Abstract
An internal development project at Bristol-Myers Squibb (BMS) led to invention of a family of organic chemistry synthesis blocks for both parallel synthesis in drug discovery and parallel reaction optimization in pharmaceutical development. The internal demand for these synthesis blocks became so great that the original development team was challenged by the burden of ongoing manufacture, support, and supply chain management. As a result, BMS entered into a unique industry partnership with Mettler-Toledo AutoChem (MT), Newark, DE, formerly Bohdan Automation, to commercialize the reactor blocks and extend the product family, now known as the MiniBlock line. This manuscript describes the initial development drivers, the overall technical design, and the ultimate successful commercialization of the MiniBlock synthesis family.
Introduction
In industries such as pharmaceuticals, agrochemicals, and chemical materials, optimization of chemical structure with respect to properties is key to success. Before about 1995, this was carried out by serial synthesis of individual chemical compounds, testing of their properties, and iterative synthesis of the next generation of compounds. In the latter half of the 1990s, this paradigm was changed by the introduction of parallel synthesis techniques that enabled synthesis of libraries or collections of new compounds in essentially a single operation. 1 At the start of this industry transformation, effective tools to enable parallel synthesis did not exist. This, in turn, triggered a rush to develop and commercialize tools to enable parallel synthesis. 2 –6 Ultimately, one of the most successful of those tools proved to be the MiniBlock Synthesizer and its product line family members 7 (Mini-Block is a registered trademark of Mettler-Toledo, Inc.). The MiniBlock is the result of an uncommon collaboration between companies in the pharmaceutical (Bristol-Myers Squibb) and laboratory instrumentation (Mettler-Toledo) industries. This article recounts the story of development and commercialization of the MiniBlock products from an historical perspective and illustrates key success factors for such successful collaborations.
The introduction of solid-phase synthesis for small nonpeptidic organic compounds in 1992
8
led to an explosion of interest in parallel or combinatorial chemistry. In actual practice, the term
Against this dynamically shifting backdrop, BMS made three key decisions: (1) to embark on an aggressive internal technology development program modeled on the biotechnology industry (rather than wait for commercial instrument vendors to provide turnkey solutions); (2) to focus its efforts exclusively on tools to enable synthesis of individual, pure compounds rather than mixtures; and (3) to focus initially on developing tools for synthesis of libraries of modest size for lead optimization rather than on very large libraries for lead discovery.
Miniblock Design
The most basic requirements for a parallel chemical reactor are the ability to manipulate a number of chemical reactions in parallel using a single device and the ability to control environmental conditions inside the reactor (temperature, atmosphere, agitation). For solid-phase synthesis, and sometimes for solution-phase synthesis (for example, when using scavenging or trapping resins), it is desirable to be able to filter and wash the reaction mixture and to subsequently direct the filtrate either to waste or to collection vials.
BMS formed a multidisciplinary team of chemists and engineers in 1995 to develop a parallel synthesis reactor based on the above requirements. Because there was a business need for immediate impact (that is, the ability to synthesize at least some compounds in parallel), the team adopted a rapid iterative design approach 23 that led to a sequence of reactors with increasing capability. The team noted that solid-phase synthesis of peptides using simple polypropylene syringes fitted with filter frits as reaction vessels had been reported as early as 1990, 24 and that a variety of commercial vacuum systems for solid-phase extraction (SPE) enabled collection of products from SPE (done in polypropylene Luer fitted syringe barrels) into either a waste stream or a collection vessel. Thus, an SPE vacuum box fitted with fritted syringe barrels could conceivably be used as a simple parallel synthesis device.
The BMS team fabricated a simple syringe barrel/vacuum box reactor and demonstrated its utility for simple solid-phase synthesis procedures in which strict atmosphere and temperature control were not required. 25 The reactor consisted of a Varian VacElut SPS 24 vacuum box (Varian Inc., Palo Alto, CA) fitted with an array of syringe barrels connected to the box via Teflon stopcocks (Fig. 1). The user could contain or release fluids from the reactor by simply turning the stopcocks, and filtrates could be directed to either waste or collection tubes by rotating the collection plate of the vacuum box. The entire assembly was agitated via an orbital shaker.
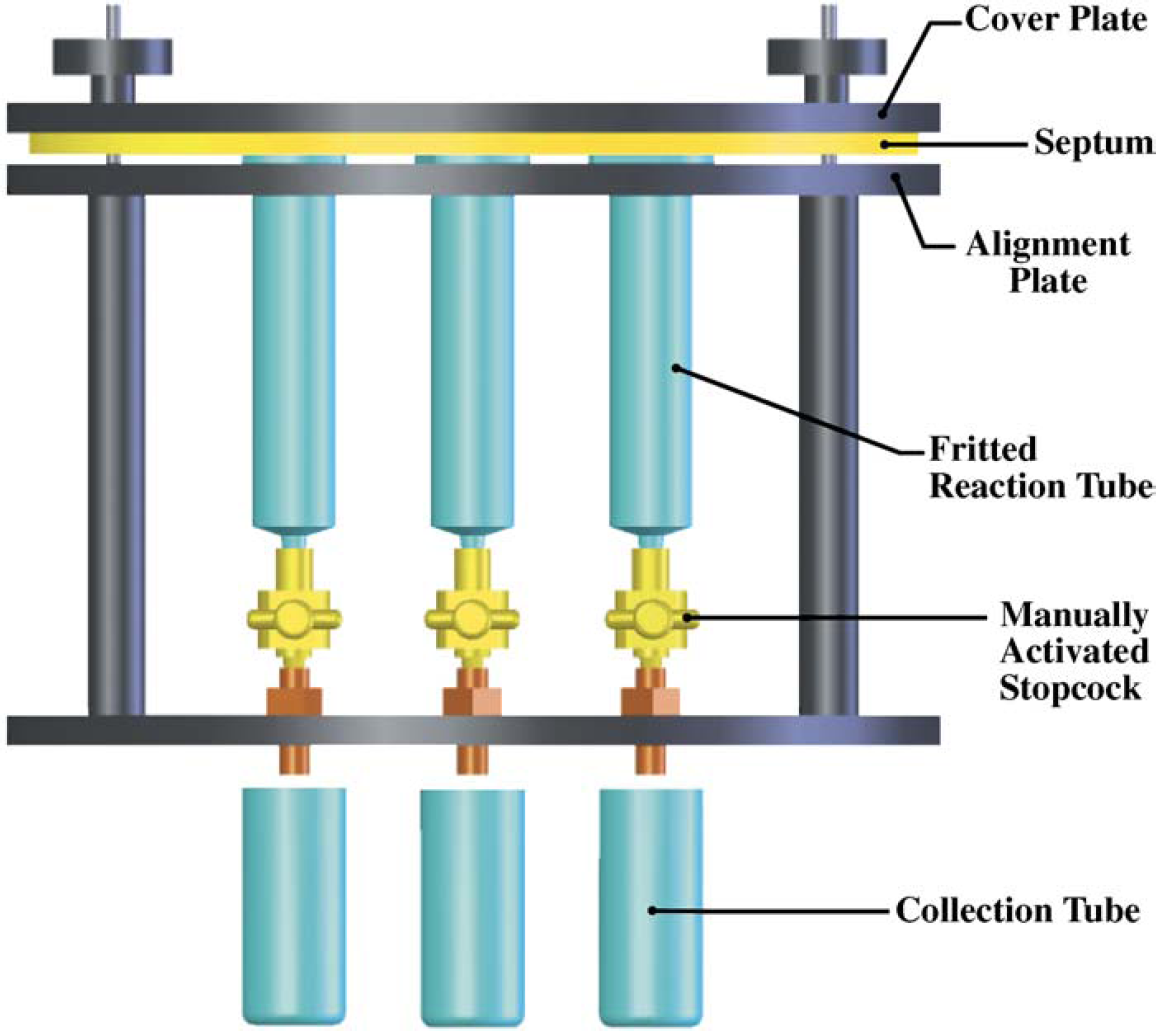
VacElut Reactor cross-section.
Although simple solid-phase reactions could be performed using the vacuum box reactor, there were several drawbacks. The reaction environment (atmosphere and temperature) was difficult to control, draining of the reactors required manual manipulation of a large number of individual stopcocks (one per reaction), and the circular reaction array and overall stack height of the VacElut were not well suited for reagent addition by common Cartesian liquid handlers.
These shortcomings led to the next iterative design of the reactor system (Fig. 2). 26 The new reactor contained 48 reaction wells, and so, in consideration of both its utility and the name of one of its inventors, became known at BMS as the “48-Weller” reactor. The reactor maintained the vacuum box concept by using a Cartesian array of syringe barrel reactors connected to a vacuum box via Luer tip Teflon stopcocks. In this case, a rail system was designed to allow actuation of all 48 stopcocks with a single motion. The Cartesian array facilitated reagent addition by conventional liquid handling robots. An aluminum heat transfer jacket that could be heated or cooled by circulating fluid added temperature control capability to the new reactor. Products were collected by opening the stopcocks and applying vacuum to a solid base with provision to hold 48 individual test tubes (Fig. 2); waste was collected using a similar tub base without test tubes (not shown). The top of the reaction vessel array was septum sealed so that an inert atmosphere manifold could be added. The manifold (Fig. 3) allowed reagent addition through off-centered reagent addition ports while maintaining inert atmosphere via separate off-centered gas inlet ports.
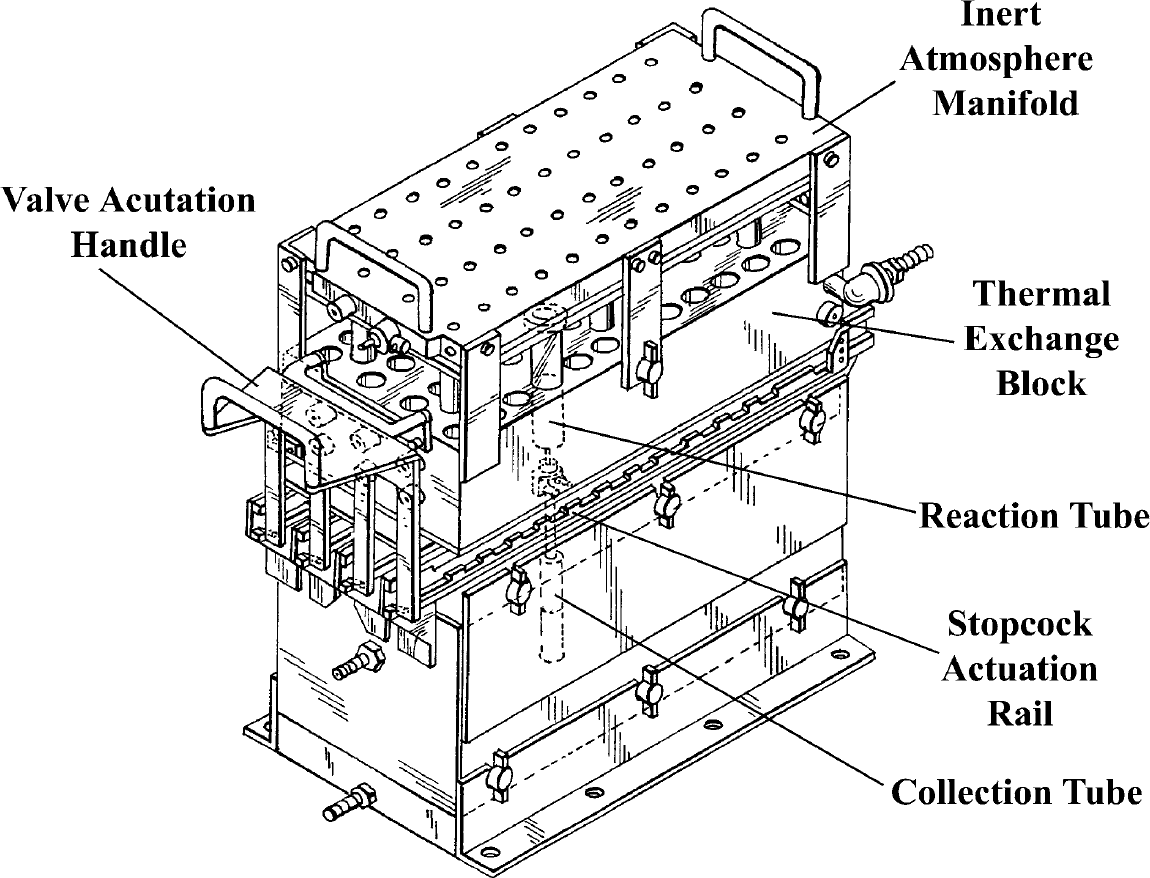
48-Weller assembly drawing.
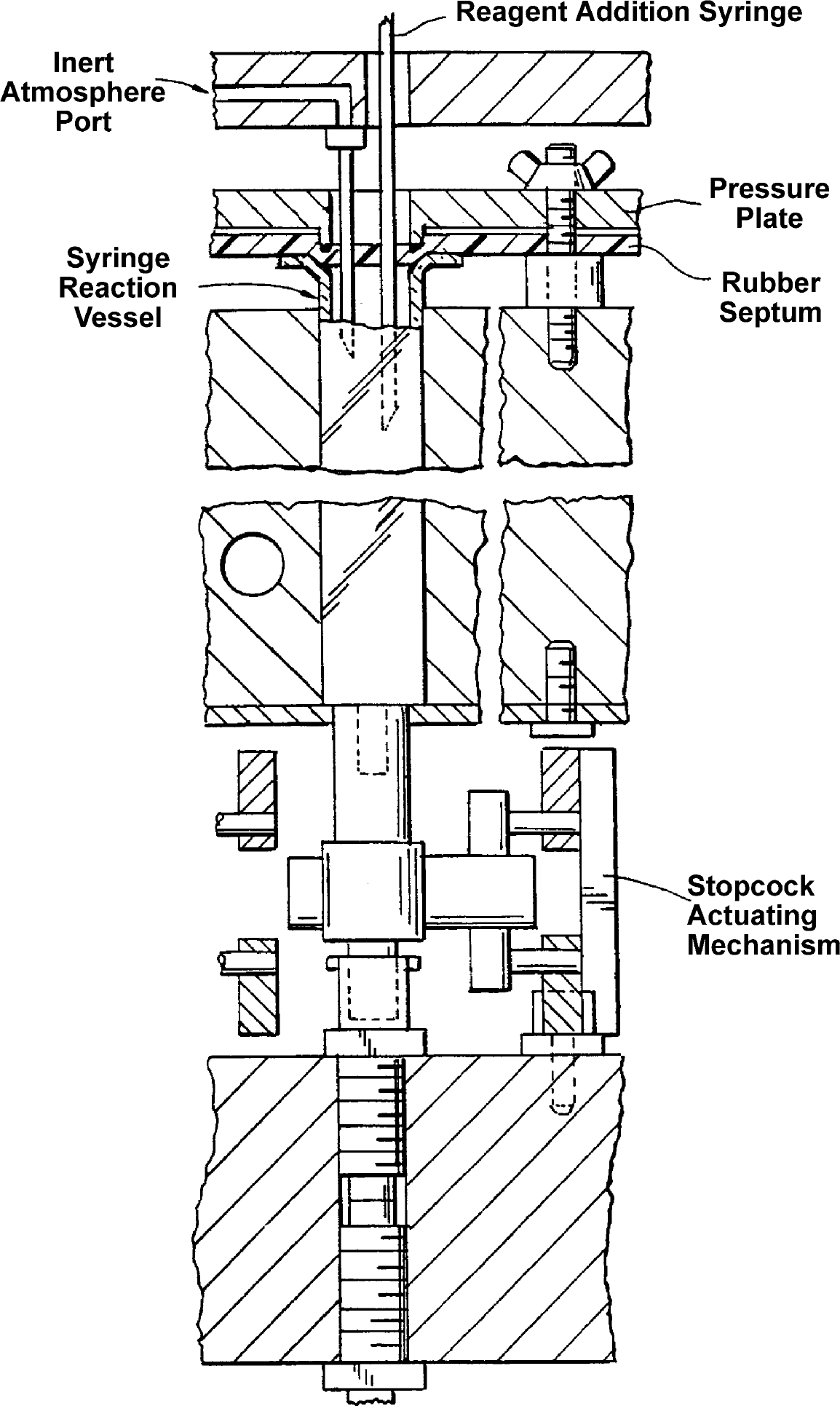
Non-centered inert atmosphere manifold and stopcock actuation mechanism.
The 48-Weller reactor thus overcame the three most significant drawbacks of the original vacuum box reactor. Dozens of reactors were built, and it became the workhorse of parallel synthesis at BMS from the time of its introduction until its displacement by the next-generation reactor, thus satisfying the business need for increasing parallel synthesis capability. Many thousands of new compounds were prepared in the 48-Weller during its reign, including reactions requiring heating and cooling and reactions requiring careful inert atmosphere control. One reaction in particular was quenched by atmospheric oxygen and could not be run in an open reactor. By using the 48-Weller with inert atmosphere manifold, this reaction could routinely deliver its desired product in >90% yield, 27 28 thus demonstrating the integrity of the atmosphere.
Successful and widespread parallel synthesis using the 48-Weller reactor quickly pointed out its drawbacks as well as its strengths. As noted above, its strengths included the ability to actuate all 48 valves simultaneously, its ability to provide controlled atmosphere and temperature, and its ability to direct filtrates to either waste or into product collection tubes. Aside from its large weight and size, the primary drawback of the 48-Weller was that its tube layout, or format, was unique in the laboratory and unlike that of any other robot or system in use. After synthesis, products were contained in a 4 × 12 array of 16 × 100-mm test tubes; individual tubes had to be labeled and transferred into a centrifugal concentrator for further processing. After dry-down, individual tubes had to again be manipulated into downstream robotic formats. By developing an efficient synthesis reactor, the team quickly mastered parallel synthesis only to shift the bottleneck from synthesis to postsynthesis processing. In this way, they learned the importance of overall workflow automation as opposed to individual process optimization. The concept of developing a well-integrated overall synthesis and postsynthesis workflow thus led to development of the next-generation synthesis reactor that eventually became the MiniBlock synthesizer.
At the same time that BMS was developing and using the 48-Weller reactor for parallel synthesis, a new deterministic split-and-pool synthesis technique was developed and commercialized. 29 –32 This method, introduced to the market by IRORI (now Discovery Partners International, Inc., San Diego, CA) in the second half of 1996, entailed solid-phase synthesis using 8.6 × 17.5-mm porous MicroKans as synthesis vessels. The MicroKans contained both synthesis resin and a radio frequency-encoded transponder for identification. MicroKans were manipulated during synthesis steps with standard laboratory glassware. At the end of synthesis, however, the MicroKans needed to be exposed to a cleavage cocktail (generally containing trifluoroacetic acid) and then filtered to deliver the filtrate to product collection vessels. At the time of the original MicroKan introduction, no commercially available cleavage device existed. 33 Because MicroKan technology was being adopted at BMS for synthesis of large libraries, alongside parallel synthesis in the 48-Weller for smaller libraries, it was decided that one design requirement for the next-generation synthesizer would be that it could be used for the cleavage step of MicroKan synthesis.
As discussed above, a major driver for development of the next generation synthesizer was the need to deliver products into an array that was compatible with other automated formats used in the lab. The most obvious choice would be the industry standard 96-well plate, with an 8 × 12 layout on a 9-mm (center to center) grid. Delivery of products into plates allows manipulation of samples as a group but precludes manipulation of individual samples. To overcome this limitation, it was decided to deliver products into an array of polypropylene tubes in the standard 96-well format. The diameter and volume requirements of MicroKan cleavage, however, precluded a simple 8 × 12 layout on 9-mm centers. Ultimately, a staggered 48-well layout with 9.7-mm (inner diameter) tubes on 18-mm centers was chosen for the reactor format. For product collection, custom high-volume microtubes were molded in a 96-position format. 34,35 Such a staggered reactor layout would normally require manufacture of mirror image reactor sets 36 ; to avoid that complication, the reactor was constructed as a 52-well reactor that could be used in either of two 48-position formats. The merger of two 52-well reactors into a single 96-well plate or tube holder is shown in Fig. 4.

Merger of mirror image plates into a single 96-well plate.
Once the format of the next-generation reactor was determined, other constraints had to be considered. It was desired to reduce cost and complexity of the reactor, and weight was a consideration. The rail-driven stopcock array used in the 48-Weller reactor was not compatible with these new constraints. Other contemporary reactors 5,36 –39 used a siphon flush mechanism, simple back pressure, or an elaborate valving mechanism. We felt that only a true valve would allow for reliable actuation without having to be concerned with delicate pressure balance. Our desire for simplicity led us to perfect a sliding valve mechanism with only a single moving part (Fig. 5). In this design, a sliding plate applies pressure to a resilient section of O-ring type cord that, in turn, pinches against a Teflon drain tube connected to the syringe barrel reactor outlet. When properly designed, this valve applies the proper amount of pressure to close the valve and prevent drainage of liquid from the reactor while allowing the Teflon outlet tube to reopen when pressure is released.

Sliding pinch valve mechanism.
Once the reactor format (Fig. 4) and valve design (Fig. 5) were determined, the overall modular reactor design was finalized (Fig. 6). The new reactor, now called the MiniBlock, retained key features of the 48-Weller, including a septum seal and inert gas manifold. The heat exchanger retained the original design but was made optional and modular. A single collection base was used for either waste collection (without insert) or product collection (by placing an appropriate collection rack into the collection base); heights could be adjusted by using inserts, either inside the collection base or between the collection base and the reactor bottom. An assembly drawing of the MiniBlock reactor is shown in Fig. 6. Complete mechanical details have been published. 40
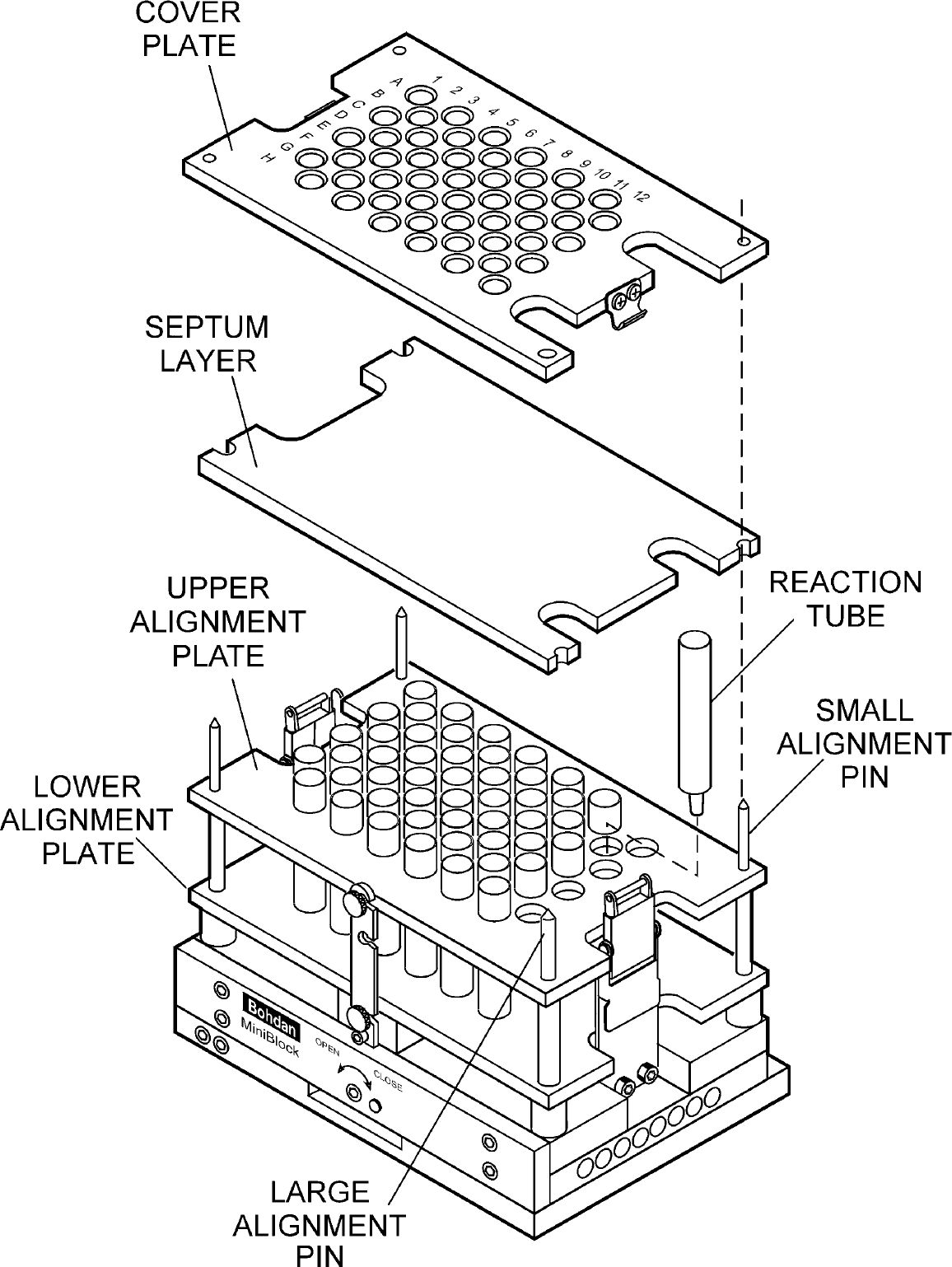
MiniBlock assembly drawing.
Upon its introduction to the chemistry community at BMS, the MiniBlock immediately displaced the 48-Weller as the standard synthesis platform for both parallel solid-phase and solution-phase synthesis and also for MicroKan cleavage. Chemistries requiring both temperature and atmosphere control were successfully carried out in the MiniBlock. Later, after its commercial introduction, an even wider array of chemistries was demonstrated successfully. 41 –43
Extension of the Miniblock Concept into Process R&D: Development of a Solution-Phase Reactor
Introduction of the 48-Weller and the MiniBlock synthesizer (and associated automation) to the discovery chemistry community enabled rapid execution of parallel library synthesis, and as a result, the bottleneck shifted from synthesis execution to synthesis rehearsal and optimization. Chemists in the drug discovery environment found themselves spending increasingly more time optimizing reactions and synthesis routes and came to realize that this activity is exactly the same activity that is engaged in daily by their counterparts in process research and development (PR&D). Meanwhile, productivity improvements in drug discovery were putting increased pressure on PR&D organizations to more quickly identify pathways to better and cheaper processes for drug synthesis. 44 –48 At Bristol-Myers Squibb, parallel experimentation strategies such as statistical design of experiments (DOE) were taking center stage as the PR&D team was attempting to catalyze its own paradigm shift from a “learn while doing” to a “learn before doing” organization 49 (for a discussion on optimizing process development, see 50 ). The result of this common interest was formation, in 1998, of a unique collaboration between chemists from discovery and process research, along with the original MiniBlock synthesizer design team, to develop tools for parallel optimization of solution-phase reaction conditions for process development.
Laboratory automation tools and concepts had been creeping into process research and development (PR&D) laboratories intermittently since the early 1990s, 51 and by the time of the formation of the BMS collaboration, commercial equipment vendors had began to specifically target PR&D applications. 52,53 As in the drug discovery area, the early commercial PR&D solutions focused heavily on fully automated systems designed to address a rather rigid interpretation of a process optimization workflow. Inflexibility was a common drawback of these initial systems, preventing the generalized adaptation of the equipment to parallel experimentation. In addition, commercially available workstations almost universally defined an array of large volume reactions (50 mL or more), requiring significant amounts of often-precious starting materials whereas, early in the development of a process, it was not uncommon to only have only a few grams of material available for experimentation. The BMS team, therefore, sought a more general and efficient approach to using automation concepts to support the new parallel experimentation workflows that confronted them. Adapting the modular design and iterative improvement concepts from the original MiniBlock team, the PR&D team decided to focus on development of a parallel reactor environment for small volume, solution-phase reactions. 54 –58
Minimum requirements for a simple process screening and optimization reactor are similar to those for parallel synthesis: the ability to execute a set of parallel reactions with control of atmosphere, temperature, and agitation. In contrast to drug discovery, however, the goal of process research and development is the generation of process knowledge and not the manufacture of material. Process knowledge is a broad understanding of the factors affecting the quality, characteristics and yield of the target chemical product and the tolerances of the process to variation of those factors. Thus, the requirements for temperature and agitation control are more rigid in PR&D than in parallel synthesis (that is, temperature and agitation must be controlled and known more precisely in process research than in parallel synthesis). In addition, the isolation of material is far less critical to the acquisition of process knowledge than control of parameters, so filtration is not a requirement for most PR&D parallel experimentation applications. The team added the requirement that equipment used to support a typical parallel experiment should not demand more than about a gram of input material to conduct an array of experiments.
With removal of the filtration requirement, it was possible to select a simple glass tube as the primary reaction vessel for the new reactor, with its size scaled such that it would be appropriate for a 50-mg reaction. Magnetic stirring was chosen as the agitation method and the useable working surface of a typical commercial magnetic stirrer was used to define the footprint of the device. The useable working surface area of a stirrer was determined experimentally by finding the boundary on its surface within which a slurry of potassium carbonate and acetone could be kept reasonably suspended. This experiment was also used to screen for the best magnetic stir bar for this application. An important discovery from this evaluation was the use of a long, thin magnetic stir bar whose length was greater than the diameter of the tube. With such a setup, the bar leaned against the wall of the tube because it could not lay flat. This “end-on” orientation prevented the stir bar from getting stuck and promoted more reliable agitation of the contents.
With the key spatial demands of the reactor block established, the first prototype was constructed (Fig. 7). The first generation prototype consisted of three parts: the heating and cooling base, the reflux jacket, and the inert gas blanketing component. The aluminum base had places for 24 reactor tubes, and recesses were deep enough that a 1-mL reaction could be effectively heated or cooled. Heating and cooling were enabled through a series of connected channels in the base that passed around the recesses and that terminated with quick connects at each end. The quick connects allowed facile connection (and disconnection) of the base to a circulating bath for temperature control. The reflux jacket was constructed similarly to the base with exception that recesses were open at both ends to allow the tube to pass through and rest in the base. The reflux jacket rested on four Teflon posts located at each corner of the base. As with the base, cooling was accomplished by connecting, again with quick connects, the reflux jacket to a circulating bath. Finally, the inert gas component allowed the array of tubes to be blanketed with an inert gas.

First-generation prototype PR&D reactor.
In accordance with the rapid iterative design principle, the first prototype reactor had immediate impact by allowing BMS process development scientists to quickly and reliably carry out parallel experiments. Its impact is illustrated by its use in the development of matrix metalloprotease inhibitor BMS-275291, a BMS oncology candidate. 59,60 The BMS team was exploring a cost-cutting strategy for the preparation of the drug. With the assistance of the reactor block, a single scientist was able to screen more than 300 experimental conditions under the pressure of short timelines, which ultimately led to the selection of a route that cut the raw material cost more than 80% from the original discovery synthesis.
Dozens of the first-generation reactor blocks were manufactured and distributed throughout BMS PR&D, and it wasn't long before success stories like the one described above began to stack up. Along with this success, however, came an understanding of the limitations of the first-generation reactor block: reactor size selection, lack of modularity, and liquid handler compatibility. An immediate challenge was that BMS process scientists demanded a wider range of reactor volumes than were available in the original design. The BMS design team initially responded by designing larger volume 10- and 6-position reactor blocks that held incrementally larger reaction volumes. The introduction of the new reactor block sizes, however, emphasized the second drawback of the original design—the block's inherent lack of modularity. None of the components for any of the different size blocks were interchangeable; thus, the design necessitated the manufacture of each part for every block. Accordingly, adequately outfitting an individual laboratory with a sufficient number of blocks of each size became prohibitively costly and impractical to realize. Finally, as in drug discovery, introduction of the reactor block shifted the research bottlenecks to other areas including evaporation and liquid handling—once again highlighting the importance of overall workflow automation. These new requirements were the technical drivers for development of the second-generation PR&D reactor block.
The PR&D team used the MiniBlock as the platform for the next iterative design of the solution-phase (PR&D) reactor (Fig. 8), sharing the same footprint and center to center spacing. This was possible because the recent extension to the MiniBlock line included parts that could be used for reactions of up to 50 mL in volume, a necessity for PR&D applications. In addition, the MiniBlock was already designed to enable heating and cooling of the reactions through a modular temperature control component and to enable inert atmosphere chemistry through use of an optional gas manifold. The team envisioned that the heating and cooling jacket for the MiniBlock could be used (in cooling mode) as the reflux condenser in the new solution-phase reactor block. To maintain the modular nature and footprint of the MiniBlock, the team designed a novel removable insert to position reactor tubes in a common reactor block base for use with existing MiniBlock components.

Second-generation PR&D reactor (24 position).
Removable aluminum inserts were thus designed with circular cavities to hold the glass reactor tubes. The inserts could be inserted into a common aluminum base frame that was drilled with channels to enable hot or cold fluid to pass through and control the base's temperature (similar to the original reactor block design). In this implementation, the temperature of the reactor tubes was controlled by thermal conductivity from the base to the insert and finally to the stirred reactor contents. Experiments showed that excellent thermal homogeneity (±pT2&° C) could be achieved across reactors using the removable insert concept. When needed for reflux capability, the original MiniBlock heat exchange jacket could be used as a condenser by cooling the upper part of the glass tubes. Inert atmosphere could be controlled using the original MiniBlock atmosphere manifold. Other MiniBlock components, such as the cover and alignment plates, could be used without modification in the new reactor block.
A suite of inserts (Fig. 9) was developed to fit into the new reactor block's circulating base, and the circular cavities were made according to the format and footprint of the staggered 48-well design of MiniBlock. The inserts were color coded (red, green, silver, and gold, in order from smallest to largest volume) according to the color coding of the existing Mettler-Toledo AutoChem MiniBlock parts. The inserts themselves had the footprint of a standard 96-well screening plate; thus, they were compatible with a wide variety of laboratory equipment, including liquid handlers and centrifugal concentrators. In addition, the entire reactor would fit within the working envelope of many standard liquid handlers, including the Gilson 215 and Tecan Genesis platforms.

An assortment of reactor inserts.
The second-generation solution-phase reactor block, eventually known as the MiniBlock-XT (XT), exceeded many of the expectations set for its design. The use of mostly commercial parts from the original MiniBlock synthesizer reduced cost by eliminating the need to manufacture custom parts and introduced an avenue through which solid-phase and solution-phase chemists could share a common inventory of parts. 61 In addition, the new XT block benefited from the modularity built into the commercial MiniBlock design, including optional reaction inserts and thermal control components. The innovation of the insert mechanism enabled a single circulating base to support a wide array of reactor sizes. The insert also significantly reduced cost associated with outfitting a single scientist for parallel experimentation, because one or two circulating bases and a set of inserts could enable a wide array of reaction volumes. Finally, basing the insert on the microplate standard offered the advantage of compatibility with commercial automated equipment. 62 Although originally designed to meet the needs of PR&D to optimize solution-phase reaction conditions, the new device also turned out to be well suited for solution-phase parallel synthesis. In particular, the ability to remove the reactor inserts and transfer them to other automated devices, such as liquid handlers or centrifugal concentrators, for postsynthesis processing was a key advantage in parallel synthesis. As a result, the new reactor quickly became a standard in both PR&D and discovery synthesis labs, where it was used for both reaction optimization and for parallel synthesis. 63,64 Thus, in combination with the original MiniBlock, a single reactor family could be used for reaction optimization, solid-phase synthesis, MicroKan cleavage, or solution-phase synthesis on a scale ranging from 0.5-mL to 50-mL volume. No other synthesis platform provided this versatility at such a low cost.
Concept to Market: Commercialization of the Miniblock
By 1998, the original MiniBlock synthesizer had become the standard for solid-phase synthesis at BMS, and demand for more reactor blocks was exceeding the capacity of the internal engineering group. The internal group was intended primarily to provide custom engineering support for pharmaceutical research, but it was neither equipped for nor intended to become a manufacturing group. In late 1998, this engineering team supervised the manufacture of more than 100 MiniBlock reactors, including parts sourcing, manufacture scheduling, and quality control. This effort placed demands on the group that prevented other key projects from moving forward—including the PR&D reactor development project outlined above. Going forward, the team was going to be challenged even further by having to maintain spare parts inventory for ongoing support of the reactor. It thus became apparent that BMS needed to identify a partner to manage the manufacture, distribution, and parts inventory associated with the MiniBlock. In addition, the MiniBlock offered opportunities for workflow automation that far exceeded the capacity of the internal development team. Although the MiniBlock offered some immediate technology advantage to BMS, it was felt that eventually parallel reactors of some sort would become common in drug discovery laboratories. By commercializing the BMS-designed reactor, there was an opportunity to drive the creation of a standard while simultaneously eliminating the burden of manufacture and support. The alternative was to maintain an increasingly costly internal manufacture and support infrastructure while the industry eventually adopted some other standard that would make the proprietary internal device obsolete. Facing these options, commercialization through a partner in the laboratory equipment business became the clear choice. After discussions with a number of potential partners, BMS eventually entered into an agreement with MT to commercialize the MiniBlock reactor.
From a vendor's perspective, there are significant advantages in partnering with a pharmaceutical company. Product ideas that originate from chemists and other scientists who are actively engaged in the drug discovery process tend to be very different from those conceived by instrument manufacturers. This originates from the fact that instrument companies are often technology and capability focused, resulting in products that may not necessarily reflect the scientists' needs accurately. Often, these products may lack key features that would be crucial to a user but that at the same time may seem unnecessary to anyone who has not used them in real-world situations. When products originate from users, such as pharmaceutical chemists, the entire product concept is driven by users' needs to improve the way that they carry out their work. In this case, the technology required to do the job only comes in at a later stage, and as a result, any features built into the product tend to be relevant to real-world use of the equipment. Instrument companies are aware of the shortcomings of designing products without significant customer input and try to address this issue by having close and frequent customer contact, as well as alpha and beta testing of product concepts. Entering into a commercialization agreement with a customer offers vendors the opportunity to bypass these steps and to offer products that have already been proven in actual applications.
At the time of commercialization discussions with BMS, MT's range of products for drug discovery largely consisted of custom robotics and automation products, most of which were related to chemistry and synthesis. All of these products involved an automation platform in the form of an XYZ workstation built on a chassis. These products could, to varying degrees, be customized to match a client's specific needs, and as such, they involved a high degree of engineering, which resulted in fairly long lead times and a high selling price. Products for synthesis applications included devices for heating, cooling, and shaking, as well as more sophisticated chemical synthesizers, which incorporated robotic functions such as picking and placing of containers, reagent dispensing, weighing, mixing, and so forth. The concept was to carry out as many of the steps in synthesis as possible, allowing the chemist a great deal of walk-away time. A number of other companies were also offering automated synthesizers to the rapidly growing parallel synthesis community.
A drawback of highly automated systems was their lack of flexibility in a dynamically changing customer environment that was in the process of embracing combinatorial chemistry and parallel synthesis in earnest. Laboratories were constantly reevaluating their synthesis techniques (loose-resin solid phase vs. trapped-resin solid phase vs. solution phase), as well as their general approach to library synthesis. As a result, synthesis parameters such as library size, sample throughput, and sample size were changing beyond the capabilities of the relatively inflexible automated equipment that was available at the time. Another limitation of highly centralized automation was that access to the instrument was by definition limited to a relatively small number of users. Because of the complexity and price of such systems, it would be impractical to provide them in large enough numbers for use by the majority of chemists. Hence, they tended to be tools for specialized high-throughput lead discovery laboratories, whereas the majority of chemists in lead optimization groups were still using traditional non-parallel tools such as round-bottom flasks. Although this divergence in methodology had some merit in terms of the goals of these diverse groups (Table 1), pharmaceutical companies were striving to increase productivity by cross-pollinating parallel-synthesis techniques into traditional lead optimization groups. This task would only be feasible by utilizing simpler, less costly, and more flexible tools for synthesis.
Divergent requirements for lead discovery and lead optimization

The requirements of lead optimization thus heralded the advent of “personal” synthesis tools, which were modular tools that could be used by any chemist involved in parallel chemistry. The MiniBlock synthesizer developed by BMS was an example of such a tool. It promised easy manual synthesis, while being compatible with other automation devices such as liquid-handlers and parallel evaporators. This provided a perfect opportunity for MT to complement its line of automated synthesis support products with a modular tool that had the potential of having a very wide appeal in the drug discovery community. The MiniBlock had the potential of lowering the automation barrier for medicinal chemists by allowing them to embrace automation one step at a time. Indeed, BMS's own experiences suggested significant productivity improvements through the use of personal parallel-synthesis technology. 65
Once the licensing agreement had been signed, the first commercial goal was to offer the existing product and accessories in such a way that the customer could painlessly begin adopting the technology. Since the MiniBlock had originally been designed to meet the very specific requirements found in the BMS environment, a number of accessories were quickly added to broaden the appeal to a more general laboratory environment. As an example, BMS was making use of a high-capacity, six-reactor incubation platform to maximize the number of compounds that could be synthesized in parallel. For many other customers, however, a smaller, entry-level version was attractive because of its lower cost and smaller footprint.
As a result, a compact two-place incubation platform was introduced, which significantly reduced the barrier to entry into smaller labs.
Because of its high degree of versatility, low cost, and timely introduction, the MiniBlock became a highly successful product within a mere 12 months of its commercialization—so much so that production often strained to keep up with demand. At the time, many customers were asking for variants of the original reactors, including larger volume formats and versions that would be more “medchem-friendly.” To reduce product development time, MT made an exception to the classical organizational structure by teaming up Marketing, Product Engineering and Chemistry staff into a single multi-functional unit. This close relationship, combined with regular dialogue and advice from BMS scientists, allowed for rapid, iterative design cycles, which would have been improbable within a conventional organizational chart. In this sense, the notion of rapid iterative design begun at BMS continued into the commercialization stage at MT.
Soon after, the established 48 + 48 format was supplemented with three large-volume versions of the reactor in 24-, 12-, and 6-position arrays for lead optimization, medicinal chemistry, and process chemistry applications. Another significant capability of the MiniBlock synthesizer was workup of reaction products by using SPE resins. This was made possible through a simple stacking arrangement, whereby the synthesized products were passed from one MiniBlock into a second reactor containing SPE media. 66 –68 Other commercial innovations included a simple-to-use manual resin-dispensing device that could simultaneously deliver powders to all reaction tubes at the same time. Staying true to the original modular concept of the product line, all these new additions were fully interchangeable, thereby minimizing the risks of obsolescing initial investments in the product line and allowing cost-efficient manufacturing through economies of scale. The price of reactor components was subsequently reduced, giving more customers access to the technology, which in turn increased the volume of manufactured parts.
Although the MiniBlock had proven to be a successful tool for solid-phase and mixed-phase synthesis, many customers had expressed interest in applications that were carried out purely in solution phase. Interestingly, these needs were being expressed by two distinct groups: lead optimization chemists in drug discovery and reaction optimization chemists in process research. Even though their respective chemistries and applications were quite different, they had many commonalities in terms of their requirements, namely larger reaction volumes, better agitation, and the ability to run reactions under reflux conditions. Fortuitously, BMS had just completed the final phase of their development of the second-generation solution-phase block for process research. Once again, BMS and MT entered into a licensing agreement, allowing the latter to develop and commercialize the technology.
Although BMS had primarily developed the solution-phase block for its process chemists, MT AutoChem made a business decision to introduce the resulting product first to discovery chemists and then to process chemists. Because the BMS design incorporated a recirculated base, it required the availability of a circulating bath for heating and cooling. For many discovery chemists, recirculators were not readily available, and, as a result, the first commercialized product was instead designed to be placed on a conventional hotplate stirrer, to which discovery chemists had easy access. The new commercialized product, named the XT, was thus launched to discovery chemists, alongside the original MiniBlock (Fig. 10). Once again, the new product became successful within a short period of time, providing access to parallel chemistry for traditional chemists, who were more accustomed to using round-bottom flasks. After the success of the XT, MT collaborated with BMS to launch the process version of the block, named the XT-Plus, which, as a closer relative of the original BMS solution-phase block, incorporated a recirculating base. The new family of personal reaction blocks, comprising the MiniBlock, the XT, and the XT-Plus now provided a wide range of solutions for applications ranging from lead generation, through optimization, to process screening.

Commercialized XT synthesizer.
As BMS had found, the MiniBlock and XT families successfully addressed the synthesis bottleneck in many laboratories, but there was still the issue of all the supporting activities such as weighing, reagent preparation, dispensing, and so on. With the MiniBlock and XT lines firmly established as the leading product for parallel synthesis, the commercial focus at MT was repositioned to providing affordable workflow automation devices that could be used to support these reactors. This new concept, the
Summary
The MiniBlock synthesizer family began as an internal development project meant to meet an immediate technology need within a large pharmaceutical company (BMS). By adopting a rapid iterative design philosophy, the team was able to meet immediate business production needs while simultaneously driving toward a final design that was optimized for the intended application—in this case, parallel synthesis. The burden of maintaining production and support for a proprietary internal technology led BMS to seek a commercial partner (MT). After commercialization, the partners worked together to extend the product line. MT focused on new features to appeal to a wider user base (larger volume formats, temperature control and agitation accessories, and so on), while BMS focused on optimizing a companion reactor for an entirely new application (the XT-Plus).
The result of this uncommon collaboration is that the MiniBlock and XT families are now used by almost every one of the top 30 major pharmaceutical companies in the world. Many companies in more than 20 countries have based their compound synthesis workflow around these platforms, confirming the applicability of the reaction blocks beyond the confines of a single pharmaceutical company. The collaboration and the reactor have been acknowledged with several industry awards including the 1999 ALA Achievement Award from the Association for Laboratory Automation and the 2003 Thomas Alva Edison Patent Award from the Research and Development Council of New Jersey. In addition, they have been featured in a number of industry trade publications. 73 –77 We believe that this collaboration may be a useful model for future technology innovation and adoption in the laboratory environment.
Acknowledgment
The authors would like to thank the many individuals at both Bristol-Myers Squibb and Mettler-Toledo AutoChem who contributed to the success of this project. While too many to name individually, this group includes the extended engineering team at BMS who participated in fabrication, production management and quality control; external collaborators such as KPMC fabrication, A. L. Miller fabrication, and United Chemical Technologies injection molding; the Combinatorial Chemistry and Medicinal Chemistry teams atBMSwho evaluated many prototype designs and provided invaluable feedback; the Process Research & Development team at BMS who also provided significant design feedback; the Engineering, Marketing, and Management teams at MT who enabled implementation of the commercialization plan; the BMS Legal and External Science & Technologies teams who developed the licensing agreement; the legal staff at Epstein, Drangel, Bazerman & James LLP who helped secure patent protection for these inventions; and finally the management teams of BMS Drug Discovery and Process Research&Development who supported and encouraged this project
