Abstract
Background
The aim of this study was to evaluate the landscape of gene mutations and the clinical significance of tumor mutation burden (TMB) in patients with soft tissue sarcoma who underwent surgical resection and received conventional adjuvant therapy.
Methods
A total of 68 patients with soft tissue sarcoma were included. Postoperative tumor tissue specimens from the patients were collected for DNA extraction. Targeted next-generation sequencing of cancer-relevant genes was performed for the detection of gene mutations and the analysis of TMB. Univariate analysis between TMB status and prognosis was carried out using the Kaplan–Meier survival analysis, and multivariate analysis was adjusted by the Cox regression model.
Results
No specific genetic mutations associated with soft tissue sarcoma were found. The mutation frequency of TP53, PIK3C2G, NCOR1, and KRAS of the 68 patients with soft tissue sarcoma were observed in 19 cases (27.94%), 15 cases (22.06%), 14 cases (20.59%), and 14 cases (20.59%), respectively. With regard to the analysis of TMB, the overall TMB of the 68 patients with soft tissue sarcoma was relatively low (median: 2.05 per Mb (range: 0∼15.5 per Mb)). Subsequently, TMB status was divided into TMB-Low and TMB-Middle according to the median TMB. Patients with TMB-Low and TMB-Middle were 37 cases (54.41%) and 31 cases (45.59%), respectively. Overall survival analysis indicated that the median overall survival of patients with TMB-Low and TMB-Middle was not reached, and 4.5 years, respectively (P=0.015).
Conclusion
This study characterizes the genetic background of patients with STS soft tissue sarcoma. The TMB was of clinical significance for patients with soft tissue sarcoma who underwent surgical resection and received conventional adjuvant therapy.
Introduction
Soft tissue sarcoma (STS) is a relatively rare malignant tumor that originates from mesenchymal tissue with a heterogeneous category comprised of more than 50 histologic subtypes. 1 , 2 It accounts for approximately 24,000 new cases annually in China and the incidence is higher in adolescents. 3 , 4 The most common histologic subtypes of STS are undifferentiated sarcoma (US), liposarcoma (LPS), synovial sarcoma (SS) and leiomyosarcoma (LMS), which are associated with different molecular characteristics and behavior features. 5 , 6 According to the National Comprehensive Cancer Network guidelines of STS, surgical resection is the curable treatment option. However, other treatment regimens for STS are extremely scarce and the prognosis remains dismal. 7 Diagnostic and therapeutic factors should be investigated to improve the survival of patients with STS. 8
Different from KIT and PDGFRA genes used as the diagnostic and therapy guidance for patients with gastrointestinal stromal tumors, 9 genetic testing of STS emerges just as an ancillary technique. 10 However, in view of the heterogeneity of tumor genomics and diversity of tumor categories, patients with STS have been classified broadly into two groups according to genomic alterations: simple karyotype with balanced translocation, and complex karyotype without aberrant dislocation. 5 , 11 A previous study suggested that sarcomas with the simple karyotype were predictive for clinical outcomes, although targeted drugs against the fusion gene were not successful. 12 Sarcomas with the complex karyotype accounted for the larger category of STS, which were heterogeneous, unstable, and profoundly altered in copy number. 5 , 13 The landscape of gene mutations in STS was performed in several research studies previously, 4 , 5 , 14 and the results indicated that just a few genes (TP53, ATRX, and RB1) were recurrently mutated across sarcoma types. The mutation frequency of TP53 gene ranged from 28%–61%. 13 To our knowledge, there have been no studies examining the landscape of gene mutations of Chinese patients with STS so far.
In recent years, great progress has been achieved in the development of immune checkpoint inhibitors, which may bring new strategic directions for anti-tumor therapy. 15 , 16 To the best of our knowledge, the objective response rate of immune checkpoint inhibitors is approximately 20%. 17 Consequently, biomarkers for immunotherapy efficacy prediction are a hot spot in research. 18 PD-L1 expression and tumor mutation burden (TMB) are potential biomarkers for the efficacy of PD-1/PD-L1 checkpoint inhibitors. 19 PD-L1 expression has been proven as a biomarker in patients with non-small cell lung cancer in several studies. 20 , 21 However, the prediction value of TMB was controversial. The standards for TMB testing were scant and it was unclear what the detection of mutations was by whole exome sequencing (WES), as well as the appropriate TMB threshold. 19 In consequence, the assessment of TMB—which was defined as the total number of nonsynonymous mutations in the coding regions—was needed urgently. Additionally, the influence of TMB status on the prognosis of patients with STS who received conventional adjuvant treatment instead of immunotherapy was unknown, and it was unclear whether TMB status could be used as a biomarker for patients with STS receiving conventional adjuvant therapy. 22
The aim of this study was to investigate the landscape of gene mutations and the clinical significance of TMB in patients with STS who underwent surgical resection and received conventional adjuvant therapy.
Material and methods
Study design
Patients with STS from October 2012 to December 2018 in the Department of Orthopedic Surgery of Chinese Academy of Medical Sciences Cancer Hospital were retrospectively screened and enrolled in this study. The eligibility criteria included: (a) histological diagnosis of STS confirmed by pathological expert in accordance with the WHO Classification of Tumors of Soft Tissue and Bone; (b) age ≥18 years; (c) an eastern cooperative oncology group (ECOG) performance status of 0∼2; (d) pathological staging of II and III according to the 7th edition of the American Joint Committee on Cancer guideline of TNM classification; and (e) underwent surgical resection and received conventional adjuvant therapy (including radiotherapy, chemotherapy, and chemoradiotherapy). The exclusion criteria were: (a) diagnosed with gastrointestinal stromal tumors, or rhabdomyosarcoma, or chondrosarcoma, or osteosarcoma, or Ewing sarcoma, or primitive neuroectodermal tumor; (b) absence of appropriate specimens for DNA extraction and next generation sequencing (NGS) analysis; (c) presence of concomitant tumors or serious disease; (d) diagnosed with metastatic disease; and (e) follow-up data were not available. The flow chart of our study is illustrated in Figure 1. A total of 68 patients with STS met the study criteria and were included in this retrospective analysis. The primary analysis of this study was the landscape of gene mutations and the clinical significance of TMB. The landscape of gene mutations was defined as somatic gene mutations according to the sequencing data, and for which details were available in the analysis of the gene mutation landscape. TMB was defined as the number of somatic, coding, base substitutions, and indels per megabase of genome examined, and the clinical significance of TMB was defined as the survival difference between patients with TMB-Low and TMB-Middle.
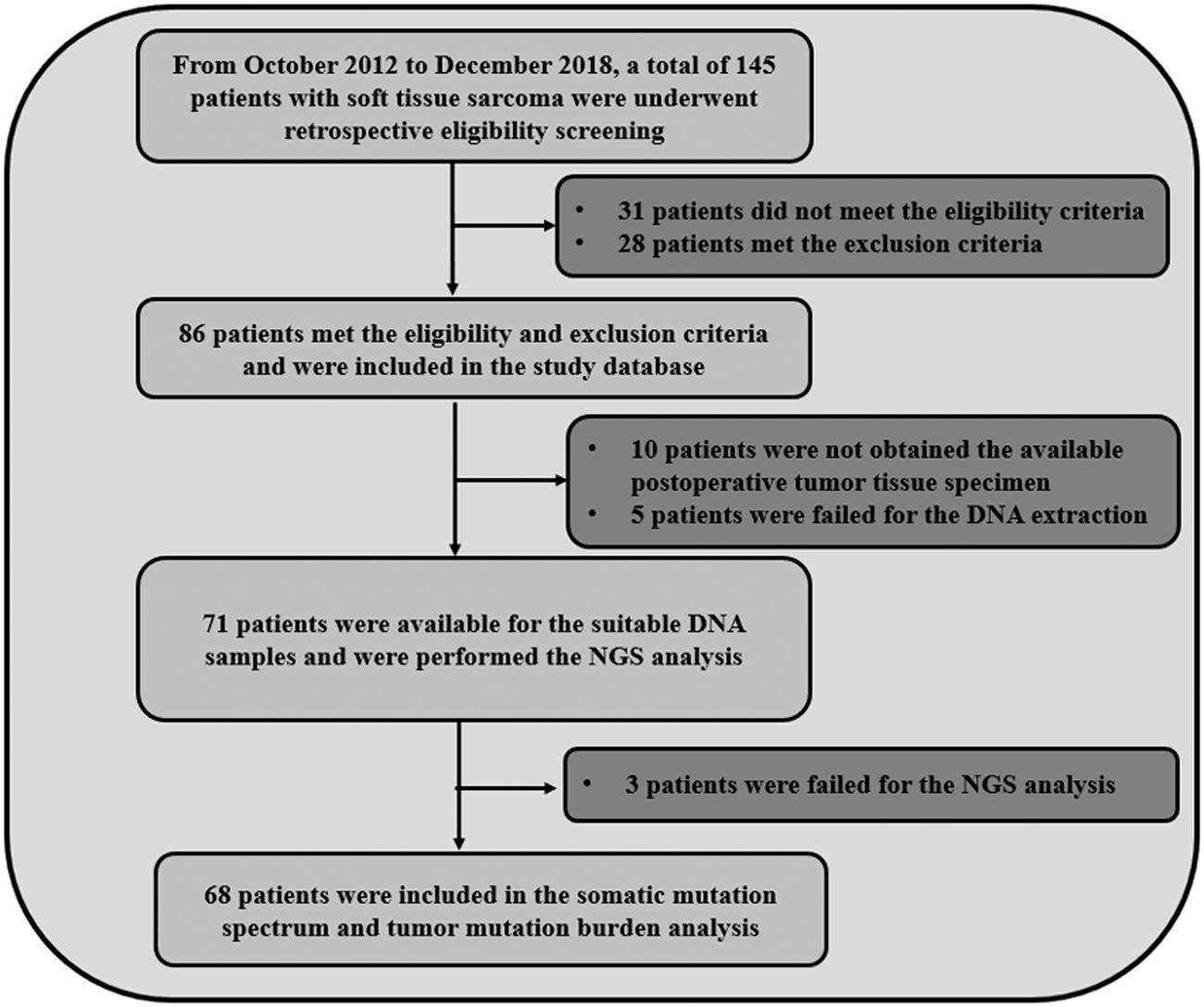
Flow chart of the retrospective study of the landscape of gene mutations and clinical significance of tumor mutation burden in 68 patients with soft tissue sarcoma.
This study was approved by the Ethics Committee of the Chinese Academy of Medical Sciences Cancer hospital. Written informed consent was obtained from all patients or their family members in accordance with the recommendations of the Declaration of Helsinki.
Collection of postoperative tumor tissue specimens and targeted DNA sequencing
Postoperative tumor tissue specimens were collected after patients underwent surgical treatment. DNA from specimens was extracted using the DNA FFPE tissue kit (QIAGEN, Valencia, CA). DNA concentration was measured and the gDNA quality was assessed to make sure that A260/A280 was within the range of 1.8–2.0.
At the time of biopsy, 10 mL of peripheral blood was obtained and stored in tubes containing ethylenediaminetetraacetic acid and incubated at room temperature for 2 hours. The supernatant was transferred to 15 mL centrifuge tubes and centrifuged for 10 minutes at 16,000 g and 4°C. The supernatant was transferred to a new tube and stored at −80°C for further analysis. Circulating cfDNA was recovered from 4 to 5 mL of plasma by using the Circulating Nucleic Acid kit (QIAGEN, Valencia, CA). Quantification of cfDNA was assessed using the Qubit 2.0 fluorimeter (Thermo Fisher Scientific). A minimum of 50 ng of cfDNA is required for construction of anNGS library.
Targeted DNA sequencing was performed for patients with available DNA. DNA was profiled by using a capture-based targeted sequencing panel (Burning Rock Biotech, Guangzhou, China), targeting 520 genes and spanning 1.67M of human genomic regions. The concentration of the DNA samples was measured with the Qubit dsDNA assay to make sure that genomic DNA was greater than 40 ng. Fragments of 200–400-bp sizes were selected followed by hybridization with the capture probe baits, hybrid selection with magnetic beads and PCR amplification. A bioanalyzer high-sensitivity DNA assay was then used to assess the quality and size range. Available indexed samples were then sequenced on a Miseq (Illumina, San Diego, CA) with pair-end reads.
Analysis of the landscape of gene mutations
The landscape of gene mutations was based on the sequencing data, which were mapped to the human genome (hg19) using BWA aligner 0.7.10. PCR duplicate reads were removed before base substitution detection. Local alignment optimization and variant calling was performed using GATK v3.2-2. DNA translocation analysis was performed. Insert size distribution and library complexity of each sample were computed to assess the level of DNA degradation. Different mutation calling thresholds were applied on samples with different DNA quality to avoid false positive mutation calls due to DNA damage. SNV and indels identification were annotated using the dbSNP database.
Integrative Genomics Viewer (Broad Institute, USA) was used to visualize variants aligned against the reference genome to confirm the accuracy of the variant calls by checking for possible strand biases and sequencing errors. Gene-level copy number variation (CNV) was assessed using a statistic after normalizing read depth region by total read number and region size and correcting GC-bias using a LOESS algorithm.
Analysis of tumor mutation burden
TMB was defined as the number of somatic coding base substitution and indels per megabase of genome. Fusions, CNVs and non-coding mutations were not counted. Synonymous mutations were counted in order to reduce sampling noise. DNA extracted from white blood cells was used to filter germline mutation. TMB status was divided into TMB-Middle (TMB-M) and TMB-Low (TMB-L) according to the median TMB value.
Statistical analysis
All of the variables in this study were statistically analyzed using the statistical analysis software SPSS version 25.0 (IBM, USA). The significance of proportion variables and continuous variables according to TMB status of the patients was performed using the chi-square test and the Mann-Whitney U non-parametric test, respectively. The overall survival (OS) was calculated from the time patients received surgical resection to patient’s death from any cause. For those without death event by the end of the study follow-up, survival end points were censored at the date of the last follow-up. The Kaplan–Meier curve was drawn using Stata 14.0 to compare OS according to TMB status and the survival difference was compared using log-rank test. For the multivariable analysis, a Cox proportional hazards model was constructed for OS. Statistical significance was accepted when P<0.05.
Results
Baseline characteristics of the 68 patients with STS according to the tumor mutation burden status
The baseline characteristics of the 68 patients with STS are shown in Table 1. The median age of the patients was 45 years (range: 18∼65 years), and 42 patients were male (61.76%). ECOG 0 score was observed in 45 patients (66.18%). The histologic subtypes included SS 18 cases (26.47%), LMS 14 cases (20.59%), US 11 cases, LPS 9 cases (13.24%), and other sarcoma types 16 cases (23.53%). The major primary tumor sites were extremities with 38 patients (55.88%) and abdominal cavity with 15 patients (22.06%). Pathological stages II and III were reported in 22 (32.35%) and 46 cases (67.65%), respectively. Patients who received adjuvant radiotherapy and chemotherapy and chemoradiotherapy were noted in 31 cases (45.59%), 19 cases (27.94%), and 18 cases (26.47%), respectively. The median TMB status of the patients was 2.05 per Mb (range: 0∼15.5 per Mb).
Baseline characteristics of the 68 patients with soft tissue sarcoma according to TMB status.
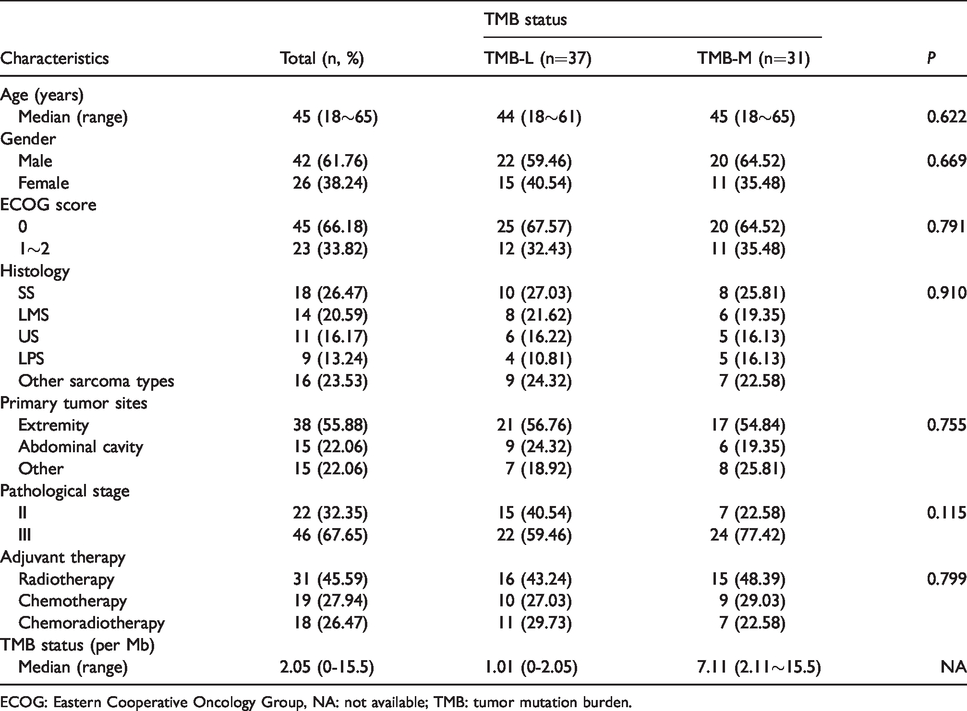
ECOG: Eastern Cooperative Oncology Group, NA: not available; TMB: tumor mutation burden.
TMB status was divided into two groups according to the median TMB value. The median TMB status of the 68 patients with STS was 2.05 per Mb. Those with TMB ranging from 0 to 2.05 per Mb were divided into TMB-Low (TMB-L) and those with TMB ranging from 2.06 to 15.5 per Mb were divided into TMB-Middle (TMB-M). The patients of TMB-L and TMB-M were confirmed in 37 cases and 31 cases, respectively. As shown in Table 1, patients with TMB-L and TMB-M were well balanced with similar baseline characteristics. Nevertheless, it should be noted that in terms of pathological stage, TMB-M patients had more stage III compared with the TMB-L patients (77.42% versus 59.46%), although the difference was not statistically significant (P=0.115).
Landscape of gene mutations of the 68 patients with STS
We further evaluated the landscape of gene mutations of the 68 patients with STS. The landscape of gene mutations is illustrated in Figure 2 according to the results of the NGS analysis. No specific genetic mutations associated with STS were found. TP53, PIK3C2G, NCOR1, and KRAS were mutated with relatively high prevalence ≥20%. The mutation frequency of TP53, PIK3C2G, NCOR1 and KRAS of the 68 patients with STS were noted in 19 cases (27.94%), 15 cases (22.06%), 14 cases (20.59%), and 14 cases (20.59%), respectively. Only those with a mutation frequency of more than 2 cases (2.94%) are shown in Figure 2, although a considerable number of genes were detected in less than 2 cases.
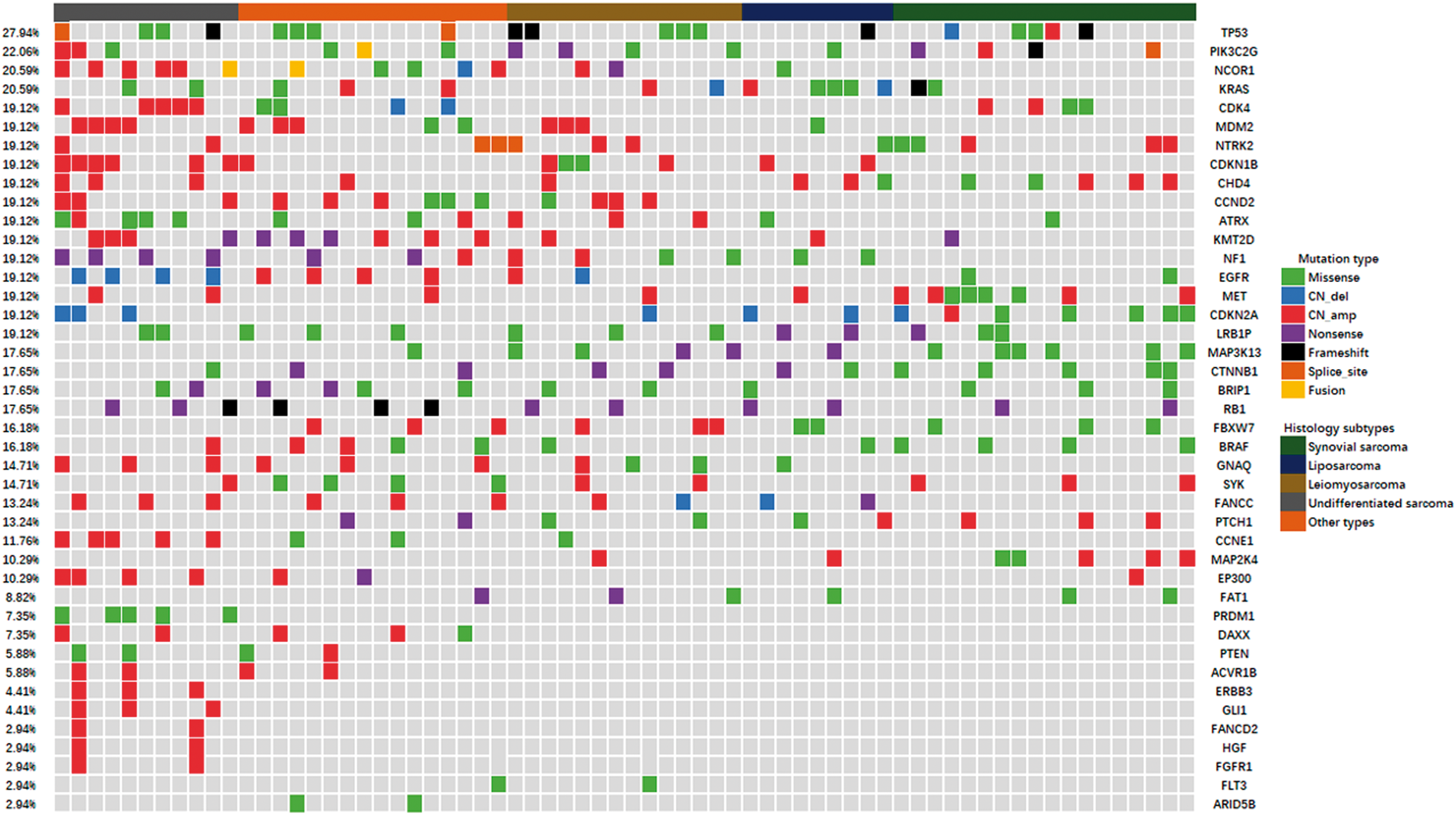
The landscape of gene mutations in 68 patients with soft tissue sarcoma.
Clinical significance of the 68 patients with STS according to TMB status
The last follow-up time was March 2019. The median follow-up of all patients from the time of enrolment to the last follow-up was 3.8 years (follow-up range: 0.5–5.9 years).
Patients with TMB-L and TMB-M were 37 cases and 31 cases, respectively. Given that the TMB was not a kind of therapy regimen, we used OS as endpoints in the prognosis analysis. As illustrated in Figure 3, the OS of patients with TMB-Low did not reach the median, and the median OS of patients with TMB-Middle was 4.5 years, which was statistically significant (P=0.015). Furthermore, in order to adjust the other confounding factors, a Cox regression model was constructed. The univariate analysis was introduced in the Cox regression model with age, gender, ECOG score, tumor location (extremity or other location), pathological stage, adjuvant therapy (radiotherapy versus chemotherapy or chemoradiotherapy), and TMB status. As shown in Table 2, age (hazard ratio (HR)=1.51, P=0.022), ECOG score (HR=1.92, P=0.009), pathological stage (HR=2.45, P=0.001), and TMB status (HR=1.68, P=0.015) were statistically significant in univariate analysis, and were introduced in the multivariate analysis. The results of multivariate analysis are shown in Table 2. A statistically significant difference was observed for the influence of TMB status on OS after adjustment for other confounding factors. This finding indicated that TMB status was an independent factor for OS (HR=1.63, P=0.021). Additionally, age (>45 versus ≤45, HR=1.48, P=0.030), ECOG score (1∼2 versus 0, HR=1.88, P=0.011) and pathological stage (III versus II, HR=2.38, P=0.004) were independent factors for OS, respectively.
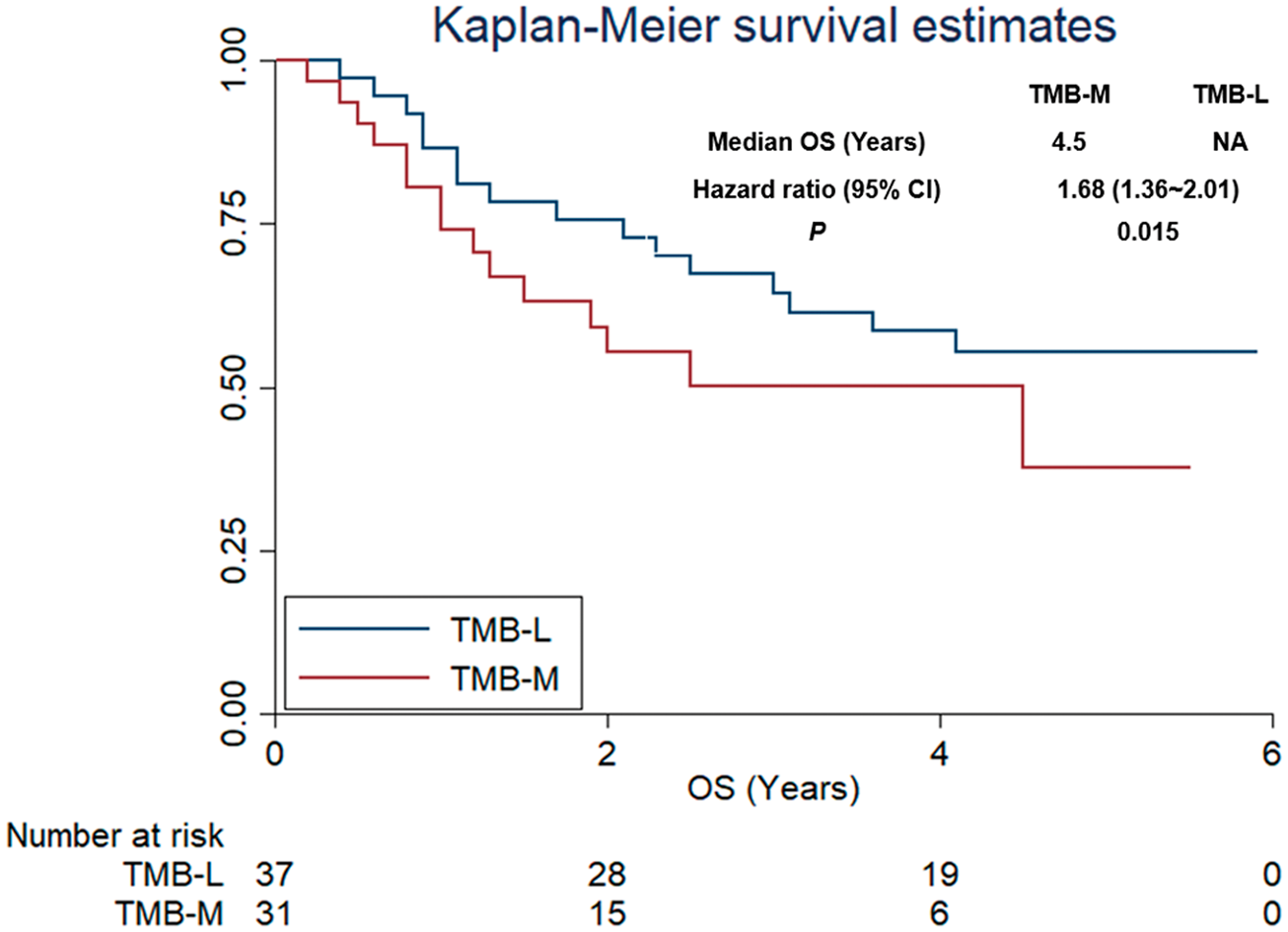
Overall survival of 68 patients with soft tissue sarcoma according to tumor mutation burden status.
Univariate and multivariate Cox regression analysis for OS according to baseline characteristics and TMB status.
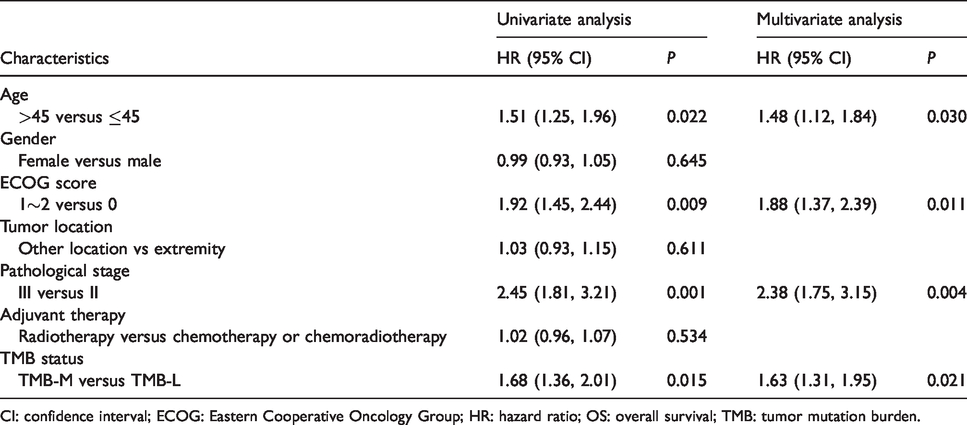
CI: confidence interval; ECOG: Eastern Cooperative Oncology Group; HR: hazard ratio; OS: overall survival; TMB: tumor mutation burden.
Discussion
This retrospective study provides evidence for the landscape of gene mutations and clinical significance of TMB in patients with STS. The landscape of gene mutations and TMB analysis was based on targeted NGS technology, which has proved to be an accurate and clinically available approach for detecting targeted gene mutations with a greater depth and less time compared with whole exome sequencing. 23 The 68 patients with STS consisted of 18 synovial sarcoma, 14 leiomyosarcoma, 11 undifferentiated sarcoma, 9 liposarcoma, and 16 other subtypes, which were in line with previous studies. 14 , 24 Nevertheless, it should be noticed that the proportion of SS in Chinese patients with STS seemed to be higher than that in Caucasian patients, 24 which may be attributed to racial differences. With regard to the landscape of gene mutations, we found that no specific genetic mutation was associated with STS, which was consistent with previous research. 5 , 14 , 24 These results indicate the wide genetic heterogeneity of STS to some extent. 25 Furthermore, the landscape of gene mutations was investigated and presented in our study. To our knowledge, our study was the largest somatic landscape of gene mutations study involving the largest number of genes in patients with STS. The previous work initiated by Zheng and colleagues 4 in China explored the pathogenic and targetable genetic alterations in resected recurrent undifferentiated pleomorphic sarcomas. However, the sample sizes were small and their conclusions were limited. TP53 was the most common somatic mutation in our study with a mutation frequency of 27.94%, which was similar to the results of the study initiated by Lee and colleagues. 26 They investigated the spectrum of mutations in leiomyosarcomas identified by clinical targeted next-generation sequencing and the results showed the frequency of TP53 was 36%. TP53 is a tumor suppressor gene that regulates cell division by keeping cells from growing and proliferating. Missense mutation was the major mutation type of TP53 in our study. 27 In addition, PIK3C2G, NCOR1, and KRAS were the other common somatic mutation genes in our study with a mutation frequency ≥20%. PIK3C2G belongs to the phosphoinositide 3-kinase (PI3K) family and has been reported to act as a second messenger involved in regulation of proliferation, transformation, differentiation, and intracellular trafficking. 28 A previous study suggested that the PIK3C2G copy number was associated with clinical outcomes of CRC patients treated with oxaliplatin. 29 NCOR1 encodes a protein that mediates ligand-independent transcription repression of thyroid-hormone and retinoic-acid receptors. 30 NCOR1 was confirmed as a tumor suppressor gene in hormone receptor negative breast and lung adenocarcinomas. 31 The exact role of both PIK3C2G and NCOR1 in patients with STS is unknown. Interestingly, the KRAS gene plays a role in a considerable number of tumors, 32 and the mutation frequency of KRAS was more than 20% in our study. The KRAS gene encodes a signaling protein that moderates the response to various extracellular signals via down-regulation of the mitogen-activated protein kinase (MAPK) or phosphoinositide-3-kinase (PI3K) pathway. 33
The prognosis analysis suggested that the median OS of patients with TMB-L was longer than that of patients with TMB-M. The reason why we divided TMB status into two groups according to the median TMB value was because there is currently no uniform threshold for TMB evaluation. 34 , 35 Interestingly, the TMB status in our study ranged from 0 to 15.5 per Mb, which was consistent with a previous study. 14 The overall TMB in 206 patients with sarcomas was low (average: 1.06 per Mb, range: 0.2–33.5 per Mb). It seemed that the TMB in rhabdoid tumor and Ewing sarcoma was significantly lower than that in lung adenocarcinoma and melanoma. 36 To our knowledge, it is unknown whether higher TMB predicts better efficacy of immunotherapy. 37 More importantly, higher TMB status has recently emerged as an additional powerful biomarker to select patients for PD-1/PD-L1 therapy. 38 However, the prognosis of patients who received conventional adjuvant therapy rather than immunotherapy according to TMB status is still controversial. 39 , 40 Previous research suggests that higher TMB is associated with worse OS in non-small cell lung cancer, melanoma, multiple myeloma, hepatocellular carcinoma, and breast cancer. However, higher TMB correlated with superior prognosis in patients with ovarian cancer when they received surgery and chemotherapy treatment.41–43 The clinical significance of TMB status in patients with STS who received conventional adjuvant therapy was unknown. 44 Our study is the first to evaluate the influence of TMB status on the prognosis of Chinese patients with STS who underwent surgical resection and received conventional adjuvant therapy. The results indicate that higher TMB may predict a worse prognosis for patients with STS receiving conventional adjuvant therapy. In view of the outstanding advantage of immunotherapy clinically, our study preliminarily suggests that immunotherapy instead of conventional therapy is a better option for STS with higher TMB status. However, this hypothesis needs to be validated in clinical trials.
From an objective view, a number of limitations exist in our study. First, the sample size was small. Second, our study was designed as a retrospective analysis, so some bias may be unavoidable. However, our study is the first to evaluate the landscape of gene mutations and the clinical significance of TMB status in Chinese patients with STS, and we show clinical utility for prognostic evaluation in patients with STS who underwent surgical resection and received conventional adjuvant therapy.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by CSCO-Junshi Tumor Immunology Research Fund (Y-JS2019-017).
Informed consent
Informed consent was obtained from all participants.
