Abstract
Soft tissue sarcomas remain one of the rarest malignancies with numerous subtypes that go undiagnosed. The PDGFRα antagonist Olaratumab (Lartruvo) was withdrawn from the market due to disappointing findings in the phase III studies. We share our experience with this medication in a tertiary care center in the Middle East and North Africa region. Monitor the effect of Olaratumab on sarcomas when it was used prior to its withdrawal, and compare our findings with the literature. We performed a retrospective analysis of adult patients with advanced-/metastatic soft tissue sarcomas treated with at least two cycles of Olaratumab at a tertiary care center in Lebanon during the period from January 1, 2017 to December 31, 2018. Fifteen patients were included in the study. The mean age was 49 with a range of 26–75 years. The median duration of the use of Olaratumab was 21.3 months with a range of 7.3–37 months. The average number of number of cycles received per patient was four. Five patients were deceased. Median PFS was 7.87 months (95% CI 5.28–10.45), and mean OS was 12.26 months (95% CI 8.47–16.05) Median OS was 9.8 months (95% CI 6.07–13.53). Olaratumab has been withdrawn from the market, and it is currently being investigated as part of the phase II ANNOUNCE 2 trial. Our experience from a tertiary care center shows results similar to those reported in the literature. The immunogenicity and heterogeneity of soft tissue sarcomas pose a challenge to the treatment of soft tissue sarcomas, but they also allow a wide array of possible management solutions.
Introduction
Soft tissue sarcomas (STS) are rare tumors that originate from mesenchymal cells, accounting for approximately 1% of all adult cancers. The standard management for most histological subtypes of localized STS consists of complete surgical resection with or without radiation, and/or chemotherapy based on relapse risk. Therapy for advanced STS is primarily based on systemic chemotherapy depending on the histological type, treatment intent, and the patient’s performance status. Some of the recommended therapies include Gemcitabine/Docetaxel, Trabectedin, Pazopanib, and Eribulin.1–6 However, the prognosis for such patients remains poor, with median survival limited to 12–16 months.
Olaratumab, a recombinant human immunoglobulin G1 monoclonal antibody targeting the extracellular domain of the platelet-derived growth factor receptor alfa (PDGFRα). Moreover, it causes certain alterations in the tumor microenvironment, which potentiate the effect of cytotoxic agents such as Doxorubicin. 7 Several Phase I trials have studied the safety and pharmacokinetics of different doses of Olaratumab and observed no dose-limiting toxicity,8,9 including the phase Ib trial by Tap et al., 10 where patients received 75 mg/kg Doxorubicin on day 1 and 15 mg/kg Olaratumab on days 1 and 8 of 21-day cycles.
In the randomized phase II trial involving 133 patients with metastatic STS, patients who were treated with Olaratumab and Doxorubicin combination had a longer progression free survival (PFS) compared to patients treated with Doxorubicin alone by a median of 2.5 months. Although this increase was statistically insignificant, the overall survival (OS), a secondary outcome in this study, was significantly higher in the Olaratumab and Doxorubicin arm compared to the Doxorubicin alone arm, with a median increase of 11.8 months. 9 Given the favorable outcomes from this phase II trial, Olaratumab received accelerated approval from the Food and Drug Administration (FDA) as well as conditional marketing authorization from the European Medicines Agency (EMA) for the treatment of advanced stage STS.11,12
However, in a following phase III randomized double-blinded clinical trial (known as the ANNOUNCE trial) involving 509 patients, it was observed that there was no statistically significant difference in the OS between the Olaratumab and Doxorubicin combination (experimental) group compared to the Doxorubicin and placebo (control) group. Moreover, the PFS, a secondary outcome in this trial, was slightly lower in the experimental group compared to the control group. 13 Given the absence of survival benefit, Olaratumab started to be withdrawn from the market and the recommendations were against its use in advanced STS patients.14,15
In this study, we share our center’s experience with olaratumab, prior to its withdrawal and we compare our findings with those reported in clinical trials as well as real-world data from other centers.
Methods
We conducted a retrospective study for all adult patients (⩾18 years old) with an advanced malignancy who received Olaratumab as a single agent or as combination therapy as Olaratumab ± other chemotherapy/radiation therapy/targeted therapy at our institution (AUBMC) during the period from January 1, 2017 to December 31, 2018. A total of 15 patients were included in the study. Data regarding baseline characteristics, type of malignancy, adverse side effects encountered, management of these AEs, type of treatment received, and disease status were collected and analyzed. A Kaplan–Meier curve for survival was done to calculate the median survival time for all patients; the follow-up period was extended from the date of diagnosis till the date of death or the date of the last encounter up to 2.5 years. All Statistical analysis was done using SPSS v.23. The study was conducted according to the ethical principles stated in the Declaration of Helsinki (2013). IRB (Institutional Review Board) at the American University of Beirut Medical Center reviewed the study proposal (BIO-2019-0058), and IRB approval was granted in March 2019 prior to data collection. Informed consents were taken, and data collected was kept confidential and no patient identifiers were used throughout the study.
Results
The median duration of follow-up was 21 months (95% CI 12.79–29.83) with a range of 7–68 months. The mean age was 49 with a range of 26–75 years. (Table 1). Most patients were of ECOG status 0–1. Only four patients had and ECOG of two. Figure 1 shows the histological subtypes of STS tumors in our study. The two most common histological subtypes were leiomyosarcoma and liposarcoma. The most common primary tumor site was the retroperitoneum (n = 4), followed by the lung (n = 2) and then one of each: liver, kidney, uterus, abdomen right hamstring, right calf, right paravertebral, and esophagus. Depicted in Figure 2, the inner circle represents the primary site of the tumor and the outer circle represents the metastatic site from the primary site. For example, four patients had their primary tumor site in the retroperitoneum. Three out of these four patients had metastasis to the lung while one had metastasis to the bone. Metastatic sites were mainly lungs (n = 11, 73.33%). Two patients (14.3%) had bone metastases, one patient had metastasis to the retroperitoneum, and one patient had metastasis to both the lungs and lymph nodes. In 10 patients (66.67%), Olaratumab was given in the first line setting, and three patients received it as a monotherapy. Among the patients who received it as a second therapy, one was started on 800 mg/day of Pazopanib, and she was later on switched to Olaratumab after her disease progressed. Another patient had a uterine sarcoma removed by hysterectomy in 2016, she was started on Gemcitabine and Taxotere for a leiomyosarcoma in the abdomen, and finally she was switched to Olaratumab after her disease progressed. This patient was one of the five patients who were deceased on the last follow-up date (31.12.2018).
Patients’ characteristics and workup.


Histologic subtype of soft tissue tumors.
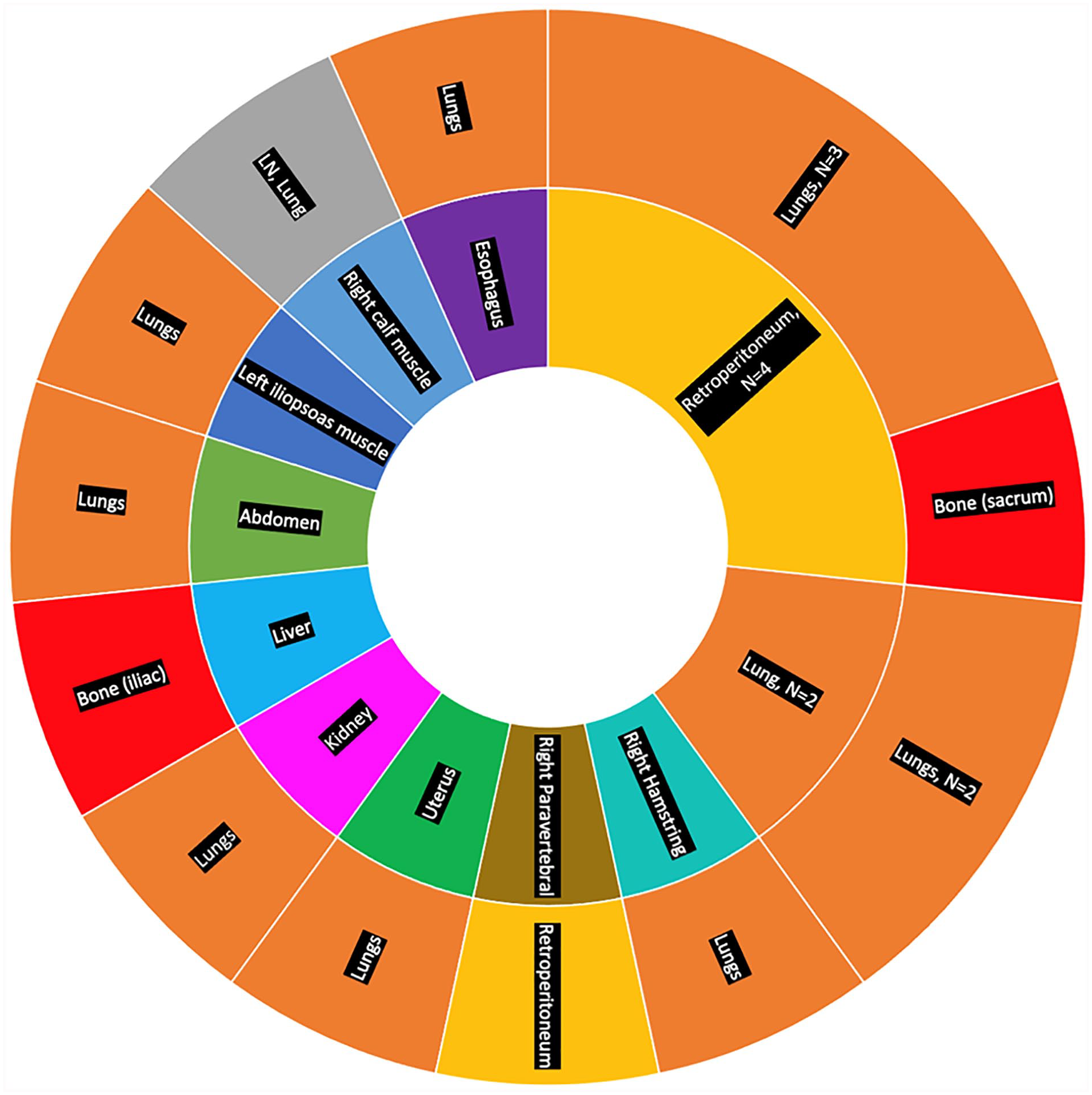
Primary tumor site (inner ring) along with metastasis site (outer ring), LN: lymph nodes.
Median PFS was 7.87 months (95% CI 5.28–10.45), and mean OS was 12.26 months (95% CI 8.47–16.05) (Figures 3 and 4). Median OS was 9.8 months (95% CI 6.07–13.53). Mean PFS was 9.36 (95% CI 5.47–13.24). The median duration of the use of Olaratumab was 21.3 months with a range of 7.3–37 months. The average number of number of cycles received per patient was four. Five patients received five cycles, five patients also received three cycles, two received four cycles, one patient received two cycles, and only one patient received. Five patients were deceased. One passed away due to septic shock and the other four due to the progression of their disease. Seven patients took other regimens after Olaratumab administration or along with Olaratumab (Table 2). Four patients reported side effects after Olaratumab use, with the main toxicities being poor tolerance with a general feeling of malaise and fatigue, followed by decreased appetite. No severe hematologic and oncologic side effects were noted.

Kaplan–Meier curve of overall survival.

Kaplan–Meier curve for progression free survival.
Patients’ regimens along with PFS.

Discussion
In this study, we report our center’s experience with Olaratumab prior to its withdrawal. The median PFS of 7.87 months (95% CI 5.28–10.45) as well as the median OS 9.8 months (95% CI 6.07–13.53) seen in our study were both similar to those reported in real-world studies from other centers around the world. In a retrospective analysis of 32 patients treated with Olaratumab and Doxorubicin in Germany, only 35% of the patients experienced some response, whereas 66% of the patients experienced disease progression. The median PFS in this group was 3.1 months and median OS was 4.6 months. 16 The low response rate was also observed in a retrospective analysis of 55 patients from different centers in Austria who received the Olaratumab and Doxorubicin combination. In this cohort, only 11.4% of the patients experienced some response, with a reported median PFS of 2.6 months of and a median OS of 11.4 months. 17 In the most recently published real-word experience from multiple centers in England and Northern Ireland, the retrospective analysis of outcomes from 172 patients treated with Olaratumab and Doxorubicin revealed that only 21% of the patients had complete or partial response, whereas 33.7% had disease progression. The median PFS in this retrospective cohort was 6.8 months. 18
Combination of Olaratumab with chemotherapeutic agents other than doxorubicin have also been studied; however, they did not show any improvement in patient outcome either. For example, combining Olaratumab with Doxorubicin and Ifosfamide as a first line treatment in metastatic STS had a lower PFS and a similar response rate to that of Doxorubicin and Ifosfamide combination without Olaratumab. 19 Moreover a pilot study reported that treatment with Doxorubicin, Ifosfamide, and Mesna (AIM) combination resulted in longer PFS and OS when compared to treatment with Olaratumab and Doxorubicin; however this difference was not statistically significant. 20 Finally in the most recent Phase Ib/II trial studying the addition of olaratumab to Gemcitabine plus Docitaxel found that there was no statistically significant improvement of OS in this treatment group compared to a control group receiving Gemcitabine, Docitaxel and Placebo, whether patients were or not previously treated with olaratumab. Moreover, the improvement in the PFS and overall response rate seen in the experimental group was not statistically significant.
Regarding the adverse effects, patients in our study did not experience serious adverse effects such as anemia or neutropenia and all the effects were limited to fatigue, nausea and decreased appetite. This was similar to the events observed in the ANNOUNCE trial, where although around 15% of the patients experienced grade 3 or higher adverse effects, the rate and type of adverse events were similar in both Olaratumab plus Doxorubicin and Doxorubicin alone groups, failing to identify any toxicity that could be attributed to the effect of Olaratumab alone. The only exception was that related to transfusion reactions that occurred slightly more frequently in the Olaratumab plus Doxorubicin group (11.7% vs 7.2%). 13 On the other hand, the real-wolrd data from England and Northern Ireland revealed a higher rate of adverse events (95%), which was similar the rate observed in the phase II trial (98%).10,18
Although the conductors of the ANNOUNCE trial could not offer a clearly defined cause for the divergent results between the phase II and phase III trials, they highlighted several factors that could have attributed to this discrepancy. These factors included the differences in study design (open-label vs blinded); study population; heterogeneity of STS in terms of histologic and biologic subtypes and crossover of the patients from the control arm to the Olaratumab containing arm in the phase II but not phase III trial. 13 Here it is worth noting that Olaratumab is not the only cancer medication to be withdrawn from the market after gaining an accelerated FDA approval. In fact, a review by Beaver et al. 15 highlighted that out of the 93 cancer drugs that got accelerated approval from FDA between 1992 and 2017, five were later withdrawn from the market as their clinical benefit could not be confirmed by subsequent confirmatory trials. One of these drugs, Gemtuzumab (anti-CD33 initially approved for AML relapse in adults) was re-approved by FDA for AML treatment, but after adjustment of its dosage as well as target population. Similarly, Olaratumab is currently being investigated as part of an ongoing, international, double-blind, placebo-controlled, randomized phase II ANNOUNCE 2 trial in combination with gemcitabine and docetaxel in patients with advanced STS (NCT02659020). 21
As a final note, the immunogenicity and the heterogeneity of STS are two heavily studied topics in the last few years which may explain why Olaratumab and other drugs are still incapable of achieving adequate responses. First, STS are less immunogenic than different malignancies since spontaneous regression is rarely observed in them. 22 PD1 and PD-L1 expression on T-cells and tumor cells is decreased, and HLA class I is downregulated on sarcomas. Some of the possible mechanisms of targeting STS’s immunogenicity is through combination therapy contain peptide vaccination and immune checkpoint inhibitors. 23 Another technique is by targeting T-cell responses against tumor-associated antigen-derived peptides.24,25 Second, STS heterogeneity also makes these tumors a great candidate for individualized treatment modalities. Derived from mesenchymal cells, STS vary on an intertumoral level and intratumoral level as well. Having complex and unstable karyotypes, STS biology stemness reflects genetics, epigenetics, and tumor microenvironment. 26 Despite these two factors, some of the molecular mechanisms of stemness are alike in different sarcoma types such as inhibition of classical tumor suppressor pRb and p53, de-inhibition of Sox-2, de-activation of Wnt/β-catenin signaling. 26 In addition, some stem cell markers function similarly in different leukemia and carcinomas as well as sarcomas. Unraveling the biology of sarcoma stem cells could help us ameliorate patient care and move us toward better-individualized treatment. 26
Our study has several limitations. Having a small sample size is the most critical limitation of our analysis. The retrospective nature of our study is another limitation. This might have affected our ability to find and demonstrate statistically significant results. Documenting the time of AEs occurrence in relation to the time the treatment was given could have also been useful. Large cohorts with more extended follow-up periods are required in order to investigate further, but this paper represents our experience as a tertiary care center in Lebanon.
Conclusion
In conclusion, our study provided an overview about the application of Olaratumab in STS in the Middle East. We found that patients in our population have reacted in a similar manner to those reported in the literature. The diagnosis and management of sarcomas is a changing and exciting field due to the genetic and biologic heterogeneity of the tumors. Gene expressions profiles are possible tools for prognostication allowing us to understand the stemness, progression and biology of tumors, which paves the way for individualized treatment. Olaratumab was not the first drug to be withdrawn from the market and probably will not be the last.
Footnotes
Author contributions
H.I. conceived the study. M.B.Z and C.E. were involved in protocol development, gaining ethical approval, patient recruitment and data analysis. M.B.Z and A.B wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
The study was conducted according to the ethical principles stated in the Declaration of Helsinki (2013). IRB (Institutional Review Board) at the American University of Beirut Medical Center reviewed the study proposal (BIO-2019-0058), and IRB approval was granted in March 2019 prior to data collection.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
