Abstract
Background
Several studies in solid tumors have shown that expression of excision repair cross-complementation group 1 (ERCC1) and class III β-tubulin (TUBB3) can predict response to chemoradiotherapy and might be prognostic factors. We assessed the role of ERCC1 and TUBB3 expressions as predictive and prognostic factors in locally advanced cervical squamous cell carcinoma (LACSCC) patients treated with different neoadjuvant regimens.
Methods
ERCC1 and TUBB3 were detected in 88 patients with LACSCC by immunohistochemical analysis. Sixty-two patients were included in 3 different prospective trials and grouped as follows: vinorelbine or docetaxel (group A, n = 44) and ifosfamide-vinorelbine-cisplatin (group B, n = 18). Both groups were compared with standard cisplatin chemoradiotherapy (group C, n = 26). Clinical data at baseline, disease-free survival (DFS) and overall survival (OS) were also collected. Univariate and multivariate Cox models were used to analyze the risk factors.
Results
Thirty-five patients (39.8%) and 18 (20.5%) had high ERCC1 and TUBB3 expression, respectively. Both proteins were overexpressed in tumors with unfavorable characteristics. High ERCC1 was associated with advanced FIGO stage (p = 0.034) and progressive disease (49% vs. 28%). Poor DFS (p = 0.021) and OS (p = 0.005) were observed in group C patients with high ERCC1 expression. Multivariate analysis showed that ERCC1 expression, FIGO stage and pretreatment hemoglobin level were significant prognostic factors (p = 0.002, p = 0.008 and p = 0.005, respectively).
Conclusions
ERCC1 expression could be a predictive and prognostic factor in LACSCC patients who receive cisplatin monotherapy. Conversely, TUBB3 had no impact on survival in patients treated with antimicrotubule agents.
Introduction
Cervical cancer is a global health concern. In developed countries it represents the tenth most common type of cancer in women and is ranked below the top 10 causes of cancer mortality; in contrast, in developing countries, it is the second most common type of cancer and cause of cancer deaths among women (1).
The benefit of chemoradiotherapy (CRT) for women with locally advanced cervical squamous cell cancer (LACSCC) rather than radiation therapy (RT) alone was demonstrated recently in a meta-analysis carried out by the Chemoradiotherapy for Cervical Cancer Meta-analysis Collaboration (2). Compared with primary RT, the use of CRT resulted in a reduction of the risk of recurrence (local and distant) and death. This reduction was seen in trials using both platinum-based and non-platinum-based regimens.
Few randomized and phase II studies have used neoadjuvant chemotherapy prior to definitive hysterectomy as an alternative to primary CRT. Vinorelbine, taxanes (paclitaxel or docetaxel) or ifosfamide alone or combined have been tested in this setting (3-7) as well as radiation sensitizers (8-10). Although a randomized phase III trial showed no additional benefit for the use of neoadjuvant chemotherapy in patients with stage Ib disease (11), 2 ongoing phase III trials (EORTC-55994 and a study sponsored by the Department of Atomic Energy of India, both started in 2011) will hopefully provide further evidence on the role of this treatment in women with advanced disease.
Despite the improvements in cancer therapies, there are still a large number of patients who succumb from their disease. This may be due to underlying mechanisms of chemoresistance and/or radioresistance in their tumors; several reports have described different pathways that allow cancer cells to survive in conditions of poor nutrient supply and low oxygen levels (12). Among these survival pathways, a prominent role is played by the overexpression of the excision repair cross-complementation group 1 (ERCC1) and class III β-tubulin (TUBB3).
ERCC1 participates in homologous recombination, interstrand crosslink repair and DNA double-strand breaks repair (13, 14). RT kills tumor cells by inducing DNA damage, the most lethal of which is double-strand breaks. Given the role of ERCC1 in DNA damage repair, it is possible that its expression level may be associated with RT response. In fact, high ERCC1 expression has been associated with poor treatment response and survival in head-and-neck, esophageal and cervical cancer patients treated with RT and platinum-based chemotherapy (15-18).
Although originally identified as a mechanism of drug resistance to taxanes and vinorelbine (19, 20), recent studies have shown that TUBB3 is involved in an adaptive response to low oxygen levels and poor nutrient supply in a growing number of solid tumors (21). This explains the involvement of TUBB3 in drug resistance regardless of whether the disease is treated with a regimen that includes a microtubule targeting agent or not (22).
The present study was developed to clarify the role of ERCC1 and TUBB3 as predictive and prognostic factors in LACSCC patients treated with different therapeutic regimens.
Patients and Methods
Patients and Samples
The study included 88 women with histologically proved diagnosis of squamous cell carcinoma of the uterine cervix, treated at the Grupo Oncologico Cooperativo del Sur (G.O.C.S.) between January 1993 and December 2007; follow-up was documented for all patients. Patients presented with International Federation of Gynecology and Obstetrics (FIGO) stages IIB to IVA.
Among the 88 patients, 62 participated in 1 of 3 different prospective phase II trials conducted by our group: 19 received vinorelbine (VNB) 30 mg/m2 intravenous (i.v.) weekly for a total of 12 cycles; 18 received ifosfamide (IFX) 2,000 mg/m2 i.v. days 1-3, mesna 400 mg/m2 i.v. bolus at hours 0 and 4 and 800 mg/m2 orally at hour 8, VNB 25 mg/m2 i.v. days 1 and 8 and cisplatin (CDDP) 75 mg/m2 i.v. day 3 (these cycles were repeated every 28 days for a total of 3 courses); 25 women received docetaxel (DTX) 100 mg/m2 every 3 weeks for 3 courses. Eligibility criteria, clinical staging, response criteria, treatment strategy and toxicity evaluation were described in previous reports from our group (3-5). In the subset of patients who required RT (n = 51), the classical “box” technique was used to deliver 50 Gy to the whole pelvis in 25 daily fractions over 5 weeks. When anatomically feasible, this treatment was followed by 2 intracavitary insertions scheduled 3 weeks apart. Insertions were intended to deliver 30 Gy to point A. Nine patients underwent radical hysterectomy with curative intent, and 2 had palliative surgery. The remaining 26 patients received CDDP 40 mg/m2 i.v. weekly with concurrent pelvic RT with curative intent.
Formalin-fixed paraffin-embedded tissues were obtained from 88 primary cervical carcinomas before treatment initiation.
All procedures followed the World Medical Association Declaration of Helsinki (Finland, 1964) and its further modifications. Informed consent was obtained from all patients included in this study. This study was approved by the regional ethics committee of the province of Neuquen, Argentina (CAIBSH # 2520/11). The Reporting Recommendations for Tumor Marker Prognostic Studies (REMARK) were followed (23).
Cervical cancer patients’ clinical and histological information are summarized in Table I.
Patients’ clinical and histological characteristics
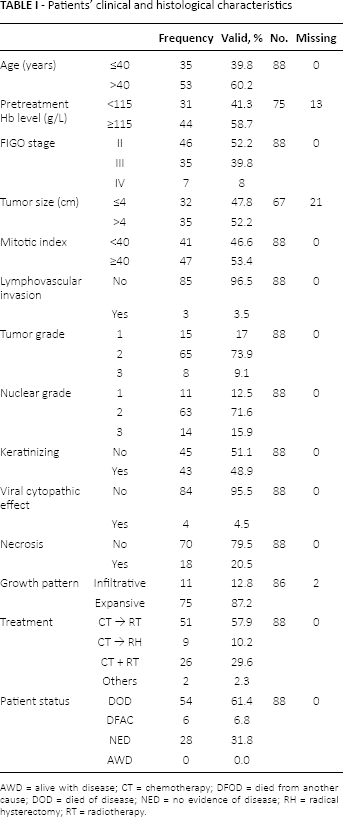
AWD = alive with disease; CT = chemotherapy; DFOD = died from another cause; DOD = died of disease; NED = no evidence of disease; RH = radical hysterectomy; RT = radiotherapy.
Monoclonal Antibodies
ERCC1 and TUBB3 antigens were detected using the monoclonal antibodies (mAbs) clone 8F1 (Santa Cruz Biotechnology, Santa Cruz, CA, USA), raised against full length of ERCC1 and the clone TU-20 (Santa Cruz, Biotechnology, Santa Cruz, CA, USA), raised against amino acids 441-448 of β3 tubulin of human origin, respectively.
Immunohistochemical Analysis
The technique was performed following standard procedures. All specimens were fixed in formalin and embedded in paraffin. Sections were deparaffinized in xylene and hydrated in a graded ethanol series; dewaxed sections were placed in methanol with hydrogen peroxide (3%) for 15 minutes to block endogenous peroxidase activity. After 3 washes with phosphate-buffered saline (PBS), sections were blocked for nonspecific binding with 50 μL of normal horse diluted 1:10 in 1% bovine serum albumin/PBS for 15 minutes, and rinsed. Before immunostaging of the samples with mAbs, tissues were treated with 10 mM sodium citrate buffer at 100°C for 5 minutes for antigenic retrieval. Subsequently, the sections were incubated with 50 µL of mAbs against ERCC1 (dilution 1:200) and TUBB3 (dilution 1:200) at room temperature 60 minutes in a moist chamber. After 3 rinses with PBS, the sections were incubated with peroxidase-conjugated anti-mouse/rabbit Igs (HiDef Detection™ HRP Polymer System, Cell Marque) for 60 minutes. After being washed, slides were counterstained with hematoxylin, dehydrated in ethanol, washed with xylene and coverslipped with mounting media. Negative controls were performed by adding PBS instead of the primary antibody.
Evaluation of the Staining
Specimens were examined with a light microscope by 2 independent observers (G.G. and A.Z.), who had no knowledge of the patient′s clinical data. Antibody staining patterns were scored in a semiquantitative manner. Staining intensities (cytoplasmic staining for TUBB3 and nuclear staining for ERCC1) were graded as negative (-), low (+), moderate (++) and strong (+++), using adjacent nonmalignant cells for reference. The number of low power (×10) optical fields in a specimen with a positive reaction was expressed as a percentage of the total number of optical fields containing tissue. Percentage of positive cells was graded as follows: 0, no positive cells; 0.1, positive in less than 9% of cells; 0.5, positive in 10%-49%; 1, positive in more than 50% of cells.
A semiquantitative scoring system (H-score) was calculated as the product of intensity and percentage of positive cells. The median value of all of the H-scores was a priori chosen as the cutoff point to classify ERCC1 and TUBB3 into high or low expression (17, 24, 25).
The pattern of reaction was categorized as linear (membrane), cytoplasmic or mixed (cytoplasmic with plasma membrane staining). Apical and nonapical staining, staining of the nucleus and adjacent normal tissue, and focal or diffuse staining were all evaluated.
Statistical Analysis
Patients were grouped according to the type of chemotherapy received: antimicrotubule agents alone such as VNB or DTX (group A); mixed agents such as IFX, VNB and CDDP (group B); and CDDP alone (group C).
The associations of ERCC1 and TUBB3 expression with the variables studied were tested with nonparametric tests. For categorical variables, the Wilcoxon rank sum test or Kruskal-Wallis test, including a Wilcoxon-type test for trend across ordered groups, were used.
Disease-free survival (DFS) was estimated from the date of diagnosis until the date of first tumor recurrence. Overall survival (OS) was calculated from the date of diagnosis until the date of death or last follow-up. The evaluation of treatment response was performed using the International Union for International Cancer Control (UICC), standard method used in solid tumors until 2000.
DFS and OS univariate associations with ERCC1 and TUBB3 expression were evaluated by Kaplan-Meier estimator with log rank test. A multivariable Cox regression model addressed DFS and OS associations among the scores of ERCC1 and TUBB3 expression and the variables studied. A multivariable Cox regression model addressed OS and PFS associations among ERCC1 score, TUBB3 score, age (≤40 vs. >40), pretreatment hemoglobin level (<115 g/L vs. ≥115 g/L), FIGO stage (early II vs. advanced III-IV), tumor size (≤4 cm vs. >4 cm), mitotic index (<40 vs. ≥40), histological grade (low 1-2 vs. high 3), nuclear grade (low 1-2 vs. high 3), keratinizing (yes vs. no), viral cytopathic effect (yes vs. no), tumoral necrosis (yes vs. no) and growth pattern (infiltrative vs. expansive). All variables where normalized and significance was set at a p value of <0.05. All calculations were done using the Statistix v8.0 software package.
Results
Patients’ Characteristics
The study included 88 stage IIB-IVA cervical cancer patients: median age was 43.5 years (range 24-74 years). Forty-six patients were FIGO stage II, whereas advanced stage was observed in 42 patients (35 patients were FIGO stage III and 7 patients stage IV). All tumors were squamous cell carcinoma. The other clinicopathological characteristics are summarized in Table I.
ERCC1 Staining
ERCC1 was localized to the nucleus, as shown in Figs. 1: A1, and B1. Notably, the majority of tumors expressed ERCC1 (65/88, 73.9%) predominantly in a diffuse area (54/65, 83%). Infiltrating inflammatory cells such as polymorphonuclear leukocytes served as internal control. Assessment of protein expression disclosed that 23 out of 88 tumors (26.1%) were score 0, 17 (19.3%) score 0.1, 19 (21.6%) score 0.5 and 29 (33%) score 1. In terms of staining intensity, 34 out of 88 tumors (52.3%) were weak, 18 (27.7%) moderate and 13 (20%) strong. The median value of H-score for ERCC1 was 0.5. Tumors with H-score exceeding 0.5 were classified as high ERCC1 expression. Among the 88 tumors examined, 53 (60.2%) were classified as low ERCC1 expression and 35 (39.8%) as high ERCC1 expression.
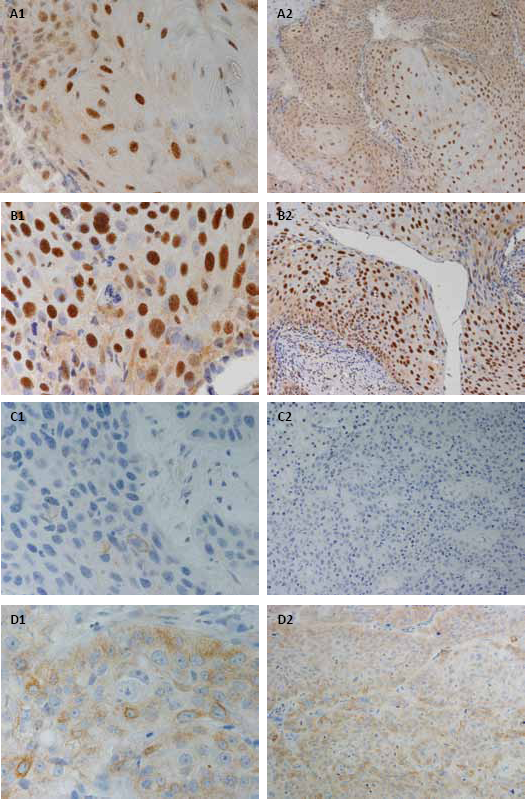
Representative examples of excision repair cross-complementation group 1 (ERCC1) and class III β-tubulin (TUBB3) immunostaining (magnification ×20 and ×60).
TUBB3 Staining
TUBB3 immunoreaction was observed in 52 of 88 cases (59.1%). TUBB3 was always found in the cytoplasm (Figs. 1: C1 and D1), and mainly in a focal area (80.7%) of the tumor sample. As internal control, TUBB3 was expressed in nerve tissue. Thirty-six tumors (40.9%) were percentage score 0, 38 (43.2%) score 0.1, 13 (14.8%) score 0.5 and only 1 tumor had score 1 (1.1%). Regarding the intensity, 44 tumors (84.6%) were weak, 7 (13.4%) were moderate and only 1 (1.9%) was strong. The median value of H-score for TUBB3 was 0.1. Tumors with H-score exceeding 0.1 were classified as high TUBB3 expression. Seventy of 88 cases (79.5%) showed low TUBB3 expression and 18 (20.5%) high TUBB3 expression.
Correlation with Clinicopathological Parameters
Table II presents clinicopathological characteristics and protein expression. A positive correlation was found between high ERCC1 tumor expression and patients with FIGO stage IV (p = 0.034). Tumors with unfavorable characteristics such as tumor size greater than 4 cm, mitotic index equal o greater than 40, elevated nuclear grade, presence of keratinization and viral cytopathic effect showed predominantly high ERCC1 expression. Infiltrative growth pattern more frequently showed low expression of ERCC1, whereas the expansive pattern had a high ERCC1 expression (p = 0.027). Cervical tumors of greater than 4 cm showed significantly higher TUBB3 expression (80% vs. 20%, p = 0.014). Similarly, high TUBB3 expression was seen more frequently in tumors with high mitotic index, poor differentiation and presence of necrosis. Although not statistically significant, TUBB3 expression showed similar characteristics with respect to growth pattern to those observed for ERCC1.
Clinicopathological characteristics and distribution of cases according ERCC1 and TUBB3 expression
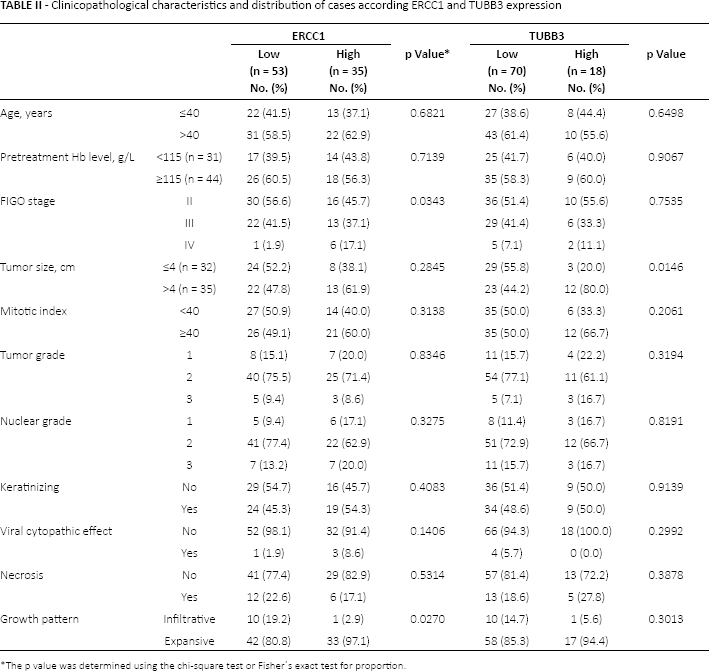
The p value was determined using the chi-square test or Fisher′s exact test for proportion.
There was no correlation (p = 0.320) between coexpression of both proteins, only 9/88 patients (26%) showed high expression of ERCC1 and TUBB3 in the same tumor.
ERCC1 and TUBB3 Status and Response to Treatment
Fifteen of 88 patients (17%) underwent surgery and 74/88 patients (84%) received external beam radiotherapy and brachytherapy.
In the whole series, progression of disease (PD) was more frequently observed in patients with high ERCC1 expression (49% vs. 28%, p = 0.094) (Tab. III). Patients who received CDDP-based schemes whose tumors overexpressed ERCC1 had higher rates of PD compared with those who received non-CDDP regimens (62% vs. 29%, respectively). TUBB3 did not show any correlation with clinical response.
Distribution of cases (%) with low/high of ERCC1 and TUBB3 expressions according to clinical response to treatment, recurrence and type of recurrence
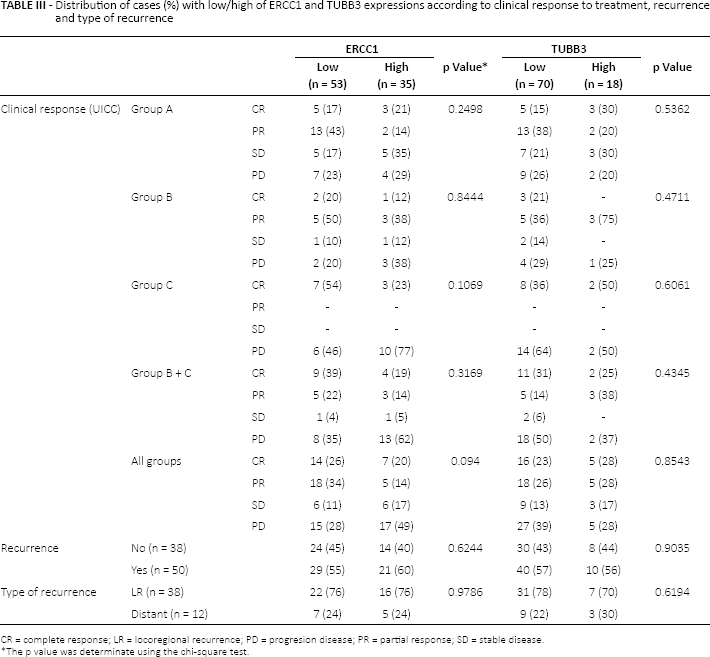
CR = complete response; LR = locoregional recurrence; PD = progresion disease; PR = partial response; SD = stable disease.
The p value was determinate using the chi-square test.
Neither ERCC1 nor TUBB3 expression was associated with development or type of recurrence.
Survival analysis
The median follow-up for all patients was 2 years (range 0.1-18.4 years). During the follow-up period, recurrence and death from disease occurred in 50 and 44 cases, respectively. The median DFS for all cases was 1.4 years (range 1.1-3.9 years) and OS was 2.3 years (range 1.6-4.5 years).
Advanced FIGO stage, presence of tumor necrosis and high ERCC1 expression (Figs. 2 and 3; Tab. IV) were associated in group C with poor DFS (p = 0.007, p = 0.040 and p = 0.021, respectively) and OS (p = 0.018, p = 0.054 and p = 0.005, respectively). In this group, low pretreatment hemoglobin level (<115 g/L) was also correlated with a poor OS (p = 0.031). On the other hand, age at diagnosis was the only factor associated with worse OS in group A (p = 0.038). TUBB3 expression did not correlate with either DFS or OS in patients who received antimicrotubule agents (Figs. 4 and 5). Group B did not show any association with the variables studied.
Univariate survival analysis according type of treatment received
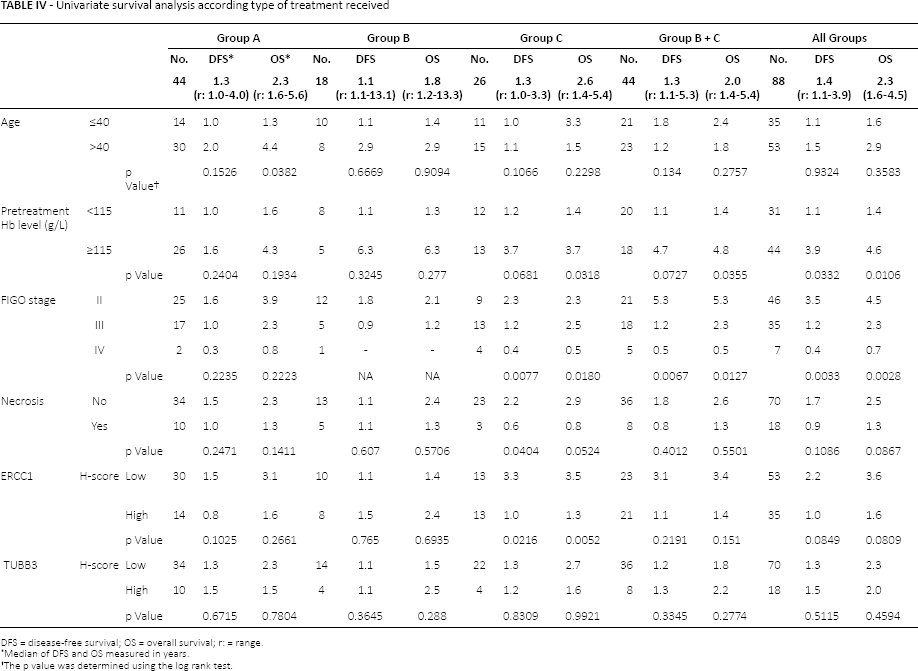
DFS = disease-free survival; OS = overall survival; r: = range.
Median of DFS and OS measured in years.
The p value was determined using the log rank test.
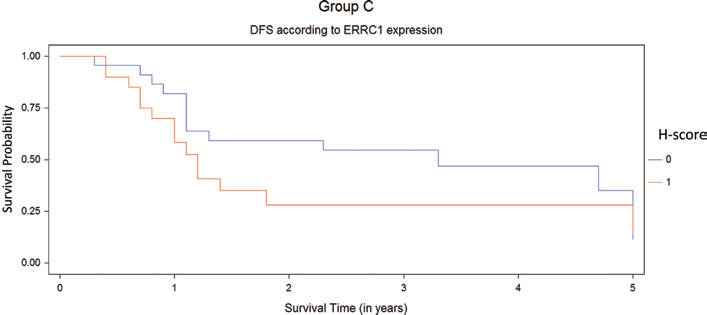
Disease-free survival (DFS) for platinum regimen population (group C) by Kaplan-Meier test. High H-score ERCC1 (1, red line) in the primary tumor was correlated with worse DFS (p = 0.021) in patients than in those with low H-score ERCC1 (0, blue line).
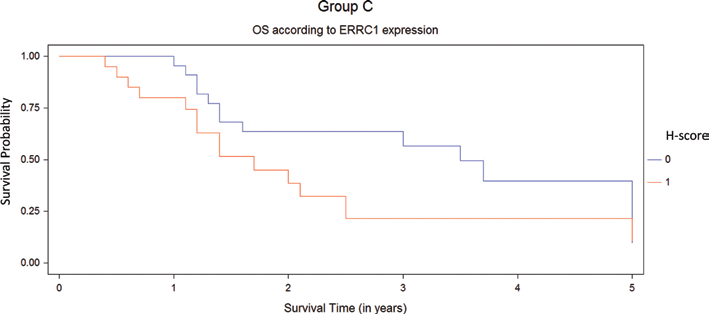
Overall survival (OS) for platinum regimen population (group C) by Kaplan-Meier test. High H-score ERCC1 (1, red line) in the primary tumor was correlated with worse OS (p = 0.005) in patients than those with low H-score ERCC1 (0, blue line).
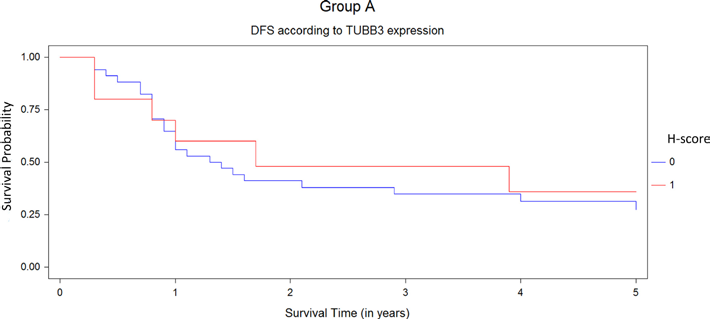
Disease-free survival (DFS) for antimicrotubule regimen population (group A) by Kaplan-Meier test. The survival analysis did not show any differences between the tumors with high (1, red line) or low expressions of TUBB3 (0, blue line).
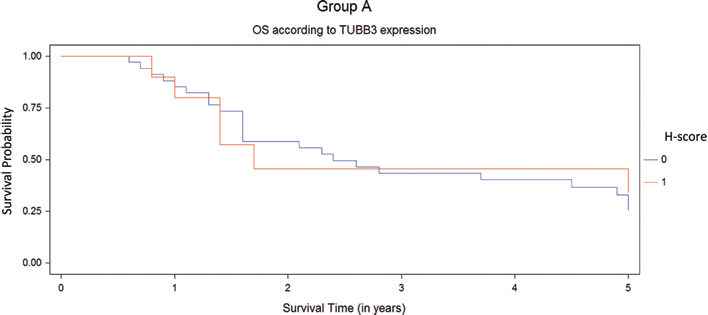
Overall survival (OS) for antimicrotubule regimen population (group A) by Kaplan-Meier test. The survival analysis did not show any differences between the tumors with high (1, red line) or low expressions of TUBB3 (0, blue line).
A subanalysis (Tab. IV) was made between CDDP-based chemotherapy groups (group B + C, n = 44) vs. non-CDDP (group A, n = 44). Low hemoglobin level and advanced FIGO stage were statistically significant prognostic factors in CDDP-based schemes (p = 0.035 and p = 0.012, respectively).
Patients whose tumors overexpressed with 1 or both proteins (Figs. 6 and 7) had a median DFS of 1 year (95% confidence interval [95% CI], 0.9-5 years) and median OS of 1.7 years (95% CI, 1.4-5 years); whereas patients with low expression of both proteins had a median DFS and OS of 2 years (95% CI, 1.1-5 years) and 3.3 years (95% CI, 1.6-5 years), respectively. Nonetheless, this difference was not significant (p = 0.468 and p = 0.355, respectively).
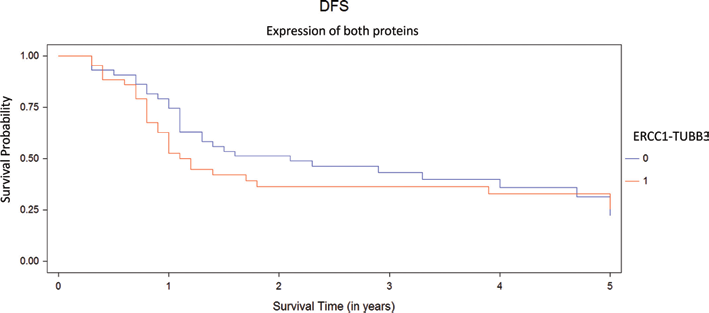
Disease-free survival (DFS) according expression of proteins, ERCC1-TUBB3 low/low (0, n = 44) and ERCC1-TUBB3 high/high or high/low or low/high (1, n = 44).
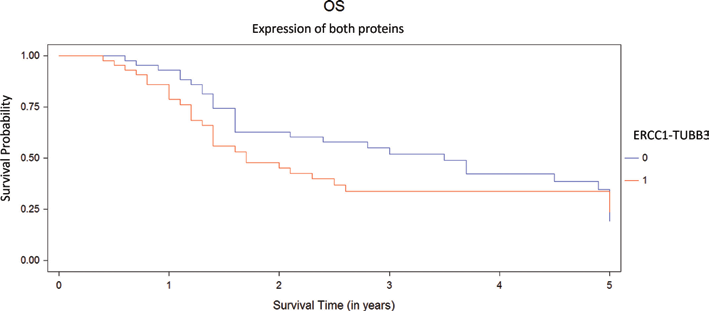
Overall survival (OS) according expression of proteins, ERCC1-TUBB3 low/low (0, n = 44) and ERCC1-TUBB3 high/high or high/low or low/high (1, n = 44).
For all patients, low pretreatment hemoglobin level and advanced FIGO stage remained as the only negative predictive (p = 0.032 and p = 0.003, respectively) and prognostic factors (p = 0.010 and p = 0.002, respectively); whereas high ERCC1 showed a tendency (p = 0.084 and p = 0.080, respectively) in the univariate analysis. In addition, by Cox regression analysis (Tab. V, Figs. 8 and 9), these variables were independent prognostic indicators of survival. There were no differences in DFS or OS between high and low TUBB3 expression.
Cox regression for disease-free survival and overall survival
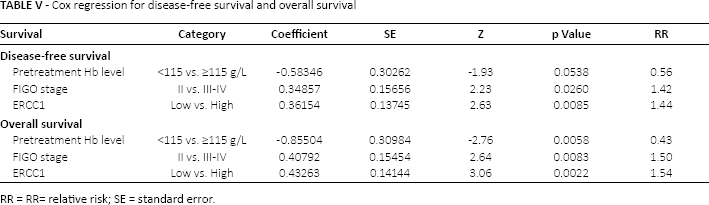
RR = RR= relative risk; SE = standard error.
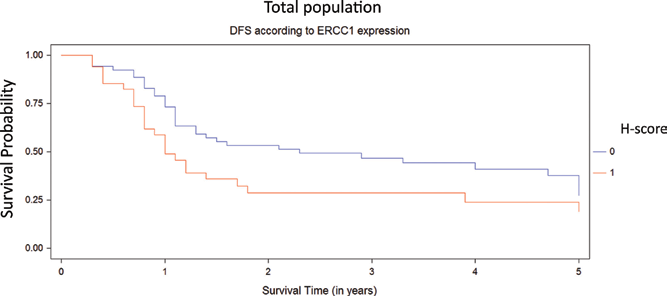
Kaplan-Meier disease-free survival (DFS) curves stratified by ERCC1 expression (high and low H-score represented in red and blue line, respectively) for total population.
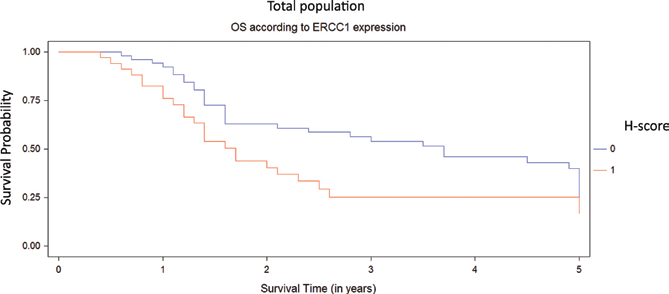
Kaplan-Meier overall survival (OS) curves stratified by ERCC1 expression (high and low H-score represented in red and blue line, respectively) for total population.
Discussion
The addition of concurrent cisplatin-based chemotherapy to standard radiotherapy could reduce the risk of recurrence and disease-related death from LACSCC by as much as 50% (26). However, those without a response will have toxicity without recourse to alleviation. It would be useful to predict response and to identify novel approaches for nonresponders (27).
The roles of ERCC1 and TUBB3 are under active investigation in several solid tumors. They are considered predictive and prognostic markers for chemotherapy at the same time. Because both molecules confer significant information for the selection of chemotherapy, they are of particular interest. In this study, we evaluated the clinical significance of ERCC1 and TUBB3 expression in pretreatment biopsy specimens from 88 patients with LACSCC exposed to different chemotherapeutic agents.
Our results are consistent with studies evaluating ERCC1 expression and response to cisplatin chemotherapy in other tumor sites. This study showed that patients with overexpression of ERCC1 have worse DFS and OS when treated with cisplatin (or cisplatin-based chemotherapy). Compared with patients who received noncisplatin agents, we found higher PD and lower complete response (CR) rates when ERCC1 is overexpressed. These data are in agreement with reports by other authors (17, 28). Liang et al assessed ERCC1 expression in 50 patients with LACSCC who received cisplatin-based concurrent CRT, by immunohistochemistry (29), and found that ERCC1-positive patients had lower CR rates than ERCC1-negative patients (75% vs. 97.1%, p = 0015); the 5-year OS rates for the ERCC1-positive and ERCC1-negative groups were 50% and 85.3%, respectively. Recently, Bai et al showed in a similar setting a significantly lower CR rate in patients with high ERCC1 mRNA levels than those with low expression levels (19.3% vs. 86.2%, p = 0.001) (30). These results could explain our findings of overexpression of ERCC1 in tumors with unfavorable characteristics such as tumor size >4 cm, high tumor proliferation index, expansive growth pattern and advanced stages. It is possible that ERCC1 status may be representative of a cell's intrinsic DNA damage repair ability, and represent the extent of accumulated intratumoral DNA damage, which may increase as tumors progress (31).
Preclinical and clinical studies have shown a relationship between ERCC1 expression and radioresistance (32, 33). Murray et al postulated that decreased ERCC1-XPF activity in tumors could increase hypoxic radiosensitivity (34). This could be the biologic reason behind the improvement in survival among ERCC1-low patients with squamous cell carcinoma of the head and neck treated with radiotherapy alone (35, 36). It would explain in part why our data showed poor DFS and OS in the univariate and multivariate analysis for all studied patients when ERCC1 was overexpressed regardless of the chemotherapy agent administered (87% of patients underwent radiotherapy). However, we must take into account that our study groups underwent heterogeneous chemotherapy regimens, and the cisplatin group (30% of total population) could influence the final statistical results.
A recent study conducted by Muallem et al failed to demonstrate a correlation between low levels of ERCC1 expression and poor outcomes in 112 patients with LACSCC (37). This was, however, a retrospective study and contradicts previous findings from the literature. Nonetheless, the study did show improvement in prognosis with increased expression of ERCC1.
TUBB3 was initially identified to confer antimicrotubule resistance in several solid tumors, but recent studies showed also an increased resistance to DNA-damaging agents such as cisplatin, doxorubicin and etoposide (22). This finding lends support to the hypothesis that TUBB3 acts as a survival factor for cancer cells, which prevents cell death after exposure to chemotherapeutic agents (38).
To the best of our knowledge, this is the first study to evaluate TUBB3 expression in LACSCC patients who received antimicrotubule agents as neoadjuvant treatment. In contrast to reports in other solid tumors (i.e., lung, ovarian, gastric, breast and unknown primary), we did not find any relationship between tumoral TUBB3 expression and survival either in patients who received antimicrotubule agents or in the total population. Despite the role of TUBB3 also being tested with diverse cutoffs, we did not find meaningful results. Furthermore, there was no association of this protein with clinical response. Similar results were reported by Ferrandina et al in 98 LACSCC patients who underwent neoadjuvant chemotherapy with cisplatin plus 5-fluorouracil (39).
Similar to ERCC1, univariate analysis showed a significant association between high TUBB3 with tumor size >4 cm and expansive growth pattern, as well as predominance in tumors with high mitotic index, poor differentiation and presence of tumor necrosis. These findings support the theory that TUBB3 expression could be a mediator of a more aggressive tumor phenotype through multiple cell survival pathways active under stress conditions such as hypoxia, ischemia and poor nutrient supply (21, 40). Interestingly, we observed worse DFS and OS when TUBB3 and ERCC1 were co-overexpressed in LACSCC. The true meaning of this finding requires further analysis in a large number of patients.
In summary, our study confirmed that ERCC1 status is a predictive and prognostic factor in LACSCC patients who receive a cisplatin regimen. This protein could be helpful to identify poor prognosis in LACSCC patients who are potential candidates to more aggressive and/or targeted therapy. In contrast to findings in other tumors, the assessment of TUBB3 status provides no advantage in identifying LACSCC patients with a poor chance of response to antimicrotubule agents and unfavorable prognosis. Because both molecules are associated with aggressive tumor behavior, their co-overexpression and relationship with survival should be evaluated in further studies.
Although the number of patients included in this study was relatively small and the findings thus need to be interpreted with caution, confirmation of our results in a large prospective study will pave the way for personalized therapy by helping to select LACSCC patients more likely to benefit from CRT.
Footnotes
Acknowledgement
The authors want to express their appreciation to Dr. Angelo Paradiso and Dr. Amada Segal-Eiras for their collaboration in the preparation of this manuscript, and to Dr. Maria May for her help in the editing of figures.
Financial support: This study was carried out thanks to a CISNe grant awarded by the Ministry of Health of the Province of Neuquén, Argentina.
Conflict of interest: The authors declare that they have no financial relationship that may lead to a conflict of interest in relation to the submitted manuscript.
