Abstract
Introduction
Skin cancers, particularly melanoma, are initiated and maintained by a subpopulation of tumor cells expressing stemness markers that are called cancer stem cells (CSCs). This study aimed to evaluate the expression levels and clinicopathological significance of the putative CSC markers CD44 and ALDH1A1 in patients with skin cancer.
Methods
The expression levels of CD44 and ALDH1A1 were investigated in 107 skin cancer specimens including 58 (54%) basal cell carcinomas (BCC), 37 (35%) squamous cell carcinomas (SCC), and 12 (11%) melanomas using the tissue microarray (TMA) technique. The correlation of the expression levels of these markers and clinicopathological parameters was then analyzed.
Results
The expression levels of CD44 and ALDH1A1 were significantly higher in melanoma patients than patients with SCC or BCC (p<0.001 and p = 0.002, respectively). A higher level of CD44 expression was more often found in melanoma tumor cells with a higher rate of recurrence (p = 0.029) and in SCC cases with ulceration (p = 0.01), while there was no significant correlation between ALDH1A1 expression and other clinicopathological parameters. Similarly, coexpression of CD44 and ALDH1A1 (CD44high/ALDH1A1high) was significantly observed in melanoma samples (p<0.001).
Conclusions
These findings suggest that a CD44high/ALDH1A1high phenotype in melanoma and a CD44high phenotype in SCC can be considered candidates for targeted therapy of skin cancers aiming at CSCs.
Introduction
Skin cancers are mainly divided into 2 major groups: non-melanoma skin cancer and melanoma. Non-melanoma skin cancer is the most common type of cancer and includes basal cell carcinoma (BCC), which comprises nearly 80% of all non-melanoma skin cancer cases, and squamous cell carcinoma (SCC), which is observed with a lower frequency (20% of all non-melanoma skin cancer cases) (1, 2). BCC and SCC are rarely fatal, whereas melanoma, which accounts for only 4% of skin cancers, is the lone culprit in 74% of all skin cancer-related deaths (1, 2). Although early-stage melanoma is curable, the prognosis of patients with advanced melanoma remains poor, with a median survival of only a few months (3, 4). The high mortality and morbidity rates of melanoma result from the high metastatic potential and the tumors’ resistance to chemo- and radiotherapy regimens (5). According to the cancer stem cell theory, only a distinct subset of cells, termed cancer stem cells (CSCs) or tumor-initiating cells (TICs), can drive tumor initiation, maintenance and progression as well as drug resistance (6, 7). CSCs have been isolated from a wide spectrum of malignancies using cell surface markers such as CD44 and ALDH1A1 (8-9-10).
CD44 is a transmembrane glycoprotein that plays an important role in growth, cell proliferation, regulation of cell adhesion and migration (11, 12). CD44, alone or in combination with other putative stem cell markers, is extensively applied for identifying CSCs in breast (13), prostate (14) and colon (15) cancers, while very limited studies have specifically investigated the differential expression of CD44 in melanoma tissue samples as a putative CSC marker (16). Although there is increasing evidence for the role of CSCs in melanoma progression (5, 17, 18), their relative importance is still unclear; for example, Quintana et al (16) showed that an average of 1 in 4 isolated cells from primary or metastatic melanoma was able to form tumors in non-obese diabetic/severe combined immunodeficiency (NOD/SCID) mice.
ALDH1A1 has been increasingly recognized in recent years as a potential CSC marker (9, 10, 19-20-21). The enzymatic activity of ALDH1A1 catabolizes the oxidation of intracellular aldehydes and thus provides resistance to some alkylating agents (22). Through the evaluation of human melanoma specimens it has been suggested that ALDH1A1-positive melanoma cells have higher tumorigenicity than ALDH1A1-negative cells, and thus ALDH1A1 may be a promising melanoma CSC marker (8-9-10); other studies, however, did not confirm these findings (23).
Given the lack of data regarding immunohistochemical expression of ALDH1A1 and CD44 as potential CSC markers in skin cancer, the present study was designed to explore the expression patterns of these markers by tissue microarray (TMA) in a well-defined series of skin cancer specimens including BCC, SCC and melanoma. We also evaluated the association between the level of expression of each marker and clinicopathological characteristics.
Material and methods
Patients and Tumor Characteristics
Archival tissue samples (107 tumor specimens including 58 BCCs, 37 SCCs and 12 melanomas) from patients with primary skin cancer diagnosed in the Shohada-e-Tajrish Hospital, a referral and teaching hospital in Tehran, Iran, were included in this study. Patients were diagnosed between 2003 and 2011 and medical records were reviewed to collect clinicopathological data including age, gender, tumor size, ulceration, tumor-infiltrating lymphocytes (TILs), Breslow thickness and Clark level (in melanoma), and histological grade (in SCC). Patients’ data were kept fully anonymous. The study protocol was approved by the Research Ethics Committee of Iran University of Medical Sciences.
Construction of TMA
Skin cancer TMAs were prepared as described previously (24, 25). Briefly, all hematoxylin and eosin-stained slides were reviewed to determine the best area for preparing the TMA from each specimen. Tissue arrays were then constructed by placing 0.6-mm diameter samples from 107 different tumor samples per single block, with 1-mm spacing separating TMA dots. The TMA blocks were constructed in 3 copies, each containing 1 sample from a different region of the tumor using a tissue-arraying instrument (Minicore; ALPHELYS, Plaisir, France). These TMAs were used for immunohistochemical staining of CD44 and ALDH1A1. The mean score of 3 cores was calculated as the final score.
Immunohistochemistry
Immunohistochemistry was performed on 4-µm-thick sections of skin cancer TMA slides (Superfrost Plus, Thermo Scientific, Germany) as previously described (26, 27). The sections were deparaffinized in xylene and rehydrated by passing them through graded ethanol series. The endogenous peroxidase activity was blocked by incubating in 3% hydrogen peroxide for 20 minutes at room temperature. Antigen retrieval was achieved by autoclaving slides in citrate buffer (pH = 6.0) for 10 minutes for CD44 antibody and 20 minutes microwaving for ALDH1A1 antibody. After 3 washes with Tris-buffered saline (TBS), the sections were incubated overnight at 4°C with specific antibodies against mouse monoclonal anti-human CD44 (Product code = NCL-CD44-2, Novocastra, UK) or anti-ALDH1A1 (ab52492, Abcam, Cambridge, UK) antigens with optimal dilution, 1:40 and 1:200, respectively. After washing, the TMA slides were incubated with anti-rabbit/anti-mouse EnVision reagent (Dako, Denmark) as secondary antibody for 10 minutes. The immunohistochemical staining was visualized with 3, 3’-diaminobenzidine (DAB, Dako) substrate as chromogen for 15 minutes at room temperature. The sections were lightly counterstained with hematoxylin, dehydrated through graded ethanol followed by xylene and mounted. Normal human tonsil tissue was used as positive control for CD44 antibody and normal liver was used as positive control for ALDH1A1 antibody. Incubation with only TBS was used as negative control.
Immunostaining Evaluation
Stained slides were examined by a semiquantitative scoring system on a multiheaded microscope by 2 observers (AR and ZM), and scoring was performed in a blinded manner to all the clinical data. In difficult cases, the scoring was confirmed by 2 observers and consensus was reached. Initially, the slides were scanned at 10× magnification to obtain a general impression of the overall distribution of the tumor cells. Positive cores were then assessed for localization and semiquantitatively for expression level at higher magnifications, and the final scores were given. Intensity of staining was scored as 0 (absent), 1 (weak), 2 (moderate) or 3 (strong). Also the percentage of positive tumor cells was scored as 1 (positive tumor cells <25%), 2 (positive tumor cells 25%-50%), 3 (positive tumor cells 50%-75%) and 4 (positive tumor cells >75%). Finally, the histochemical score (
Statistical Analysis
Statistical analyses were performed using the SPSS software version 20 (SPSS, Chicago, IL, USA). Associations between CD44 and ALDH1A1 status and clinicopathological parameters were assessed with Pearson's chi-square and Pearson's R tests. Moreover, CD44 and ALDH1A1 expression in different skin cancer types was compared using the Mann-Whitney
Results
Study Population
Of 107 skin cancer samples, 95 (89%) were non-melanoma skin cancer including 58 (54%) BCC and 37 (35%) SCC, whereas 12 (11%) specimens were melanoma (Tabs. I, II and III). There was a male predominance in all groups of skin cancer with different male/female ratios: 1.52 (35/23) in BCC, 3.11 (28/9) in SCC, and 3 (9/3) in melanoma. Of the 58 BCC samples, only 4 (7%) patients showed local recurrence without any metastasis. Among the SCC samples, 13 (36%) patients showed local recurrence, 12 (32%) had no local recurrence and there were no available data for 12 (32%) specimens. Nine (24%) SCC patients had metastases, 18 (49%) had no metastasis, and no data were available for 10 (27%) cases. From the data available for local recurrence in melanoma cases, 5 (42%) patients had local recurrence and 4 (33%) had no local recurrence. Dermal invasion was reported in 8 (67%) melanoma specimens, whereas 4 (33%) patients had in situ melanoma. TILs, as a prognostic factor, were categorized as absent, moderate and brisk levels (28, 29). In melanoma cases, tumor size ranged from 1 mm to 13 mm (mean = 4.1 mm); 8 (67%) had a mean size ≤4.1 mm and 4 (33%) a mean size >4.1 mm. Breslow thickness was categorized as thin melanoma (Breslow thickness ≤1 mm) and thick melanoma (Breslow thickness >1 mm) (30). Thin melanoma was found in 8 (67%) cases and thick melanoma in 4 (33%) cases. Mitotic count ≤6 and mitotic count >6 had similar frequencies (50%). The Clark levels in melanoma were also divided into 2 groups: group 1 (Clark levels I and II) and group 2 (Clark levels III through V) (28). Nine (75%) melanoma sample were categorized as group 2 and 3 (25%) as group 1 (Tab. III).
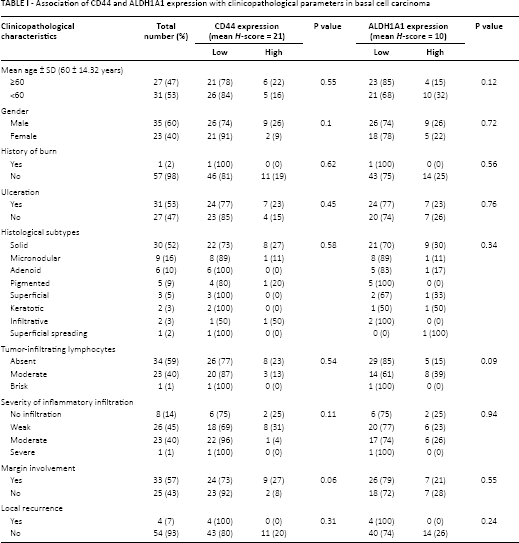
Association of CD44 and ALDH1A1 expression with clinicopathological parameters in basal cell carcinoma
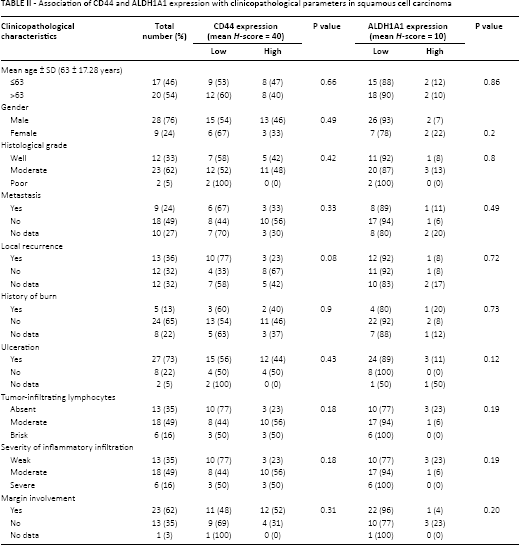
Association of CD44 and ALDH1A1 expression with clinicopathological parameters in squamous cell carcinoma
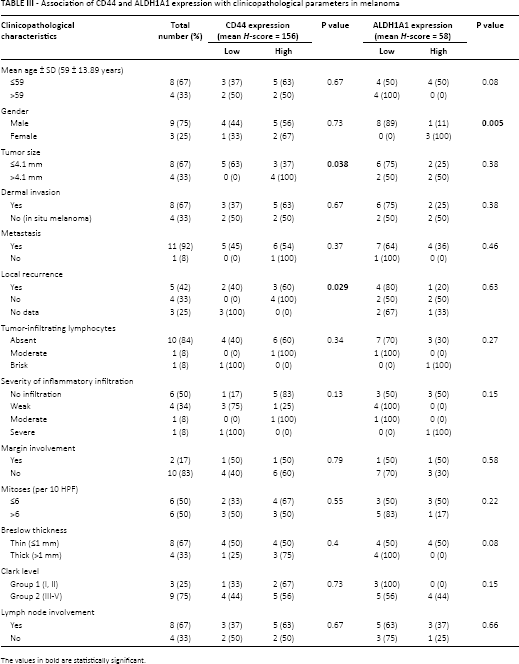
Association of CD44 and ALDH1A1 expression with clinicopathological parameters in melanoma
The values in bold are statistically significant.
Analysis of CD44 Expression and Its Correlation with Clinicopathological Features
Each tumor type was divided into either lower (≤ mean of
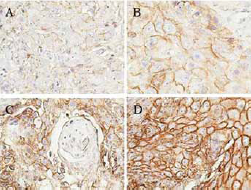
Expression of CD44 in different skin cancer subtypes. In each type of tumor, intensity of staining was graded as (
Analysis of ALDH1A1 Expression and its Correlation with Clinicopathological Parameters
We also performed immunohistochemical analysis to investigate the expression of another putative CSC marker, ALDH1A1, in the same series of skin tumors. The pattern of ALDH1A1 staining was cytoplasmic. The mean
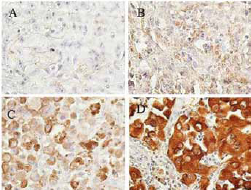
Expression of ALDH1A1 in different skin cancer subtypes. In each type of tumor, staining intensity was graded as (
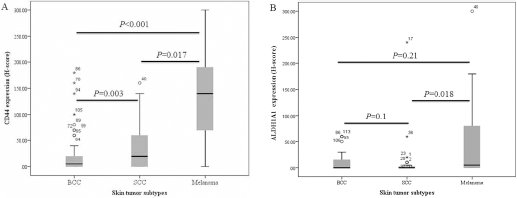
Box-plot diagram of CD44 and ALDH1A1 expression in skin tumor subtypes. In each panel the vertical axis shows the total immunostaining score (
Combined Analysis of CD44 and ALDH1A1 Expression
Evaluation of the relation between the expression of CD44 and ALDH1A1 showed a significant reciprocal correlation in our series of skin tumors (p<0.001). In the current study, a CD44high/ALDH1A1high phenotype was designated as group 1, a CD44low/ALDH1A1high or CD44high/ALDH1A1lowphenotype was designated as group 2, and a CD44low/ALDH1A1low phenotype was designated as group 3. The CD44low/ALDH1A1lowphenotype was observed in 58 (54%) cases, whereas 10 (9%) samples showed the CD44high/ALDH1A1high phenotype. Expression of any marker (CD44low/ALDH1A1high or CD44high/ALDH1A1low) was found in 39 (37%) skin cancer specimens. The majority of BCC samples (67%) displayed the CD44low/ALDH1A1lowphenotype, while coexpression of the markers (CD44high/ALDH1A1high) was seen in 5 (9%) BCC cases (Fig. 4). Although expression of any marker (CD44low/ALDH1A1high or CD44high/ALDH1A1lowphenotype) and the CD44low/ALDH1A1low phenotype were observed in 20 (54%) and 17 (46%) of SCC cases, respectively, the CD44high/ALDH1A1highphenotype was not found in SCC samples (0%) (Fig. 4). In melanoma, the phenotype groups 1 and 2 had similar frequencies (42%) (Fig. 4).
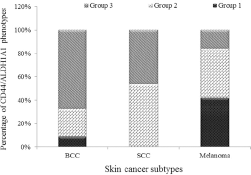
Distribution of CD44/ALDH1A1 phenotypes in skin cancer subtypes including basal cell carcinoma (BCC), squamous cell carcinoma (SCC) and melanoma. Group 1: CD44high/ALDH1A1high phenotype; group 2: CD44low/ALDH1A1high or CD44high/ALDH1A1low phenotype; group 3: CD44low/ALDH1A1low phenotype.
The correlation of CD44/ALDH1A1 phenotypes with clinicopathological parameters was investigated by 1-way ANOVA and Tukey's post hoc analysis. We found that CD44/ALDH1A1 status significantly correlated with tumor type (p<0.001). Also among the different subtypes of skin tumors, the CD44/ALDH1A1 status was significantly different between melanoma versus BCC (p<0.001) and SCC (p = 0.003) (Fig. 3). No significant association was found between CD44/ALDH1A1 phenotypes and other clinicopathological parameters in BCC and melanoma.
Discussion
Among various markers for identification of CSCs, ALDH1A1 has been reported to be a melanocytic stem cell marker (8-9-10). ALDH1A1 is an enzyme that accelerates oxidation of aldehydes to their corresponding carboxylic acids (31). ALDH1A1 has been widely used for the identification and isolation of CSCs from many types of cancers including melanoma (9, 19, 20, 23, 32, 33). For the first time (34), this study showed that ALDH1A1 had higher expression levels in melanoma than in BCC and SCC. In line with our data, previous studies showed that high expression of ALDH1A1 correlated strongly with aggressive tumor behavior and poor prognosis in breast, colon and lung cancer (19, 20, 33), whereas for melanoma there is no consensus about the biological role(s) of ALDH1A1 in CSCs (8, 9, 23, 34). There is some evidence that ALDH1-positive melanoma cells fulfill the CSC criteria based on
The CD44 antigen is a cell-surface molecule involved in cell proliferation and survival, cell-cell interactions, cell adhesion and migration (11, 36). There is still insufficient data regarding the role of CD44 as a promising CSC marker in melanoma (11, 13, 26, 37-38-39). We found that the highest percentages of CD44-positive tumor cells were detected in melanoma samples, followed by SCC and lastly BCC samples. Our study confirmed previous data showing low expression of CD44 in BCC samples and intermediate expression levels in SCC specimens (40). In the current study, higher CD44 expression was more often found in melanoma samples with larger sizes. Additionally, a positive correlation was observed between the expression of CD44 and recurrence in melanoma cases. These findings may suggest that high CD44 expression confers aggressiveness and metastatic potential on melanoma cells. In several previous reports, the role of CD44 as a potential CSC marker was explored in melanoma samples using flow cytometry; the results showed that melanoma cells expressing CD44 and those that were CD44 negative had similar CSC-like properties in
We also evaluated the coexpression of ALDH1A1 and CD44 in the examined skin cancer specimens. The CD44high/ALDH1A1high expression pattern was predominantly found in melanoma cases compared to the other types of skin cancer, which might account for the fact that melanoma is the most aggressive skin tumor.
CTLA-4 (CD152) is encoded by the
In the present study, overexpression of ALDH1A1 and CD44 was found in melanoma samples compared to non-melanoma skin cancer samples including BCC and SCC. Increased expression of ALDH1A1 and CD44 appears to confer aggressive behavior on melanoma, and these markers may therefore be considered potential candidates for targeting melanoma CSCs.
Conclusions
The distinct differential expression of the potential CSC markers CD44 and ALDH1A1 in melanoma compared with SCC and BCC showed that expression of CD44/ALDH1A1 is associated with aggressive tumor behavior and metastasis. The CD44high/ALDH1A1high phenotype in melanoma samples and CD44high cells in SCC can be considered as reliable targets for diagnosis and therapy of skin cancers.
Footnotes
Financial support: This study was supported by a grant from Iran University of Medical Sciences (Grant #12417-12-04-89).
Conflict of interest: The authors declare that they have no conflict of interest.
