Abstract
Background
Several mechanisms are involved in the development of resistance to therapy in locally advanced cervical squamous cell carcinoma (LACSCC). Studies have shown that CD44 and Lewis Y antigen (LeY) form a complex that is associated with chemoresistance, tumor invasion and metastasis. We assessed the role of CD44 and LeY in the outcome of LACSCC patients treated with different chemotherapy regimens.
Methods
126 LACSCC patients at FIGO stages IIB-IVA were selected from the GOCS database: 74 patients included in 3 different prospective phase II trials in the neoadjuvant setting (vinorelbine, docetaxel, ifosfamide-vinorelbine-cisplatin) and 52 patients treated with standard radiochemotherapy based on cisplatin (RCBC). Clinical data at baseline, disease-free survival (DFS) and overall survival (OS) were recorded. Univariate and multivariate Cox models were employed.
Results
Median age was 45.6 years (range: 24.9-80.5). Sixty-three and 47 tumors were CD44+ and LeY+, respectively. Tumors with expansive growth showed higher grade (p = 0.0024), mitotic index (p = 0.0505), tumor necrosis (p = 0.0191), LeY+ (p = 0.0034) and CD44+/LeY+ coexpression (p = 0.0334). CD44+ cells were present in 91.3% of patients with local recurrence (p = 0.0317). Advanced stage was associated with LeY+ tumors. Patients treated with RCBC had worse DFS and OS when their tumors expressed LeY (p = 0.0083 and p = 0.0137, respectively). Pre-treatment hemoglobin level, FIGO stage and tumor response remained the most significant prognostic factors in Cox regression.
Conclusions
In our cohort of LACSCC patients, the coexpression of CD44 and LeY was not associated with worse outcome. However, in the subgroup of patients receiving RCBC, LeY expression was correlated with shorter DFS and OS.
Introduction
Cervical cancer is one of the most frequent malignancies in women and a considerable cause of morbidity and mortality. Currently, cervical cancer is the fourth most common cancer among women globally (1). Each year approximately 500,000 women around the world are diagnosed with invasive cervical cancer and more than half of them die from their disease. Eighty percent of these deaths occur in developing countries. Therefore, it is essential for us to reach a deeper understanding of the biology of this disease in order to develop more effective therapies.
The development of resistance to chemo- or radiotherapy involves multiple mechanisms in locally advanced cervical squamous cell carcinoma (LACSCC). One of them could be the presence of a subpopulation of cells with regenerative abilities under cytotoxic stress. Such cancer stem or clonogenic cells tend to repopulate tumors during the course of chemo- or radiotherapy (2, 3). CD44 is a cell adhesion glycoprotein functioning as a transmembrane receptor for the extracellular matrix component hyaluronan (HA). It participates in epithelial cell-stroma interactions that are important in tumor invasion and metastasis (4). CD44 expression characterizes a subset of cancer cells with stem-cell-like properties and could be involved in cell adhesion-mediated drug resistance (CAM-DR) through interaction with its ligand HA (5, 6). It is known that CD44 is modified posttranslationally by glycosylation, which has been shown to influence CD44-mediated CAM-DR (7).
Recently, Gao et al (8) found that the difucosylated Lewis Y antigen (LeY) is part of the composition of CD44 and its increased expression is correlated with enhanced CD44-mediated cell adhesion and migration. Also, increased expression levels of LeY and CD44 were associated with elevated resistance to chemotherapeutic drugs such as platinum agents, taxanes, 5-fluorouracil, doxorubicin and mitomycin (9-10-11-12).
Although the effects of alternative splicing and posttranslational glycosylation of CD44 on its interaction with HA have been studied in several tumor types, few data are available on the effect of these alterations in LACSCC. The aim of this study was to evaluate the role of CD44 and LeY in the outcome of LACSCC patients.
Patients and Methods
Patients
One hundred and twenty-six women with a histologically proven diagnosis of squamous cell carcinoma of the uterine cervix treated at the Grupo Oncologico Cooperativo del Sur (GOCS) between January 1993 and December 2007 were included. Patients presented with International Federation of Gynecology and Obstetrics (FIGO) stages IIB to IVA.
Among the 126 patients, 74 participated in 1 of 3 different prospective phase II trials conducted by our group in the neoadjuvant setting: 23 received vinorelbine, 25 docetaxel (antimicrotubule agents), and 26 were treated with ifosfamide plus vinorelbine plus cisplatin (mixed agents). The remaining 52 patients received cisplatin at standard doses concurrent with pelvic radiotherapy. Eligibility criteria, clinical staging, response criteria, treatment strategy, toxicity evaluation as well as dose schedules, number of chemotherapy cycles and criteria for surgery or radiotherapy have been described in previous reports by our group (13-14-15).
Formalin-fixed paraffin-embedded tissues were obtained from 126 primary cervical carcinomas before treatment initiation. The hematoxylin slides of all cases were reevaluated to corroborate the original diagnosis and to classify the cases according to the original epithelial tissue. Samples with any of the following characteristics were excluded: excessive necrosis and bleeding, insufficient tumor material, poorly preserved tumor material, and those cases in which there were no corresponding paraffin blocks. Ten benign exocervical and endocervical tissue samples were included as a control group to compare the immunohistochemical reactivity of cervical cancer.
All procedures followed the World Medical Association Declaration of Helsinki (Finland, 1964) and its further modifications. Informed consent was obtained from all patients. This study was approved by the regional ethics committee of the province of Neuquén, Argentina (CAIBSH # 2520/11). The REMARK guidelines (Reporting recommendations for tumor MARKer prognostic studies) were followed (16). The clinical and histological information of the patients is summarized in Table I.
Clinical features and survival in 126 patients with LACSCC according to the investigated variables
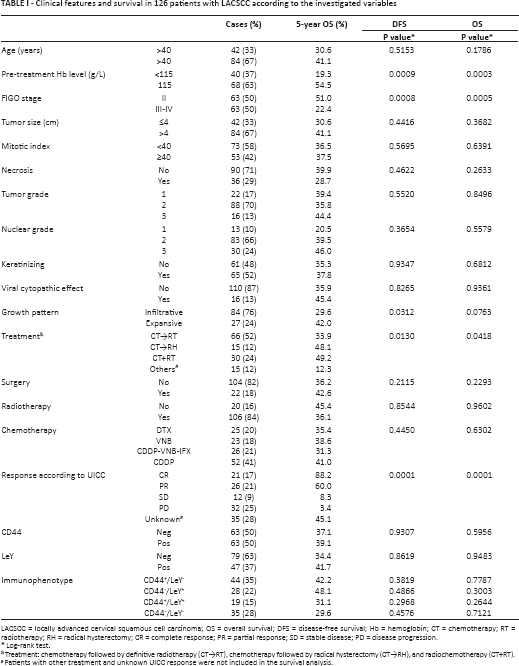
LACSCC = locally advanced cervical squamous cell carcinoma; OS = overall survival; DFS = disease-free survival; Hb = hemoglobin; CT = chemotherapy; RT = radiotherapy; RH = radical hysterectomy; CR = complete response; PR = partial response; SD = stable disease; PD = disease progression.
Log-rank test.
Treatment: chemotherapy followed by definitive radiotherapy (CT→RT), chemotherapy followed by radical hysterectomy (CT→RH), and radiochemotherapy (CT+RT).
Patients with other treatment and unknown UICC response were not included in the survival analysis.
Immunohistochemical Analysis
The immunohistochemistry technique was performed following standard procedures. All specimens were fixed in formalin and embedded in paraffin. Sections were deparaffinized in xylene and hydrated in a graded ethanol series; dewaxed sections were placed in methanol with hydrogen peroxide (3%) for 15 minutes to block endogenous peroxidase activity. After 3 washes with PBS, sections were blocked for nonspecific binding with 50 mL normal horse serum diluted 1:10 in 1% bovine serum albumin/PBS for 15 minutes and rinsed.
Prior to immunostaining with monoclonal antibodies (MAbs), tissues were treated with 10 mM sodium citrate buffer at 100°C for 5 minutes for antigen retrieval. Afterwards, the sections were incubated with 50 µL of the mouse MAbs against standard CD44 (clone DF1485, 1:200; Santa Cruz) and Lewis Y (clone A70-C/C8, 1:200; Santa Cruz) at room temperature 60 minutes in a moist chamber. After 3 rinses with PBS, the sections were incubated with peroxidase-conjugated anti-mouse/rabbit Igs (HiDef Detection™ HRP Polymer System, Cell Marque) for 60 minutes. After washing, slides were counterstained with hematoxylin, dehydrated in ethanol, washed with xylene and coverslipped with mounting media. Negative controls were performed by adding PBS instead of the primary antibody. A positive reaction against the antibodies in the study was represented by the presence of brown precipitation in the membrane or cytoplasm or in both cell compartments.
Evaluation of Staining
Specimens were examined with a light microscope by 2 independent observers (GG and AZ) who had no knowledge of the patient's clinical data. The antibody staining patterns were scored in a semiquantitative manner. Low-power images of sections were scored based on the staining intensities: negative (-), low (+), moderate (++) and strong (+++), using adjacent nonmalignant cells for reference. Normal tonsils were used as positive control for CD44 and colon cancer tissue was used as positive control for LeY, according to the manufacturer's recommendations. Negative controls for immunostaining were prepared through serial sections of selected tissue samples in the absence of the primary antibody.
Subsequently, a total of 5 high-power fields in series were selected from each slide for scoring of individual cells. The mean percentages of positively stained cells were calculated for each field. The percentage of positive cells was graded as follows: 0, no positive cells; 0.1, positive in less than 9% of cells; 0.5, positive in 10%-49%; 1, positive in more than 50% of cells. The pattern of reaction was classified as linear (membrane), cytoplasmic, and mixed (cytoplasmic with plasma membrane staining). Apical and non-apical staining, adjacent normal tissue, and focal or diffuse staining were all evaluated.
Different cutoff values according to previous reports, taking into account the intensity and percentage of CD44 and LeY, were tested. No meaningful results were found when considered as positive or negative the immunostaining of both antigens. For prognostic investigation and survival analysis, each individual immunophenotype pattern was evaluated: CD44+ (CD44-positive cells), LeY+ (LeY-positive cells), CD44+/LeY+, CD44+/LeY-, CD44-/LeY+ and CD44-/LeY-.
Statistical Analysis
The associations of CD44 and LeY expression with the variables studied were tested with nonparametric tests. For categorical variables, the Wilcoxon rank-sum test or Kruskal-Wallis test, including a Wilcoxon-type test for trend across ordered groups, was used.
Disease-free survival (DFS) was estimated from the date of diagnosis until the date of first tumor recurrence. Overall survival (OS) was calculated from the date of diagnosis until the date of death or last follow-up. The evaluation of treatment response was performed using the Union for International Cancer Control (UICC) criteria, the standard method used in solid tumors until 2000.
DFS and OS univariate associations with CD44 and LeY expression were evaluated employing the Kaplan-Meier estimator with the log-rank test. A multivariable Cox regression model addressed OS and DFS associations with CD44+, LeY+, CD44+/LeY+, CD44+/LeY-, CD44-/LeY+, CD44-/LeY-, age (≤40 vs. >40), pre-treatment hemoglobin level (<115 g/L vs. ≥115 g/L), FIGO stage (early II vs. advanced III-IV), tumor size (≤4 cm vs. >4 cm), mitotic index (<40 vs. ≥40), histological grade (low 1-2 vs. high 3), nuclear grade (low 1-2 vs. high 3), keratinizing (yes vs. no), viral cytopathic effect (yes vs. no), tumor necrosis (yes vs. no), and growth pattern (infiltrative vs. expansive).
All variables where normalized and significance was set at p<0.05. All calculations were done employing the Statistix v.8 software package.
Results
Patient Characteristics Associated with Prognosis
One hundred and twenty-six patients with LACSCC with a median age of 45.6 years (range, 24.9-80.5 years) were included. Sixty-three patients (50%) had FIGO stage IIB and the remainder had advanced stages (52 patients FIGO stage IIIB, 11 stage IVA). Fifty-six patients (44.4%) developed local recurrence and 71 patients (56.3%) died of their disease. In all serie studied, the univariate analysis showed that a hemoglobin level lower than 115 g/L, advanced stage, an expansive tumor growth pattern, treatment, and poor response by UICC criteria were related to worse DFS and OS. The other clinicopathological characteristics are summarized in Table I.
Relationship of CD44 and LeY Expression to Resistance to Therapy
Membrane immunoexpression was seen in all CD44+ cervical squamous cell cancer samples, predominantly with weak intensity and in a focal area of the tumor (Fig. 1). Only 23/63 (36.5%) tumors expressed CD44 in more than 10% of tumoral tissue. In the underlying stroma, the endothelial CD44+ cells were taken as internal positive control. LeY expression was observed in 47/126 (37.3%) of tumor tissues. Twenty-nine (61.7%) of these showed membrane immunostaining (Fig. 1), 13 (27.7%) cytoplasmic immunostaining, and 5 (10.6%) showed mixed (membrane and cytoplasmic) immunostaining. In 36 (76.3%) tumors the staining intensity of LeY was weak and in 40 (85.1%) it was in a focal area (Fig. 2). Positive staining of polymorphonuclear leukocytes was taken as internal positive control. The immunophenotypes of the tumors analyzed according to the clinicopathological features are summarized in Table II.
Relationship between immunomarkers and clinicopathological features of LACSCC patients
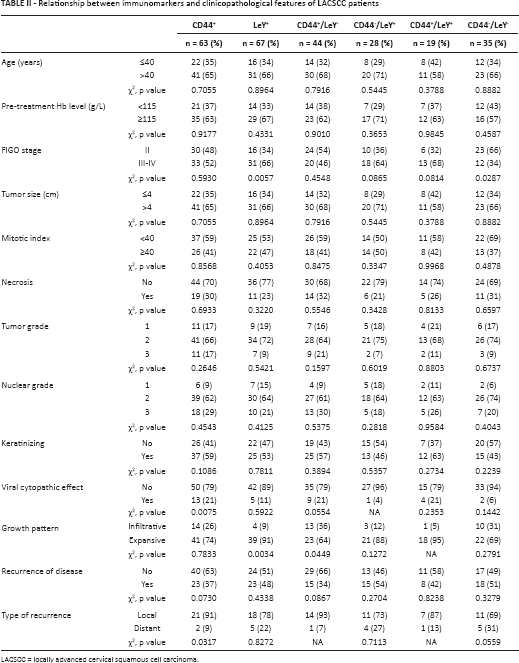
LACSCC = locally advanced cervical squamous cell carcinoma.
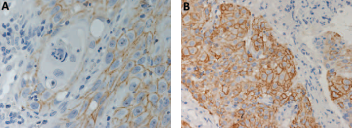
An invasive cervical squamous cell carcinoma sample showing membranous staining of CD44 (
DFS (
The predominant immunophenotypes were CD44+ (50%), LeY+ (37.3%) and CD44+/LeY- (34.9%). CD44+ cells were present in 13/16 (81.3%) of tumors with a viral cytopathic effect and in 21/23 (91.3%) of those with local recurrence. Patients with advanced stages of disease expressed higher LeY+ (66% vs. 34%, p = 0.0057) and CD44+/LeY+ rates (68.4% vs. 31.6%, p = 0.0814) than those with early stages.
Tumors with expansive growth presented significantly higher grade (p = 0.0024), mitotic index (p = 0.0505), tumor necrosis (p = 0.0191), LeY+ (p = 0.0034) and CD44+/LeY+ coexpression (p = 0.0334) (data not shown).
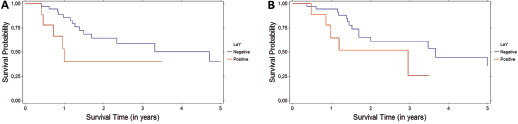
Mean follow-up was 3.4 years (range 0.1 to 18.4 years). The median OS time was 1.6 years. The 3-year DFS rate for all 126 patients was 40.8%, and the 5-year OS rate was 37.1%. There was no difference in DFS or OS according to CD44 or LeY status (Tab. II). However, patients treated with radiochemotherapy based on cisplatin had worse DFS and OS when their tumors expressed LeY (p = 0.0083 and p = 0.0137, respectively) (Tab. III, Fig. 2). Also, LeY-/CD44+ tumors showed a trend towards better DFS than LeY+/CD44- tumors, but this was not statistically significant.
Survival analysis of tumor phenotypes according to chemotherapeutic agents used. LeY expression is associated with shorter DFS and OS in patients treated with cisplatin
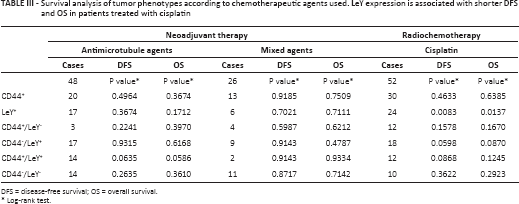
DFS = disease-free survival; OS = overall survival.
Log-rank test.
Disease progression was registered in 56/126 (44.4%) LACSCC patients, 43/56 (76.8%) of whom presented local recurrence and 13/56 (23.2%) distant spread. The CD44 expression in pretreated samples was significantly higher in those tumors that progressed locally than in those developeding distant metastases (91.3% vs. 8.7%, p = 0.0317).
Multivariate Analysis
The Cox proportional hazards regression model confirmed the variables that showed a significant association with outcome in univariate analysis (Tab. IV).
Multivariate analysis showing the impact of hemoglobin level, FIGO stage and response on the prognosis of LACSCC patients
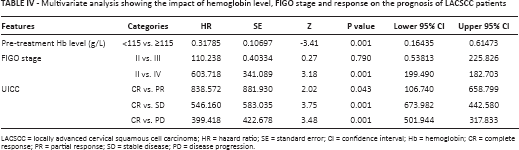
LACSCC = locally advanced cervical squamous cell carcinoma; HR = hazard ratio; SE = standard error; CI = confidence interval; Hb = hemoglobin; CR = complete response; PR = partial response; SD = stable disease; PD = disease progression.
Discussion
Few advances have been accomplished in cervical cancer therapy over the past 20 years. In the late 1990s, investigators proved that the addition of concurrent cisplatin-based chemotherapy to standard radiation therapy protocols could reduce recurrence and disease-related death rates by as much as 50% (17). Other trials suggested that there might be a benefit for neoadjuvant chemotherapy using different therapeutic agents (18). A decade later, an added survival advantage was reported when concurrent and adjuvant gemcitabine were combined with weekly cisplatin (19). Today, the combination of radiation therapy and chemotherapy is standard for patients with locally advanced cervical cancer, with an established improvement in OS. Although a significant reduction in the relative risk of cancer death has been achieved, the absolute gains are relatively small for patients with early tumors, many of whom would have been cured with radiation alone, and recurrence rates are still high for patients who have very large or advanced-stage tumors. As with other tumor locations (e.g., breast, colon or lung), in-depth knowledge of the biological behavior of cervical squamous cell cancer and its response to different chemotherapeutic drugs could help us select the best treatment for individual patients.
To evaluate chemoresistance to diverse chemotherapeutic agents used in cervical cancer, we selected 126 patients with LACSCC from the GOCS database. The objective of this study was to determine by immunohistochemical methods in pretreated samples whether CD44 or LeY or their coexpression contribute to the resistance to diverse agents used in the treatment of LACSCC patients.
In our series, we found a negative correlation between LeY expression and the outcome of LACSCC patients who received radiochemotherapy based on cisplatin. Multiple mechanisms are involved in the resistance of cancer cells to cisplatin, including the expression of multidrug resistance-associated protein, enhanced DNA repair activity, and alterations in signal transduction pathways (20). Studies have reported that protein glycosylation could play an important role in drug resistance. For example, in head and neck squamous cell carcinoma the aberrant glycosylation of α5β1-integrin is involved in the resistance to cisplatin as well as tunicamycin (21); in ovarian cancer overexpression of LeY might be related to carboplatin/paclitaxel-resistant tumors (22). Also, in colon carcinoma cells the α1,2-fucosyltransferase and histoblood group antigen H type 2 are involved in resistance to 5-fluorouracil (23). Recently, Gao et al showed that the LeY antigen is part of the composition of CD44 and increased levels of LeY are associated with increased CD44-mediated epithelial ovarian cancer cell adhesion and migration as well as increased resistance to carboplatin and paclitaxel (8, 24). In our cervical squamous cell cancer study, we could not confirm this result. We did not find a clear association between CD44+/LeY+ tumors and resistance to different chemotherapy regimens used neoadjuvantly or in combination with radiotherapy. However, CD44+/LeY- and CD44-/LeY+ phenotypes showed an interesting trend towards better and worse response to cisplatin-based radiochemotherapy, respectively. We also found a trend towards worse responses to docetaxel and vinorelbine (antimicrotubule agents) in CD44-/LeY+ tumors. Taken together, these findings suggest that the presence of difucosylated LeY antigen in cervical squamous cell cancer plays a negative role in chemoresistance, regardless of whether CD44 is present.
Studies have shown that glycosylated proteins are related to some malignant cell behaviors including adhesion, recognition and signal transduction, and that increased LeY levels promote the invasion and migration of tumor cells (25). Cervical tumors that extended to the pelvic wall or compromised the vagina, bladder or rectum, or extended beyond the true pelvis (FIGO stages III-IV) were associated with a significant increase in LeY expression in our study. Overexpression of LeY could be responsible for the upregulation of matrix metalloproteinases (MMPs), which lead to breakdown of the extracellular matrix (ECM), and downregulation of specific tissue inhibitors of MMPs (TIMPs), promoting the process of invasion although the specific mechanism needs to be studied further (26).
Several studies have analyzed the differences in CD44 expression in tumor areas and its correlation with prognosis of squamous tumors. Ostwald et al (27) observed major differences in the intensity of CD44 expression depending on the tumor area studied, with high intensity of expression by cells of the external peripheral area and low or absent expression in internal areas. The authors concluded that CD44 can be a valid proliferation marker and attributed its higher expression at the periphery to the proliferative activity of malignant cells in these areas, which could indicate a tendency to expansive growth of the tumor (27). One reason for this growth pattern could be the increased expression of difucosylated antigen in the adhesion molecule CD44 (28). In line with this hypothesis, we observed significantly high LeY expression and a predominant CD44+/LeY+ phenotype in tumors with an expansive growth pattern, which showed also a significantly higher mitotic index and more necrosis than those with an infiltrative pattern. Recently, Hotta et al (29) observed in squamous cell tumors of the head and neck an increased expression of LeY in superficial areas and decreased or absent expression in invasive regions, which correlated with the prognosis of patients. However, other studies have demonstrated the opposite (30).
A number of clinical and morphological factors have been well established to predict local recurrence in cervical cancers after radiochemotherapy; these include tumor size, histology and regional spread. In addition, nonmolecular biomarkers (tumor hypoxia, interstitial tumor pressure, vascular density, anemia) and molecular biomarkers (single genes such as CAXII, ERCC1 and EGFR, or HPV) seem to be associated with local disease recurrence. In this context, we observed a positive correlation between higher expression of CD44 in the pretreated samples of LACSCC patients and the development of local recurrence. High CD44 expression was associated with local recurrence in many other tumors such as HER2-positive breast cancer (31), hepatocellular carcinoma (32), gastric cancer (33), rectal cancer (34) and larynx cancer (35). A potential explanation for this phenomenon may include the following factors: 1) CD44 acts as a coreceptor for the ErbB family which can lead to activation of the PI3K/AKT pathway, a pathway known to promote survival after cytotoxic damage, including after radiotherapy, suggesting a possible link between CD44 expression and intrinsic radiosensitivity; 2) CD44 could be linked to hypoxia or to the repopulating ability, both known to influence radiotherapy outcome; and 3) CD44 expression could reflect the number of stem cells or cancer-initiating cells needed to be killed independent of whether the putative stem cells are more or less radioresistant than the bulk tumor cells.
The majority, that is, 75% to 95%, of cervical tumors are positive for human papillomavirus (HPV). Interestingly, among populations with rare cases of cervical cancer that do not have detectable HPV, the prognosis is poor (36). A worse prognosis in HPV-negative tumors has also been reported in squamous oropharynx cancer, in which 30%-40% of cases do not have detectable HPV. Five-year survival rates are 45%-50% in patients with HPV-negative tumors versus 75%-80% in those with HPV-positive tumors. According to these results, we found a higher 5-year survival rate in LACSCC patients whose tumors presented a viral cytopathic effect than in those without this characteristic, although the difference was not statistically significant. The reason for the survival differences according to HPV status is not known, but HPV status correlates with multiple molecular abnormalities including chromosomal changes and p53 mutation status (37). Among the tumors with a viral cytopathic effect found in our series, we observed predominant expression of CD44 and the CD44+/LeY phenotype. Recently, Näsman et al (38) observed in 290 oropharyngeal squamous cell carcinoma patients a poor DFS when the tumors were HPV+ and showed medium-/strong-intensity CD44 staining. Also, Hufbauer et al (39) showed in a cutaneous keratinocyte cell line (PM1) that exposure to HVP led to a shift of the total population from low toward higher CD44 cell surface expression and a reduction of the number of terminally differentiated cells. The significant increase found in the number of CD44+ cells suggests that HPV enlarges this population of proliferative stem cell-like cells. The authors concluded that the expression of HPV inhibits the entry of keratinocytes into differentiation and that the maintenance of basal cells in an undifferentiated state may increase the pool of cells available for the accumulation of damage that can persist and lead to the generation of stem cells with malignant properties. The genetic mutation resulting from high-risk HPV infection can lead the stem cells to undergo uncontrolled proliferation, invasion and metastasis (39).
In our entire sample studied, a low pre-treatment hemoglobin level, expansive growth pattern, advanced FIGO stage, treatment, and poor response to therapy were associated with shorter DFS and OS in the univariate analysis. Patients treated with radiochemotherapy based on cisplatin had worse DFS and OS when their tumors expressed LeY. Pre-treatment hemoglobin level, FIGO stage and tumor response remained the most significant prognostic factors in Cox regression. In our series, we did not find any associations between CD44+/LeY+ tumors and outcome. In line with our results, Lindström et al (40) assessed different biomarkers in 128 squamous cell cervical cancers and concluded that CD44 was not a valid prognostic marker.
In summary, in our cohort of LACSCC patients, the CD44+/LeY+ immunophenotype was not associated with worse outcome. However, in the subgroup of patients receiving radiochemotherapy based on cisplatin, LeY expression was correlated with shorter DFS and OS.
Footnotes
Acknowledgment
The authors want to express their appreciation to Dr. Maria Virginia Croce for her collaboration in the preparation of this manuscript, and to Dr. Maria May for her help in the editing of figures.
Financial support: This study was carried out thanks to a CISNe grant awarded by the Ministry of Health of the Province of Neuquén, Argentina.
Conflict of interest: The authors declare that they have no financial relationship that may lead to a conflict of interest in relation to the submitted manuscript.
Meeting presentation: The results of this research were presented in part at the ASCO Meeting, May 29-June 2, 2015, Chicago, IL, USA. J Clin Oncol. 2015;33(suppl):abstr e16590.
