Abstract
Introduction
Oncogenic hotspot mutations in the promoter region of the TERT gene have been identified in several cancer types as being associated with a worse outcome. Additionally, a polymorphism (rs2853669) in the TERTpromoter region was reported to modify the survival of TERT-mutated patients. Our aim is to determine the frequency of c.-124 C>T and c.-146 C>T TERT mutations and to genotype the rs2853669 polymorphism in a series of 68 soft tissue sarcomas (STS) comprising 22 histological subtypes.
Methods
PCR was performed, followed by direct sequencing of a fragment of TERT containing the hotspots and the rs2853669.
Results
We found TERTmutations in 4/68 (5.9%) STSs including 1 pleomorphic liposarcoma (1/1), 1 dedifferentiated liposarcoma (1/1) and 2 myxoid liposarcomas (2/9). The variant C allele of rs2853669 was found in 54.8% (34/62) of all STSs and in 75% (3/4) of TERT-mutated cases. TERT mutations were associated with younger age, and the C allele of the rs2853669 was associated with high histological grade (2 and 3). No association was found between TERT mutation status or rs2853669 genotype and patient prognosis.
Conclusions
We showed that TERT promoter mutation is not a recurrent event in STS and is present in particular histological subtypes.
Introduction
The human telomerase reverse transcriptase (TERT) gene encodes the catalytic subunit of telomerase which, together with an RNA component, TERC, maintains the genomic integrity by telomere elongation (1). Telomere maintenance is an important step in tumorigenesis because it confers unlimited proliferative capacity on cancer cells (2). Telomerase upregulation has been shown to be a ubiquitous feature in human cancer, with over 90% of cancers showing activation of this enzyme (3). A newly discovered genetic alteration consisting of mutations in the promoter region of TERT represents a new mechanism of this reactivation (2).
Hotspot somatic mutations in the promoter region of TERT, located c.-124 and c.-146 bp upstream of the ATG start site (c.-124 C>T and c.-146 C>T), have been reported in several human cancers (2). They were first described in melanomas with a frequency of 22%-85% and subsequently in bladder cancer (~85% of mutated cases), gliomas (~50%) and thyroid tumors (~15%) (4-5-6-7-8). The consequence of these TERT mutations is the creation of new binding motifs for E twenty-six/ternary complex factors (Ets/TCF) transcription factors, which results in an up to 4-fold increased TERT promoter activity in reporter gene assays (4, 5). In bladder and thyroid cancer, melanomas and gliomas, the mutations were associated with risk of tumor recurrence, metastasis (9) and/or poor survival (7, 10). Interestingly, recent studies in the context of bladder cancer and clear cell renal cell carcinoma reported that TERT promoter mutations affect the survival in a manner that is modified by a single nucleotide polymorphism (SNP) (T>C) at position -245 bp (rs2853669) (7, 11).
Soft tissue sarcomas (STSs) are a histologically and genetically heterogeneous group of malignant tumors of mesenchymal origin. They can occur anywhere in the body but are most frequent in the extremities (75%), followed by the trunk (10%) and retroperitoneum (10%) (12). STS occur at any age and are relatively rare, compared with carcinomas and other neoplasms, accounting for only 0.7% of all adult malignancies and approximately 6.5% of childhood cancers (13). Although, in general, STS occur more commonly in older patients and in males, gender and age-related incidences vary among the histologic types (14).
Biologically, events such as gene amplification of the oncogenes MDM2, N-MYC and ERBB2, and mutations of members of the RAS family are implicated in tumor development and have been associated with adverse outcomes in STS patients (15, 16). Mutations in the promoter region of the TERT gene are among the most common somatic genetic lesions in human cancers, but knowledge about their frequency in STS is limited because of the heterogeneity of STSs. Therefore, the present study was conducted to investigate the frequency of TERT promoter hotspot mutations in a series of 68 cases of STS.
Material and methods
Patients
In the present study we analyzed 68 patients with a diagnosis of STS who underwent surgery at the Barretos Cancer Hospital in Brazil. Importantly, all STS were diagnosed based on histological and immunohistochemical analysis by 2 expert pathologists (LFAM and CSN). When necessary, fluorescent in situ hybridization (FISH) was performed to confirm diagnosis by searching for specific genomic rearrangements (e.g. FUS-DDIT3 gene fusion in myxoid liposarcoma). Relevant clinicopathological data available included the patient's age, race and gender, tumor location, histology and grade (according to the French Federation of Cancer Centers Sarcoma Group (FNCLCC) (17), disease progression, presence of metastasis and disease recurrence, as specified in Table I. This study was approved by the local ethics commitee (CEP-331/2010).
Clinicopathological features of STS’ patients

DNA Isolation
Paraffin-embedded tumor samples from 68 patients were retrieved from the Pathology Department of Barretos Cancer Hospital. The tissues were deparaffinized by serial extraction with xylene and ethanol, and selected tumor areas (with neoplastic cellularity higher than 50%) were macrodissected using a sterile needle. DNA was isolated using the QIAamp DNA Micro Kit (Qiagen, Hilden, Germany) according to the manufacturer's instructions and as previously described by our group (18, 19). The quality and concentration of DNA were measured in a NanoDrop 2000 UV-Vis Spectrophotometer (Thermo Fisher Scientific, Wilmington, USA) followed by storage at -20°C until molecular analysis.
Sequence Analysis
A fragment of the TERT promoter was amplified by PCR using the primers 5’-AGTGGATTCGCGGGCACAGA-3’ and 5’-CAGCGCTGCCTGAAACTC-3’, resulting in a PCR product of 235 bp, which contained the sites of the c.-124 C>T and c.-146 C>T mutations and the T>C (A>G) SNP rs2853669 as previously described (8, 20). PCR was performed with initial denaturation at 95°C for 15 minutes, followed by 40 cycles of denaturation at 95°C for 30 seconds, annealing at 64°C for 90 seconds, elongation at 72°C for 30 seconds, and final elongation at 72°C for 7 minutes. The quality of PCR products was confirmed by gel electrophoresis. DNA sequencing of the PCR product was performed using the BigDye Terminator version 3.1 Cycle Sequencing Kit (Applied Biosystems, USA) and an ABI PRISM 3500xL Genetic Analyzer (Applied Biosystems, USA). Electropherogram analysis of the same 235-bp PCR product allowed the genotyping of rs2853669 SNP.
Statistical Analysis
The SPSS 19.0 software (IBM Corp, Armonk, NY, USA) was used for all statistical analyses. To assess the relationship between variables, we used Fisher's exact test. Overall survival was defined as the time between the date of admission to Barretos Cancer Hospital and death or last follow-up and was calculated by the Kaplan-Meier method. The differences between curves were assessed using the log-rank test. The p value established for statistical significance was <0.05.
Results
Sequencing analysis was successful in all 68 STS cases. We found the c.-124 TERTpromoter mutation in 5.9% (4/68) of STSs (Tab. II and Fig. 1), specifically in 1 pleomorphic liposarcoma, 1 dedifferentiated liposarcoma and 2 myxoid liposarcomas (Tab. II). The present series was previously analyzed by immunohistochemistry for several molecular markers that are summarized in Table II (19, 21, 22). No TERT promoter mutation was found in the other histological subtypes. TERT promoter mutation was associated with younger age (≤51 years) (p = 0.004), and no difference in overall survival was found between patients harboring tumors with and without TERT promoter mutation (Tab. III).
Clinicopathological, immunohistochemical and molecular data of the STSs with TERT promoter mutation
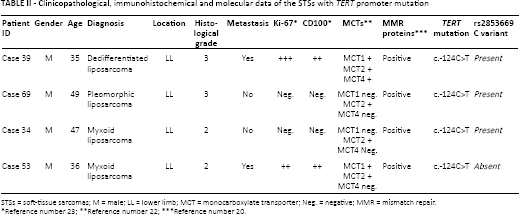
STSs = soft-tissue sarcomas; M = male; LL = lower limb; MCT = monocarboxylate transporter; Neg. = negative; MMR = mismatch repair.
Reference number 23;
Reference number 22;
Reference number 20.
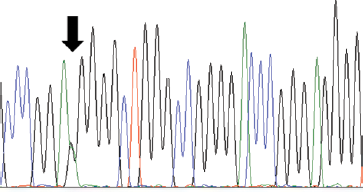
Electropherogram showing a heterozygous c.-124 C>T TERT promoter mutation (
Correlation between clinicopathological patient characteristics and TERT promoter genotype and rs2853669 polymorphism
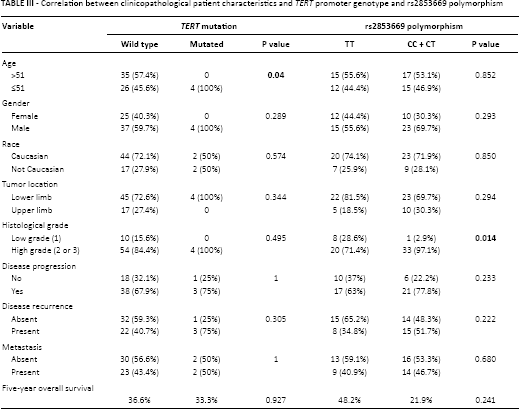
The variant C allele of the rs2853669 polymorphism was found in 54.8% (34/62) of STS cases. The SNP was found in all cases of the following STS subtypes: synovial sarcoma (9/9), malignant peripheral nerve sheath tumor (7/7), clear cell sarcoma (1/1) and solitary fibrous tumor (1/1). In addition, considering only the TERT promoter-mutated cases, 75% (3/4) showed the rs2853669 polymorphism (Tab. II). We observed a statistical correlation between the variant T>C allele of rs2853669 and high histological grade (2 and 3) (p = 0.014) (Tab. III).
Discussion
This study describes the occurrence of the c.-124 TERT promoter mutation in ~6% of Brazilian STSs. Despite the distinct background, all mutated cases were liposarcomas: 1 pleomorphic, 1 dedifferentiated and 2 myxoid. The absence of germline tissue of the mutated cases hampers the determination of its somatic or germline nature; however, the absence of these mutations in the 1000 genome project and the many publications that performed such paired (tumor versus normal) analysis (4, 8, 23-24-25) strongly suggest its somatic nature.
The prevalence of TERT promoter mutations in STSs is reported to vary extremely by sarcoma subtype (Tab. IV). Previous studies identified high frequencies of TERT promoter mutations in a specific liposarcoma subtype, the myxoid type (6, 26). In myxoid liposarcomas, studies have shown 74% to 79% of cases with TERT mutations (6, 26). In our series, only 22.2% of myxoid liposarcomas exhibited TERT promoter mutations. This discrepancy could be due to the low number (n = 9) of myxoid liposarcomas analyzed in our series. We also found mutations in other liposarcoma subtypes, namely pleomorphic and dedifferentiated liposarcomas, which were previously reported to be TERT wild-type by Koelsche et al (26) (Tab. IV).
Summary of described TERT promoter mutation in soft tissue sarcomas
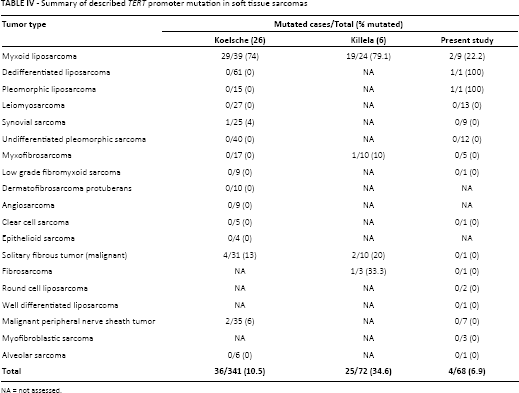
NA = not assessed.
In some malignancies such as thyroid tumors and gliomas, TERT promoter mutations are associated with several clinicopathological features, like older age and worse outcome (8, 27). In our study, we found a correlation between the presence of a mutation and younger age. Koelsche et al (26) showed in myxoid liposarcomas a high prevalence of TERT promoter mutations (29/39; 74%) and no association with the phenotype (myxoid vs. round cell), tumor grade, tumor site and patients’ median age and gender. In a study by Killela et al (6), patients were relatively young, with a peak age range between 30 and 50 years.
While somatic mutations in the TERT promoter region create Ets/TCF binding sites, it has been described that the variant T>C allele of the rs2853669 polymorphism disrupts a preexisting noncanonical Ets2 binding site adjacent to an E-box in the proximal region of the TERT promoter (7, 10). In bladder cancer, 47.1% of tumors showed the SNP (7) and in clear cell renal cell carcinoma the prevalence was 75.7% (10). In both types of tumors the presence of the rs2853669 polymorphism increased the survival of patients with the TERT promoter mutation (7, 10). In the present study, we also found that the C allele was present in approximately half of the cases. The presence of TERT mutation in only 4 cases limited the statistical impact of rs2853669 in this subset of patients. Neverthless, we observed a statistical association between the variant T>C allele of rs2853669 and high histological grades.
In human cancers, maintenance and/or increase of telomere length occur by reactivation of telomerase activity or by alternative lengthening of telomeres (ALT) (28). In the ALT mechanism, the telomerase dynamics are consistent with the recombination-based mechanism (29) that is more frequently present in tumors of mesenchymal origin, including osteosarcomas (30) and STSs (31), than in those of epithelial origin. Additionally, it was reported that ALT not only supports the tumorigenesis of liposarcomas but was also associated with an unfavorable outcome (28). The importance of the ALT mechanism in STS is one possible reason for the low frequency of TERT promoter mutations observed in our and other studies.
Therapeutically, several strategies of telomerase inhibition including small molecular inhibitors, gene therapy and immunotherapy have entered clinical trials. It will be important to find out whether TERT promoter mutations and the rs2853669 polymorphism can predict patients’ response to therapy (32).
In conclusion, we found a low incidence of the c.-124 TERT promoter mutation in STS, which, moreover, was restricted to the liposarcoma subtype. In addition, we found a high incidence of the rs2853669 C variant in STS, and its association with histologically high-grade lesions. Further studies involving larger patient series are warranted to clarify the prognostic relevance of TERT promoter mutations and of the rs2853669 C variant in these tumors.
Footnotes
Financial support: This project was partially supported by Barretos Cancer Hospital Internal Research Funds (PAIP) and CNPq Universal Grant (476192/2013-7) to Rui Manuel Reis. Nathália Cristina Campanella is the recipient of a FAPESP doctoral fellowship (2013/25787-3).
Conflict of interest: The authors declare they do not have any competing interests.
