Abstract
Aberrant regulation of BCL6 plays crucial oncogenic roles in various malignant tumors; howbeit, the function of BCL6 in tumorigenesis of ovarian cancer remains unclear. The aim of this study is to investigate the role of BCL6 in ovarian cancer. The methods of immunohistochemical staining, quantitative real-time polymerase chain reaction, immunocytochemical staining, and gene expression profile enrichment analysis were performed to identify the possible role of BCL6 in ovarian cancer. We observed that the expression of BCL6 was significantly higher in ovarian cancer tissues and correlated with higher tumor burden including advanced International Federation of Gynecology and Obstetrics stages, poor differentiation, Type II ovarian cancer, the presence of >1 cm residual tumor size, and appearance of recurrence or death (all p < 0.05). The expression patterns of Lewis y were similar to these of BCL6. Multivariate Cox analysis demonstrated that advanced International Federation of Gynecology and Obstetrics stage, lymph node metastasis, residual tumor size >1 cm, as well as high expressions of BCL6 and Lewis y antigen were independent factors of worse progression-free survival and overall survival (all p < 0.05). There was a positive correlation of the expressions of BCL6 and Lewis y antigen. The associated genes with BCL6 in response to Lewis y antigen were identified, including four upregulated genes (SOCS3, STAT1, PPARG, and GADD45A) and three downregulated genes (ACAN, E2F3, and ZBTB7B). In conclusion, the high expressions of BCL6 and Lewis y antigen are associated with development, high tumor burden, and worse prognosis of ovarian cancer and targeting BCL6 could be a novel therapeutic strategy for ovarian cancer treatment.
Introduction
Ovarian cancer is the second cause of death among female reproductive malignancies and claims 140, 200 lives every year worldwide. 1 Most of the ovarian cancers are diagnosed at an advanced stage due to its innocuous symptoms such as abdominal distension and discomfort at the onset. 2 Thus, searching the early detector and prognostic predictor of ovarian carcinoma is vitally important to facilitate the early diagnosis or screen, surveillance of the reaction to the clinical therapy, and disease recurrence.
B-cell CLL/lymphoma 6 (BCL6), a highly conserved zinc finger (ZF) transcriptional factor, was first identified as an oncogene by gene expression profile in non-Hodgkin’s lymphoma in 2000; 3 it exhibits pivotal roles in germinal center formation and regulation of lymphocyte function, differentiation, and survival. 4 In recent years, more and more studies identified that BCL6 was overexpressed not only in B-cell lymphoma 5 but also in gastric cancer, 6 gallbladder cancer, 7 breast cancer, 8 colorectal cancer, 9 head and neck cancer, 10 and so on. However, there are quite a few studies on the function of BCL6 in ovarian cancer, and it remains unclear whether BCL6 may aberrantly express or be a potential prognosis marker in ovarian cancer. As an important component of glycoproteins and glycolipids, glycosyl antigen is widely expressed in the cell membrane, 11 and changes in the antigens are significantly associated with the variations of characteristics on tumor cell surface, resulting in the cell carceration, invasion, and migration.11,12 Lewis y antigen, a tumor-associated carbohydrate antigen (TACA), expressed in over 75% of ovarian epithelial neoplasm. 11 α-1,2-Fucosyltransferase (FUT1) is a key enzyme in the synthesis of Lewis y. 12 In our previous studies, we have already transfected FUT1 gene into ovarian cancer cell line RMG-1 to establish RMG-1-hFUT cell line with high Lewis y expression, and we noted that the RMG-1-hFUT cells exhibit increased malignant biological behaviors, such as cell proliferation, adhesion, metastasis, and drug resistance.13–15 Moreover, recently, we performed an item of whole genome microarray analysis to identify the alteration of gene expression profile in response to Lewis y antigen in ovarian cancer cells, and we discovered that BCL6 was the one upregulated gene that exhibited significantly differential expression. 16 We speculated that BCL6 would exert an oncogene role in ovarian cancer.
In this study, we detected the expression of BCL6 and Lewis y antigen in ovarian cancer tissues and cells, and we also analyzed how the expressions of two markers correlate with clinical significance including prognosis; moreover, we conducted an enrichment analysis on basis of gene expression profile to find out the associated genes of BCL6 in ovarian cancer.
Materials and methods
Ethics statement
Samples were fully encoded to protect patient confidentially. The study was approved by the Ethical Committee of Shengjing Hospital of China Medical University (number of approval: 2010PS84K).
Patients and tissue samples
Selected paraffin-embedded samples (170 cases in total) were obtained from the operations performed from 2000 to 2012 in the Department of Gynecology of Shengjing Hospital of China Medical University. All tissue sections were examined by specialists to obtain a definitive diagnosis, in which 103 cases were malignant, 22 borderlines, 25 benign, and 20 normal ovarian tissues. A total of 20 normal ovarian samples were obtained from tissue excised in squamous cervical cancer operations. The mean age of overall patients was 50.2 (16–78) years. The median and age range of the ovarian cancer group was 53.0 (16–77) years; the borderline, benign, and normal ovarian group was 39.0 (19–78) years, 45.0 (28–68) years, and 46.0 (31–61) years, respectively (Table 1). There is no statistical difference among the ages of these groups (p > 0.05). Among the 25 benign ovarian tumors, 14 cases were mucinous and 11 cases were serous cystadenomas. Among the 22 borderline tumors, 12 cases were mucinous and 10 cases were serous cystadenomas. All the 103 ovarian malignant samples were obtained from primary debulking operations; no patient received chemotherapy or radiation therapy before operation. According to the 2010 International Federation of Gynecology and Obstetrics (FIGO) staging system for ovarian cancer, there were 33 patients in stage I, 11 patients in stage II, 55 patients in stage III, and 4 patients in stage IV. In total, 10 cases were well-differentiated, 32 were moderately differentiated, and 61 were poorly differentiated. By histological analyses, 17 45 patients were serous, 15 were mucinous, 8 were endometrioid, 10 were clear-cell, and 25 were undifferentiated carcinomas. According to dualism model classification, 18 54 patients were Type I ovarian cancer (including 21 cases of low-grade serous, 15 cases of mucinous, 8 cases of endometrioid, and 10 cases of clear-cell carcinomas) and 49 patients were Type II ovarian cancer (including 24 cases of high-grade serous and 25 cases of undifferentiated carcinomas). There were 17 patients who had lymph node metastasis, and 37 patients whose residual tumor size were more than 1 cm. General clinical and pathological information of patients was shown in Tables 1 and 3.
Ovarian tissue patient characteristics.

SD: standard deviation.
Immunohistochemical staining and quantification
Consecutive paraffin-embedded sections of these cases were prepared and immunohistochemistry (IHC) was used to analyze the expression of BCL6 and Lewis y antigen in all these ovarian tissues. Streptavidin–biotin complex (SABC) and streptavidin–peroxidase (SP) kits were used and performed according to their manuals. Rabbit monoclonal anti-BCL6 antibody (EP529Y, ab33901; Abcam, USA) and mouse monoclonal anti-Lewis y antibody (F3, ab3359; Abcam, USA) were used, and the working dilutions were 1:150 and 1:200, respectively. Negative controls were performed by omission of the primary antibody or incubation with an isotype control antibody, whereas the positive controls were performed as follows: a normal tonsil tissue sample for BCL6 and a colon cancer sample for Lewis y antigen. The presence of brown-colored granules in the cells was taken as a positive staining and was classified according to color intensity as follows: not colored, light yellow, brown, and tan were scored as 0, 1, 2, and 3, respectively. Five high-power fields in series from each slice were observed; the mean percentages of the stained cells were recorded and scored as follows: a positive cell rate of less than 5% was 0, 5%–25% was 1, 26%–50% was 2, 51%–75% was 3, and more than 75% was 4. The final score was determined by multiplying the two values: 0–2 was considered negative (−), 3–4 was (+), 5–8 was (++), and 9–12 was (+++); − and + was considered as low expression, ++ and +++ as high expression. Two observers read the sections to control error.
Cell lines and cell culture
Human epithelial ovarian cancer cell line OVCAR3 was purchased from American Type Culture Collection (Manassas, VA, USA). α-1,2-Fucosyltransferase (FUT1) gene was transfected to OVCAR3 cells to establish cell line OVCAR3-H with high expression of Lewis y antigen, and OVCAR3-M was the cell line transfected with the empty plasmid alone. Transfection was carried out with a vector transfection kit according to the instructions that were shown in our previous studies.19,20 All cells were cultured in Dulbecco’s Modified Eagle Medium (DMEM) supplemented with 10% fetal bovine serum (FBS) at 37°C, 5% CO2 in humidified air.
RNA isolation and quantitative real-time polymerase chain reaction
Total messenger RNA (mRNA) was extracted from ovarian cancer cells using Trizol reagent. Complementary DNA (cDNA) was then synthesized using the real-time polymerase chain reaction (RT-PCR) kit (Invitrogen, China) according to manufacturer’s protocol. Quantitative RT-PCR was performed on Roche LightCycler 480 (Roche Diagnostics, Mannheim, Germany) sequence detection system. The PCR primer sequences were designed (Invitrogen) and chemically synthesized. The sequences of the primers for BCL6 was, sense, CTGCAGATGGAGCATGTTGT, and antisense, TCTTCACGAGGAGGCTTGAT; FUT1, sense, AGGTCATCCCTGAGCTGAAACGG, and antisense, CGCCTGCTTCACCACCTTCTTG. The housekeeping gene GAPDH (sense, AATCCCATCACCATCTTCCA, and antisense, TGGACTCCACGACGTACTCA) served as the constitutive control. PCR reactions of each sample were done in triplicate. Data were analyzed through the comparative threshold cycle (CT) method.
Immunocytochemistry
Cells cultured on slides were fixed with 4% paraformaldehyde for 30 min and then stained according to the instructions for the SABC kit. The working dilutions for BCL6 and Lewis y antigen were 1:150 and 1:50, respectively. The primary antibody was replaced by rabbit IgG for negative control. Images were obtained using a fluorescence microscope at magnification ×400.
Gene expression profile analysis and enrichment analysis
Using human whole genome oligonucleotide microarrays, gene expression profile alteration in response to Lewis y antigen was investigated and 215 differentially expressed genes (DEGs) were identified and validated in Caov-3 transfected with FUT1 compared with its empty-plasmid–transfected cells, in which 122 genes were upregulated and 93 genes were downregulated. 16 BCL6 was among the upregulated genes in response Lewis y. Gene Ontology (GO) term enrichment analysis was performed ulteriorly in the 122 upregulated genes to find out the gene functions that BCL6 involved. Finally, the relationship between BCL6 and the other DEGs in response to Lewis y antigen in ovarian cancer was evaluated by uploading all these DEGs into the Qiagen’s Ingenuity® Pathway Analysis IPA®.
Statistical analysis
SPSS program (Version 22 for Mac; SPSS Inc., Chicago, IL, USA) and Graph Pad Prism 6 (version 6.0 h for Mac; Graph Pad Prism Software Inc., San Diego, CA, USA) were used for statistical analysis. Independent t test, chi-square (χ2) and Spearman correlation were employed. Quantitative data are presented as mean ± standard deviation (SD). Survival analysis was analyzed using Kaplan–Meier curves, and significant differences among different clinicopathological variants and immunomarkers were tested using the log-rank test. Cox proportional hazards regression models were used to control for confounding variables. Follow-up time was calculated from the date of surgery to the date of progression, death, and last visit or contact with the patient. Overall survival (OS) was defined as the time interval between the date of surgery and the date of death; progression-free survival (PFS) was defined as the time interval between the date of surgery and the date of identification of progressive disease (disease not treatable with curative intent). For all these end points, the last date of follow-up (September 2015) was used for censored subjects. A p < 0.05 was considered statistically significant.
Results
Expression patterns of BCL6 and Lewis y in overall ovarian tissues
BCL6 was mainly localized in the cell nucleus and cytoplasm (Figure 1), and the high expression rates in malignant, borderline, benign, and normal ovarian tissues were 69.9%, 59.1%, 28.0%, and 5%, respectively (Table 2). Malignant group showed the highest positive expression rate and significantly higher than that of the benign and normal groups (p < 0.001, Table 2). Similarly, the high expression rate in borderline was apparently higher than that in benign and normal groups, respectively (p < 0.05, Table 2). Lewis y antigen was mainly detected in the cell membrane and cytoplasm (Figure 1). Similar expression inclinations were also observed in Lewis y antigen, the high expression rates in malignant, borderline, benign, and normal ovarian tissue groups were 67.0%, 54.5%, 16.0%, and 5.0%, respectively (Table 2), and the malignant group maintained the highest high expression rate and obviously higher than that in benign and normal groups (p < 0.001); however, no significant difference was observed between the benign and normal groups (p = 0.243).

Representative images of BCL6 and Lewis y antigen staining in ovarian tissues. Immunopositivity for BCL6 and Lewis y antigen is represented by brown staining. (a and e) malignant ovarian tumor, (b and f) borderline ovarian tumor, (c and g) benign ovarian tumor, (d and h): normal ovarian tissue. (a–d) BCL6 and (e–h) Lewis y antigen. Scale bar: 50 μm.
The expression of BCL6 and Lewis y in different ovarian tissues.

Compared with the benign and normal group, p = 0.00011, p < 0.001, respectively.
Compared with the benign and normal group, p < 0.001, p < 0.001, respectively.
Compared with the benign and normal group, p = 0.031, p = 0.0002, respectively.
Compared with the benign and normal group, p = 0.005, p = 0.001, respectively.
Compared with the normal group, p = 0.045.
Correlation of BCL6 and Lewis y antigen expression with clinicopathological features of ovarian cancer
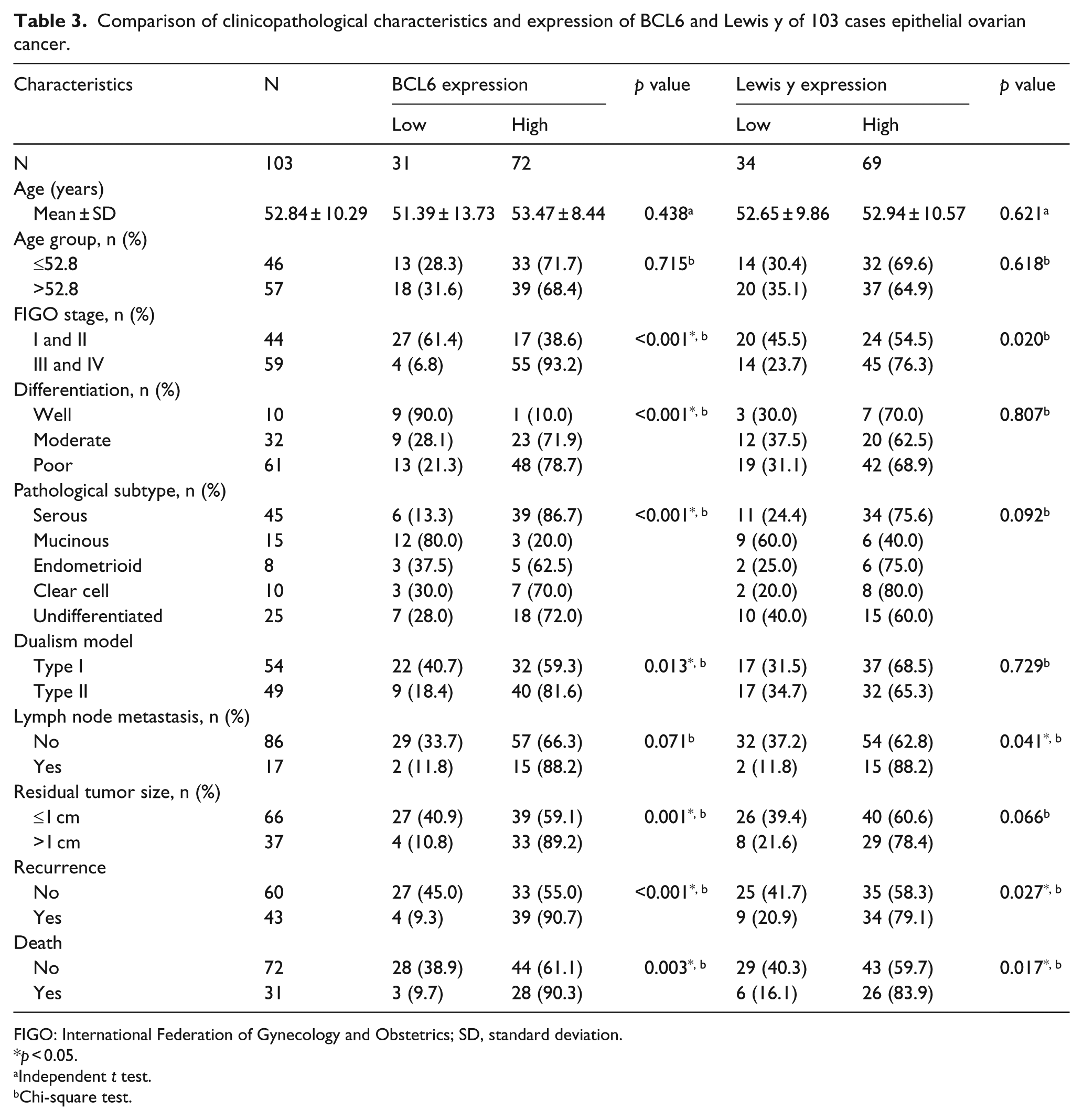
The high expression patterns of BCL6 were upregulated in tumors with a higher tumor burden, as defined by advanced FIGO stages (vs Stages I and II, p < 0.001), poor differentiation (vs moderate or well, p < 0.001), Type II ovarian cancer (vs Type I, p = 0.013), presence of >1 cm residual tumor size (vs ≤1 cm, p = 0.001), as well as the appearance of recurrence (p < 0.001) or death (p = 0.003). The high expression rate of BCL6 in serous carcinoma was significantly higher than that of in mucinous (86.7% vs 20%, p < 0.001); moreover, the endometrioid, clear-cell, and undifferentiated carcinomas maintained the high expression rates of BCL6. The high expression of BCL6 in lymphatic node group (88.2%) was higher than that of the non-lymphatic node metastasis group (66.3%), although the difference was not significant (p = 0.071). There was no obvious correlation between the expression of BCL6 and age (p > 0.05).
The high expression levels of Lewis y antigen were also upregulated in advanced FIGO stages (p = 0.020), the existence of recurrence (p = 0.027) or death (p = 0.017), as well as the presence of lymph node metastasis (p = 0.041). The high expression rate of Lewis y in serous carcinoma was significantly higher than that of in mucinous (75.6% vs 40.0%, p = 0.011). However, no obvious differences were observed in the high expression of Lewis y as to age, differentiation, dualism model classification, or residual tumor size, although high expression of antigen was observed in the >1 cm (78.4%) compared with ≤1 cm residual tumor size (60.6%) (Table 3).
Comparison of clinicopathological characteristics and expression of BCL6 and Lewis y of 103 cases epithelial ovarian cancer.
FIGO: International Federation of Gynecology and Obstetrics; SD, standard deviation.
p < 0.05.
Independent t test.
Chi-square test.
Relevance of BCL6 and Lewis y expression in ovarian tissues
Totally, there were 54 and 16 cases simultaneously showing high and low expression patterns both in BCL6 and Lewis y antigen in all the 103 cases of ovarian cancer and Spearman correlation analysis revealed that these two markers positively correlated (r = 0.260, p = 0.008); moreover, when pooled all these ovarian tissues, there was 64 and 55 cases simultaneously showing high and low expression patterns in all the 170 cases of ovarian tissues and Spearman analysis further showed their positive correlation (r = 0.401, p < 0.001) (Table 4).
The relevance of BCL6 and Lewis y expression in ovarian tissues.

Spearman correlation, r = 0.260, p = 0.008.
Spearman correlation, r = 0.401, p < 0.001.
Follow-up visit and prognostic factors analysis
During the period of follow-up for the ovarian cancer patients, 31 patients (30.1%) were dead, 12 patients (11.6%) were alive with recurrence of the disease, 52 patients (50.5%) were alive without evidence of disease, and 8 patients (7.8%) were lost. The median follow-up was 60.0 (range: 33.2–85.5) months, the 5-year PFS was 59.5% (Figure 2(a)), and mean survival time was 58.7 months (95% confidence interval (CI): 51.8–65.6); the 5-year OS was 68.7% (Figure 2(d)), and mean survival time was 70.0 months (95% CI: 64.5–75.6).

Kaplan–Meier curves for progression-free survival (PFS) and overall survival (OS) in 103 ovarian cancer patients. (a) PFS curve and (d) OS curve for all the ovarian patients, the 5-year survival rate and mean survival time are shown in figures. PFS and OS according to the low and high expression of (b and e) BCL6 and (c and f) Lewis y.
Cox survival analyses indicated that among various prognostic factors, FIGO stages III and IV, lymph node metastasis, residual tumor size >1 cm, high expressions of BCL6, and Lewis y antigen were found to be significant risk factors that correlated with PFS (hazard ratio (HR): 8.466, 2.329, 4.889, 5.752, and 2.124, respectively, all p < 0.05, Table 5). As to OS, similarly, FIGO stages III and IV, lymph node metastasis, residual tumor size >1 cm, high expressions of BCL6, and Lewis y antigen were also found to be significant risk factors (HR: 6.342, 2.519, 3.414, 5.191, and 2.799, respectively, all p < 0.05, Table 5). Taken together, except the clinical pathological parameters, the high expressions of both BCL6 and Lewis y antigen were independent risk factors and significantly correlated with prognosis (Kaplan–Meier analysis, log rank, p < 0.001 and p = 0.002 as to PFS, Figure 2(b) and (e); p = 0.038 and p = 0.027 as to OS, Figure 2(c) and (f), respectively).
Cox survival analysis of factors affecting patient’s progression-free survival or overall survival for clinicopathologic variables and the IHC expression in 103 patients of EOC.

IHC: immunohistochemistry; EOC: epithelial ovarian cancer; PFS: progression-free survival; OS: overall survival; HR: hazard ratio; CI: confidence interval; FIGO: International Federation of Gynecology and Obstetrics.
p < 0.05.
The mRNA and protein expression levels of BCL6 in ovarian cancer cells after Lewis y overexpression
The mRNA expression levels of FUT1 and BCL6 were significantly elevated after FUT1 gene transfection (p < 0.001, p = 0.002, respectively), whereas there was no obvious change between OVCAR3 and OVCAR3-M cell lines (p >0.05, Figure 3). As to the protein expression level, both the Lewis y antigen and BCL6 showed the positive staining in ovarian cancer cells OVCAR3, Lewis y antigen was mainly localized in cytoplasm and membrane (Figure 4(a)), and BCL6 was mainly localized in the nucleus and cytoplasm (Figure 4(c)). In the FUT1-transfected cell line OVCAR3-H, the Lewis y antigen showed significant higher expression (Figure 4(b)), whereas the BCL6 was also obvious highly expressed (Figure 4(d)).
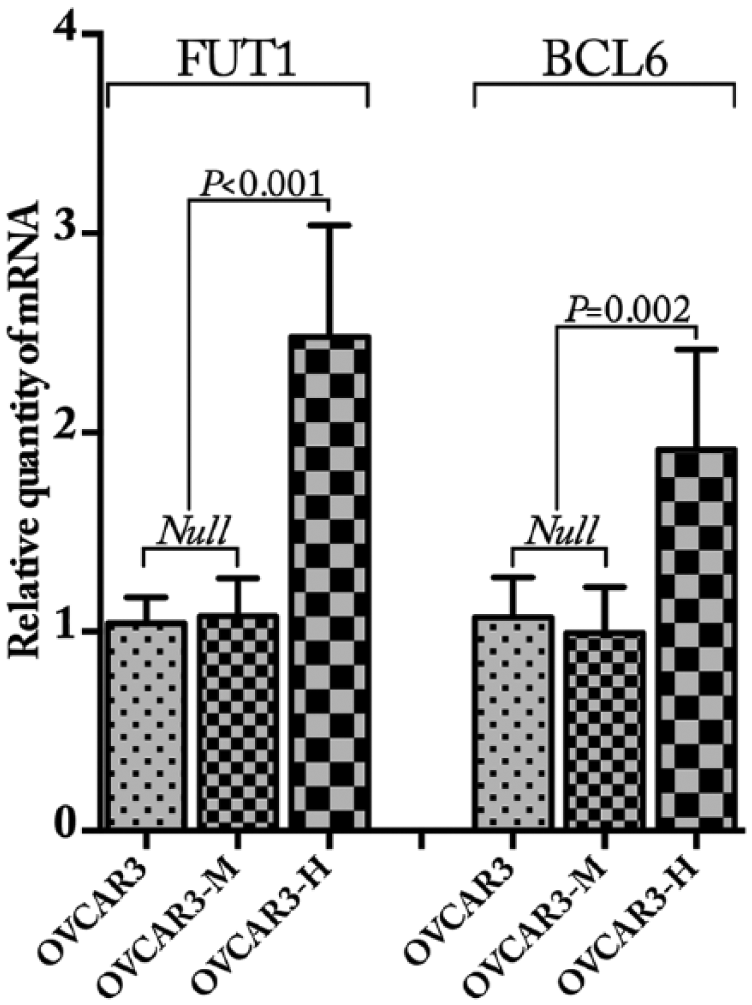
The mRNA expression of FUT1 and BCL6 in OVCAR3 cells after FUT1 gene transfection. Quantitative real-time PCR revealed that the mRNA expression levels of FUT1 and BCL6 were significantly increased after the transfection of FUT1 (p <0.001, p =0.002, respectively), whereas there was no change between OVCAR3 and OVCAR3-M cell lines.

Representative images of immunocytochemical staining of BCL6 and Lewis y antigen in OVCAR3 cells. Immunopositivity for BCL6 and Lewis y antigen is represented by brown staining, Lewis y antigen was mainly localized in cytoplasm and membrane, where BCL6 was predominantly localized in nucleus and cytoplasm. (a and c) OVCAR3 cells, (b and d) OVCAR3-H cells. (a and b) Lewis y antigen, (c and d) BCL6. Scale bar: 50 μm.
GO function analysis and interaction network of BCL6 involved in the DEGs in response to Lewis y
GO function enrichment analysis was performed in the upregulated DEGs based on molecular function, cellular component, and biological process (Table 6). The interaction network of genes associated with BCL6 was presented in Figure 5, including four upregulated genes (SOCS3, STAT1, PPARG, and GADD45A) and three downregulated genes (ACAN, E2F3, and ZBTB7B).
GO enrichment analysis that BCL6 involved for upregulated DEGs in response to Lewis y antigen in ovarian cancer cells.

GO: Gene Ontology; DEGs: differentially expressed genes.

Gene network generated by the IPA® platform for BCL6-associated genes. Genes marked with red and green represent the upregulated and downregulated genes, respectively. Dotted lines indicate indirect interactions and solid lines indicate direct interaction between the genes.
Discussion
BCL6, a nuclear transcriptional repressor, can regulate transcription of various genes. Located on chromosome 3q27, the BCL6 gene encodes a 706 amino acid protein composed of an amino-terminal bric-a-brac, tramtrack, broad complex/Pox virus ZF (BTB/POZ) domain, and a carboxy-terminal six ZF domain,4,21 these domains regulate transcription of target genes via distinct interactions. BCL6 exerts its pivotal roles for regulating germinal center development, proliferation/differentiation of lymphocytes, inflammation, and cell cycle/apoptosis. 4 Initially, researchers found BCL6 mainly abundantly expressed in B cell and CD4+ T cell in germinal centers of normal lymphoid tissues, 22 and the association of BCL6 with lymphoma has been widely investigated and well established,3,5,22 especially in diffuse large B-cell lymphoma, 23 the pathogenesis mechanisms attribute to rearrangements or somatic mutations of BCL6, and so on.4,24,25 BCL6 inhibits p53 expression in germinal center of B lymphocyte 26 and interacts with pro-oncogene c-myc to accelerate the carcinogenesis of lymphoma. 27 Based on these findings, BCL6 inhibitors (compound 79-6 28 or retroinverso BCL6 peptide inhibitor (RI-BPI) 29 ) have been developed and showed delightful potential application prospect as a therapeutic strategy for patients with hematological malignancies.28,29 In recent years, overexpression of BCL6 was reported to be relevant to somatic malignant tumors, such as gastric cancer, 6 gallbladder cancer, 7 breast cancer, 8 colorectal cancer, 9 and head and neck cancer, 10 suggesting that the tumorigenicity of BCL6 is not only limited to lymphomas.
BCL6 protein was highly expressed both in cell lines and tissue specimens of breast cancer 8 and it enhanced the proliferation, growth, migration and survival of breast cancer cells through the regulation of epithelial–mesenchymal transition (EMT) by enhancing the expression of ZEB1 which bound to the E-cadherin promoter and repressing the E-cadherin transcription. 30 The ZEB1, a crucial transcriptional repressor, inhibits the transcription of E-cadherin to regulate the EMT response, which is also prominent in non–small-cell lung cancer (NSCLC) 31 and colorectal cancer. 32 At the gene level, BCL6 was a target gene of miR-339-5p8,33,34 or miR-187-3p, 35 these microRNAs inhibit proliferation, migration, and invasion in breast cancer, 34 NSCLC, 35 or ovarian cancer cells. 33 In gallbladder cancer, BCL6 overexpression correlated with high tumor, node, and metastasis (TNM) stage, higher histological grade, vascular invasion, decreased p19ARF expression, and worse survival. 7 However, until now, there are quite a few studies on the potential biological roles of BCL6 in ovarian cancer. BCL6 was found to be bound to the cytoplasmic domain of heparin-binding epidermal growth factor–like growth factor (HB-EGF) 36 to suppress cell cycle, 37 and HB-EGF played a critical role in tumor formation and growth in patients with ovarian cancer. 38 Recently, BCL6 was identified to be one of the upregulated genes in a bioinformatics study designing to identify the molecular mechanisms of NSC319726 (a small-molecule anticancer drug functioning to the p53R175 protein) in ovarian cancer, 39 which revealed that BCL6 may play an efficient role in ovarian cancer. One study reported that BCL6 is a negative prognostic factor and exerts pro-oncogenic characteristics in ovarian cancer, 40 high expression was tightly correlated with tumor grades, lymph node metastasis and recurrence, BCL6-stimulated proliferation, invasion, and migration in ovarian cancer cells. Our data further confirmed the tumorigenesis roles of BCL6 in ovarian tumor and demonstrated that BCL6 was correlated with high tumor burden, and the high expression was obviously increased along with the tumor carcinogenesis progression including late FIGO stages, poor differentiation, Type II ovarian cancer, >1 cm residual tumor size, and recurrence or death in ovarian cancer. BCL6 was a good and independent predictor both in PFS and OS. The discrepancy between expression pattern and tumor grading/differentiation in different pathological types suggests that the mechanism that relates BCL6 to carcinogenesis in ovarian cancer may be tumor-type specific.
As a kind of TACA, Lewis y antigen was upregulated at a different extent in more than 75% of epithelial ovarian cancer tissues, and patients with high expression of Lewis y usually have bad prognosis. 15 In our previous studies, we have transfected FUT1 gene into human ovarian cancer cell line RMG-1 to construct cell line RMG-1-H with high expression of fucosyltransferase and Lewis y;13,15 the overexpression of Lewis y antigen can aberrantly activate downstream signaling pathways, such as epidermal growth factor receptor (EGFR), PI3K/Akt, Ras/ERK, p38/MAPK, Smad,15,19,41–43 and anomalously regulate the expression of cellular factors, such as cell cycle proteins, autophagy-related factors, and apoptosis-related factors; thus enhance the malignant biological behaviors including proliferation, adhesion, apoptosis, drug resistance, autophagy, invasion, and metastasis; and ultimately promote the occurrence and development of ovarian cancer.41–47 For further comprehending the fundamental molecular mechanism of Lewis y antigen in ovarian cancer, we have performed a gene expression profile analysis using human whole genome oligonucleotide microarrays in ovarian cancer cell line Caov-3 transfected with FUT1 compared with its empty-vector transfected cells and we identified 215 DEGs according to the selection criteria, in which 122 genes were upregulated and 93 genes were downregulated. 16 BCL6 gene was among the upregulated DEGs in response to Lewis y. And here, in this study, we further demonstrate that the expression of BCL6 and Lewis y antigen is positively correlated with each other both at mRNA and protein levels; moreover, our enrichment analysis including GO term analysis and interaction network may provide crucial elements for successive concentrated investigation on BCL6 in ovarian cancer. Lewis y antigen could enhance the adhesion and metastasis capabilities of ovarian cancer cells by glycosylated modification of CD44 structure; 48 intriguingly, CD44s, one of CD44 splice isoforms, can activate the expression of ZEB1, resulting in a CD44s–ZEB1 regulatory loop to accelerate EMT process in cancer cells. 49 Likewise, BCL6 can increase the expression of ZEB1 to regulate the EMT process for enhancing the proliferation and migration in breast cancer cells; 8 moreover, BCL6 can interact with HB-EGF by binding to its cytoplasmic domain. 36 Increased expression of Lewis y promotes cell proliferation and migration through activation of EGFR signal pathway.15,50,51 Accordingly, there is a good possibility that Lewis y regulates the expression of CD44 via BCL6 by activation of EGFR signal to enhance the malignant behaviors in ovarian cancer, whereas the mechanisms are still warranted more in-depth and concentrated investigations.
Collectively, our results demonstrate that there is a positive correlation between the expression of BCL6 and Lewis y, and high expression of these two markers are significantly associated with adverse clinicopathological factors and bad prognosis, and BCL6 is an independent prognosticatory factor for ovarian cancer. Gene expression enrichment analysis shows the associated genes of BCL6, which can provide the fundamental data for in-depth investigation. These results suggest that BCL6 play a critical role in tumorigenesis of ovarian cancer, thus to provide new ideas for early detection and surveillance for ovarian cancer treatment.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (no: 81172491, 81101527); the Research Fund for the Doctoral Program of Higher Education (no: 20112104110016, 20112104120019); Scientific Research Projects of Liaoning Province Department of Education (no: L2011129); and Outstanding Scientific Fund of Shengjing Hospital (no: 201303).
