Abstract
Activation of proinflammatory cytokines in seizures has been well characterized. However, role of cytokines in epilepsy and association with different clinical phenotype has not been well investigated. Reports on possible link between proinflammatory molecules and epilepsy are very limited. In this study, we performed a hospital-based case control study to investigate the association of plasma cytokines and their expression with different clinical categories of epilepsy. Patients admitted to Neurology Department of Renmin Hospital were enrolled in this study after clinical investigations. In all, 92 patients with temporal lobe epilepsy (TLE) and 45 with extra-temporal lobe epilepsy (XTLE) were included in this study. Furthermore, we included 86 healthy controls from the similar geographical population. Plasma levels of interleukin (IL)-6, tumor necrosis factor (TNF)-α, and IL-1β were quantified by enzyme-linked immunosorbent assay (ELISA). All plasma cytokines were elevated in TLE and XTLE compared to healthy controls (
Introduction
Epilepsy is a typical chronic disorder showing symptoms of unpredictable seizures with long-lasting convulsions caused due to disturbance in nerve cells of brain and has serious impact on patient’s behavior and attitude. Studies indicate that about 65 million people are affected with epilepsy worldwide, and this disorder constitutes about 1% of global burden of the disease. 1 According to International League Against Epilepsy (ILAE) guidelines, based on anatomical origin of seizures, epilepsy can be classified into temporal lobe epilepsy (TLE), parietal lobe epilepsy (PLE), occipital lobe epilepsy (OLE), or multifocal epilepsies based on electroclinical findings. 2 TLE constitutes an uncontrollable focal epilepsy syndrome, the seizures being originated from temporal lobe of brain, often associated with hippocampal atrophy and exhibits extensive neuronal loss. Seizures triggered in the rest of the brain (parietal and occipital regions) constitute extra-temporal lobe epilepsy (XTLE). Earlier reports suggested a significant association of inflammation induced during epilepsy with pathogenic outcome of the disease. Many proinflammatory signals such as cytokines, chemokines, prostaglandins, toll-like receptors (TLRs), and complement factors are rapidly induced during epilepsy. 3 Among different cytokines, neuroinflammation during epilepsy is mostly characterized by higher levels of interleukin (IL)-1β, IL-6, tumor necrosis factor (TNF)-α, and IL-17 secreted by activated astrocytes. 4 It has also been reported that IL-1β, IL-6, and IL-1Rα are associated with pathophysiological relevance in epilepsy. 4
Although many studies have been made to correlate the inflammatory status with neuropathology during epilepsy, no critical attempt has been taken to examine any biomarker(s) to differentiate TLE from XTLE. Finding accurate marker(s) to differentiate between TLE and XTLE is essential from clinical point of view to allow proper diagnosis followed by suitable treatment options. Cytokines have been used as biomarkers for many neurological diseases such as Parkinson’s disease, Alzheimer’s disease, and other central nervous system (CNS) disorders. 5 Among different cytokines studied during epilepsy, IL-6, IL-1β, and TNF-α have drawn attention in most of the clinical investigations to examine their role in mediating neuropathogenesis. Therefore, we tried to examine whether any of these cytokines can be used as a potential marker(s) to differentiate TLE and XTLE.
Materials and methods
Patients and controls
Patients admitted to Neurology Department of Renmin Hospital, Wuhan University, were included in this investigation. All patients were diagnosed in accordance with approved criteria of the ILAE. 2 Patients with infectious diseases, inflammatory disorders, autoimmune disorders, kidney or heart dysfunctions, and defect in CNS were excluded from this study. Based on clinical and different investigations such as seizure semiology, electrocardiography (ECG), and magnetic resonance imaging (MRI) of the brain, patients were divided into TLE and XTLE. Age- and sex-matched healthy controls (HCs; n = 86) were enrolled in this study, those free from history of seizure. From all patients, about 3 mL of intravenous blood was collected with ethylenediaminetetraacetic acid (EDTA) and plasma samples were separated by centrifugation at 3000 r/min for 20 min. All plasma samples were stored at −80°C till quantification of cytokines by enzyme-linked immunosorbent assay (ELISA). The study protocol was approved by Institutional Human Ethical Committee, and written informed consent was obtained from patients or their attendant. All patients were treated according to standard guidelines by National Institute of Health and Care Excellence with lamotrigine, topiramate, sodium valproate, and/or carbamazepine.
Cytokine quantifications
The stored plasma samples were thawed and levels of proinflammatory cytokines, namely, TNF-α, IL-6, and IL-1β, were measured by ELISA as instructed by manufacturer (eBiosciences). Inter- and intra-assay coefficients of variation for all cytokines are as follows: TNF-α: 7.4% and 6.0%; IL-6: 8.4% and 6.8%; and IL-1β: 8.1% and 6.7%, respectively.
Statistical analysis
GraphPad prism V7.04 was used for all statistical analyses. Baseline characteristics among patients or with HCs were compared by student’s t-test. D’Agostino-Pearson normality test was used to investigate distribution of baseline and cytokines data. Difference in mean plasma cytokines levels among different clinical groups was analyzed by analysis of variance (ANOVA) and followed by Tukey’s post test. Correlation of cytokines was investigated by Pearson rank coefficient. A
Results
Baseline characteristics of epileptic patients and controls
The demographic features of study subjects are mentioned in Table 1. A total of 137 subjects with epilepsy was classified into TLE (n = 92) and XTLE (n = 45) based on clinical phenotype. The mean age was comparable among both clinical categories. HCs (n = 86) were taken for comparison. Mean age and gender distribution among HC and epilepsy patients were comparable. The duration of TLE was slightly higher as compared to XTLE, although the difference was not statistically significant. Furthermore, a similar frequency of epileptic episodes (seizures per month) was observed between TLE and XTLE subjects. Generalized seizure was more prevalent in XTLE than TLE. Distributions of other seizure type were comparable among both TLE and XTLE. Furthermore, possible etiology of seizure (CNS infections, traumatic brain injury, and depression) was investigated in TLE and XTLE, and the distribution remained comparable (Table 1).
Demographic characteristics of patients and controls.

HC: healthy control; TLE: temporal lobe epilepsy; CNS: central nervous system; TBI: traumatic brain injuries; XTLE: extra-temporal lobe epilepsy; SD: standard deviation.
Plasma levels of proinflammatory cytokines in patients
We compared the plasma levels of TNF-α, IL-6, and IL-1β among three study groups (Figure 1). Although the levels of TNF-α were significantly higher in TLE and XTLE subjects as compared to HCs (

Plasma levels of proinflammatory molecules in epilepsy patients and controls. Plasma levels of (a) TNF-α, (b) IL-6, and (c) IL-1β were quantified by ELISA in enrolled subjects and compared among different clinical categories by analysis of variance (ANOVA) followed by Tukey’s post test. A
Correlation of proinflammatory cytokines
We examined the correlation of plasma levels of TNF-α, IL-1β, and IL-6 in patients to understand their association among each other. There was a strong correlation between IL-6 versus TNF-α (Figure 2(a)), IL-1β versus IL-6 (Figure 2(b)), and IL-1β versus TNF-α (Figure 2(c);
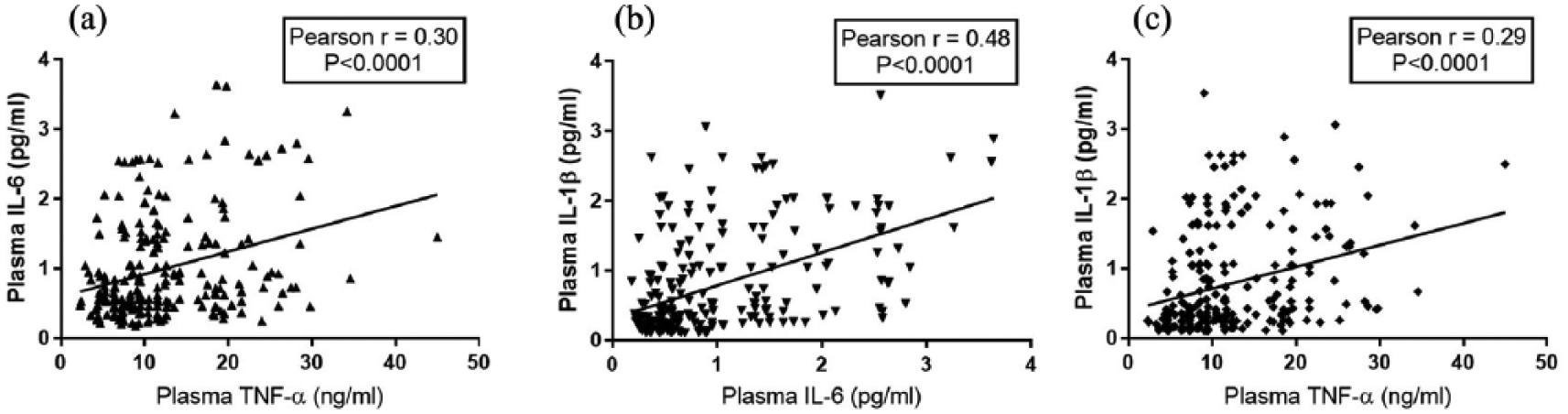
Correlation of plasma cytokines in epilepsy patients. Plasma cytokines (TNF-α, IL-1β, and IL-6) were quantified by ELISA. Correlation of plasma TNF-α with (a) IL-6 levels and (c) IL-1β was analyzed by Pearson rank correlation coefficient test. A positive correlation of TNF-α was noticed with IL-6 and IL-1β. (b) Furthermore, plasma IL-1β and IL-6 was also positively correlated with each other. A
Discussion
TNF-α, IL1-β, and IL-6—the three potent proinflammatory cytokines—play an important role in mediating pathogenesis for many diseases and are also associated with CNS disorders. These three key molecules, secreted by mostly microglia and astrocytes in brain, play a role as neuromodulators in CNS of healthy subjects and are also involved in inflammatory processes during neurological disorders like Alzheimer’s disease and Parkinson disease. 6 In our hospital-based study, we found elevated levels of these cytokines in plasma of patients with epilepsy as compared to HCs, and these findings are convergent with earlier reports. For instance, plasma IL-6 level was found to be higher in subjects with treatment-resistant epilepsy. 7 An increased basal level of IL-6 in plasma was observed in subjects with chronic localization-related epilepsy as compared to HCs. 8 Similarly, IL-1β and IL-6 levels were found to be significantly enhanced in plasma of patients during interictal period of epilepsy as compared to HCs. 9 Besides human studies, in an experimental epilepsy model, a significant upregulation in IL-1β, IL-6, and TNF-α levels occurred rapidly after seizures in hippocampus. 10
The major intention of our study was to compare levels of these three proinflammatory molecules between TLE and XTLE subjects, and we found elevated levels of IL-1-β and IL-6 in TLE subjects as compared to XTLE group among Chinese population. An inconsistent result was shown in earlier studies on plasma levels of IL-1β and IL-6 between TLE and XTLE patients. In a hospital-based study at Izmir, Turkey, a higher level of IL-1β in patients with TLE was observed as compared to XTLE, 11 whereas a prospective 2-year follow-up investigation in Finland revealed no significant difference in plasma level of IL-1β and IL-6 between two study groups. 12
It has been demonstrated that seizures induce cytokines during epilepsy. Levels of IL-6 and IL1-Rα cytokines in plasma and cerebrospinal fluid (CSF) are rapidly induced after onset of tonic–clonic seizures as well as generalized seizures 8 , but not IL-1β and TNF- α. 13 Few cytokines are also elevated in body fluids during febrile seizures. The frequency of seizures (seizures/month) in TLE and XTLE subjects was found to be 3.5 ± 2.9 and 3.7 ± 1.9, respectively, as observed in our studies, and we expect that the induction of cytokines in subjects with epilepsy is due to onset of seizures. Since there was no significant difference in seizure frequency between TLE and XTLE subjects, the enhanced levels of IL-1β and IL-6 in TLE patients cannot be due to higher occurrence of seizures in these subjects. Although the exact reason for upregulation of IL-6 and IL-1β is not known in our study, the assumption is, since TLE represents a group showing refractoriness to drugs, the higher levels of cytokines in these patients may be due to some active epileptic processes occurring in the brain. The role of cytokine other than IL-6, IL-1β, and/or different cellular factors cannot be ruled out. However, further studies are required to address this issue.
Previous studies indicate a minor role played by TNF-α in subjects with epilepsy. Studies showed no significant difference in levels of this cytokine in plasma or CSF of epileptic patients 24 h after acute tonic–clonic seizures or generalized seizures. 13 Other studies also failed to reveal possible role of TNF-α as the levels were comparable in CSF of patients with febrile seizures and that of HCs. 14 Interestingly, we found an elevated level of this cytokine in epileptic subjects as compared to HCs. Production of TNF-α from activated microglia or astrocytes in brain during neuroinflammation is adequately reported. 4 We, therefore, assume that higher levels of plasma TNF-α in epileptic subjects could be due to the onset of neuroinflammation mediated by activated cells of brain.
Defining biological markers to diagnose epilepsy is a critical part of research. An important study tried to find out possible biomarkers for diagnosis of epilepsy and quantified 14 different cytokines in plasma and CSF showed that plasma levels of IL-6, interferon gamma (IFNγ), IL-17α, and IFNλ3 as well as CSF IL-6, IL-17 α, and IFNλ3 can be used as potential biomarkers to detect severe epilepsy. 15 There are no specific marker(s) available to differentiate TLE from XTLE. Our observation on significantly higher levels of IL-6 and IL-1β in TLE subjects as compared to XTLE possibly suggests using these cytokines as biomarkers of the disease.
Footnotes
Acknowledgements
The authors would like to thank all patients and healthy controls for their volunteer enrollment in this study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
