Abstract
The optimization of synthesis conditions plays an important role in chemistry and life sciences. A wide variety of reaction parameters should be explored to minimize the use of educts and maximize the yields of desired products in high purity. A fully automated system for combinatorial synthesis has been developed and used for the optimization of synthesis conditions of a multicomponent single-pot reaction. In order to explore optimal reaction conditions for the synthesis of pure endo isomer of 3-(2-bromo-benzoylamino)-cyclohex-4-ene1,2-dicarboxylic acid diethyl ester, an intermediate in the synthesis route of lycorizidine analogues, several hundred experiments were carried out with varying reaction temperature, times, concentrations, and ratios of educts. In this case study, financial aspects also were considered in order to minimize synthesis costs.
The system was integrated using a Zymate XP robot as a system integrator. In addition to a GC/MS and an HPLC system, a multicomponent solid dispenser workbench and a liquid handling system are located within this fully automated system.
Introduction
The optimization of synthesis conditions plays an important role in chemistry and life sciences. Exploring a wide variety of reaction conditions, optimal reaction parameters are sought for minimizing the use of educts with maximum yields of desired products in high purity. 1 –3
Modern organic synthesis emphasizes the utilization of reactions that require minimal amounts of labor and allow for the construction of complex molecules in a timely and cost effective manner. During the past decade great efforts have been devoted to the development of domino reactions, which constitute highly efficient transformations by virtue of their ease of operability and complexity, and diversity-generating potential. 4
With the utilization of sequential multicomponent reactions of suitably functionalized starting materials, phenanthridone derivatives can be synthesized in a straight-forward manner. In this regard it is interesting to note that during the past decade, intensive research activities have been subjected to the synthesis of phenanthridone-based alkaloids such as the Amaryllidaceae alkaloids hippadine and lycoricidine via palladium catalysis methodologies.
Fully automated methods are necessary to enable the handling of large numbers of samples. Whereas automated systems are known for solid phase synthesis, 5 there is still a lack of suitable systems for complex liquid-phase synthesis. The need to generate useful information on a large number of reaction products requires the integration of sample pretreatment, reaction, sample preparation, and analytical measurements.
Experimental Details
Sample preparation (e.g., addition of liquids, solids, and reactant stock solutions), reaction processing (e.g., heating, mixing), and workup (e.g., cooling, dilution, analytical pretreatment, analysis) steps were performed by a laboratory automation system based on a Zymate XP robot (Caliper Life Sciences, Hopkinton, MA).
The reactions were carried out in 2 mL screw cap vials (VWR International, Darmstadt, Germany). Starting materials and solvents were used as received from commercial suppliers. N-methyl pyrrolidone was used as the solvent, and the concentration of the catalyst toluol sulfonic acid was 1 mol %.
Analysis Parameters
The analysis of the educts and products was carried out using GC-MS (Agilent 5973) with HP5/MS column (30 m × 0.25 mm × 0.25 μm).
System Design
A fully automated system for combinatorial synthesis has been developed at our institute 6 (Fig. 1). A Zymate XP robot mounted on a linear track is used to integrate the system instrumentation. It is equipped with additional devices, such as storage racks for 2 mL vials; a modified Büchi syncore reaction system for heating, cooling, and shaking of the reaction mixtures; a crimper/decrimper station; a centrifuge; filtration units; and liquid delivery stations. A specially designed PAL robot (CTC Analytics AG, Zwingen, Switzerland) is used for liquid handling tasks, even under inert conditions. For the handling of very small amounts of solids, a many-to-many solid dispensing system (Autodose S.A., Geneva, Switzerland) is included. To ensure a fully automated process including automated analysis, a GC/MS system, as well as an HPLC, is integrated into the system. This complex system is controlled by novel graphical software. Complete reaction procedures including sample preparation, reaction, preanalytical sample treatment, and analysis can be programmed. The current status of the system and the samples processed can be monitored at any time (Fig. 2).
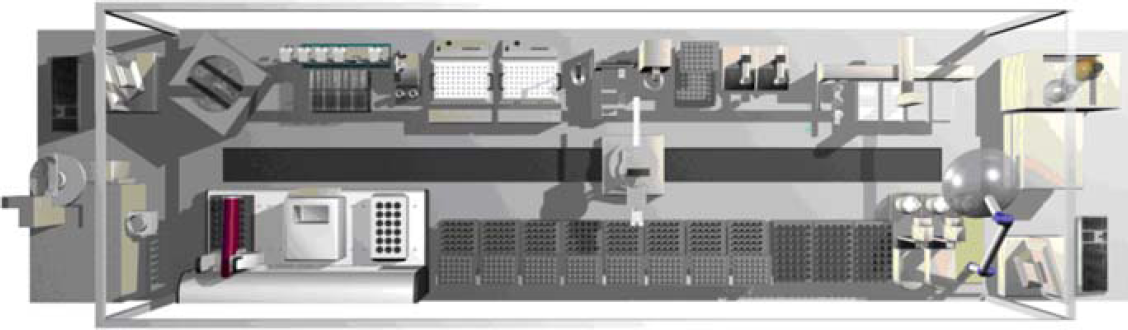
Fully automated system for chemical synthesis optimization.
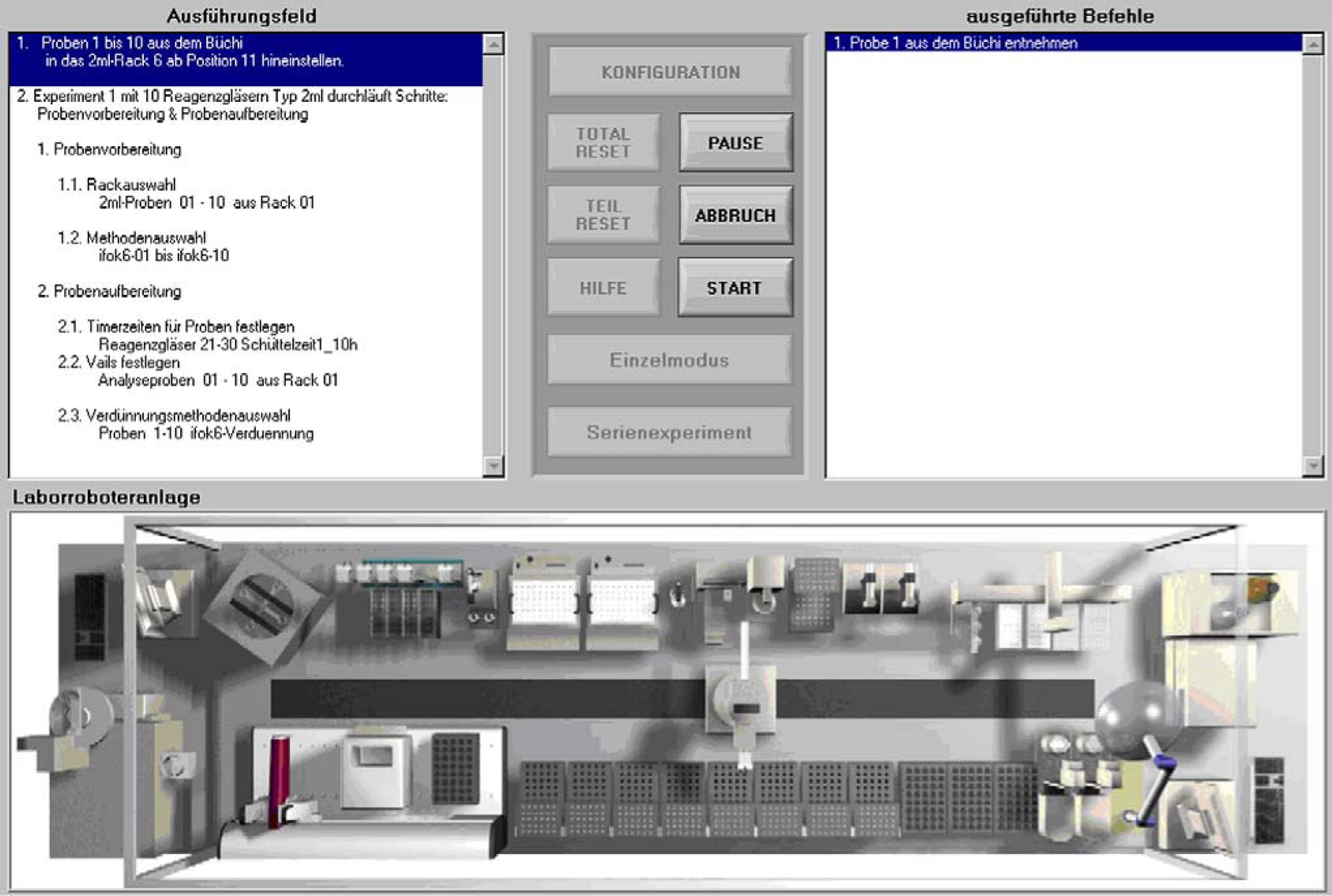
System control software.
Automated Synthesis Optimization
Investigated Reaction and Parameters
The system was optimized for yield and purity of the endo-isomer of 3-(2-bromo-benzoylamino)-cyclohex-4-ene-1,2-dicarboxylic acid diethyl ester, an intermediate in the synthesis route of lycorizidine analogues. In this multicomponent, single-pot reaction, an aldehyde, an amide, and a dienophile are combined under catalytic conditions 7,8 (Fig. 3). Initial investigations showed very low yields of the desired product. Side reactions such as the synthesis of the exo-isomer were detected.
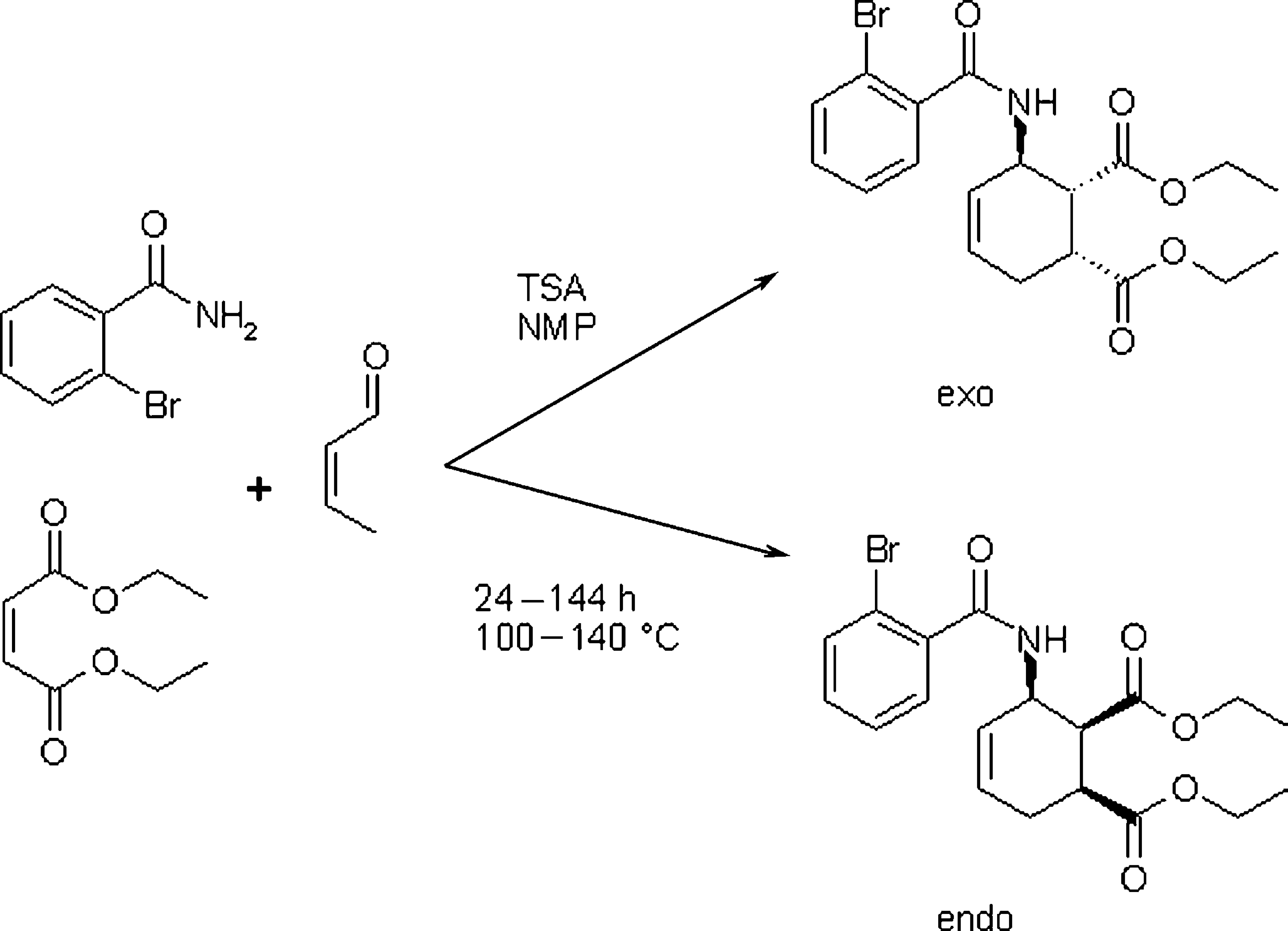
Multicomponent reaction for the synthesis of 3-(2-bromo-benzoylamino)-cyclohex-4-ene-1,2-dicarboxylic acid diethyl ester.
For synthesis optimization, the following parameters were explored: (1) reaction temperature (100 to 140°C), (2) reaction time (24 to 120 h); (3) compound ratios of the reactants, and (4) concentrations of the reactants (0.15 to 0.6 mol/L).
Results and Discussion
Table 1 summarizes the composition of the reaction mixtures. Figure 4 shows a typical series of automated screening experiments at 120° C. It demonstrates the influence of compound composition and reaction time on the yield of the endo isomer.
Example for investigated compound mixtures and educt prices (equivalents used in the reactions are given)
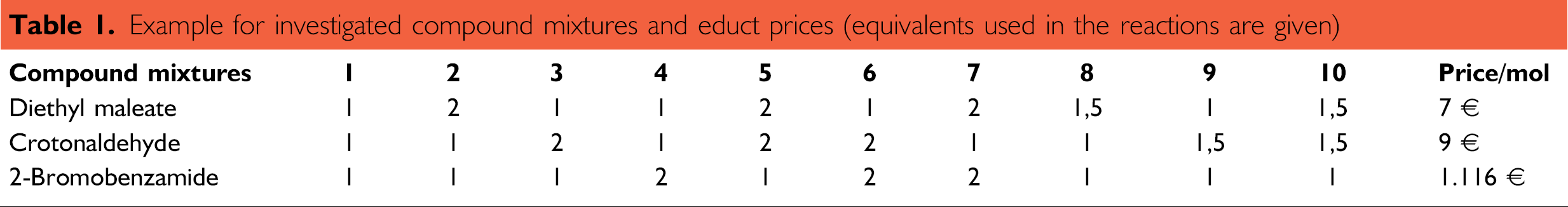
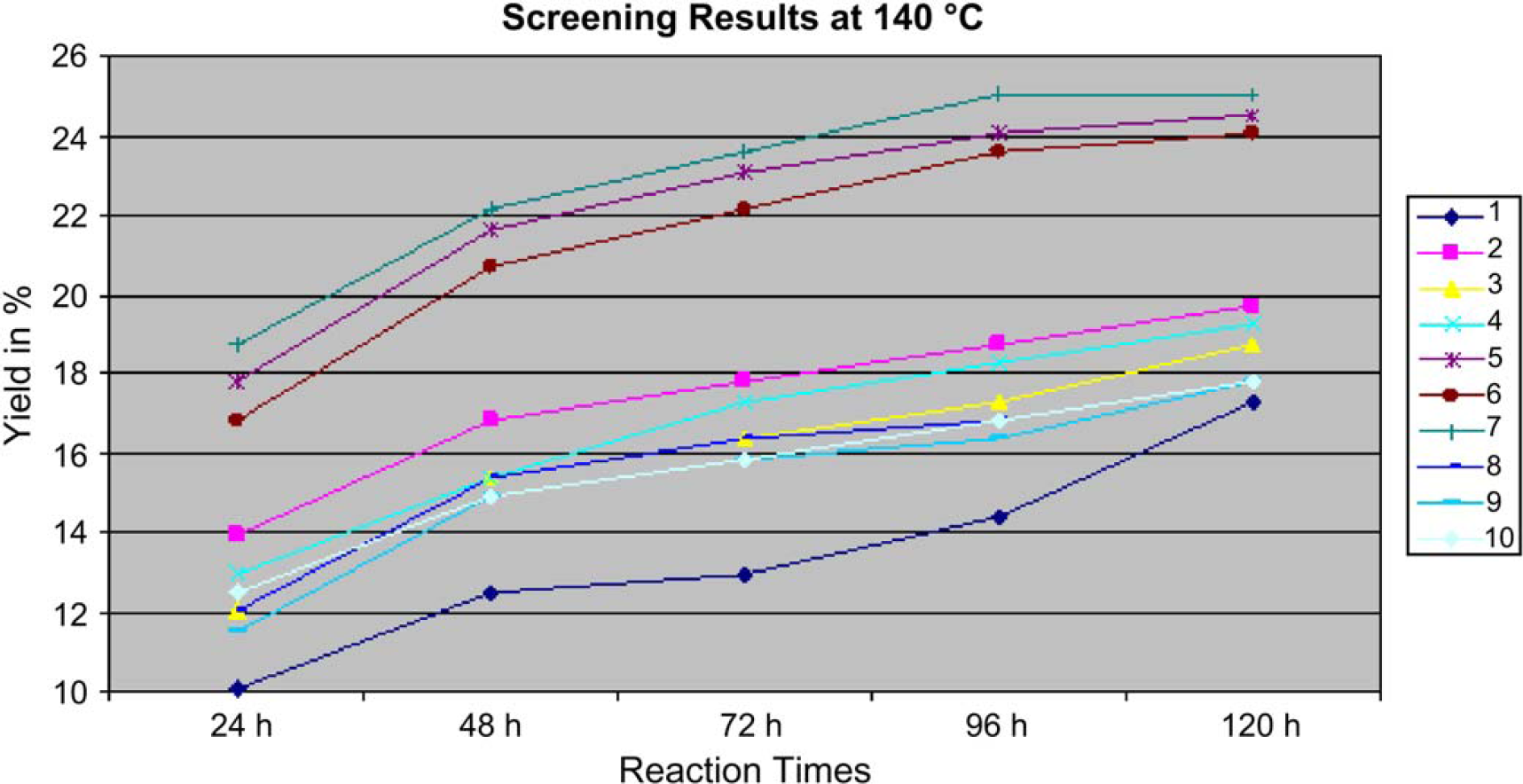
Screening results at 140°C.
As can be seen, the 20% yields of compound mixtures 5, 6, and 7 show the best results. Increased temperatures up to 140° C led to an increasing yield for these reaction mixtures of approximately 25% using long reaction times up to 96 to 120 h(Fig. 5). When one considers reaction yield and costs, the results also demonstrate that only one economically relevant hit (mixture number 5) can be found.
Screening results at 140°C. Yields are increasing up to 25% at reaction times of 120 h.
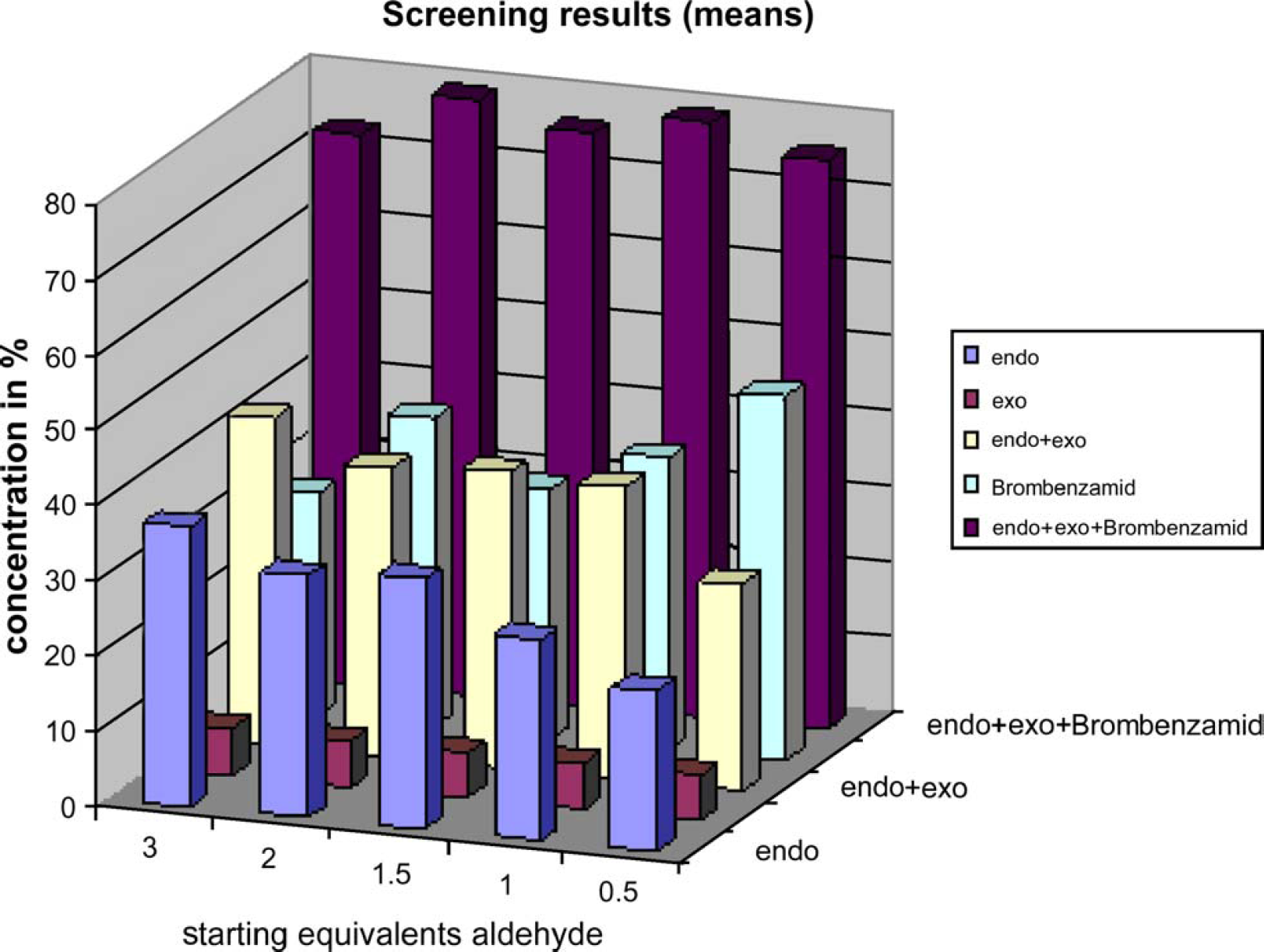
To reduce side reactions, a second screen was performed to explore the time-dependent addition of 3 equivalents to crotonaldehyde (one to six dosing steps, Fig. 6). Compound mixture 5 as well as the reaction temperature of 140° C were selected for further experiments in the second screening step, as described above. The reaction time was increased to 144 h.
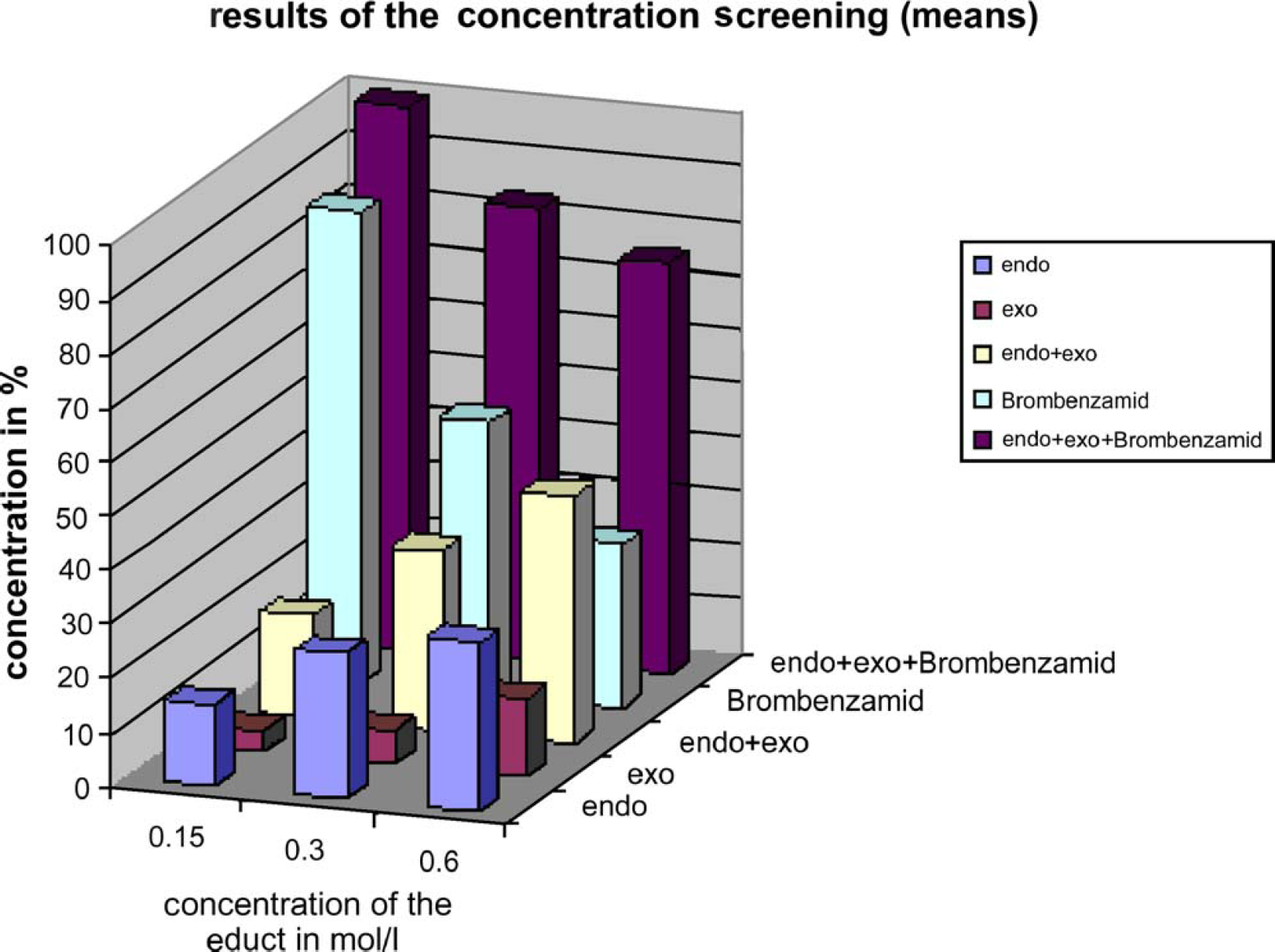
Screening results with variation of educt concentrations.
Figure 5 Screening Results of Step 2
The highest yield for the endo isomer was found to be 37% using 3 equivalents aldehyde without temporary dosing. Nevertheless, due to reduced side reactions, 1.5 start equivalents of crotonaldehyde were selected for a third concentration screening step to explore suitable educt concentrations. While the highest yield of the endo isomer could be achieved using educt concentrations of 0.6 mol/L, lower educt concentrations led to higher purity without side reactions.
Conclusions
A fully automated system was used for the synthesis optimization of a multicomponent reaction to determine optimal reaction conditions for high purity and high yields of 3-(2-bromo-benzoylamino)-cyclohex-4-ene-1,2-dicarboxylic acid diethyl ester. Several hundred reaction combinations were screened within a short time. Thanks to the flexible structure of the system, adaptation to different liquid-phase reactions, including analytical measurements, is possible. Other applications that were performed with the system include the synthesis of substituted pthalic acid derivatives in one-pot reactions or sequential three-component AAD reactions, and Heck cyclization for the synthesis of phenanthridones. 5
Acknowledgments
The project was supported by the European Union and the Ministry of Cultural Affairs of Mecklenburg/Westpommerania.
