrical nature of the tubing, monitoring the cell growth with microscopy was very difficult. In an attempt to improve upon the production of such endothelium mimics, the channels of microfluidic devices were employed as the “circulatory vessel walls” in place of the microbore tubing. 2 The microfluidic devices, as mentioned previously, provide the same dimensionality as standard cell culture flasks, thus facilitating the immobilization of the cells to the patterned channels. Moreover, the planar structure and transparent nature combined make it much easier to monitor the cells in the channels using standard inverted microscopes, often employed for cell culturing methods. In addition, it is also possible to stain cells or load them with fluorescence probes to monitor various states of the cells or processes occurring within the cells, measurements that would be difficult in cylindrical microbore tubing. Figure 2 shows the fluorescence of NO reacting with diaminofluorescein (DAF), a reactive fluorescent probe for NO, upon stimulation of endothelial cells with ATP.
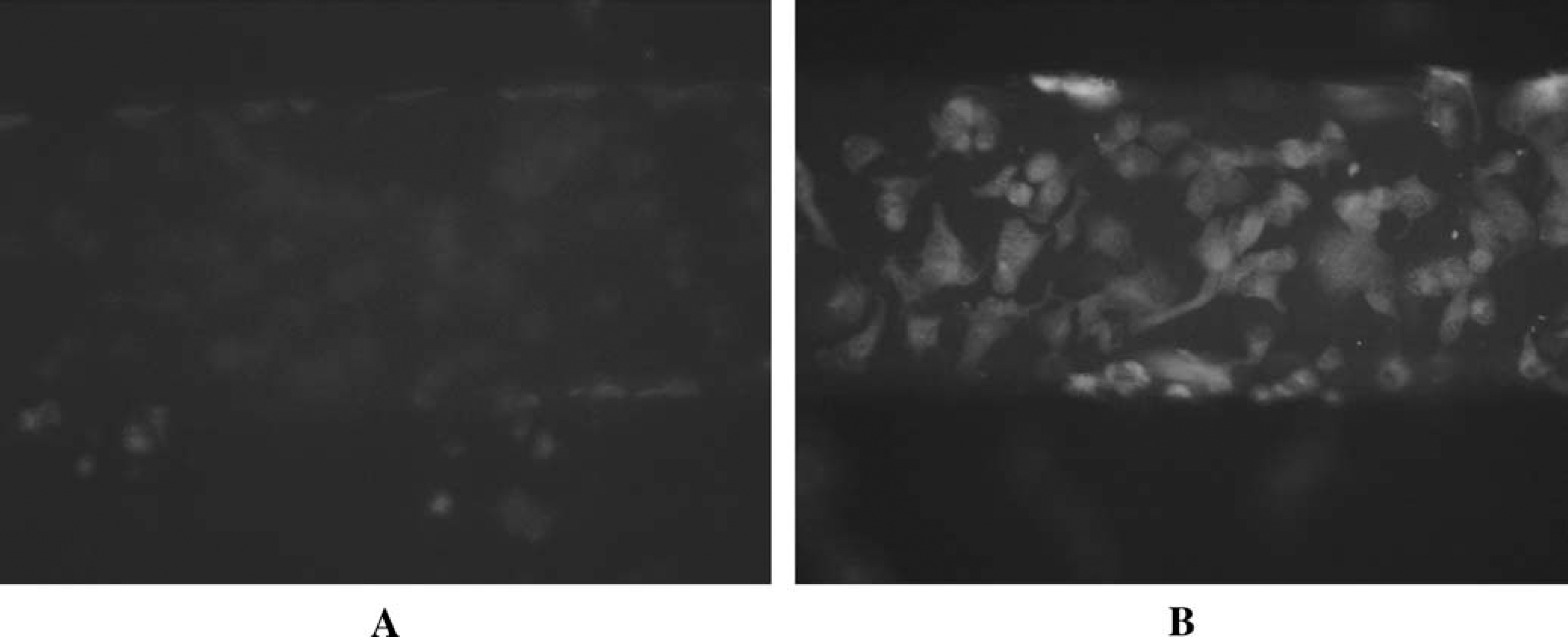
Fluorescence microscopy reveals NO production in endothelial cells. The cells, immobilized to a channel in a microfluidic device, were loaded with a fluorescence probe, DAF2 in (A), and stimulated with physiological levels of ATP (12 μM). The resultant ATP-stimulated NO production, which results in an increase in fluorescence, can be seen in (B).
Advancing Microfluidic-Based Physiological Mimetic Processes through Automation
There are numerous steps that could be taken in the attempt to create an improved microfluidic-based microcirculation including, but not limited to, extended life span of the immobilized endothelial cells, recycling of the RBCs flowing through the microfluidic channels, periodic measurements of analytes of interest, and periodic recalibration. All of the above improvements would benefit from automation. For example, cell culture success on a microfluidic platform would benefit from an automated constant feedback system whereby the cell environment could be constantly monitored for pH levels, gas content, temperature, and nutrients. Any change from optimal conditions could be corrected using automated control of the aforementioned parameters. This type of automated technology would benefit all types of cell culture techniques on a microfluidic platform. Advances in cell culture on microfluidic devices have already been reported. 20
In addition to improving cell culturing methods on microfluidic platforms, automation may also help to improve certain in vitro models of in vivo processes. Recently, our group has employed microfluidic technology in an attempt to mimic the microcirculation. To date, our studies have involved pumping erythrocytes through the microchip channels (the diameters of which approximate those of resistance vessels, in vivo) and performing the measurement portion of the analysis, after which the erythrocytes exit to waste. In vivo, red cells are present in the bloodstream for approximately 120 days; thus, it would be more realistic to have the cells cycle through the microfluidic channels for periods longer than a few minutes, which is the current practice. In order to perform such cell recycling on a micro-fluidic device and manipulate the direction of the erythrocyte flow, on-chip valving will be required. Such valving has been described by Quake 21 –24 and is being implemented by other groups. However, in order to direct the opening and closing of these valves successfully and with proper timing, automation will be required to control the actuators that close and open the valves.
A final area in which automation would clearly benefit certain microfluidic-based assays is in the measurement portion of the analysis and in calibration. Ramsey's group has demonstrated the ability to perform a serial dilution of a standard and generate a calibration curve. 25 However, the ability to recalibrate a device in an automated and time-dependent fashion has yet to be demonstrated. Such a calibration will be important for those processes occurring in a microchip format that occur over an extended period of time (e.g., a determination of an analyte in blood that circulates through the device for a period of hours to days).
Potential Impact of an Automated in Vitro Mimic of in Vivo Processes: Beyond Molecular Pharmacology
Can automation take microfluidic processes to a new level of usefulness? This author believes the answer to this question is yes, and improved drug-discovery methods may be the beneficiary of this usefulness. That is, currently, at some initial stage of drug discovery, there is often an investigation of the molecule's activity or behavior in a cell culture environment. However, if there is one thing that our research group has learned in the short time that we have been focused on bioanalytical measurements, it is that there is a large gap between in vitro pharmacological studies and in vivo pharmacological studies. In other words, not unlike the Kolthoff statement that “theory guides, experiment decides”, one could also say that “pharmacology guides, physiology decides.” In our group, the goal of using microfluidics for biomedical studies is, due to blood flow through the channels and other cells immobilized to the channel walls, to enable one to come closer to monitoring physiological responses to pharmacological input. An example of how automated microfluidics could help us learn more than automated molecular pharmacological studies in, say, 96-well (or greater) microtiter plates is the effect of iloprost in vivo. It is known that iloprost, the stable analog of prostacyclin, in the presence of endothelial cells, is capable of inducing a relaxation of smooth muscle cells. Interestingly, Olearczyk et al. have shown that iloprost induces ATP release from RBCs through its ability to increase cyclic adenosine monophosphate levels in the RBCs. 26 ATP is a known stimulus of NO (a potent vasodilator) production in endothelial cells. In other words, is iloprost, in the presence of endothelial cells, directly involved in the relaxation of smooth muscle cells, or is this effect mediated in vivo by the RBC (via the ability of iloprost to induce ATP release)? This is the type of question that would be difficult to answer with a simple cell culture of endothelial cells and the subsequent addition of iloprost. However, an improved in vitro model involving RBC flow and an immobilized endothelium could provide an intermediate model between conventional in vitro cell culture models and animal models. Figure 3 attempts to illustrate how a microfluidic device with multiple parallel channels incorporating blood flow and other immobilized cells may be able to bridge in vitro and in vivo pharmacological studies.
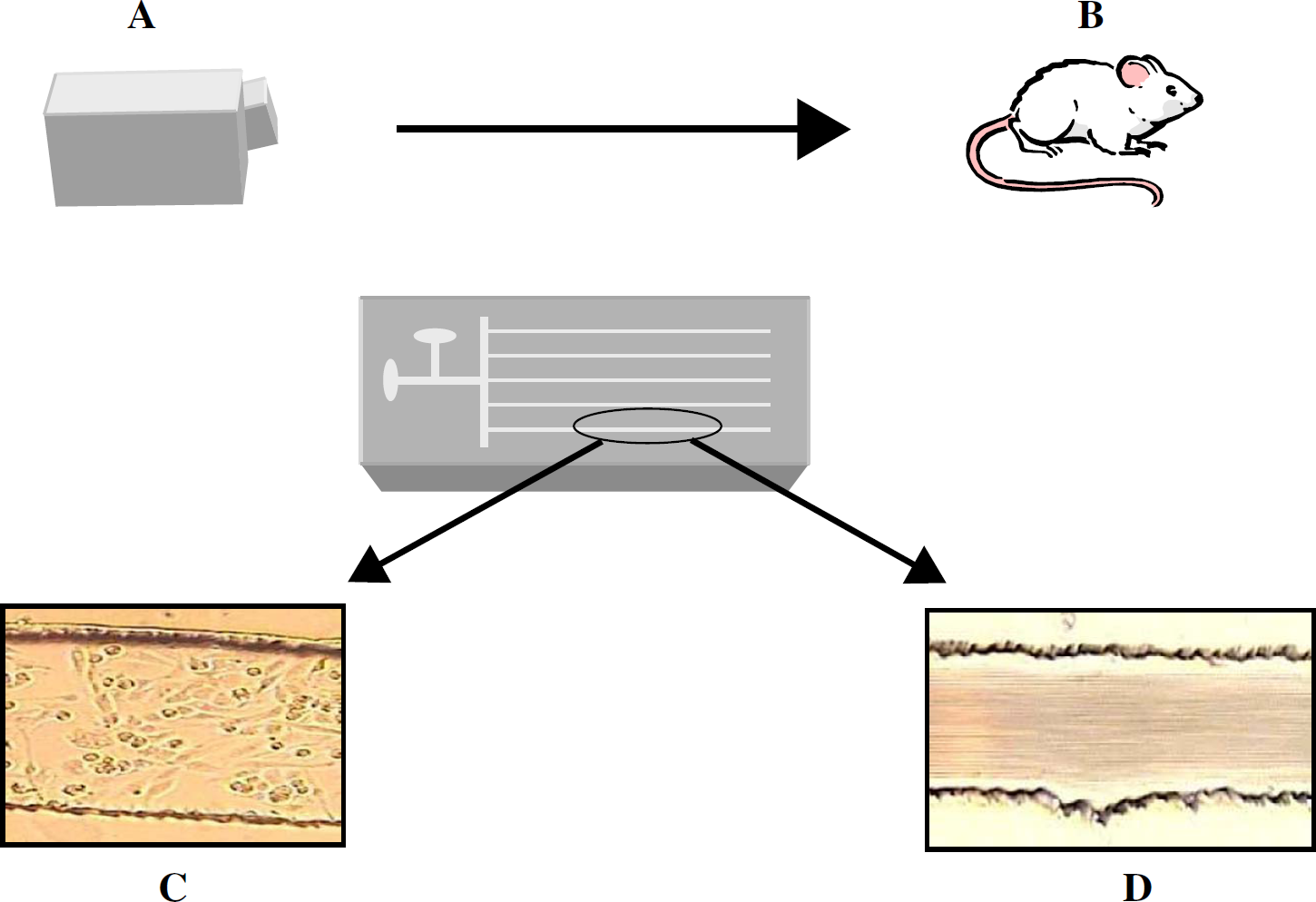
Quite often, biological studies begin in culture flasks (A) and eventually move to animal models (B). As depicted here, a microfluidic device with reagent and sample reservoirs may be able to flow through channels of a microfluidic device that represents an intermediate between the cell culture flask and the animal model. Importantly, the device may incorporate an endothelium (C) and blood flow (D).
Another example where an improved in vitro model incorporating blood flow would provide more meaningful data than a simple static in vitro study involves the role of antioxidants in protecting RBC deformability. That is, upon oxidant insult with diamide, an oxidant that is somewhat specific for oxidizing the reduced form of glutathione (GSH) to the oxidized dimer (GSSG), the RBC membrane stiffens. While an in vitro study of the RBCs in a cuvette would reveal a decrease in GSH and a subsequent increase in the level of GSSG, the physiological consequence of this oxidant insult would probably go undetected. However, in a microfluidic device incorporating RBC flow through patterned channels, one can actually monitor the effect of the added oxidant via chemiluminescence determination of ATP. That is, the continuous flow of RBCs enables for a constant monitoring of the physiological consequence of the diamide-induced stiffening of the cell membrane, specifically, the RBC-derived and deformation-induced release of ATP. In sum, the microfluidic device incorporating RBC flow enables an in vivo event to be witnessed on an in vitro platform. Figure 4 shows the ATP release from RBCs obtained from male, white New Zealand rabbits at various points in time after diamide was introduced to the RBC sample. Note that the dynamic ATP release from the RBCs can be monitored continuously over a predetermined amount of time (here, 23 min), and that the dynamic membrane properties can be investigated via detection of the ATP release from the cells. That is, as the RBCs stiffen, the ATP release decreases; however, at 18 min and beyond, the membrane deformability (and ATP release) have somewhat recovered. These types of measurements would not be possible in a typical in vitro cell culture or static system.
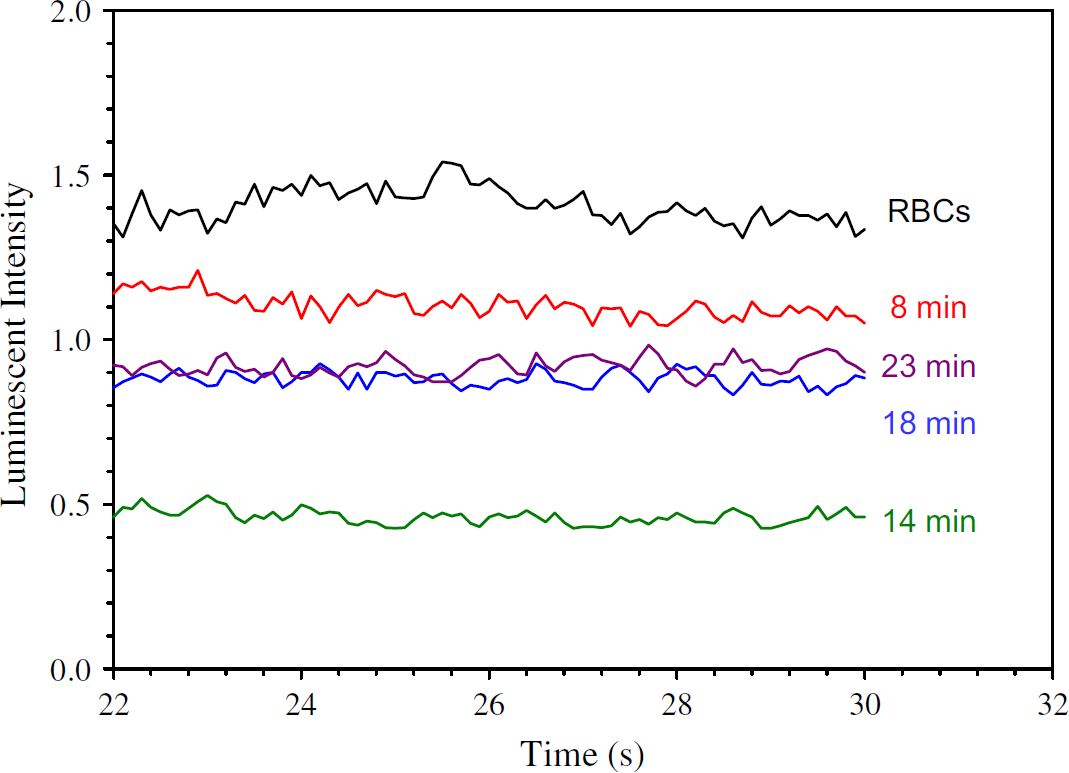
ATP release from RBCs that were stiffened with diamide. Note the ability of the RBCs to regain their ability to release ATP after approximately 18 min.
If determinations on microfluidic devices are going to find a place in the toolbox of pharmaceutical drug discovery, or any other scientific field, sample throughput will need to be on par with that of multiwell plates. Microfluidic devices have the ability to exceed the throughput of multiwell plates. For example, consider a microfluidic device that is 5-cm wide having 100 μM wide channels; one could potentially have 250 channels (if 100 μM spacing is left between each channel). Importantly, sample preparation, reactions, and washing steps are all possible with microfluidic devices because they incorporate flow. Taken collectively, it is not unreasonable to conceive that the properties of microfluidic devices may enable thousands of samples per hour to be determined in an environment that more closely resembles in vivo conditions. However, a great deal of automation will be required before such microfluidic devices can operate at this type of sample throughput. Detection schemes will need to be automated to measure the analytes of interest in each of the parallel channels on the microfluidic device. Moreover, if washings, reactions, or any other type of preparative work is to be completed in the microfluidic channels, on-chip valving will become an important determinant to this success (to enable the direction of fluid flow throughout the device).
Conclusions
Here, an attempt has been made to glimpse into the near future to predict the role of microfluidic devices in biomedical research. Automated detection and automated fluid manipulations will both improve the overall usefulness of such devices. Moreover, automation may also play a major role in the development of creating better in vitro models of in vivo processes. If so, the role of microfluidic devices in laboratory settings will only grow more popular, especially in those areas where high-throughput determinations and biological assays are required.
Acknowledgment
This work was supported by the NIH (HL-073942).
