Abstract
BACKGROUND:
Dynamin 2 (DNM2) involved in tumor progression in various malignancies.
OBJECTIVE:
For the first time, we evaluated DNM2 expression pattern, its association with clinicopathological characteristics and survival outcomes in RCC subtypes.
METHODS:
We evaluated the DNM2 expression pattern in RCC tissues as well as adjacent normal tissue using immunohistochemistry on tissue microarray (TMA) slides.
RESULTS:
Our findings revealed increased DNM2 expression in RCC samples rather than in adjacent normal tissues. The results indicated that there was a statistically significant difference between cytoplasmic expression of DNM2 among subtypes of RCC in terms of intensity of staining, percentage of positive tumor cells, and H-score (
CONCLUSIONS:
Our results indicated that DNM2 cytoplasmic expression is associated with tumor aggressiveness and poor outcomes. DNM2 could serve as a promising prognostic biomarker and therapeutic target in patients with ccRCC.
Introduction
Renal cell carcinoma (RCC), as the most lethal malignancy of the urologic system, represents approximately 2 to 3% of all malignancies [1]. In 2020, there were 431,288 new cases of RCC and 179.368 RCC cancer-related deaths worldwide [2]. Clear cell RCC (ccRCC) (80–90%), papillary RCC (10–15%), and chromophobe RCC (4–5%) are the main subtypes of RCC [3, 4, 5]. Stage, grade, and cancer-specific survival (CSS) varied between RCC subtypes and impacts on tumor prognosis [5]. Identification of molecules associated with histopathological characteristics is critical for survival prediction and choosing the appropriate treatment [6]. After surgical resection, which is the main curative treatment for localized RCC, the risk of recurrence is nearly
Dynamin (DNM) is a member of multidomain GTPases family introduced as a microtubule-binding protein. Dynamin, as a key regulator, is involved in the formation, fission, and remodeling of multiple organelles, specially endocytic vesicles [11]. Dynamin genes encode three tissue-specific protein isoforms, dynamin 1-3 (DNM1-3). Dynamin 1 is predominantly expressed in neuronal tissue; DNM2 is expressed at lower levels in all cell types of tissues, and dynamin 3 is present in the testis, lung, and postsynaptic nervous system [12, 13]. DNM2 is involved in endocytosis, intracellular membrane trafficking, apoptosis, and the actin network. Studies have shown that DNM2 knockout results in in early embryonic lethality [11, 14, 15].
Alterations in homeostasis and mutations in DNM2 have been associated with diseases such as Alzheimer’s and cancer [16, 17]. Recent studies indicated that increased expression of DNM2 promotes tumor progression in several tumors such as pancreatic, lung, and prostate cancers [18, 19, 20]. Also, DNM2 interacts directly with PI3K, Src, and FAK, which are all involved in cell migration, invasion, and metastasis [17, 21]. In 60% of human metastatic pancreatic tumors, DNM2 is upregulated and associated with poor prognosis in patients. DNM2 over-expressed pancreaticductal tumor cells were shown to have strong migration and invasion potentials [18]. Also, the role of DNM2 in traffickingof DNA repair enzyme has been investigated [22]. Many cases of triple-negative breast cancers (TNBC) have shown recurrence after the initial response to chemotherapy, which has been associated to upregulated homology-directed repair (HDR). HDR repairs DNA double-strand breaks (DSBs), the most critical chemotherapy-induced lesions. Inhibition of DNM2 could impair HDR and improve chemotherapy response. Thus, the level of DNM2 can predict chemotherapy outcome in estrogen receptor (ER)-negative breast cancer [22]. A DNM2 inhibitor has been shown to reduce cell migration in glioblastoma [23]. In addition, various investigations have shown that pharmacological inhibition of DNM2 with GTPase inhibitors reduced tumorigenicity and cancer cell invasion, representing DNM2 as a new therapeutic target for cancer therapy [18, 24, 25].
Considering the previous data, it is unknown whether DNM2 is involved in RCC progression or prognosis. For the first time, we used immunohistochemistry (IHC) on tissue microarray (TMA) slides to assess DNM2 expression in the membrane, cytoplasm, and nucleus of three major RCC subtypes: ccRCC, papillary RCC, and chromophobe RCC, and investigated its clinical significance as well as prognostic value.
Materials and methods
Bioinformatics study
Gene Expression Profiling Interactive Analysis (GEPIA2) (
Patient characteristics and tumor samples
In the current study, 233 paraffin-embedded tissues were collected from RCC samples between 2008 and 2015 from Hasheminejad hospital, a foremost national referral center for urology and nephrology center in Tehran, Iran. To access all clinicopathological data, all samples were collected from patients who had undergone radical nephrectomy and none of them had any history of chemotherapy or radiotherapy prior to surgery. The samples consisted of multiple subtypes of RCC including clear cell, papillary, and chromophobe RCC. The tissue samples were in the formalin-fixed paraffin-embedded (FFPE) form using a routine pathological tissue protocol (TPP) technique. The clinicopathological data of patients including age, gender, tumor size, nucleolar grade, TNM stage, microvascular invasion (MVI), lymph node invasion (LNI), renal vein invasion, tumor necrosis (TN), renal sinus fat invasion, renal pelvis invasion, perirenal fat invasion, gerota’s fascia invasion, distant metastasis, and tumor recurrence were collected from the patients’ documents. Furthermore, 20 adjacent normal tissue samples were used to compare the levels and patterns of expression of DNM2 across a range of tissue specimens. In addition, information regarding survival outcomes was also collected. The date of radical nephrectomy to the date of each death and death caused due to RCC was measured as - overall survival (OS) and disease-specific survival (DSS), respectively. Progression-free survival (PFS) was defined as the interval between the surgery and the last follow-up visit without evidence of disease, relapse, or metastasis. Tumor staging was performed according to the pTNM classification for renal cell carcinoma [31]. This study received its ethical approval (Code: IR.IUMS.REC.1398.1354.) from the Research Ethics Committee of Iran University of Medical Sciences.
Tissue microarray (TMA) construction
Renal tissue TMAs were prepared as described previously [32]. Briefly, H&E slides were examined by a pathologist to select the most representative area of the tumor in each block. Then, using a precision arraying instrument (Tissue Arrayer Minicore; ALPHELYS, Plaisir, France), selected regions of each block were punched out (0.6 mm diameter) and transferred into a new recipient paraffin block. Next, 4-
Immunohistochemistry (IHC) staining
Briefly, all TMA slides were deparaffinized at 60
Evaluation of immunostaining
Immunostaining of DNM2 was evaluated by a pathologist (M.R.) who was blinded to the patient outcome and pathological information. The intensity of staining was scored by applying a semi-quantitative system, ranging from negative to strong as follows: 0, no staining; 1, weak; 2, moderate; and 3, strong staining. The percentage of positive cells was scored from 0% to 100%, and then categorized according to the positive tumor cells as follows:
Statistical analysis
Statistical software SPSS, version 22.0 (IBM Corp, USA) was utilized to analyze the data. In this study, the categorical data were reported as
Box plot results of clinical proteomic tumor analysis consortium (CPTAC) for DNM2 protein expression on UALCAN database. The results showed a higher expression of DNM2 protein in ccRCC tissue samples compared to normal samples (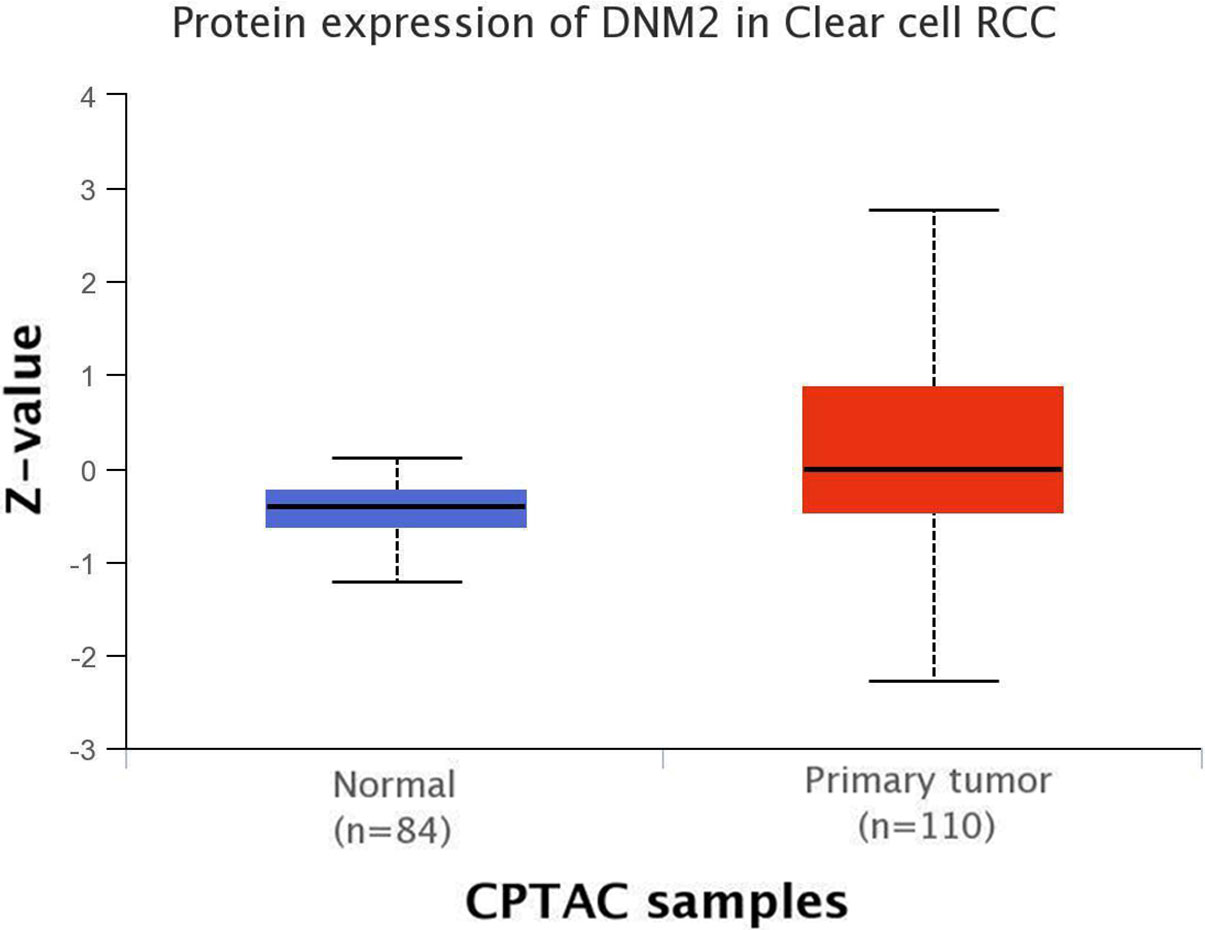
Bioinformatics approaches
The results of the TCGA database via GEPIA2 revealed that mRNA expression of DNM2 gene was higher (
Protein-protein interaction (PPI) network for DNM2. PPI network for DNM2 protein was created by StringApp plug-in using Cytoscape software with confidence score 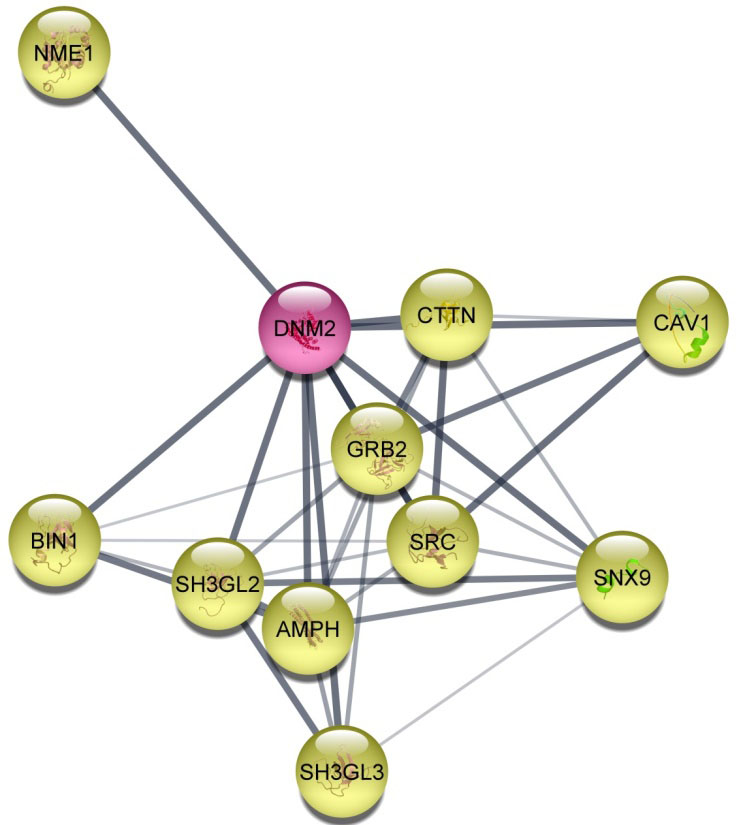
In total, 233 RCC patients, including ccRCC (182, 78%), papillary RCC (22, 9.5%), and chromophobe RCC (29, 12.5%), were evaluated in this study. The mean age at diagnosis was 55 years and male-to-female ratio was 2.4. The tumor size ranged from 1 to 21 cm, and they were classified into four groups: Group 1:
Tumor pathological features of histological subtypes of renal cell carcinoma (RCC)
Tumor pathological features of histological subtypes of renal cell carcinoma (RCC)
ccRCC; clear cell renal cell carcinoma, pRCC; papillary renal cell carcinoma, chRCC; chromophobe renal cell carcinoma.
The expression level of DNM2 protein was evaluated using IHC on TMA sections. It was assessed by three scoring methods: intensity of staining, percentage of positive tumor cells, and H-score. The expression was detected at different intensities in the cell membrane, cytoplasm, and nucleus with each analyzed separately. Note that adjacent normal renal tissues samples showed a lower expression level of DNM2 compared with RCC tissues (Fig. 3). No staining was observed in isotype control sections exposed to non-immune rabbit IgG.
Immunohistochemical analysis of DNM2 protein expression in three major subtypes of renal cell carcinomas (RCCs). DNM2 protein expression in clear cell renal cell carcinoma (ccRCC): low expression (A, A-1), moderate expression (B, B-1), high expression (C, C-1) and adjacent normal tissue sample of ccRCC (D–D1). DNM2 protein expression in papillary RCC (pRCC): low expression (E, E-1), moderate expression (F, F-1), high expression (G, G-1) and adjacent normal tissue sample of pRCC (H–H1). In chromophobe RCC (chRCC): low expression (I, I-1), moderate expression (J, J-1), high expression (K, K-1) and adjacent normal tissue sample of chRCC (L–L1). Isotype controls in immunohistochemistry staining in ccRCC (M), pRCC (N), chRCC (O) and adjacent normal tissue sample of ccRCC (p). Figures are shown with a magnification of 100X and 200X.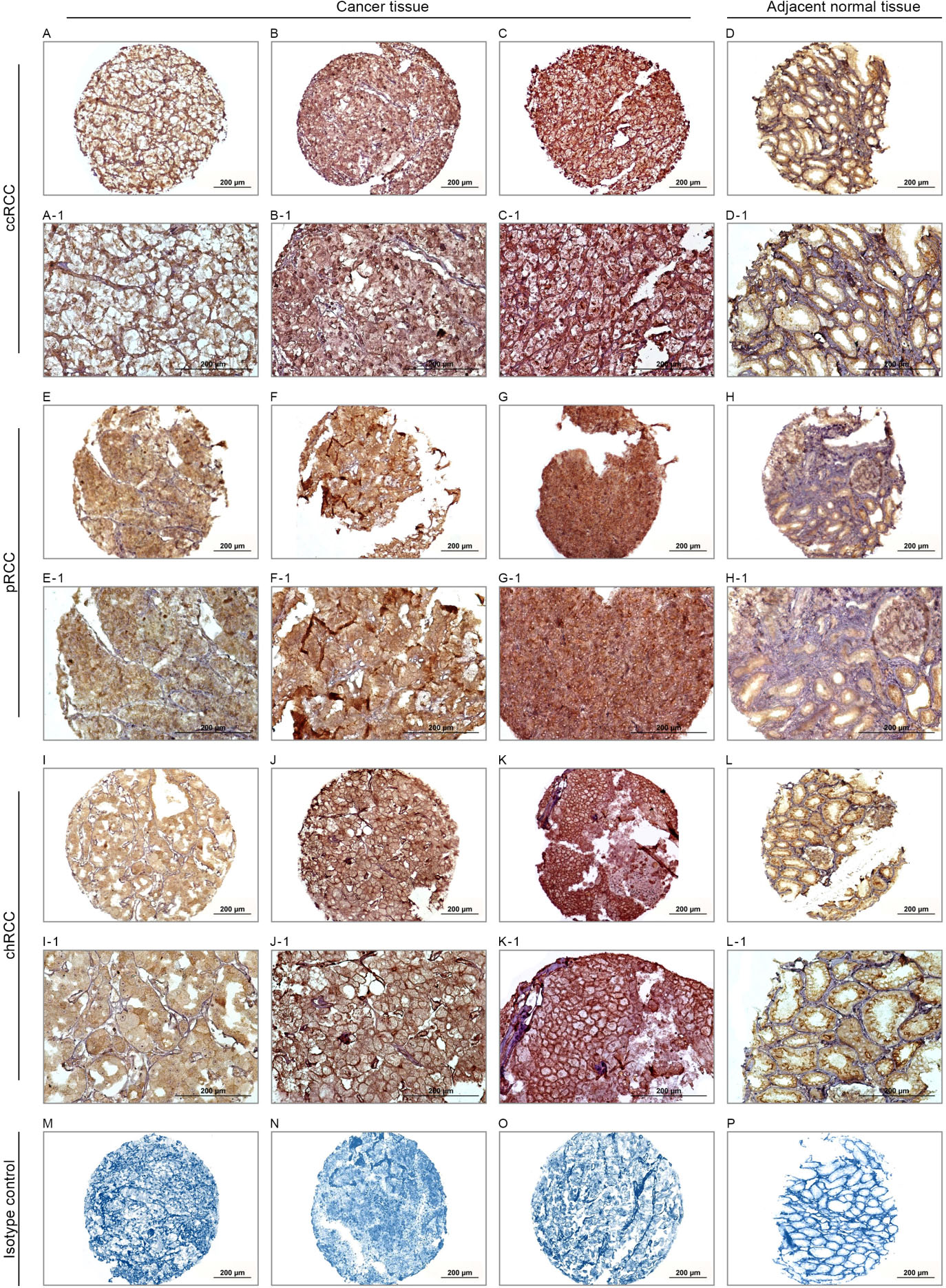
Expression of DNM2 was observed in variable intensities in the cell membrane, cytoplasm, and nucleus across the histological RCC subtypes (Fig. 3). Of 233 RCC samples stained for DNM2, 82 (35.2%) of all RCC samples did not show any staining in the membrane, while weak, moderate, and strong expression were observed in 28 (12%), 83 (35.6%), and 40 (17.2%) cases, respectively. In the cytoplasm, 10 (4.3%) did not show any staining, whereas low, moderate, and high intensity of DNM2 expression was observed in 49 (21%), 118 (50.6%) and 56 (24%) cases, respectively. Also, 123 (52.8%) of all cases did not show any staining in nucleus and DNM2 expression observed as weak, moderate and strong expression in 12 (5.2%), 82 (35.2%), 16 (6.9%) of cases, respectively.
In this study, chi-square test was applied to investigate the association between DNM2 expression and histological subtypes of RCC. All results of the analysis are reported in Table 3. The findings showed that there are statistically significant differences between DNM2 cytoplasmic expression in terms of intensity of staining, percentage of positive tumor cells, plus H-score and various histological subtypes of RCC (
Associations between DNM2 expression (membrane, cytoplasm, and nucleus) and clinicopathological characteristics of ccRCC
The results of chi-square test revealed a statistically significant association between increased cytoplasmic expression of DNM2 and histological TN (
Associations between DNM2 expression (membrane, cytoplasm, and nucleus) and clinicopathological characteristics of papillary RCC and chromophobe RCC
In papillary RCC, we found a significant association between increased membranous DNM2 expression and distant metastasis (
Expression of DNM2 for clinical outcomes in RCC subtypes
In this study, the follow-up period was 116 months in RCC patients. The mean duration of the follow-up time for OS and DSS was 54 (SD
| Tumor characteristics | Total samples | Membranous expression H-score |
|
Cytoplasmic expression H-score |
|
Nuclear expression H-score |
|
||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
|
0–100 | 101–200 | 201–300 | 0–100 | 101–200 | 201–300 | 0–100 | 101–200 | 101–200 | ||||
| ccRCC | 182 | 154 (84.6) | 18 (9.9) | 10 (5.5) | 49 (26.9) | 84 (46.2) | 49 (26.9) | 174 (95.6) | 8 (4.4) | 0 (0.0) | |||
| Mean age, years (range) | 56 (25–82) | ||||||||||||
| |
93 (51.1) | 83 (53.9) | 5 (27.8) | 5 (50.0) | 0.111 | 28 (57.1) | 38 (45.2) | 27 (55.1) | 0.335 | 87 (50.0) | 6 (75.0) | 0 (0.0) | 0.167 |
| |
89 (48.9) | 71 (46.1) | 13 (72.2) | 5 (50.0) | 21 (42.9) | 46 (54.8) | 22 (44.9) | 87 (50.0) | 2 (25.0) | 0 (0.0) | |||
| Gender | |||||||||||||
| Male | 128 (70.3) | 108 (70.1) | 15 (83.3) | 5 (50.0) | 0.179 | 35 (71.4) | 58 (69.0) | 35 (71.4) | 0.940 | 123 (70.7) | 5 (62.5) | 0 (0.0) | 0.620 |
| Female | 54 (29.7) | 46 (29.9) | 3 (16.7) | 5 (50.0) | 14 (28.6) | 26 (31.0) | 14 (28.6) | 51 (29.3) | 3 (37.5) | 0 (0.0) | |||
| Tumor size (cm) | |||||||||||||
| |
37 (20.3) | 29 (18.8) | 5 (27.8) | 3 (30.0) | 0.806 | 8 (16.4) | 17 (20.2) | 12 (24.5) | 0.542 | 33 (19.0) | 4 (50.0) | 0 (0.0) | 0.085 |
| 4.1–7 | 58 (31.9) | 48 (31.2) | 7 (38.8) | 3 (30.0) | 20 (40.8) | 25 (29.8) | 13 (26.5) | 57 (32.8) | 1 (12.5) | 0 (0.0) | |||
| 7.1–10 | 50 (27.5) | 44 (28.6) | 3 (16.7) | 3 (30.0) | 15 (30.6) | 22 (26.2) | 13 (26.5) | 47 (27.0) | 3 (37.5) | 0 (0.0) | |||
| |
37 (20.3) | 33 (21.4) | 3 (16.7) | 1 (10.0) | 6 (12.2) | 20 (23.8) | 11 (22.5) | 37 (21.2) | 0 (0.0) | 0 (0.0) | |||
| Nucleolar grade | |||||||||||||
| I | 10 (5.5) | 7 (4.5) | 3 (16.7) | 0 (0.0) | 0.185 | 1 (2.0) | 5 (6.0) | 4 (8.2) | 0.613 | 10 (5.7) | 0 (0.0) | 0 (0.0) | 0.193 |
| II | 105 (57.7) | 88 (57.2) | 9 (50.0) | 8 (80.0) | 30 (61.2) | 52 (61.8) | 23 (46.9) | 98 (56.3) | 7 (87.5) | 0 (0.0) | |||
| III | 55 (30.2) | 50 (32.5) | 4 (22.2) | 1 (10.0) | 15 (30.6) | 22 (26.2) | 18 (36.7) | 55 (31.6) | 0 (0.0) | 0 (0.0) | |||
The association between expression of DNM2 and clinicopathological features of clear cell renal cell carcinoma (ccRCC)
36 (15.5%) of samples had recurrence. During the follow-up time, cancer-related death occurred in 34 (18.7%), 2 (9.1%), and 3 (10.3%) in ccRCC, papillary RCC and chromophobe RCC, respectively. The main characteristics of patients enrolled for survival analysis based on histological subtypes of RCC are summarized in (Table 4).
In this study, the Kaplan–Meier survival analysis curves and the log-rank tests were used to assess the association between membranous, cytoplasmic, and nuclear DNM2 expression and OS, DSS, and PFS.
ccRCC
Survival analysis based on cytoplasmic DNM2 expression
The main characteristics of patients survival analysis based on subtypes of renal cell carcinoma (RCC)
The main characteristics of patients survival analysis based on subtypes of renal cell carcinoma (RCC)
Kaplan-Meier survival analysis in the cytoplasmic expression of DNM2 in ccRCC showed a significant difference between OS (Log-rank test:
Kaplan–Meier curves for overall survival (OS), disease-specific survival (DSS), and progression-free survival (PFS) based on cytoplasmic DNM2 protein expression levels in clear cell renal cell carcinoma (ccRCC). In ccRCC, a higher level of cytoplasmic DNM2 protein expression was associated with shorter, OS, DSS, and PFS compared to the tumors with moderate and low expression of this protein (Log-rank tests; 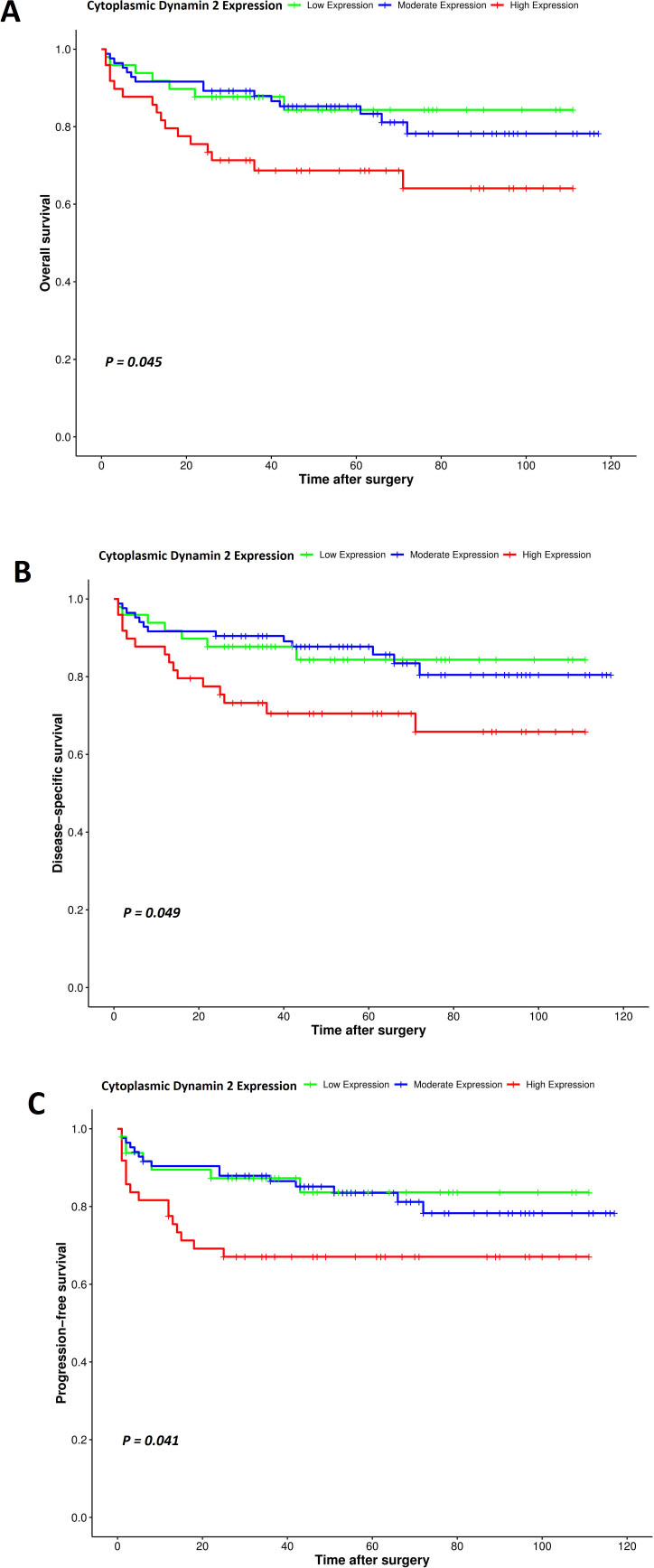
Univariate and multivariate cox regression analyses of potential prognostic factors for overall survival (OS) in patients with clear cell RCC (ccRCC)
HR; hazard ratio, CI; confidence interval The variables with
Univariate and multivariate cox regression analyses of potential prognostic factors for disease- specific survival (DSS) in patients with clear cell RCC (ccRCC)
HR; hazard ratio, CI; confidence interval The variables with
Then, univariate and multivariate cox regression analyses were performed to assess the clinical significance of various parameters that might influence OS, DSS, and PFS in patients with ccRCC. Cytoplasmic expression of DNM2, tumor size, grade, and TNM stage were significant risk factors, affecting the OS, DSS, and PFS of patients with ccRCC in the univariate analysis (Tables 5–7). As outlined in Tables 5–7, TNM stage was an independent prognostic factor for OS, DSS, and PFS in the multivariate analysis (
Univariate and multivariate cox regression analyses of potential prognostic factors for progression-free survival (PFS) in patients with clear cell RCC (ccRCC)
HR; hazard ratio, CI; confidence interval The variables with
Survival analysis based on membranous and nuclear DNM2 expression
Survival analysis revealed that membranous and nuclear expression of DNM2 did not have a statistically significant effect on OS, DSS, and PFS in ccRCC patients.
Survival analysis based on membranous, cytoplasmic, and nuclear DNM2 expression
Kaplan–Meier survival analysis revealed that expression of DNM2 did not have a statistically significant effect on OS, DSS, and PFS in papillary and chromophobe RCC patients. In addition, the results of univariate and multivariate analyses demonstrated that the clinicopathological variables were not significant factors affecting the OS, DSS, and PFS of patients with papillary and chromophobe RCC.
Discussion
Renal cell carcinoma accounts for approximately 3% of adult malignancies and 90–95% of renal neoplasms [38, 39]. The initial treatment for this disease is usually partial or complete removal of the damaged kidney, but about 30% of patients experience metastasis at the time of diagnosis and 40% have recurrence post-surgery. In cases where cancer does not metastasize or invade deep into the kidney tissue, the 5-year survival rate is relatively good, but as cancer spreads, the rate decreases significantly [40]. In addition, there is resistance to radiation therapy and chemotherapy in RCC patients, and despite the development of several targeted therapies for the treatment of RCC, the effectiveness of treatment and resistance to therapy has remained a challenge [39, 41]. Thus, finding novel molecules is crucial for assessing the prognosis as well as RCC-targeted therapy [42].
DNM2 is a GTPase molecule that contributes to cancer pathomechanisms and has well- known roles in different types of tumors based on the DisGeNET database. Previous studies have shown increased expression of DNM2 in several types of cancer such as pancreatic, lung, breast, prostate, and cervical cancer [18, 19, 20, 43, 44], and its overexpression is associated with several cancer-related processes, including migration, invasion, metastasis, cell proliferation, and patient survival [20, 25, 45, 46, 47, 48, 49]. DNM2 has an oncogenic role through several mechanisms. Based on the STRING-PPI network, DNM2 was correlated with proteins which have important roles in cancer, such as SRC and GRB2. It has been reported that DNM2 required for the activation of the downstream proto-oncogene-Src family kinase-mediated signaling cascade. It has been shown that deregulation of DNM2 may be impaired pathways of Grb2 as a tumor suppressor gene, in cancer cells [35, 36, 50]. DNM2 is required for endocytosis of several proteins and oncogenic receptors associated with cancer motility and invasiveness [51, 52, 53]. Furthermore, DNM2 has been linked to the regulation of actin-driven cytoskeletal remodeling processes, where one of the consequences is the formation of Invadopodia. Invadopodia are dynamic actin-rich protrusions of the membrane that degrade ECM in cancer invasiveness. DNM2 activation and intracellular localization are critical in the focal degradation of ECM at invadopodia [54, 55]. Regarding importance of DNM2 function in cancer, in this study, for the first time, the expression levels of DNM2 were examined in 233 RCC tissue samples of three major RCC subtypes including ccRCC, papillary RCC, chromophobe RCC, as well as adjacent normal tissue. Further, the association between its expression levels and clinicopathological characteristics plus survival outcomes was assessed.
Bioinformatics analysis revealed a modest difference in mRNA expression between RCC and normal tissues (a slightly greater expression in the ccRCC group), but a considerable difference was found in protein expression between the two groups (increase in the ccRCC group). In this current study, high expression of DNM2 in RCC samples was observed compared to adjacent normal tissues. These findings are in agreement with our previous study on breast cancer and several studies, regarding protein expression level: pancreatic tumors, cervical and prostate cancer, mRNA expression level: acute lymphoblastic leukemia and bladder cancer [18, 19, 43, 53, 56, 57]. As a result, our findings confirm the importance of the increased expression of DNM2 in cancer progression.
Cheville et al. reported that histological subtype was significantly associated with CSS and ccRCC was found to be associated with decreased survival compared with papillary and chromophobe RCC [58]. In this study, the expression patterns of DNM2 were investigated in different histological subtypes of RCC. The results indicated that the intensity of staining in each subtype was heterogeneous especially in subcellular localizations ranging from weak to strong. The results exhibited that 95% of RCCs were positive for cytoplasmic expression of DNM2. Importantly, we found a statistically significant difference between the cytoplasmic expression of DNM2 among subtypes of RCC in terms of intensity of staining, percentage of positive tumor cells, and H-score. These findings indicated that DNM2 expression patterns are different in histological subtypes of RCC, which may highlight the significance of prognostic and therapeutic potentials of this molecule in RCC patients.
In the present study, increased cytoplasmic expression of DNM2 was found to be associated with advanced histological TN. In this regard, previous studies have shown that histological TN can be an independent prognosis of poor outcomes in RCC as well as in malignancies of lung, breast, thyroid, colon, pancreatic, gastrointestinal stromal tumors, and Ewing sarcoma [59, 60, 61, 62]. It is well known that hypoxia and metabolic stress cause cell necrosis [63, 64, 65]. Increased mitochondrial fission is one of the characteristics of necrotic cells, due to stress factors such as inflammation [66]. It has been shown that a set of dynamin-related GTPases, such as Dyn2 and Drp1 (dynamin-related protein1), play an important role in the mitochondrial fission machinery [67, 68, 69, 70, 71, 72]. Thus, increased cytoplasmic expression of DNM2 in necrotic tissues can be justified.
In the present study, survival analysis revealed that cytoplasmic expression of DNM2 in ccRCC patients had a significant association with OS, DSS, and PSF. Patients who had a higher cytoplasmic expression of DNM2 had a worse prognosis than those who had moderate or low expression of DNM2. The results also indicated that high cytoplasmic expression of DNM2 was associated with poor 5-year OS, DSS, and PFS in ccRCC. Ren et al. demonstrated that DNM2 over-expression was significantly correlated with the OS rate of papillary thyroid cancer (PTC) and that higher DNM2 expression linked to more unfavorable prognosis of PTC [73]. In breast cancer, there is association between prolactin exposure and development of invasiveness. The downstream signaling pathway due to the presence of prolactin, associated with proliferation, invasion, and survival in breast cancer cells, was shown to be DNM2-dependent [35, 74]. Further, DNM2 overexpression can indirectly affect survival through concomitant upregulation of miRNA located at the DNM2 locus. In DNM2 overexpressing cells, overexpression of miR-638 may reduce the expression of PTEN and p53 and subsequently deregulate their specific signaling pathways governing cell growth and survival [75, 76]. Importantly, the cytoplasmic expression of DNM2 was found as an independent prognostic factor affecting DSS and PFS in multivariate analysis, indicating an unfavorable prognosis. Hence, these results exhibited that cytoplasmic DNM2 expression compared with membranous and nucleus expression, is related to the degree of malignancy and progression of disease in ccRCC cases.
To understand the significance of increasing DNM2 cytoplasmic expression compared to the membrane and nuclear expression in ccRCC, it should be noted that the most conspicuous morphological feature of ccRCC is glycogen and lipid-rich cytoplasmic deposits [77]. Reprogramming of glucose and fatty acid metabolism is a key factor for carcinogenesis and progression of ccRCC [77, 78, 79, 80]. Insulin-like growth factor receptor1 (IGF1R) pathway has been shown to play an important role in the accumulation of glycogen and lipids and tumor development in ccRCC [81, 82]. Further, the most common molecular features of ccRCC are changes in the VHL tumor suppressor, a molecule that has been shown to inhibit IGF1R cell signaling. Thus, in ccRCC, the IGF1R signaling pathway is chronically activated, resulting in increased cancer growth, progression, and chemoresistance [77, 83, 84, 85]. IGF1R is endocytosed after ligand binding, a process mediated by the endocytosis machinery in which dynamin plays an important role [86, 87, 88]. When IGF1R receptors are activated, two signal-transduction pathways, Ras-Raf-MEK-ERK and PI3K – Akt are involved [85, 89, 90]. In Ras-Raf-MEK-ERK pathway, activated MEK in the presence of DNM2 could phosphorylate the MAPK molecule, a process that is inhibited in dynamin mutants. These pathway activate gene expression causing cell proliferation [35]. Translocation of GLUT4 via a PI3K – Akt dependent pathway activates glycogen synthase and DNM2 plays important functional role in the endocytosis of glut4 [91]. All these highlight the importance of the DNM2 molecule in internalization and the initiation of the IGF1R pathway ultimately leading to the accumulation of glycogen, lipids, and proliferation. It seems an increase in DNM2 in the cell cytoplasm might indicate increased IGF1R signaling pathway activation and ccRCC progression.
In general, our findings revealed the importance of DNM2 in tumor cell migration and cancer progression, and introduce it as a candidate for targeted cancer therapy. Indeed, a study has shown that the DNM2 inhibitors reduce migration and invasion by reducing the expression of matrix metallopeptidase 9, resulting in a slower rate of cell proliferation [45, 46]. Pharmacological inhibitor of DNM2 results in reduction of the tumor volume and stable expression of DNM2-siRNA, causing diminished tumor weight and reduced number of lymph node metastases in prostate cancer and glioblastoma models [19, 23, 92]. In this regard, DNM2 inhibitor increased survival of mice in leukemia [93]. Aside from being a therapeutic target in several cancers, other studies [19, 53, 73] and our research have indicated that DNM2 may potentially be a prognostic factor. Indeed, DNM2, which presents a different expression pattern and levels in histological subtypes of RCC, could be a specific prognostic biomarker in ccRCC.
In this study, it was shown that increased expression of DNM2 in patients with ccRCC is associated with disease progression as well as poor prognosis. However, one of the limitations of this study is the lack of in vivo experiments to investigate the role of DNM2 in ccRCC.
Conclusion
In summary, our data revealed the potential role of DNM2 overexpression in predicting worse clinical outcomes of RCC patients. In silico analysis illustrated increased DNM2 protein in ccRCC tissues. Especially, the experimental results indicated a statistically significant difference between DNM2 cytoplasmic expression in RCC subtypes. These results suggest that increased cytoplasmic expression of DNM2, compared to nuclear and membranous expression, plays a key role in tumor aggressiveness and progression of ccRCC. Higher cytoplasmic expression of DNM2 in ccRCC is associated with more malignancy, disease progression, as well as poor DSS and PFS in ccRCC. According to the results, DNM2 can be used as a specific prognostic marker in patients with ccRCC and a therapeutic target.
Author contributions
Preparation of the manuscript: S.S, R.S,L.SZ.
Interpretation or analysis of data: S.S, L.SZ, M.R, F.F, G.KS.
Revision for important intellectual content: M. Ra, S.D, L.E.
Conception, supervision, preparation of the manuscript: R.G, Z.M, L.SZ.
Funding
This work was supported by the grant from Iran University of Medical Sciences (Grant Number: 98-4-28-17395).
Data availability
The analyzed data during the current study are available from the corresponding author on reasonable request.
Ethical approval
All procedures performed in this study were in line with the ethical standards of the institution at which this study was conducted. Informed consent was individually obtained from all participants. The Research Ethics Committee of Iran University of Medical Sciences issued IR.IUMS.REC.1398.1354. for this study.
Informed consent
Informed consent was obtained from all individual participants, parents or legally authorized representatives of participants under legal age years old at the time of sample collection with routine consent forms.
Footnotes
Acknowledgments
The authors would like to thank the Iran University of Medical Sciences for supporting this research.
Conflict of interest
The authors declare no conflict of interests.
