Abstract
The tumour microenvironment is characterized by pro-inflammatory profiles, including interleukin-1β (IL-1β). This profile is generated by infiltrating macrophages following interactions with tumours or their components. The objectives of this study were to identify whether tumour exosomes could induce macrophage IL-1β production and the mechanism involved. Exosomes were isolated from ovarian cancer patients and ovarian tumour cells by chromatography and ultracentrifugation. Specific exosomal proteins were defined by mass spectrometry (MS) and confirmed by Western immunoblotting. Using macrophage-like THP-1 cells, induction of IL-1β release was investigated by ELISA. RGD peptides were used to block fibronectin binding by THP-1 α5β1 integrin. Exosomes isolated from ovarian cancer patients and from ovarian cancer cells were demonstrated, by MS and immunoblotting, to express fibronectin. Incubation of THP-1 cells with these exosomes induced pro-inflammatory cytokines, in particular IL-1β. Blocking of THP-1 binding of exosomal fibronectin with RGD peptides abrogated exosome-mediated IL-1β production and down-stream phosphorylation of Akt and c-Jun. Although cancer patients generally exhibit increased levels of IL-1β, the underlying mechanism is unclear. Here, tumour-derived exosomes are demonstrated to induce pro-inflammatory cytokine in macrophages including IL-1β, whose induction is mediated by fibronectin.
1. Introduction
Macrophages have been implicated in phagocytosis and tissue remodelling and production of cytokines and chemokines [1, 2]. Unlike many cells of the innate immune system, macrophages exhibit a high degree of plasticity and can differentiate depending on the signals in their environment [3]. Based on their receptors and cytokine production, macrophages can be defined as M1 or M2 macrophages. Pro-inflammatory M1, or classically activated, macrophages exhibit up-regulation of pro-inflammatory cytokines and chemokines, including TNF-α, IL-12, IL-6, CCL2 and IL-1β, in addition to increased production of reactive oxygen species and nitrogen intermediates [4]. In contrast, anti-inflammatory M2, or alternatively activated, macrophages are polarized by stimuli such as IL-4, IL-13, IL-10 or glucocorticoid hormones [4] and up-regulate scavenger, mannose and galactose receptors. They are IL-1 receptor antagonists and downregulate IL-1β and other pro-inflammatory cytokines [5].
The malignant behaviours of tumour cells are defined by the microenvironment, where macrophages represent the major population of immunologic cells infiltrating the tumours. These infiltrating macrophages are referred to as tumour-associated macrophages (TAMs). While initially it was believed that these macrophages exhibited anti-tumour activity, the infiltration of TAM into tumours, such as breast, ovarian, prostate and cervical cancers, has been correlated with poor prognosis and short survival [6]. This adverse function of macrophages has been attributed to their immunosuppressive nature [7]. Recent studies have demonstrated that TAMs promote tumour cell proliferation, invasion and metastasis, as well as angiogenesis, which is essential for tumour growth [8]. TAMs secrete a variety of cytokines, growth factors and enzymes, which promote tumour metastasis and angiogenesis, such as vascular endothelial growth factor, IL-8, epidermal growth factor, platelet-derived growth factor, tumour necrosis factor (TNF)-α and matrix metalloproteinases [9, 10].
Cytokines coordinate the immune response and are released in response to infection, injury, stress or trauma. Cytokines regulate differentiation and activation of various immune cells and coordinate inflammatory reactions, such as the induction of acute-phase proteins. The hyper-activation of pro-inflammatory cytokines and/or the impairment of anti-inflammatory cytokines can lead to pathological conditions, such as allergic reactions, sepsis and dysplasia. IL-1, TNF-α and IL-17 are prominent proinflammatory cytokines, whereas IL-10 and TGF-β are characterized by anti-inflammatory properties. The pro-inflammatory phase is characterized by the production of pro-inflammatory cytokines, including interleukin 1β (IL-1β) [11]. IL-1β is a potent multifunctional pro-inflammatory cytokine produced by monocytes and tissue macrophages. IL-1β promotes multiple events, such as attraction and activation of immune cells, stimulation of endothelial cell adhesion molecule expression, and enhanced expression of extracellular matrices and matrix-degrading enzymes.
Our previous work has demonstrated that exosomes derived from a first trimester extravillous trophoblast were capable of inducing the migration of monocytes and “educating” these cells to produce a proinflammatory cytokine profile, including IL-1β [12]. IL-1β induction by exosomes was mediated by a ligand-receptor interaction and was shown not to require exosome uptake. Other studies have shown that interactions between components of the extracellular matrix and macrophages lead to the production in pro-inflammatory cytokines [13]. Proteins shown to induce pro-inflammatory cytokines by macrophages include Thy-1, vitronectin, integrins, thrombospondin, myosin and fibronectin. Fibronectin is present in the extracellular matrix of most tissues, where it supports basic cellular processes including cell adhesion, migration, survival and growth. Fibronectin is a modular protein composed of independently folded domains (types I, II and III), which represent regions of amino acid sequence homology [14]. Within the extracellular matrix, fibronectin is polymerized and can be subjected to tensional forces generated by the cells that expose cryptic activities within the molecule, including binding sites for integrins and growth factors [15]. In general, fibronectins are a class of large glycoproteins with known adhesive activity for collagen, fibrin, heparin and cell surfaces and are associated with wound healing, platelet aggregation and clotting, and cancer metastasis [16]. A specific function in cancer biology has not been defined for fibronectin, due primarily to its ubiquitous expression in adult human tissues and plasma. Matsuura et al. identified a group of fibronectin isoforms, containing an O-linked, N-acetyl-galactosaminylated hexapeptide within the alternatively spliced type III connecting segment [17]. These unique fibronectin iosforms have been identified within several human tumours and developmental tissues. These isoforms are designated as oncofetal fibronectin and they appear to be distinct from plasma and cellular fibronectins based on molecular size and glycosylation patterns [18].
In the present study, we demonstrate the induction of IL-1β by cancer-derived exosomes and investigate the mechanism underlying this induction. The study addressed the expression of exosomal components linked with the induction of pro-inflammatory cytokines by macrophages. Based on MS identification of fibronectin on tumour-derived exosomes, the study investigated the blockage of the exosome-mediated events by RGD analogues and the molecular mechanism linked with the exosome-associated component(s) responsible for the induction of IL-1β.
2. Materials and Methods
2.1 Cell culture conditions
The UL-O cell line was established from the ascites of a patient with Stage III papillary serous adenocarcinoma of the ovary. UL-O cells were maintained in 75cm2 tissue culture flasks in 20ml Hyclone Roswell Park Memorial Institute (RPMI) 1640 culture media (ThermoScientific) supplemented with 2mM L-glutamine, 10% exosome-depleted foetal bovine serum (FBS), 1mM sodium pyruvate, 0.1mM nonessential amino acids and 100units/mL penicillin-streptomycin in a humidified incubator at 37°C with 5% CO2. FBS-associated exosomes were removed by ultracentrifugation for 15 hours at 100,000xg. The resulting supernatant was sterile filtered using a 0.22μm filter. THP-1 acute monocytic leukaemia cells were obtained from ATCC (Rockville, MD, USA) and were maintained in RPMI 1640 supplemented with 2mM L-glutamine, 10% FBS, 1mM sodium pyruvate, 0.1mM non-essential amino acids, 0.05mM 2-mercaptoethanol and 100 units/mL penicillin-streptomycin in a humidified incubator at 37°C with 5% CO2.
2.2 Exosome isolation from culture media and patient ascites
To isolate tumour exosomes from culture media, UL-O cells were utilized. UL-O cells were cultured for three to four days (approximately 85% confluent and greater than 95% viability based on trypan blue exclusion). The conditioned medium was centrifuged at 400xg for ten minutes to remove whole cells and this supernatant was then centrifuged at 15,000xg for 20 minutes to remove large microvesicles and apoptotic vesicles. The resulting cell free medium was concentrated by ultrafiltration using an Amicon stirred cell Model 8200 with a molecular weight cut-off membrane of 500,000 Daltons (Millipore, Billerica, MA). This concentrated material was then chromatographically separated using a Sepharose 2B column (2.5×30cm). The void volume fractions were pooled and centrifuged using a Ti90 rotor (Beckman) at 100,000 x g for one hour at 4°C. The pellet was resuspended in PBS and the vesicular protein concentration determined using the DC protein assay (Bio-Rad Laboratories, Hercules, CA) according to manufacturer's instructions. This vesicle preparation from UL-O cells was termed OEX.
To isolate exosomes from patient ascites, the ascites fluids were centrifuged at 400xg for ten minutes to remove cells and the supernatant was centrifuged at 15,000xg for 20 minutes to remove cell debris and apoptotic vesicles. The resulting supernatant was concentrated by ultrafiltration using an Amicon stirred cell with a molecular weight cut-off membrane of 500,000 Daltons (Millipore). This
concentrated material was then chromatographically separated using a Sepharose 2B column (2.5×30cm). The void volume fractions were pooled and centrifuged at 100,000 x g for one hour at 4°C. The pellet was resuspended in PBS and the protein concentration determined using the DC protein assay. To exclude the possibility of endotoxin contamination in the exosome preparations for both the culture and ascites-derived vesicles, a LAL assay (Genscript, Piscataway, NJ) was performed to quantify any endotoxin in the vesicle preparations. For the exosome preparations used in these studies, endotoxin concentrations were <0.2EU/ml, including insufficient levels of endotoxin for the observed activation effects. Additionally, OEXs were demonstrated to be negative for IL-1β mRNA using specific primers (SABiosciences) and protein using a commercially available IL1-β ELISA kit (e-Biosciences, San Diego, CA, USA).
2.3 SDS-PAGE and Western immunoblot analysis of cell culture lysate and culture and ascites-derived exosomes
Vesicle-derived proteins or proteins from total cellular lysates (30μg each) were resuspended in a Laemmli sample buffer. This suspension was boiled and loaded onto 10% SDS-PAGE gels. After electrophoretic separation, the gels were stained with Imperial Purple Protein Stain (Pierce Chemical Co, Rockland, IL) at room temperature for two hours and destained overnight in double distilled water. The protein bands were imaged using a PharosFx Molecular Imager and Quantity One Software (BioRad Laboratories, Hercules, CA).
For Western immunoblot analysis [19], 10% SDS-PAGE gels were electrophoretically transferred to nitrocellulose membranes (BioRad Laboratories) for one hour, followed by blocking with 5% non-fat milk in TBS with 0.05% Tween 20 (TBST) for one hour. Membranes were incubated overnight at 4°C with the following primary antibodies: anti-fibronectin (FN-clone TV-1), anti-CD63 (clone Y-18) and anti-β-actin (clone C-4) (Santa Cruz Biotechnology, Santa Cruz, CA). After the overnight incubation, membranes were washed three times with 1xTBST and incubated for one hour with HRP-conjugated anti-mouse or anti-rabbit IgG (BioRad Laboratories) at room temperature. Blots were developed using an Immun-Star HRP substrate developing kit (BioRad Laboratories) and exposing x-ray film. Two separate western blots were performed for each target. Densitometric quantification of the resulting films for IL-1β, caspase 1 and β-actin was performed using Un-Scan-it software (Silk Scientific, Orem, UT).
2.4 Dynamic light scattering analysis
Exosome preparations were diluted to ∼50μg/mL to yield an optimum scattering intensity for dynamic light scattering measurements, which were performed on a Malvern 4700 autosizer (Malvern Instruments Ltd., UK) employing a 20mW helium/neon laser (633nm), at a constant temperature of 25°C. Light scattering from the sample was detected by a photomultiplier tube place at 90° to the incident laser beam. The translational diffusion coefficient of the solutions was calculated from the time autocorrelation of the scattered light intensity and extracted from the correlogram using the method of cumulants applied in the proprietary Malvern software. The diameter of the exosomes were obtained from the application of the Stokes-Einstein equations [20].
2.5 Mass spectrometry and proteomic analysis of ascites-derived exosomes
Specific protein bands from SDS-PAGE gels of ascites-derived exosomal proteins were reduced, alkylated and trypsinized at 37°C overnight with shaking. Briefly, tryptic peptides were loaded onto a 1D capillary chromatography column comprised of a needle tip (100 × 365μm fused silica capillary with an integrated, laser pulled emitter tip) packed with 10cm of Jupiter 5μm RP300A (Phenomenex, Torrence, CA). Peptides were eluted, ionized and sprayed into the mass spectrometer using a 120 minute gradient from 7% acetonitrile to 80% acetronitrile (plus 0.1% formic acid) at a flow rate of 200nL/min. Spectra were acquired with a LTQ Orbitrap XL Linear Ion Trap Mass Spectrometer (Thermo Fisher Scientific, Waltham, MA). During LC-MS/MS analysis, the mass spectrometer performed data-dependent acquisition with a full MS scan between 300 and 2000m/z followed by MS/MS scans (35% collision energy) on the six most intense ions from the preceding MS scan. Data acquisition was performed using dynamic exclusion with a repeat count of a one and three minute exclusion duration window.
The acquired mass spectrometry data were searched against a translated human genome database (Human-RefSeqXPs) using the SEQUEST algorithm assuming fixed modification of cysteine (+57 for carabmidomethylation) and variable oxidation of methionine (+16 to methionine). Database analysis was performed with SequestSorcerer (Sage-N Research, San Jose, CA). High-probability peptide and protein identifications were assigned from the SEQUEST results using the ProteinProphet (http://tools.proteomecenter.org/software.php) and SageN Sorcerer statistical platforms. These data were loaded onto Scaffold 3 proteomic analysis software to qualitatively and quantitatively compare individual LCMS analyses and provide approaches to functional annotation of data.
2.6 Cytokine antibody array
To define the effects of tumour-derived exosomes on macrophage chemokine and cytokine profiles, their production by THP-1 cells was quantified with duplicate arrays, each having duplicate spots for each cytokine using Proteome profiler™ Human Cytokine Antibody Array Panel A Arrays (R&D Systems, Minneapolis, MN) according to the manufacturer's instructions. THP-1 monocytic cells were incubated with 100μg/ml tumour-derived exosomes or were untreated for 20 hours. The cytokine array membranes were incubated with 1ml of conditioned media from each sample, diluted 1:3 and 15μl of Cytokine Array Panel A detection antibody at 4°C overnight. The membranes were then washed three times with 20ml of 1x wash buffer and incubated with horseradish peroxidase-conjugated streptavidin (1:2000-dilution). After 30 minutes, the membranes were washed thoroughly and exposed to a chemiluminescent peroxidase substrate for five minutes in the dark before imaging. Membranes were exposed to x-ray film (Research Products International, Mt Prospect, IL). As per the manufacturer's package insert, the cytokine array data on developed X-ray film was quantitated by scanning the film on a transmission-mode scanner and analysing the array image file using image analysis software, Un-Scan-it gel digitizing software version 6.1 (Silk Scientific Corporation, Orem, UT). Positive controls at three spots were used to identify membrane orientation and to normalize the results from different membranes. For each spot, the specific pixel level was determined by subtracting the background pixels from the total raw pixel levels. To quantify relative change in cytokine levels between samples, the average background-subtracted mean spot pixel densities of the pair of duplicate spots representing each cytokine was determined for each condition. To facilitate further analyses, all spots in the arrays were quantified and their specific intensity values were obtained by subtracting the background intensity. Only differences in cytokine levels that were ≥ two-fold compared to controls were considered significant.
2.7 ELISA determination of IL-1β release
For ELISA determination of IL-1β human THP-1, cells (1×106 cells/well) were incubated with exosomes at 100μg/ml for six hours. For the ELISA analysis, 1mM ATP was added for 30 minutes to induce mature IL-1β release. Culture supernatants for the treated human THP-1 cells were harvested and the IL-1β levels were measured using IL-1β ELISA kit (eBiosciences, San Diego, CA) according to the manufacturer's instructions.
2.8 Co-Culture of THP-1 monocytes with integrin function inhibitory peptides
To assess IL-1β production, THP-1 cells (5×106 cells/well) were cultured in RPMI 1640 supplemented media (as previously described) alone, co-cultured with 100μg/mL UL-O culture-derived exosomes (OEX), or co-cultured with 100μg/mL UL-O culture-derived exosomes (OEX) plus peptide mimics of fibronectin binding (Sigma-Aldrich, St. Louis, MO) at 0.5mg/mL for 30 minutes. Peptide mimics include: a negative control peptide (Gly-Arg-Ala-Asp-Ser-Pro-Lys) that does not block integrin binding (designated as CP), an antagonist peptide (Gly-Arg-Gly-Asp-Ser) of integrin function (designated as
2.9 Kinetics of THP-1, OEX and integrin function inhibitory peptide co-cultures
THP-1 cells were unstimulated, stimulated with OEX, or stimulated with OEX + peptide mimics of fibronectin binding (as previously described) for 0, 30, 60, 120, or 240 minutes. Total intracellular protein extracts were prepared by adding 100μL of RIPA buffer plus protease and phosphatase inhibitors/106 cells (ThermoScientific). The extracts were incubated on ice for 30 minutes and centrifuged at 12,000rpm for ten minutes. Protein concentration was determined using the DC Protein Assay (BioRad Laboratories). Cellular protein (30μg) was added to Laemmli reducing buffer and boiled for five minutes. Samples were electrophoresed at 100V for 1.5 hours and the resulting gel was transferred to nitrocellulose membranes (BioRad Laboratories) at 98V for one hour on ice. Membranes were blocked with 5% non-fat dry milk in 1xTBS + 0.05% Tween 20 (1xTBST) for one hour and incubated on a rocker overnight at 4°C with anti p-Akt2 (clone B5), anti pser73 c-Jun and anti-β-actin (clone C-4). All antibodies are from Santa Cruz Biotechnology. After an overnight incubation, membranes were washed three times with 1xTBST and incubated at room temperature for one hour with HRP-conjugated anti-mouse or anti-rabbit IgG (BioRad Laboratories). Subsequently, membranes were developed using Immun-Star HRP Chemiluminescent Kit (BioRad Laboratories) and visualized on Biomax MR film (Kodak). Western blot images for p-Akt2, pSer73 cJun and β-actin were digitized and relative change in expression of protein in comparison to the control was quantified. Statistical analyses were performed using the t-test and calculated using Graph Pad Prism-5.01 graphing and statistical software. A p-value of <0.05 was considered statistically significant.
2.10 Statistical analysis
Comparisons between exosome-treated and untreated cells were performed using a t-test or one-way Anova with Bonferroni's multiple comparison test, calculated using GraphPad Prism-5 graphing and statistical software. A p value of <0.05 was considered statistically significant.
3. Results
3.1 Characterization of vesicles derived from ascites and cell culture
Assessment of vesicles derived from both in vivo and in vitro by SDS-PAGE (Figure 1A) revealed similar composition of the major protein components. These protein bands ranged from 250kD to 20kD. The electrophoretic protein pattern for vesicles isolated from patient UL-L and from culture media of UL-O cells were virtually identical. Vesicles derived from patient UL-TB were similar to vesicles from UL-L with additional bands at 250kD and 150kD. The size of these vesicles was then analysed using Dynamic light scattering (Figure 1B)
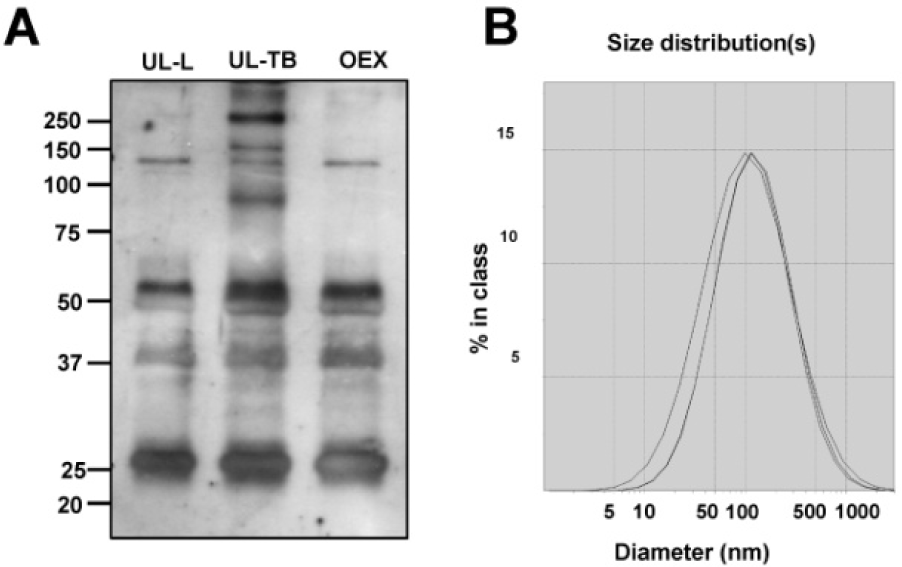
Protein profiles obtained from SDS-PAGE of vesicles isolated from patients UL-L and UL-TB ascites and from culture media from UL-O (OEX). Panel A: Imperial purple stained gel of UL-L and UL-TB ascites derived vesicles and OEX vesicles. Panel B: Dynamic light scattering size determination of vesicles isolated from cell cultures of UL-O.
Vesicles isolated from the conditioned media of UL-O cells demonstrated a mean diameter of 112.6 ± 9.0nm. This mean diameter size is within the general size range described for exosomes and is consistent with our recent findings with Nanoparticle Tracking Analysis [21].
3.2 Presence of fibronectin associated with patient ascites-derived vesicles
The protein composition of the vesicles isolated from the ascites of ovarian cancer patients was defined by MS sequencing. Proteins from
These vesicles were separated by SDS-PAGE. To identify vesicular proteins, bands were cut from the gel and subjected to MS sequencing. Three specific bands produced clear sequences with high ion scores (Figure 2). Of these 3 bands, band one was identified as fibronectin.
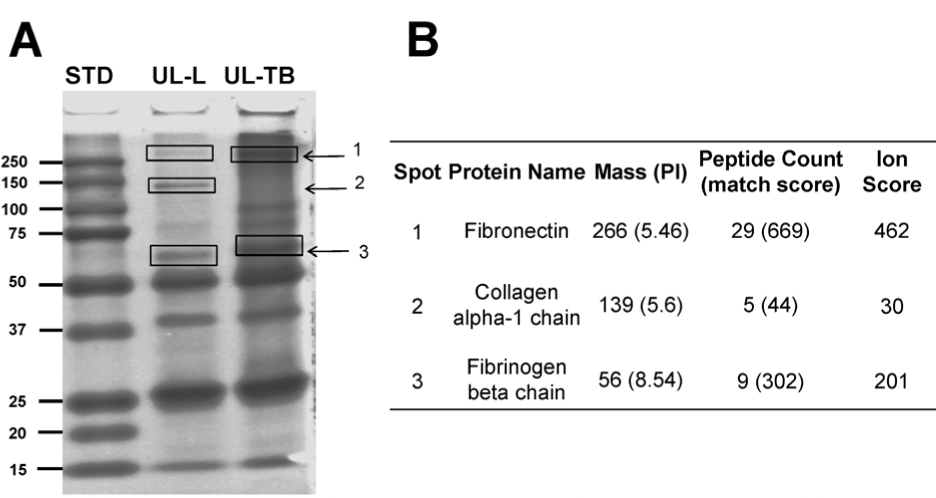
Mass spectrometric determination of specific proteins associated with patient-ascites derived vesicles. Panel
Since previous studies have demonstrated the role of tumour cell-associated fibronectin in the induction of IL-1β, the presence of fibronectin was confirmed on vesicles isolated in vitro from conditioned media of UL-O cells (OEX, Figure 3A) and in vivo from ovarian cancer patients (UL-L, UL-O and UL-TB, as well as normal control, Figure 3B). These results confirm the enhanced presence of fibronectin on these tumour-derived vesicles. In addition, specific markers were evaluated between UL-O cells and their corresponding vesicles (OEX, Figure 3A) using Western immunoblotting. These results also demonstrated the enriched presence of CD63 on the vesicles, establishing their identity as exosomes.
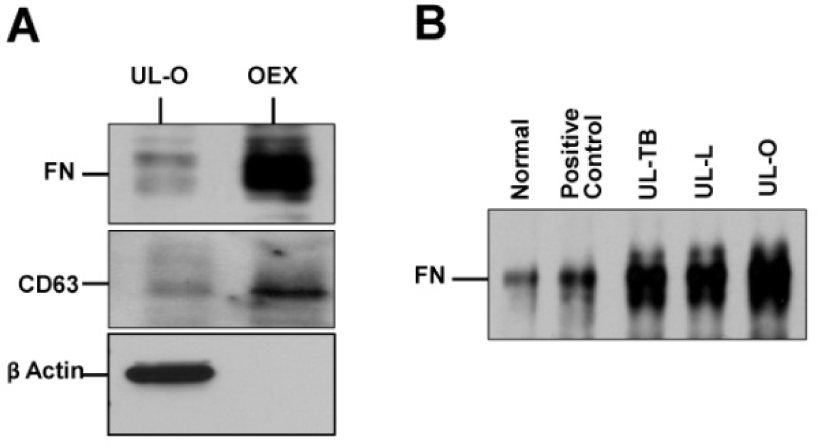
Western immunoblot identification of specific protein markers on cells and corresponding extracellular vesicles. Panel A presents markers associated with UL-O cells and extracellular vesicles released by UL-O cells (OEX). Panel B presents presence of fibronectin with extracellular vesicles isolated from normal human AB serm (normal) and three ovarian cancer patients.
3.3 Exosome induction of IL-1β release by human macrophages
Since the presence of tumours has been linked with the induction of a pro-inflammatory microenvironment. The initial question was whether exosomes derived from an ovarian tumour would induce the THP-1 cells to produce a pro-inflammatory profile. Using the Proteome profiler Human Cytokine Antibody Array Panel A Array, the induction of multiple cytokines/chemokines by tumour-derived OEX was evaluated (Figure 4). When THP-1 monocytic cells were incubated with 100μg/ml tumour-derived exosomes, 14 of the 36 cytokines/chemokines were induced by greater than two-fold. Of these, GROα, IL-1α, IL-1β, IL-8, IL-23, MCP-1 (CCL2), MIP1β and TNFα were induced by more than 20-fold (versus the control treated). Of particular interest was that IL-1β was induced by 350-fold following exposure to tumour-derived exosomes.
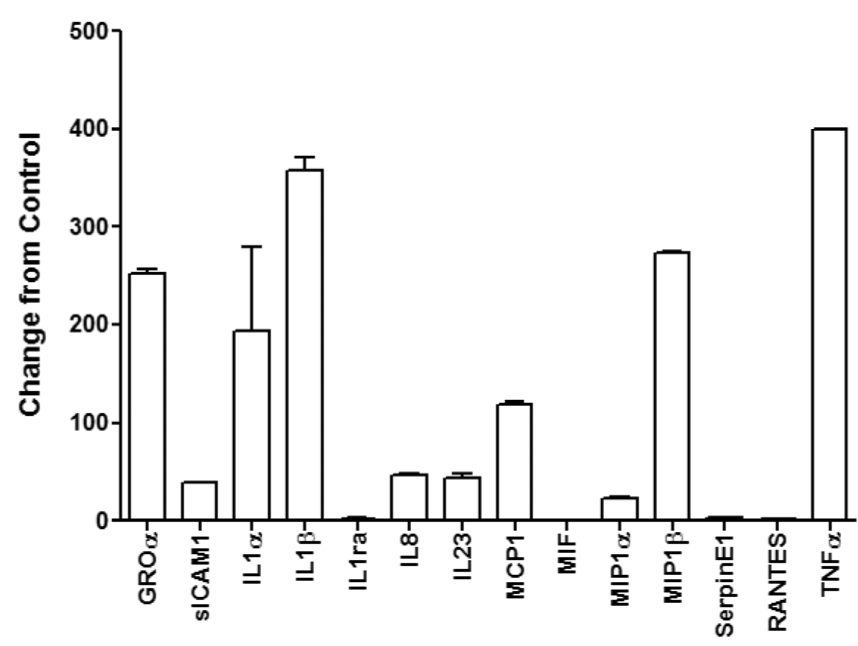
Exosome induction of cytokines/chemokines in THP-1 cells, incubated with 100μg/ml OEX (derived from UL-O ovarian tumour cells) for 20 hours. The plot presents the average background-subtracted mean spot pixel densities of the pair of duplicate spots representing each cytokine, divided by the value for control THP-1 cells. Means and standard deviations from two independent studies are shown Only values greater than two-fold were considered significant and shown on the graph.
Based on this major induction of IL-1β, the exosome component that might mediate this pathway was evaluated. Based on our previous mass spectrometric evaluation of exosomal components, the potential role of fibronectin in this exosome-induction was analysed. Fibronectin has been reported to utilize an RGD (Arginine-Glycine-Aspartic acid) binding motif for its binding to integrins (specifically α5β1) localized at the plasma membrane of macrophages to induce IL-1β production. To assess the role of exosome-associated fibronectin on IL-1β induction, commercially available RGD sequence mimics were used. Incubation of OEX with THP-1 cells resulted in the production and release of IL-1β. The negative control mimic peptide (CP), which does not block integrin binding of fibronectin, was included as a negative control. In Figure 5, CP was observed not to exhibit any effect on IL-1β production or release. In contrast, the two mimic peptides (ANT and BFCV), known to inhibit fibronectin binding to integrins, significantly suppressed IL-1β release by THP-1 cells (p<0.001). In contrast to previous studies using trophoblast-derived exosomes, IL-1β production and release is induced by tumour-derived exosomes in both the absence and presence of ATP.
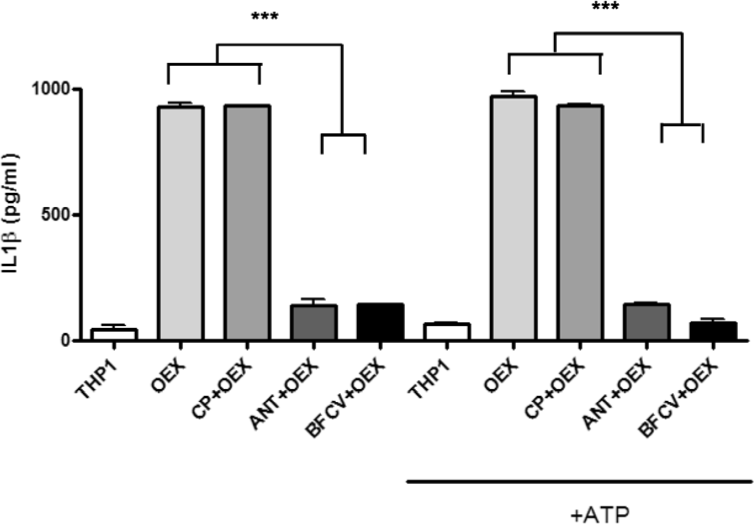
Exosome-associated fibronectin mediates the increased release of IL-1β. Human THP-1 were cultured in culture media only (THP) or pre-cultured with the various RGD mimic peptides at 0.5mg/ml for 30 minutes. OEX (exosomes from UL-O cells) were added at 100μg/ml for 6 hours, followed by 30 minutes with or without ATP (1mM) at 37°C. Supernatants of cultures were removed to determine the quality of IL1β protein released by ELISA. Means and standard deviations from three independent studies are shown with p-values calculated using Student's t-test. *** indicates level of significance (p < 0.001).
The IL-1β promoter region contains AP1 and c-Jun transcription factor binding sites. Thus, to define the kinetics and mechanisms of exosome induction of IL-1β, the time course of phosphorylation of Akt2 and c-Jun in the presence and absence of the RGD mimics was investigated (Figure 6). In untreated THP-1 cells, no phosphorylation was observed in Akt2 and c-Jun phosphorylation showed a decreasing trend in phosphorylation. Addition of OEX resulted in a time-dependent phosphorylation of both Akt2 and c-Jun. Pre-incubation with the negative control peptide (CP) did not significantly alter the time-dependent phosphorylation. In contrast, both inhibitory mimics (ANT and BVCF) suppressed the phosphorylation of both Akt2 and c-Jun to background levels.
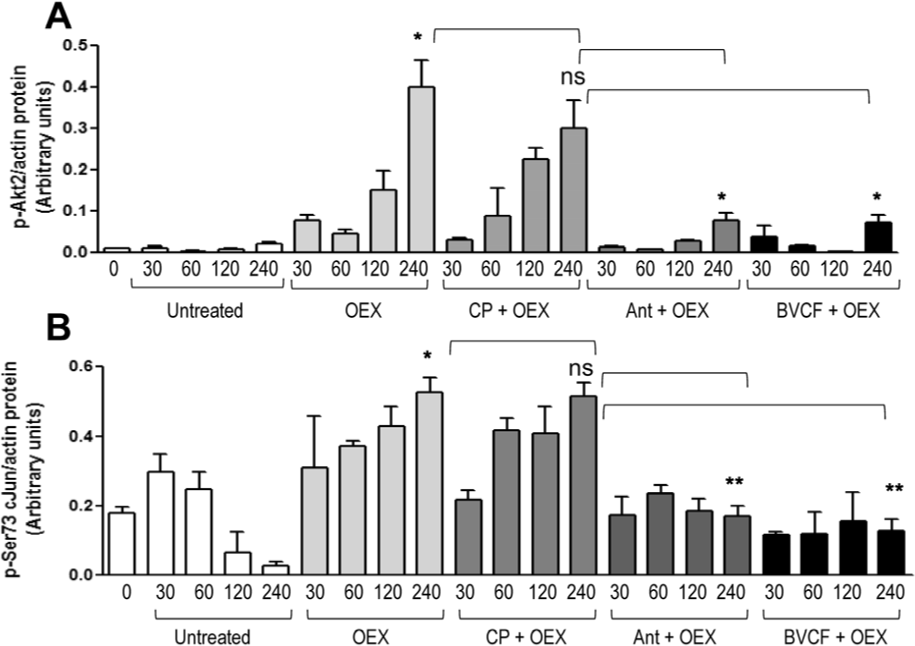
Phosphorylation of Akt2 and cJun, following exposure to OEX. Human THP-1 were cultured in culture media only (THP) or pre-cultured with the various RGD mimic peptides at 0.5mg/ml for 30 minutes. OEX were added at 100μg/ml for 0, 30 60, 120 and 240 minutes and THP-1 cells were lyzed and levels of the phosphorylated Akt and c-Jun were defined by Western immunoblotting. Means and standard deviations from three independent studies are shown are shown with p-values calculated using Student's t-test (p value significant p < 0.05). The bar at the 240 minute time point following OEX treatment was compared to untreated and the peptide mimic treated samples were compared with the OEX treated. * indicates p<0.001, ** indicated p<0.05, ns= not statistically significant.
4. Discussion
While previous studies have demonstrated that tumour-derived exosomes can directly suppress activation and proliferation of CD4 and CD8 lymphocytes [22] and induce T regulatory cells [23] and myeloid derived suppressor cells [24], this study focuses on their consequences for macrophages. Macrophages are critical to both the innate and adaptive immune response, as they rapidly respond to changes within their microenvironment. In general, inflammation from pathogens or tissue damage can activate resident macrophages to initiate or amplify the production of pro-inflammatory cytokines and other inflammatory mediators [25]. This current study addresses the mechanism underlying exosome-mediated induction of IL-1β by macrophages, observed in cancer. In vitro, macrophages can be phenotypically polarized to the M1 state by treatment with IFN-γ and inducers of TNF-α, such as lipopolysaccharide (LPS) [26]. M1 macrophages produce pro-inflammatory cytokines and chemokines, including TNF-α, IL-12, IL-6, CCL2 and IL-1β [4, 5]. Elevated levels of IL-1β are present in M1 polarized macrophages due to activation of the NF-κB and MAPK pathways [27], while no IL-1β protein is found in M2 polarized macrophages [28].
IL-1β exhibits profound effects on immune cell function during inflammation, resulting in investigations focused on factors that control IL-1β expression. Stimuli known to regulate IL-1β production in vitro include bacterial lipopolysaccharide, adherence to plastic, certain viral infections such as cytomegalovirus, phorbol myristate acetate, macrophage ingestion of asbestos fibres and certain cytokines, including tumour necrosis factor-α, granulocyte-macrophage colony stimulating factor and IL-1 itself [29]. While these factors are important in the regulation of IL-1β expression, the mechanisms by which they modulate IL-1β expression in vivo remain unclear. Extracellular matrices (ECM) are also capable of stimulating the expression of IL-1β [30]. Fibronectin is highly expressed in injured tissues and appears to be positioned to modulate the expression of IL-1β in diseased tissues [30]. Since multiple components have been implicated in the induction of the pro-inflammatory microenvironment associated the cancer, the protein composition of the vesicles isolated from the ascites of ovarian cancer patients was analysed. The presence of fibronectin of vesicles isolated from cancer patients and from vesicles obtained from ovarian cancer patients was demonstrated by mass spectrometric sequencing and confirmed by Western immunoblotting (Figures 2 and 3). The ability of this exosome-associated fibronectin to induce the release of IL-1β was further investigated. Our studies demonstrate the exposure of THP-1 cells to tumour-derived exosomes induced the significant production and release of pro-inflammatory cytokines/chemokines, including of particular note IL-1β (Figure 4).
In vitro, fibronectin stimulates the expression of IL-1β mRNA and its translation into the 31kDa intracellular precursor protein, along with secretion of the 17kDa active form in human mononuclear cells [31]. This effect of fibronectin is mediated by the specific cell surface α5β1 integrin receptor, which activates poorly understood intracellular signals to induce IL-1β expression [32]. Fibronectin contains a sequence, termed Arg-Gly-Asp (RGD), which promotes its attachment to integrin receptors. Monocytes and macrophages have been shown to possess fibronectin receptors that recognize the RGD motif and mediate pro-inflammatory cytokine production. The effect of fibronectin has been shown to be dependent on binding of the RGD sequence of fibronectin to integrin receptors, as this effect could be inhibited by integrin receptor blocking peptides (anti-RGD sequence mimics) [33].
IL-1β post-translational processing and release is a highly regulated pathway utilizing a multiprotein complex, termed the capase-1 inflammasome [34]. IL-1β is initially synthesized as a 34kD precursor protein that is cleaved to the biologically active 17kD form by active caspase-1. Inactive pro-caspase-1 is constitutively present in both M1 and M2 macrophages and is activated within M1 macrophages by self-cleavage, occurring in the inflammasome complex. The nucleotide-binding domain and leucine-rich repeat receptor containing pyrin domain 3 (NLRP3) inflammasome can be activated by specific microbial motifs, by microbial toxins, by uric acid crystals and by extracellular ATP acting through the ATP-gated plasma membrane ion channel, the P2×7 receptor. Addition of extracellular ATP to macrophages can generally increase the release of mature IL-1β. In contrast to our previous study using trophoblast-derived exosomes, tumour-derived exosomes do not require addition of ATP to induce release of IL-1β (Figure 5). It is unclear whether these exosomes carry ATP or utilize a distinct pathway to induce the production and release of IL-1β. However, these possibilities are currently under investigation.
The IL-1β promoter region contains AP1 and c-Jun transcription factor binding sites that are critical to mediate certain pathologic conditions. Our results demonstrate that tumour exosomes induce IL-1β and produce phosphorylation of Akt2 and c-Jun (Figure 6). The addition of the RGD mimics resulted in the suppression of Akt2 and c-Jun phosphorylation, following addition of UL-O exosomes. These findings confirm the essential role of exosome-associated fibronectin in the regulatory events leading to the promotion of IL-1β production and release.
While other studies have investigated the role of fibronectin and other components of the ECMs on the induction of IL-1β, in the current study, we demonstrate IL-1β induction by fibronectin associated with tumour-derived exosomes. This study with ovarian cancer-derived exosomes demonstrates that these tumour exosomes also express fibronectin, which may account for the pro-inflammatory microenvironment observed in cancer. Exposure of macrophages to ovarian tumour-derived exosomes appears to “educate” these cells to M1 macrophages. These M1 macrophages produce pro-inflammatory cytokines and chemokines, including TNF-α, IL-8, MCP-1 (CCL2) and IL-1β. While enhanced levels of IL-1β characterize M1 polarized macrophages, IL-1β is absence in M2 polarized macrophages [24]. These findings may have implications for the potential use of exosome-associated fibronectin as potential biomarkers for cancers. Exosome-associated fibronectin may also serve as a therapeutic target for cancer, by targeting the fibronectin-induced pro-inflammatory environment essential for tumour growth and proliferation.
