Abstract
Background:
The combination patterns of pathological intermediate-risk factors and the choice of adjuvant therapy for early-stage cervical cancer (CC) remain controversial.
Objectives:
To develop and validate nomogram-based prediction models incorporating pathological intermediate-risk factors to predict survival outcomes and optimize adjuvant therapy strategies in early-stage CC patients.
Design:
A multicenter retrospective study.
Methods:
A total of 1104 patients with stage IB-IIA CC who underwent primary surgical treatment and had no pathological high-risk factors were retrospectively enrolled from three tertiary medical centers in China between January 2005 and December 2017. Patients were randomly assigned to development and validation cohorts (approximately 3:1 ratio). Prognostic models for disease-free survival (DFS) and overall survival (OS) were developed by Cox proportional hazards regression and visualized using nomograms.
Results:
In this study, four prognostic models were developed incorporating different combinations of five key variables: lymphovascular space involvement (LVSI), stromal invasion (SI), tumor size (TS), histological type, and adjuvant therapy. Among these, Model 4 (LVSI + SI + TS + histological type + adjuvant therapy) demonstrated the highest discriminatory performance, with C-indices of 0.79 for both DFS and OS in the development cohort, and 0.84 for DFS and 0.77 for OS in the validation cohort. Model 4 also effectively stratified patients into prognostic risk groups in both cohorts, with high-risk patients exhibiting significantly worse DFS (development cohort: p < 0.0001; validation cohort: p = 0.0011) and OS (development cohort: p < 0.0001; validation cohort: p = 0.0036) compared to low-risk patients.
Conclusion:
The nomogram models developed in this study may provide individualized prognostic predictions for early-stage CC patients, potentially facilitating personalized decision-making regarding adjuvant therapy, though further validation in diverse patient cohorts and prospective studies is needed.
Introduction
Cervical cancer (CC) continues to be a major global health burden, accounting for about 600,000 new cases and 300,000 deaths annually, and is the fourth most prevalent malignancy among women worldwide. 1 Despite screening advances, survival improvements remain limited, particularly in low-resource countries.2,3 For early-stage CC, defined as International Federation of Gynecology and Obstetrics (FIGO) stage IB-IIA, potentially curative treatment options include radical hysterectomy with lymph node dissection or radical pelvic radiotherapy with vaginal brachytherapy.4–6 Postoperative histopathological examination often reveals critical prognostic indicators. 7 While the necessity of postoperative concurrent chemoradiation in cases with high-risk factors (such as lymph node metastasis, parametrial involvement, or positive resection margins) is well-established, 8 considerable debate surrounds the definition and management of intermediate-risk factors. 9
The landmark GOG49 study established capillary lymphatic spaces (CLS), tumor size (TS), and depth of tumor invasion as independent prognostic factors for recurrence in early-stage cervical squamous cell carcinoma (SCC) following radical surgery. 10 Building upon these findings, the GOG92 trial demonstrated that adjuvant radiation significantly reduced recurrence rates (from 28% to 15%) in patients exhibiting at least two intermediate-risk factors: >1/3 stromal invasion (SI), CLS involvement, and large clinical tumor diameter, subsequently establishing the Sedlis criteria. 11 However, these criteria demonstrated limited sensitivity, as approximately half of the recurrences occurred in patients not meeting these criteria. 12 After that, Ryu et al. 13 proposed an enhanced “four-factor model” incorporating histological type alongside lymphovascular space involvement (LVSI), TS, and SI, which showed improved predictive accuracy for post-surgical recurrence. In 2021, Levinson et al. 14 developed histology-specific nomograms based on auxiliary analyses of GOG49, 92, and 141, offering a more nuanced approach to recurrence risk prediction across different histological subtypes and enabling more targeted adjuvant treatment recommendations. Nevertheless, in this study, the retrospective nature of the analysis may introduce inherent selection and information biases; additionally, as the data were collected from GOG institutions with patient diagnoses dating back to earlier periods, the generalizability of these findings to contemporary populations and regions remains to be determined. Furthermore, the existing criteria for identifying intermediate-risk factors lack standardization and do not fully account for emerging prognostic factors, underscoring the need for more comprehensive and updated risk assessment tools. Consequently, the optimal approach to evaluating pathological intermediate-risk factors and selecting postoperative adjuvant treatments for early-stage CC patients remains a topic of ongoing discussion in clinical practice.
Given these considerations, this study sought to establish and validate nomogram-based prediction models for disease-free survival (DFS) and overall survival (OS) in early-stage CC patients without pathological high-risk factors, based on data from three tertiary medical centers. The findings are intended to provide evidence-based guidance for optimizing postoperative adjuvant treatment decisions in clinical practice.
Methods
We followed the TRIPOD (Transparent Reporting of a multivariable prediction model for Individual Prognosis or Diagnosis) guidelines when preparing this article (Supplemental Material). 15
Patients
This retrospective study enrolled patients with early-stage CC who underwent primary surgical treatment between January 2005 and December 2017 at three tertiary medical centers in China (Shandong Provincial Hospital Affiliated to Shandong First Medical University, Qilu Hospital of Shandong University Dezhou Hospital, and Shandong Provincial Maternal and Child Health Care Hospital Affiliated to Qingdao University). The inclusion criteria were: (1) FIGO (2018) stage IB-IIA disease, 16 and (2) underwent radical or modified radical hysterectomy with pelvic lymphadenectomy. Patients with the following conditions were excluded: (1) pathological high-risk factors (lymph node metastasis, parametrial involvement, or positive resection margins), (2) concurrent other primary malignancies, or (3) incomplete medical records.
All patients were clinically staged according to the 2018 FIGO system, based on comprehensive pelvic examination by two experienced gynecologic oncologists, supplemented by imaging modalities including ultrasound, computed tomography (CT), and/or magnetic resonance imaging (MRI) to assess TS, local extension, and lymph node involvement. All surgical specimens were examined independently by at least two experienced pathologists at each participating center. The pathological assessments included evaluation of histological type and grade, LVSI, SI (classified as superficial, middle 1/3, or deep 1/3), TS (measured as the largest diameter on gross examination), lymph node status, parametrial status, and resection margin status. Discrepancies between pathologists were resolved by consensus discussion.
Predictors and endpoints
Clinical and pathologic data collected included demographic characteristics (age, menopausal status), medical history (chronic comorbidities), tumor features (FIGO stage (2018), histological type and grade, LVSI, SI, TS), and treatment details (surgical approach and adjuvant therapy). The primary endpoints were DFS and OS. DFS was determined by the interval from the surgical date to the initial recurrence or the final follow-up, whereas OS was assessed based on the period from the surgery to death due to any cause or the last follow-up.
Model development and validation
The study workflow is illustrated in Figure 1. The study population (n = 1104) was randomly allocated to development and validation cohorts in approximately a 3:1 ratio. Prediction models were constructed using the development cohort and subsequently validated in the validation cohort. Univariable and multivariable Cox proportional hazards analyses were performed for both DFS and OS, with results expressed as hazard ratio (HR) and 95% confidence intervals (CI). The prediction models incorporated several combinations of histological type and established Sedlis criteria components (LVSI, SI, and TS), along with adjuvant treatment methods, based on clinical relevance and statistical significance. Model performance was assessed using the concordance index (C-index). The optimal model was selected to develop nomograms predicting 3- and 5-year DFS and OS probabilities. Each variable’s regression coefficient (β) was proportionally translated into a corresponding point value on the nomogram, with higher coefficients assigned higher point weights. The total score for each patient, corresponding to the individual risk score, was obtained by summing the points assigned to the variables included in the model. The median risk score served as the threshold for stratifying patients into low- and high-risk groups. The discriminative ability between risk groups was evaluated using Kaplan-Meier survival analysis with the log-rank test.

The workflow of this study.
Statistical analysis
Statistical analyses were performed using SPSS software (version 27.0.1; IBM Corp., Armonk, NY, USA) for descriptive statistics and R software (version 4.4.1; R Foundation for Statistical Computing, Vienna, Austria) for advanced analyses, including Cox proportional hazards regression modeling, nomogram construction, C-index calculation, and survival comparisons via the log-rank test.
Results
Patient characteristics
The characteristics of the patients are presented in Table 1. In total, 1104 patients with early-stage CC were included in this study and randomly divided into development (n = 800) and validation (n = 304) cohorts. The FIGO stage distribution was as follows: IB1 (n = 290, 26.3%), IB2 (n = 526, 47.6%), IB3 (n = 173, 15.7%), IIA1 (n = 69, 6.3%), and IIA2 (n = 46, 4.2%). Squamous histology was observed in 923 (83.6%) patients, while nonsquamous histology accounted for 181 (16.4%). LVSI was observed in 126 (11.4%) patients. Laparotomy was performed in 1042 (94.4%) patients, with laparoscopy in 62 (5.6%). A total of 897 (81.2%) patients received adjuvant therapy. During a median follow-up of 101 months (range: 2–197 months), 84 (7.6%) patients experienced recurrence, and 54 (4.9%) patients died.
Characteristics of cervical cancer patients in development and validation cohorts.

Values are presented as n (%).
FIGO, International Federation of Gynecology and Obstetrics; LVSI, lymphovascular space involvement; SI, stromal invasion; TS, tumor size.
Predictor assessment of DFS and OS
Table 2 presents the univariable and multivariable Cox proportional hazards analyses results from the development cohort. For DFS, independent prognostic factors were middle 1/3 SI (HR: 7.59, 95% CI: 2.16–26.61), deep 1/3 SI (HR: 12.84, 95% CI: 3.67–44.97), and adjuvant therapy (all p < 0.001). For OS, significant predictors included menopause (HR: 2.68, 95% CI: 1.24–5.77), diabetes (HR: 6.05, 95% CI: 1.33–27.46), LVSI (HR: 2.70, 95% CI: 1.11–6.54), deep 1/3 SI (HR: 5.18, 95% CI: 1.48–18.20), 2 cm ⩽ TS < 4 cm (HR: 10.00, 95% CI: 1.17–85.65), and adjuvant therapy (all p < 0.05).
Univariable and multivariable Cox proportional hazards regression analysis for DFS and OS.
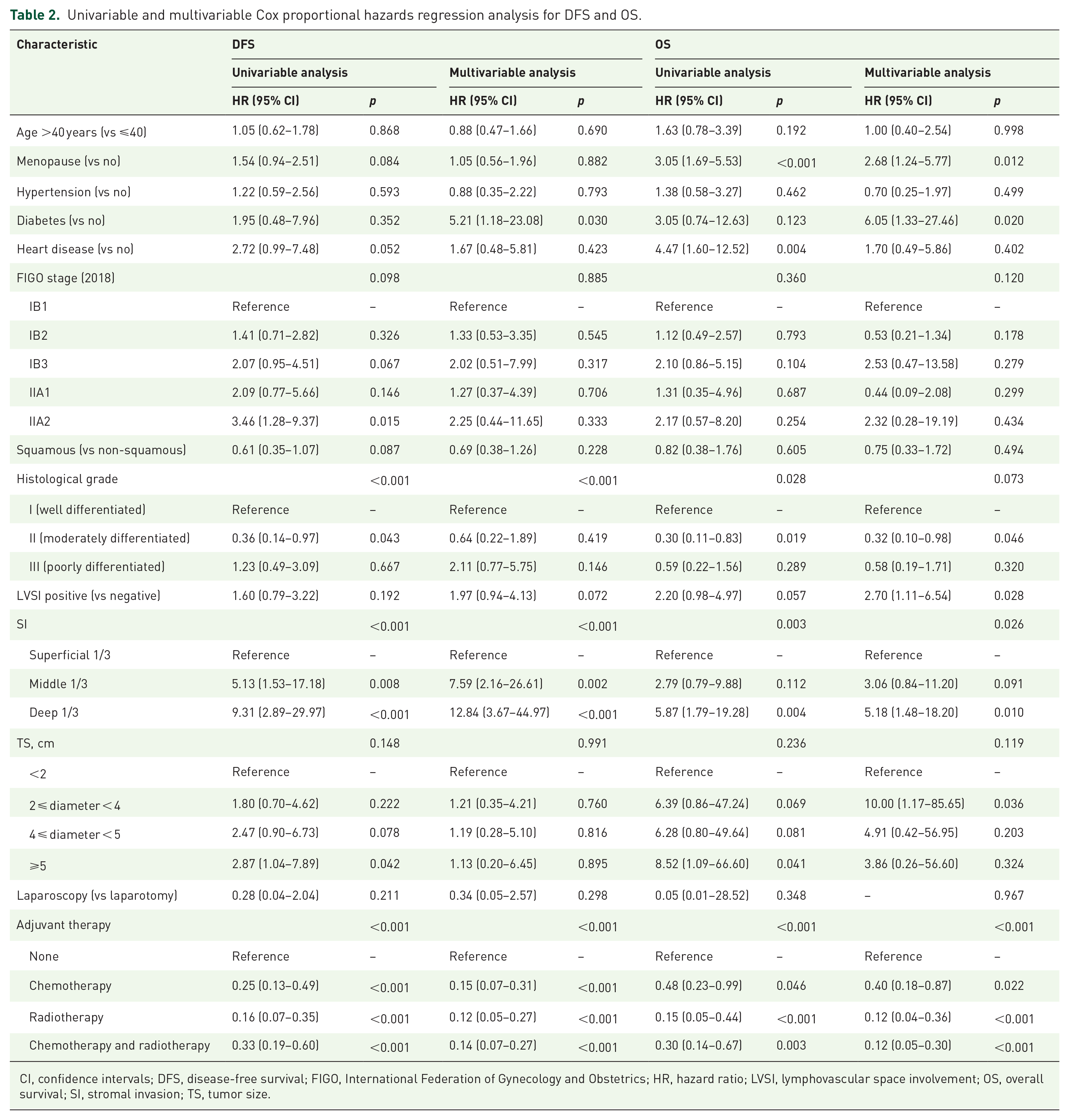
CI, confidence intervals; DFS, disease-free survival; FIGO, International Federation of Gynecology and Obstetrics; HR, hazard ratio; LVSI, lymphovascular space involvement; OS, overall survival; SI, stromal invasion; TS, tumor size.
Model development and validation of DFS and OS
Based on the clinical significance of Sedlis criteria, histological type, and adjuvant therapy in clinical practice, four prediction models were constructed incorporating different combinations of five variables (LVSI, SI, TS, histological type, and adjuvant therapy). As shown in Table 3, these models comprised: Model 1 (LVSI + SI + TS), Model 2 (LVSI + SI + TS + histological type), Model 3 (LVSI + SI + TS + adjuvant therapy), and Model 4 (LVSI + SI + TS + histological type + adjuvant therapy). Discrimination analysis showed that Model 4 performed the best, with C-indices of 0.79 for both DFS and OS in the development cohort, and 0.84 for DFS and 0.77 for OS in the validation cohort.
C-index of DFS and OS prediction models for different combinations of intermediate-risk factors.
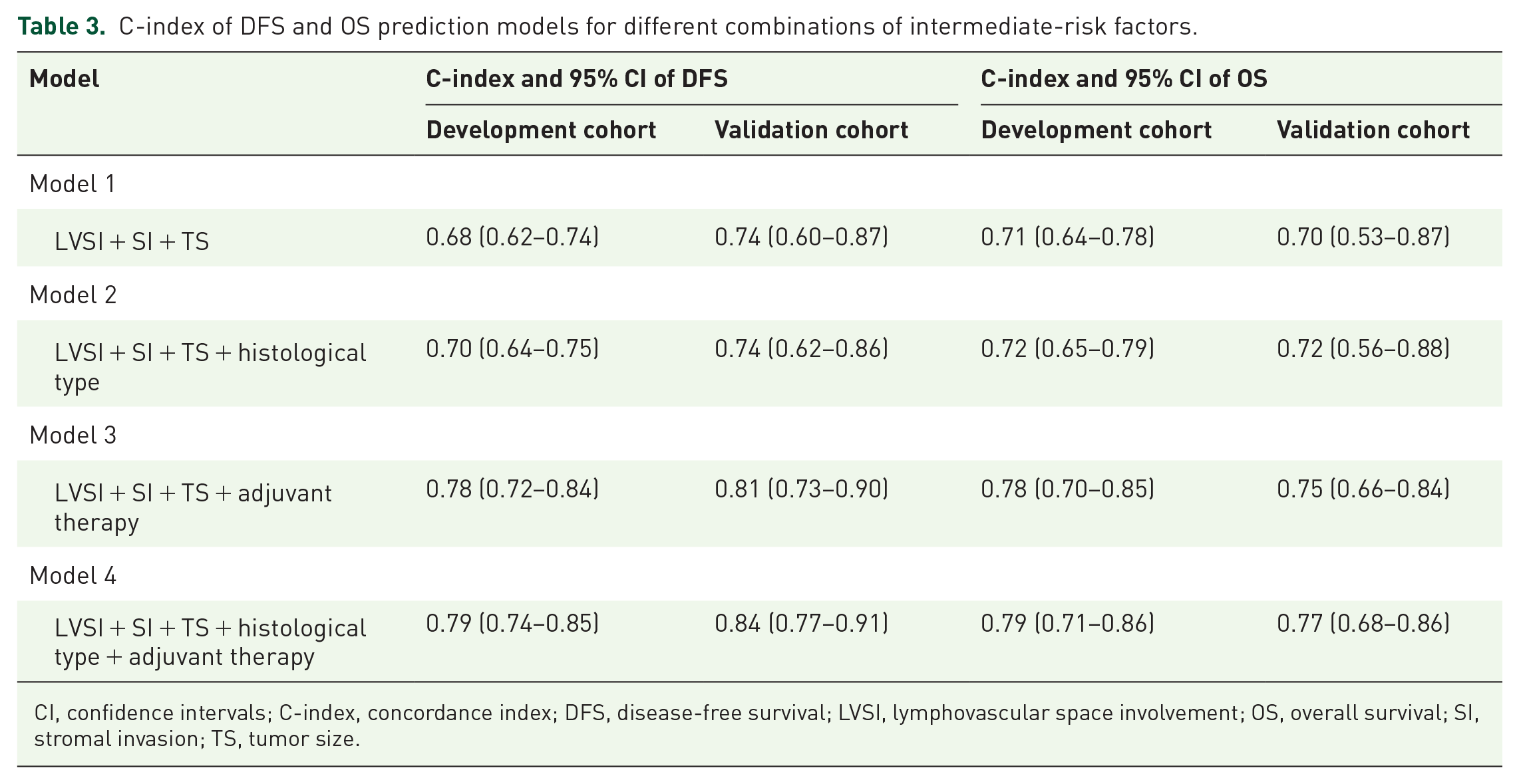
CI, confidence intervals; C-index, concordance index; DFS, disease-free survival; LVSI, lymphovascular space involvement; OS, overall survival; SI, stromal invasion; TS, tumor size.
The optimal prediction models for DFS and OS were visualized as nomograms (Figure 2). The risk scores, defined as the sum of the weighted points assigned to each variable, were calculated as follows: DFS risk score = 25.1 × (LVSI positive) + 12.4 × (2 cm ⩽ TS < 4 cm) + 17.6 × (4 cm ⩽ TS < 5 cm) + 20.8 × (TS ⩾ 5 cm) +75 × (middle 1/3 SI) +100 × (deep 1/3 SI) + 22.4 × (non-SCC) + 10.8 × (chemotherapy and radiotherapy) + 12.6 × (chemotherapy) + 82 × (no adjuvant treatment); and OS risk score = 44.9 × (LVSI positive) + 77 × (2 cm ⩽ TS< 4 cm) +68.5 × (4 cm ⩽ TS < 5 cm) + 87.5 × (TS ⩾ 5 cm) + 48 × (middle 1/3 SI) + 83.6 × (deep 1/3 SI) + 11.3 × (non-SCC) + 8 × (chemotherapy and radiotherapy) + 46 × (chemotherapy) + 100 ×(no adjuvant treatment). The risk scores corresponded to the predicted 3- and 5-year probabilities of DFS and OS as shown in the nomograms.

Nomogram lists of risk prediction models for DFS (a) and OS (b). For example, an early-stage FIGO IB1 cervical cancer patient with postoperative pathological results showing non-SCC, negative LVSI, deep SI, and a TS of 2 cm would not meet the current Sedlis criteria for adjuvant treatment. However, using our nomogram, this patient would be classified as high risk for recurrence (22.4 points for non-SCC + 100 points for deep SI + 12.4 points for 2 cm ⩽ TS < 4 cm = 134.8 points) and high risk for death (11.3 points for non-SCC + 83.6 points for deep SI + 77 points for 2 cm ⩽ TS < 4 cm = 171.9 points). Therefore, the choice of postoperative adjuvant therapy for this patient may require further discussion by the gynecologic oncologists.
Risk stratification for DFS and OS
Based on the median nomogram-derived risk scores, the cohort was divided into high- and low-risk groups. In the DFS model, the median risk score was 106.6 (range: 0–224.7), with patients having risk scores >106.6 classified as high-risk for recurrence. In the OS model, the median risk score was 162.5 (range: 0–297.0), and those with scores >162.5 were considered high-risk for death. Kaplan-Meier analysis in the development cohort demonstrated significantly worse outcomes in high-risk groups for both DFS (p < 0.0001; Figure 3(a)) and OS (p < 0.0001; Figure 3(b)). These findings were confirmed in the validation cohort, with high-risk groups showing significantly inferior DFS (p = 0.0011; Figure 3(c)) and OS (p = 0.0036; Figure 3(d)).

Kaplan-Meier analysis of risk groups with DFS and OS in the development and validation cohorts. (a) DFS in the development cohort. (b) OS in the development cohort. (c) DFS in the validation cohort. (d) OS in the validation cohort.
Discussion
This study presents validated prediction models for DFS and OS in early-stage CC patients, incorporating pathological intermediate-risk factors and postoperative adjuvant therapy. The Cox proportional hazards Model 4, which integrated histological type and adjuvant therapy with established risk factors (LVSI, SI, and TS), demonstrated superior discriminative ability. The risk stratification system effectively identified distinct prognostic groups, with significant differences in survival outcomes between high- and low-risk cohorts. These findings were subsequently confirmed in the validation cohort, supporting the model’s predictive reliability. The developed risk assessment model offers potential clinical utility for postoperative prognostication and adjuvant treatment decision-making in early-stage CC patients.
The Sedlis criteria, comprising LVSI, SI, and TS, remain the most widely adopted system for determining the treatment approach in early-stage CC patients without pathological high-risk factors. 11 And the criteria were primarily derived from research into SCC; however, the distinct biological characteristics and prognostic patterns between SCC and non-SCC have been well-documented.17,18 Furthermore, given that the study population received treatment guided by the Sedlis criteria in recent years, adjuvant therapy emerged as a crucial prognostic factor, significantly improving survival outcomes in early-stage CC patients. Therefore, based on clinical relevance and established risk factors, four prediction models were developed by systematically incorporating combinations of five variables (LVSI, SI, TS, histological type, and adjuvant therapy) in this study. Model 1 included the Sedlis criteria components (LVSI, SI, and TS); Model 2 incorporated histological type into Model 1; Model 3 added adjuvant therapy to Model 1; and Model 4 integrated all five variables.
Based on the GOG49 and GOG92 studies in patients with early-stage CC, CLS was considered an independent prognostic factor for recurrence and was included as a key component of the Sedlis criteria.10,11 As the pathological understanding of CC biology deepened, LVSI, providing a more comprehensive description of vascular and lymphatic invasion, gradually supplanted the concept of CLS. 19 Multiple studies have shown that LVSI positive is associated with lymph node metastasis, distant metastasis, and reduced survival in patients.20,21 Consistently, our multivariable Cox proportional hazards analysis demonstrated that LVSI-positive patients exhibited poorer OS (HR: 2.70, 95% CI: 1.11–6.54), further validating its adverse prognostic impact. Moreover, our study identified deep 1/3 SI as an independent prognostic factor for both DFS (HR: 12.84, 95% CI: 3.67–44.97) and OS (HR: 5.18, 95% CI: 1.48–18.20), which aligns with previous research and confirms that deep SI is associated with significantly poorer survival outcomes, emphasizing its value as a reliable prognostic indicator in CC.10,11,13
In addition, our study also confirmed that 2 cm ⩽ TS < 4 cm is strongly associated with poorer OS (HR: 10.00, 95% CI: 1.17–85.65), supporting prior studies that have highlighted the critical role of TS in prognosis.11,22 In our cohort, TS measurements were obtained through clinical pelvic examination by two experienced gynecologic oncologists, supplemented by imaging modalities including ultrasound, CT, and/or MRI, in accordance with the 2018 FIGO staging recommendations. While this approach reflects real-world clinical practice, it may still introduce some degree of subjectivity in measurement. In contrast, the study by Ryu et al. 13 employed dedicated imaging techniques such as CT and MRI for TS evaluation, providing stronger evidence that TS is correlated with recurrence and survival after CC surgery. Notably, the 2018 FIGO cancer report endorsed the use of imaging findings for clinical staging, reflecting the increasing adoption of CT and MRI in high-income countries.16,23 This evolution in tumor assessment methodology suggests that future studies might benefit from incorporating standardized imaging-based TS measurements to enhance the accuracy and reproducibility of prognostic models.
The World Health Organization reports that non-SCC of the cervix, which mainly includes adenocarcinoma, adenosquamous carcinoma, and neuroendocrine tumors, accounts for more than 25% of CC cases. 24 Non-SCC shows distinct differences from SCC in terms of risk factors, therapy response, and overall prognosis.17,18 An analysis of GOG 92 with a 10-year follow-up revealed that radiation provided a greater benefit for women with non-SCC, reducing the risk from 44% to 9%, compared to a reduction from 28% to 20% in those with SCC. 25 In addition, the introduction and widespread recognition of the “four-factor model” and the histology-specific nomogram further underscore the significant impact of different histological types on prognosis.13,14 These highlight the necessity of incorporating histological type as a key factor in clinical modeling and risk stratification.
Current guidelines for early-stage CC patients without pathological high-risk factors present varying recommendations. The European Society for Medical Oncology suggests no further adjuvant therapy (evidence level II, B), 4 while the FIGO report advocates postoperative radiotherapy without chemotherapy when any two risk factors (TS > 4 cm, LVSI, or deep SI) are present. 26 The National Comprehensive Cancer Network advises pelvic external beam radiation therapy (category 1) as the standard approach, with the addition of concurrent platinum-containing chemotherapy (category 2B) being an optional consideration, for patients with stage IA2, IB, or IIA1 CC who are lymph node-negative and exhibit large primary tumors, deep SI, and/or LVSI. 27 Although consensus on postoperative adjuvant therapy remains controversial, the patients in this study received treatment according to these established guidelines. Consistent with our previous research on intermediate-risk factors, 28 the inclusion of adjuvant therapy as a prognostic variable in the current model may facilitate more individualized treatment decisions. This distinctive incorporation of adjuvant therapy represents a key methodological difference from the prediction model developed by Levinson et al. 14
Our study was based on a retrospective cohort of early-stage CC patients treated at three tertiary medical centers, providing a representative multicenter dataset. In addition to multicenter experiences, findings from recent single-center studies have also corroborated the prognostic significance of pathological intermediate-risk factors and postoperative adjuvant therapy. Cao et al. 29 conducted a retrospective analysis of 861 patients with intermediate-risk, early-stage cervical SCC at a single institution, demonstrating that LVSI and deep 1/3 SI were significant independent predictors of recurrence and survival, while the benefit of adjuvant therapy remained controversial. Zhang et al. 30 analyzed early-stage CC patients undergoing radical surgery in a single center and found that adenocarcinoma was associated with poorer DFS compared to SCC, whereas no significant survival difference was observed between adenosquamous carcinoma and SCC. These consistent findings across both multicenter and single-center studies strengthen the clinical relevance and generalizability of our prognostic model, highlighting the necessity of developing more precise and individualized postoperative management strategies for patients with early-stage CC.
The major strength of our study lies in the systematic evaluation of various combinations of intermediate-risk factors in early-stage CC patients, ultimately developing an optimal five-factor prognostic model with superior discriminative ability. The visualization of this model through a nomogram enhances its clinical applicability and provides a practical reference tool for treatment decision-making. In this study, we specifically focused on patients without pathological high-risk factors, as this subgroup remains clinically heterogeneous with no clear consensus on risk stratification or adjuvant therapy. 7 Future investigations will extend this work to include patients with high-risk factors, enabling broader validation and refinement of prognostic models and ultimately facilitating more precise and personalized care for patients with early-stage CC.
This study also has several inherent limitations. First, its retrospective nature introduces the potential for selection bias and population heterogeneity, including variability in surgical procedures and adjuvant treatments across the three participating centers. Second, the inclusion of patients over a 12-year period may result in temporal bias, as evolving treatment strategies and clinical practices during this time could influence outcomes and affect the generalizability of our findings. Although the model demonstrated good performance in internal validation, external validation using independent cohorts is still lacking. Future large-scale, prospective, and multicenter studies are warranted to address these limitations and further validate our findings.
Conclusion
This study proposed a five-factor prognostic model (incorporating LVSI, SI, TS, histological type, and adjuvant treatment) for early-stage CC without pathological high-risk factors. The model demonstrates enhanced predictive ability for DFS and OS, offering a potential tool for individualized risk assessment and treatment optimization. However, further validation in larger, prospective studies is needed to confirm its clinical utility.
Supplemental Material
sj-pdf-1-tam-10.1177_17588359251359461 – Supplemental material for Development and validation of prognostic prediction models for early-stage cervical cancer patients based on pathological intermediate-risk factors
Supplemental material, sj-pdf-1-tam-10.1177_17588359251359461 for Development and validation of prognostic prediction models for early-stage cervical cancer patients based on pathological intermediate-risk factors by Zihan Wang, Ran Chu, Namei Wu, Ming Yuan, Xiao Song, Wei Tian, Chunrun Yang, Jipeng Wan and Guoyun Wang in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
