Abstract
METHODS:
Talin-1 protein was demonstrated as a potential prognostic marker in renal cell carcinoma (RCC) using bioinformatics analysis. We, therefore, examined the protein expression levels and prognostic significance of Talin-1 with a clinical follow-up in a total of 269 tissue specimens from three important subtypes of RCC and 30 adjacent normal samples using immunohistochemistry. Then, we used combined analysis with B7-H3 to investigate higher prognostic values.
RESULTS:
The results showed that high membranous and cytoplasmic expression of Talin-1 was significantly associated with advanced nucleolar grade, microvascular invasion, histological tumor necrosis, and invasion to Gerota’s fascia in clear cell RCC (ccRCC). In addition, high membranous and cytoplasmic expression of Talin-1 was found to be associated with significantly poorer disease-specific survival (DSS) and progression-free survival (PFS). Moreover, increased cytoplasmic expression of Talin-1
CONCLUSIONS:
Collectively, these observations indicate that Talin-1 is an important molecule involved in the spread and progression of ccRCC when expressed particularly in the cytoplasm and may serve as a novel prognostic biomarker in this subtype. Furthermore, a combined analysis of Talin-1/B7-H3 indicated an effective biomarker to predict the progression of disease and prognosis in ccRCC.

Introduction
Renal cell carcinoma (RCC) is the most common type of kidney cancer found in adults. It represents approximately 3% of adult malignancies and 90–95% of all neoplasms arising from the kidney [1, 2]. The RCC incidence rates have been on the rise during the last decade [3]. According to the American Cancer Society, an estimated 79,000 new cases (50,290 in males and 28,710 in females) and 13,920 deaths (8,960 in males and 4,960 in females) from RCC will occur in 2022 [4]. If cancer does not metastasize, the 5-year survival rate is 65–90%, but this is lowered considerably when it spreads so that the 5-year survival rates for patients with stage IV disease are less than 20% [5]. Unfortunately, approximately 20–30% of RCC patients have distant metastasis at the initial diagnosis. In addition, about 25–40% of RCCs relapse and ultimately develop into metastatic disease after resection of the primary tumor [6]. Moreover, increasing resistance to targeted therapies has occurred in advanced RCC patients [7]. Therefore, prognostic and predictive biomarkers will be an urgent requirement to assist in early diagnosis and identify novel therapeutic targets for RCC.
To date, several subtypes of RCC have been defined, of which clear cell RCC (ccRCC) is the most common subtype (75–80%), followed by papillary RCC (pRCC) 15%, and chromophobe RCC (chRCC) around 5% [8]. In this study, we have focused on the three major histological subtypes of RCC.
High throughput proteomics was introduced as an innovative novel approach that paves the way for cancer diagnostic or prognostic biomarker discovery [9]. These studies can identify effective proteins with alternative expression and cellular functions in the samples with various diseases [10]. Although a high level of variance exists in proteomics data, network and enrichment analysis explore the hub genes and their related biological significance among results [11]. In the present study, protein-protein interaction (PPI) network analysis was performed for proteomics data of the research that has reported proteins with significant differential expression in 3 major histological subtypes of RCC tissue samples compared to adjacent normal tissues [12, 13, 14]. Enrichment analysis for proteins with the highest confidence in the PPI network was carried out for a better understanding of the genes effective in the RCC network. It was found that TLN1 (Talin-1) as a key gene could act as a prognostic marker in three important histological subtypes of RCC.
Talin-1 is a ubiquitous cytoskeleton-associated protein with a high-molecular weight which has been shown to play an essential role in regulating the activity of the integrin family of cell adhesion proteins using coupling integrins to F-actin [15, 16]. Also, Talin-1 is responsible for binding to critical adhesion molecules, including integrins, vinculin, actin, and other focal adhesion kinases (FAKs) and influences cell cytoskeletal remodeling. Therefore, increased expression of Talin-1 could lead to the development of tumors and migration [17, 18]. A number of studies have demonstrated that overexpression of Talin-1 was associated with advanced disease and reduced overall survival (OS) in patients, including oral squamous cell carcinoma (OSCC) [19], prostate cancer [20], colon cancer [21], and nasopharyngeal carcinoma (NPC) [22]. Fong et al. showed both Talin-1 and Talin-2 levels correlate with tumorigenicity in human hepatocellular carcinoma (HCC), indicating that these molecules constitute important molecular targets for the diagnosis and/or treatment of HCC [23]. To date, the expression levels and clinical significance of Talin-1 in the RCC tumors remain unclear. Thus, in the current study, we have investigated the membranous, cytoplasmic, and nuclear expression levels of Talin-1 protein in three important subtypes of RCC from formalin-fixed paraffin-embedded (FFPE) tissue samples assembled on tissue microarrays (TMAs) using the immunohistochemistry (IHC) technique. Consequently, the Talin-1 protein obtained from the network analysis in proteomics studies in RCC samples can be confirmed and validated. Then, we sought to determine the association between expression levels of Talin-1 protein and clinicopathological parameters, including nucleolar grade, tumor stage, microvascular invasion, and other clinicopathological characteristics of RCC as well as survival outcomes. Finally, we carried out a combined analysis with another marker of our previous study, namely B7-H3 [24], which predicts progression and metastasis in ccRCC. B7-H3 is a member of the B7 family of immunoregulatory proteins which may be a promising target for cancer treatment. It was first identified as a co-stimulatory molecule for T cell activation, but recent studies demonstrate B7-H3 as a T cell inhibitor that promotes tumor aggressiveness and proliferation [25]. Numerous studies indicated that B7-H3 was overexpressed in many types of malignancies and was linked to poor prognosis, increased tumor grade, and metastasis [26, 27]. Our findings from the previous study have shown that B7-H3 is a useful marker for predicting progression and metastasis in ccRCC [24]. Considering the role of Talin-1 in metastasis [18, 19], for the first time, we aimed to investigate a combined analysis of Talin-1/B7-H3 markers for further research if the combined analysis of two markers could increase prognostication in ccRCC.
Materials and methods
Data source and network analysis
A search on the PRoteomics IDEntifications database (PRIDE) [28] from 2019 to 2020 led to the identification of three important subtypes of RCC studies each including proteomics information and adjacent normal tissues. The results of proteomics analysis were downloaded from the supplementary information of their published articles [12, 13, 14]. Venn diagram analysis was performed to find common significant differential proteins in three main subtypes of RCC by OriginPro 2019 (OriginLab Corporation, Northampton, MA, USA). PPI network was constructed using SrtringApp [29] based on STRING database with medium confidence
Study population
Two hundred and ninety-five FFPE tissues from RCC tumor specimens that underwent radical nephrectomy were collected from a major referral university-based urology-nephrology center, the Hasheminejad Hospital, in Tehran, Iran, during 2008–2015. It was confirmed that none of the patients had received radiotherapy or chemotherapy. Three important subtypes of RCC were comprised in this study including ccRCC, pRCC (type I & II), and chRCC. Hematoxylin and eosin (H&E) stained pathology slides and clinical and pathological information were obtained from medical archival records. The following variables included age, gender, the maximum diameter of tumor size, nucleolar grade, tumor stage, MVI, lymph node invasion (LNI), renal vein invasion, histological TN, renal sinus fat, and renal sinus invasion, perirenal fat invasion, Gerota’s fascia invasion, distant metastasis, and tumor recurrence.
Clinical follow-up was available for all patients until July 2015. The information of DSS was defined as the time from the first operation to death due to renal cancer. PFS was defined as the interval between the primary operation and the last follow-up visit without any disease, evidence of tumor recurrence or metastasis. In addition, tumor stage and nucleolar grade were defined according to the pTNM classification for RCC [39, 40, 41]. Moreover, it was agreed that chRCC should not be graded [40]. This study was approved by the Iran University of Medical Sciences Human Research Ethics Committee in Iran (Ref no: IR.IUMS.REC.1398.938). All procedures performed in this study were in accordance with the 1964 Helsinki Declaration and its later amendments. Informed consent was obtained from all individual participants included in the study at the time of sample collection with routine consent forms.
Tissue microarray (TMA) construction
The tissue samples were brought into a TMA format as follows. Briefly, H & E slides were examined and the most representative areas of the tumor were marked by pathologists. A total of 0.6-mm tissue cylinders were punched out from the selected regions of each donor tumor tissue block and transferred into a recipient paraffin block using the tissue arrayer MiniCore (ALPHELYS, Plaisir, France). The TMA blocks were constructed in triplicate for each specimen. The mean of the three scores was calculated as the final score. Previous studies have shown that three cores are highly representative of the whole section [42, 43]. Finally, 4-
Immunohistochemistry (IHC) for Talin-1 protein expression
IHC was applied to detect the expression of Talin-1 protein. Briefly, all TMA sections were deparaffinized at 60
Immunohistochemistry evaluation
The evaluation procedure of the IHC was reviewed by one independent pathologist (M.A) without being informed of clinical data. The intensity and percentage of the staining area were considered two important components of the scoring system for Talin-1 protein expression. The intensity of staining was scored by applying a semi-quantitative system, ranging from negative to strong as follows: 0 (no staining), 1 (weak), 2 (moderate), and 3 (strong). The percentage of staining area was categorized according to the positive tumor cells, less than 25% positive cells, 25–50% positive cells, 51–75% positive cells, and more than 75% positive cells. The final Talin-1 protein expression score was calculated by multiplying the value of the intensity score by the percentage of positive tumor cells which ranged from 0 to 300, namely the histochemical score (H-score). In this study, the median H-score was chosen to categorize samples with high or low Talin-1 protein expression.
Co-expression of Talin-1/B7-H3 markers in ccRCC
In this study, we used the set of RCCs TMAs that were utilized in our previous study. Our results from the previous study have shown that B7-H3 is a useful marker for predicting progression and metastasis in ccRCC [24]. Considering the role of Talin-1 in metastasis [18, 19], we aimed to investigate the combined analysis of Talin-1/B7-H3 for further investigations if this combined analysis of two markers could increase prognostication in ccRCC. We first considered the samples which have been stained by two markers (previous study; B7-H3 and current study; Talin-1) in patients with ccRCC. Then common tissue samples which have been stained with two markers were selected for this study. The number of common tissue samples was 138. Then, Spearman correlation test was applied to determine the significant correlation between expression of cytoplasmic and membranous B7-H3 and Talin-1 proteins. Finally, the association between co-expression of Talin-1/B7-H3 markers and clinicopathological parameters as well as survival outcomes were assessed.
Statistical analysis
Analyses were performed using the “statistical software SPSS, version 22.0. Armonk, NY: IBM Corp”. To verify the significance of the association and correlation between Talin-1 protein expression and clinicopathological parameters, Pearson’s chi-square and Spearman’s correlation tests were performed. Kruskal-Wallis and Mann-Whitney
(A) Venn diagram represents the overlaps of differential protein expressions between three important subtypes of renal cell carcinoma (RCC). (B) Protein-protein interaction network (PPI) of 110 common differential protein expressions in three major RCC subtypes. (A) One hundred and ten common differential protein expressions were obtained based on proteomics data from previous articles, including PMID: 30838877, PMID: 31484429, and PMID: 32694149 for clear cell RCC (ccRCC), papillary RCC (pRCC), and chromophobe RCC (chRCC), respectively. (B) PPI network was obtained by stringApp in Cytoscape. Bold nodes in the network reflect the proteins with the highest degree of connectivity (highest confidence 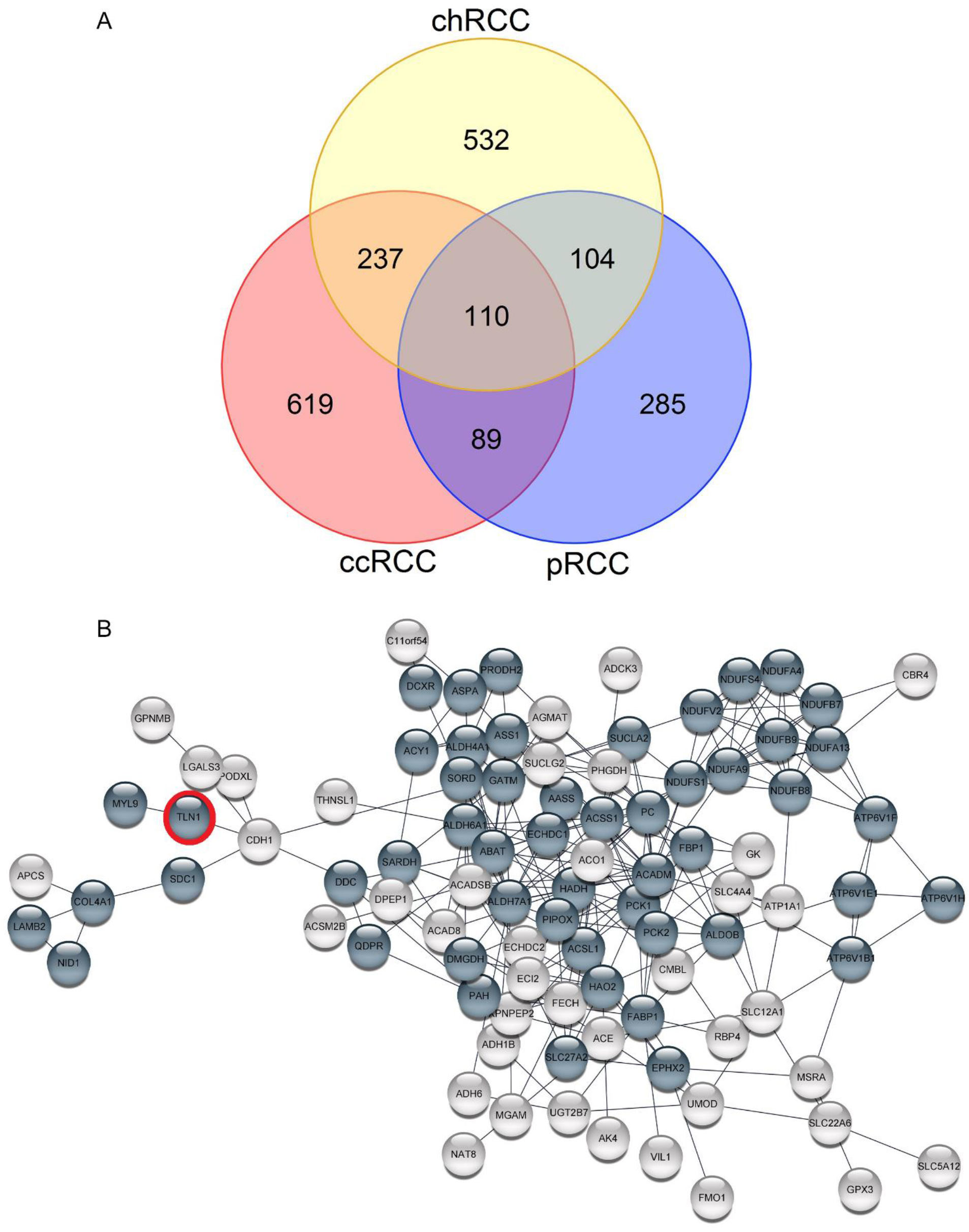
Bioinformatics approach
The significance of differential expression of proteins from tumor tissues in comparison to adjacent normal tissues was obtained from selected previous studies that include 1055, 589, and 983 proteins for ccRCC, pRCC, and chRCC, respectively (Supplementary Table 1). The results of the Venn diagram analysis revealed 110 common differential protein expressions between 3 important subtypes of RCC (Fig. 1A). The PPI network analysis led to explore 52 proteins with the highest degree of connectivity (highest confidence
Pathway analysis and crosslink figure of pathways using ClueGO for key genes. Pathway analysis indicated that several genes such as TLN1 (Talin-1) are interconnected to focal adhesion (KA) and extracellular matrix (ECM) pathways. The genes are collected together in close pathways.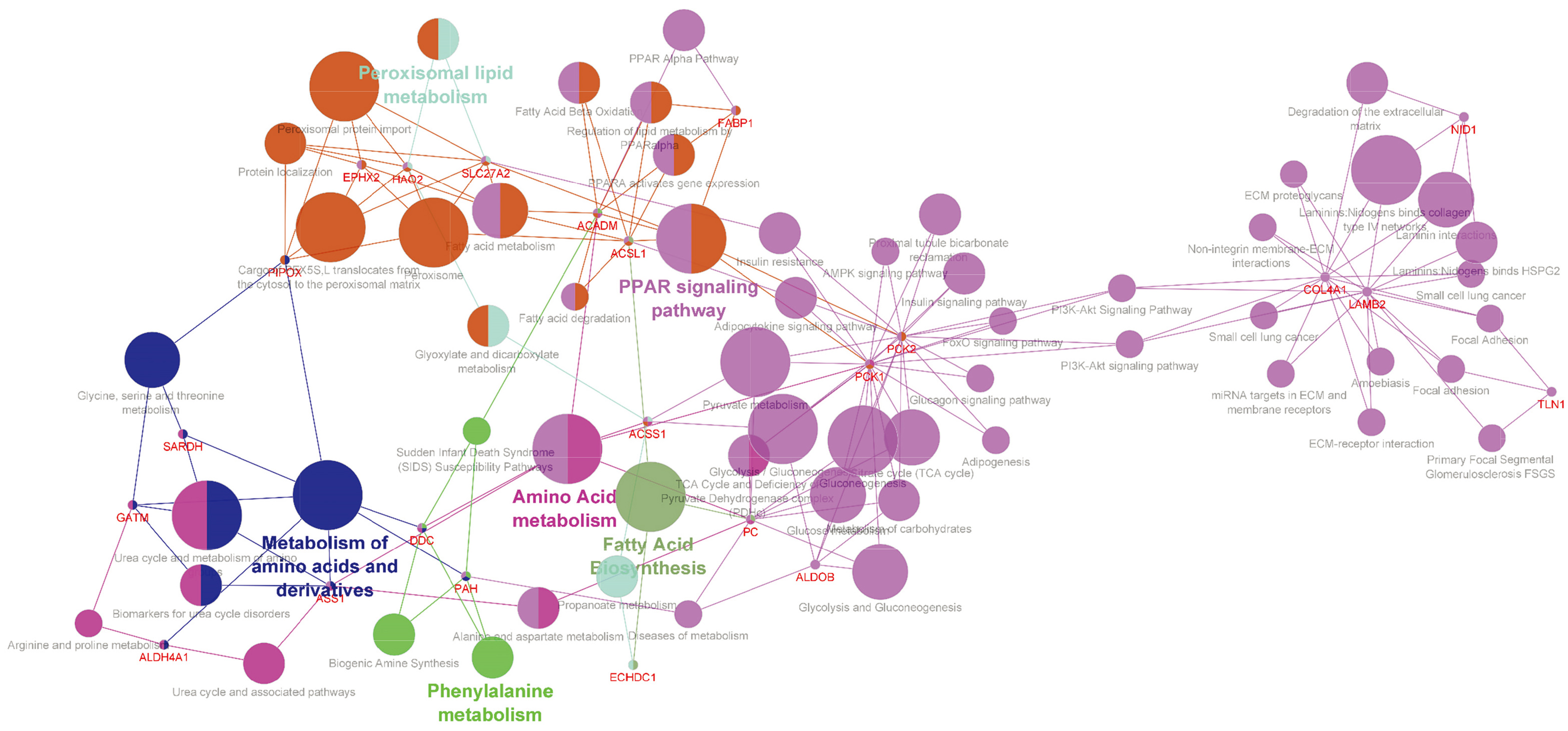
that these genes are present in the microbody and play roles in fatty acid catabolic process. Also, the basement membrane organization is a remarkable part of gene annotation results (Supplementary Fig. 1). Talin-1 was selected as our promising molecular candidate after analysis of the list including collagen type IV alpha 1 (COL4A1), laminin-
Two hundred and ninety-five RCC patients were included in this study. Since technical problems led to a loss of some cases, 269 cases remained for evaluation. These samples include 195 (73.58%) ccRCC, 20 (7.43%) pRCC type I, 20 (7.43%) pRCC type II, and 34 (12.63%) chRCCs. The patients’ clinicopathological characteristics were summarized in Table 1 based on the whole of RCC samples and histological subtypes.
Immunohistochemical analysis of Talin-1 protein expression in three subtypes of renal cell carcinoma (RCC) tissues. Talin-1 protein expression in clear cell renal cell carcinoma (ccRCC): low membranous and cytoplasmic expression (A, A-1), high membranous and cytoplasmic expression (B, B-1), low nuclear expression (C, C-1), and high nuclear expression (D, D-1). Talin-1 protein expression in papillary RCC (pRCC): low expression (E, E-1) and high expression (F, F-1). In chromophobe RCC (chRCC): low expression (G, G-1) and high expression (H, H-1). IHC staining of adjacent normal tissue (I), and human normal kidney as (J) positive and (K) negative controls. Isotype controls in immunohistochemistry staining in ccRCC (L), pRCC (M), and chRCC (N). Figures are shown with a magnification of 100 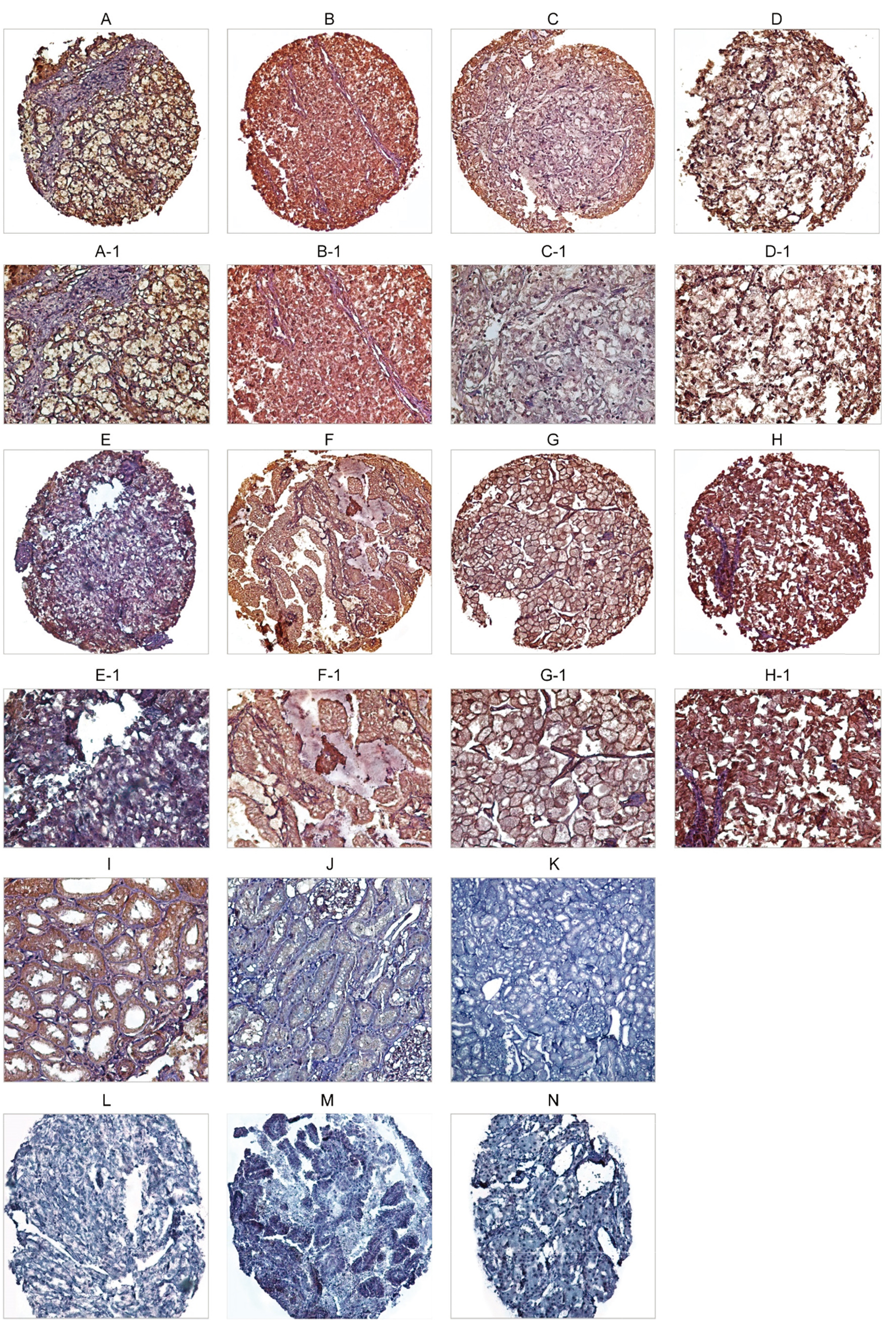
Box plot analysis of expression levels of Talin-1 protein in various subtypes of renal cell carcinoma (RCC), nuclear grade (I 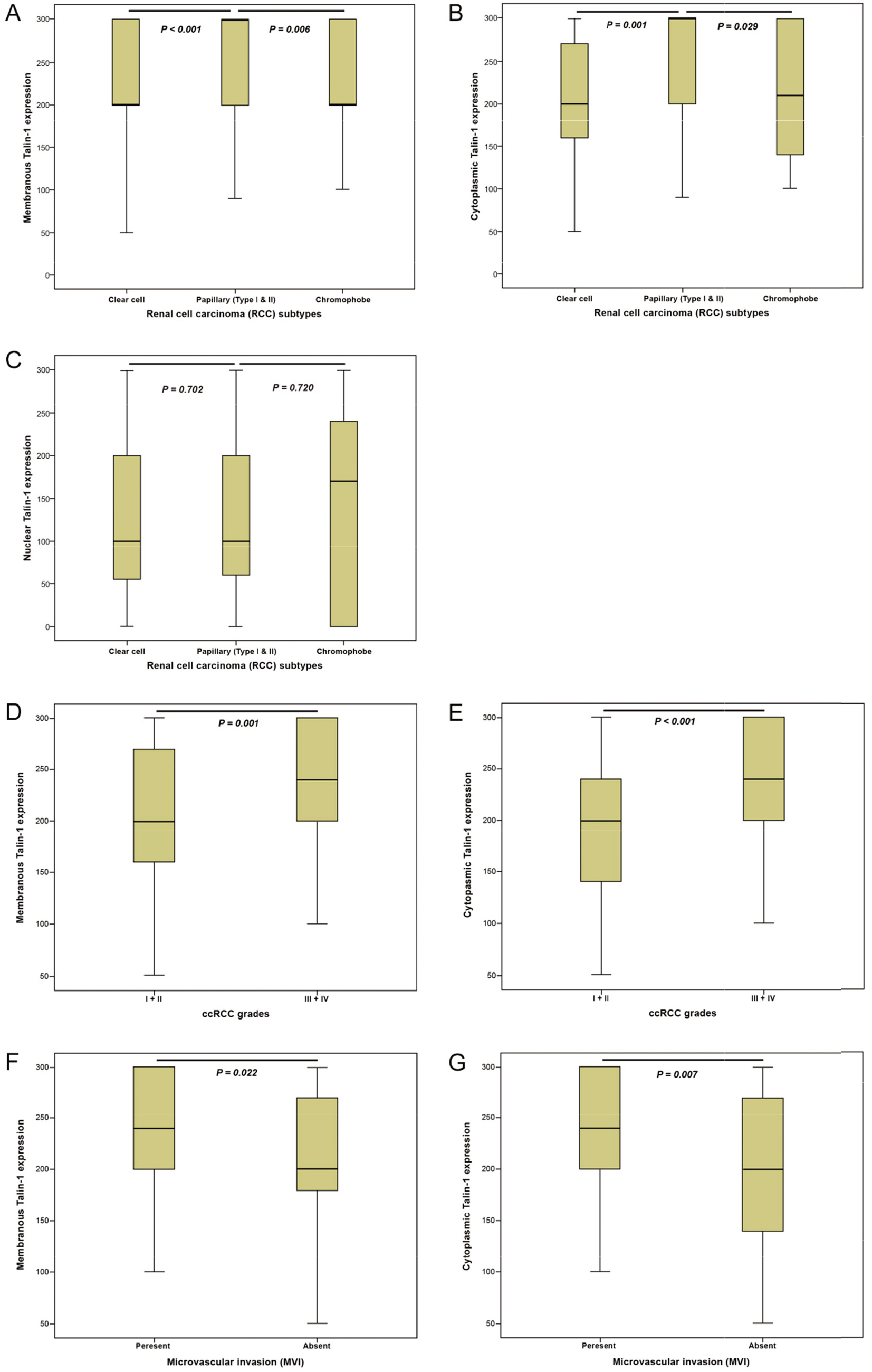
Patients and tumor pathological characteristics of histological subtypes of renal cell carcinoma (RCC)
ccRCC; clear cell renal cell carcinoma, pRCC; papillary renal cell carcinoma, chRCC; chromophobe renal cell carcinoma.
Membranous, cytoplasmic, and nuclear Talin-1 protein expression (Intensity of staining, percentage of positive tumor cells, and H-score) in histological subtypes of RCC
H-score; histological score. RCC; renal cell carcinoma. ccRCC; clear cell renal cell carcinoma, pRCC; papillary renal cell carcinoma, chRCC; chromophobe renal cell carcinoma.
The expression level of Talin-1 protein was evaluated through the IHC method on TMA sections by three scoring methods, including the intensity of staining, percentage of positive tumor cells, and H-score in histological subtypes of RCC and adjacent normal samples. Talin-1 protein was expressed at different intensities in the cell membrane, cytoplasm, and nucleus in the samples (Table 2) (Fig. 3), but the expression of Talin-1 protein in adjacent normal tissue samples was less compared to tumor samples.
Comparison of Talin-1 expression based on RCC histological subtypes
The results of the Kruskal-Wallis test revealed a statistically significant difference between the membranous and cytoplasmic Talin-1 protein expression levels in different RCC subtypes (
Associations between membranous, cytoplasmic, and nuclear Talin-1 expression and clinicopathological parameters
ccRCC
The results of Pearson’s chi-square test showed a highly significant association between increased membranous and cytoplasmic expression of Talin-1 protein and advanced nucleolar grades (I
The association between Talin-1 protein expression and clinicopathological parameters of clear cell renal cell carcinoma (ccRCC)
The association between Talin-1 protein expression and clinicopathological parameters of clear cell renal cell carcinoma (ccRCC)
Kaplan-Meier curves for disease-specific survival (DSS) and progression-free survival (PFS) based on membranous, cytoplasmic, and nuclear Talin-1 protein expression levels in clear cell renal cell carcinoma (ccRCC). (A) In ccRCC, the results showed there were significant differences between DSS and the patients with high and low membranous expression of Talin-1 protein (Log-rank test; 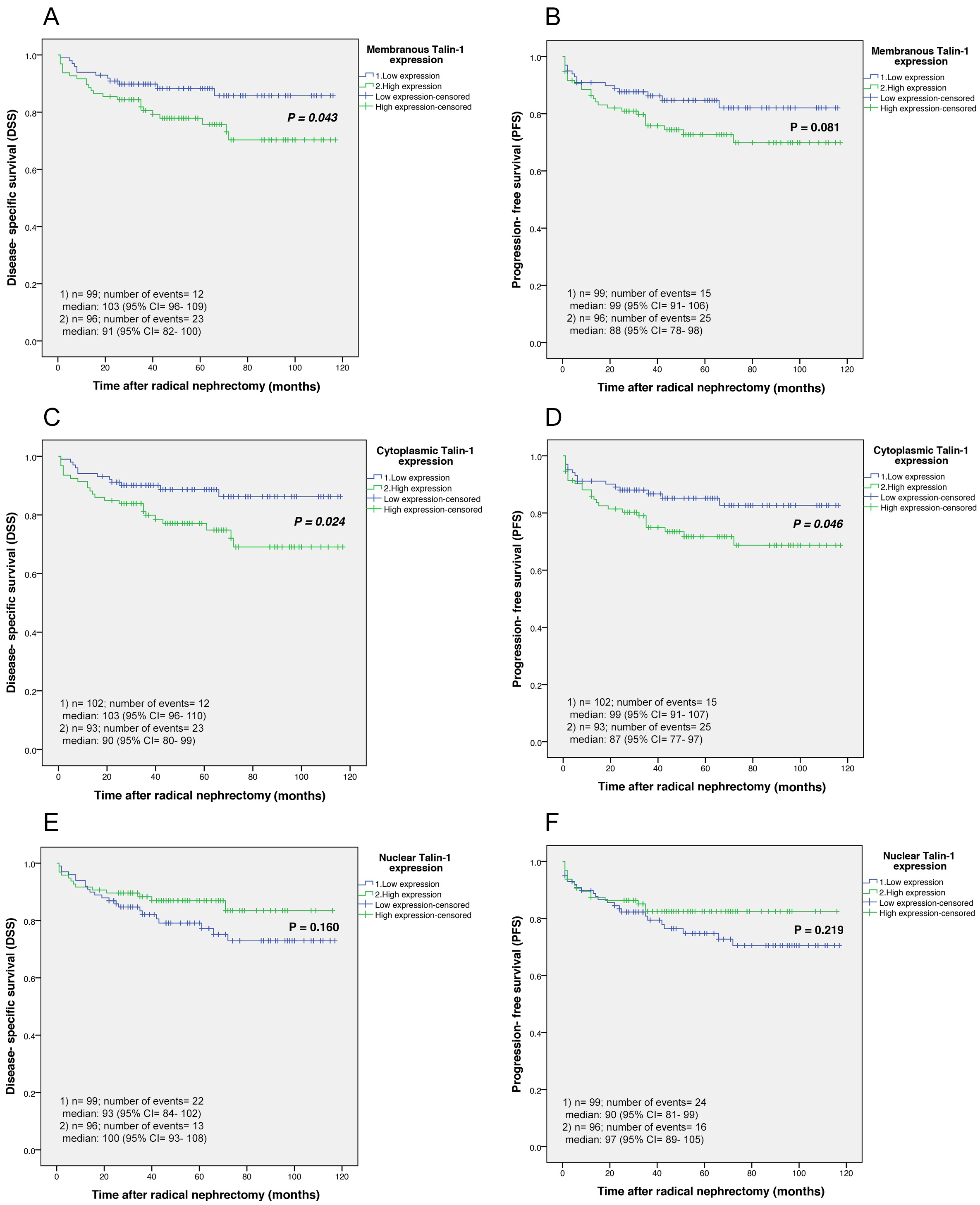
Univariate and multivariate cox regression analyses of potential prognostic factors for disease-specific survival (DSS) in patients with clear cell RCC (ccRCC)
HR; hazard ratio, CI; confidence interval. Values in bold are statistically significant.
The results of Pearson’s chi-square test showed no significant association between Talin-1 protein expression and clinicopathological parameters in pRCC (type I & II) (Supplementary Tables 3 and 4). In chRCC, we observed a statistically significant association between increased membranous expression of Talin-1 and advanced tumor stage (
Prognostic value of expression of Talin-1 for clinical outcomes in RCC subtypes
In the current study, the mean and median duration of the follow-up time for DSS were 52 (SD
Survival outcomes based on the expression of Talin-1 in RCCs
ccRCC
3.7.1.1. Survival outcomes based on membranous Talin-1 expression
According to the results of the Kaplan-Meier curve, significant differences were found between disease-specific survival (DSS) and the patients with high and low membranous expression of Talin-1 protein (Log-rank test:
3.7.1.2. Survival outcomes based on cytoplasmic Talin-1 expression
Our findings of Kaplan-Meier curves analysis in the cytoplasmic expression of Talin-1 revealed significant differences between DSS (Log-rank test:
3.7.1.3. Survival outcomes based on nuclear Talin-1 expression
Kaplan Meier survival curves showed no significant differences between DSS or PFS and the patients with high and low nuclear expression of Talin-1 protein in ccRCC cases (Log-rank test:
Univariate and multivariate cox regression analyses of potential prognostic factors for progression-free survival (PFS) in patients with clear cell RCC (ccRCC)
Univariate and multivariate cox regression analyses of potential prognostic factors for progression-free survival (PFS) in patients with clear cell RCC (ccRCC)
HR; hazard ratio, CI; confidence interval. Values in bold are statistically significant.
The association between cytoplasmic Talin-1/B7-H3 phenotypes and clinicopathological parameters of clear cell renal cell carcinoma (ccRCC)
The results from the univariate and multivariate analysis were summarized in Tables 4 and 5. As demonstrated in Tables 4 and 5, the nucleolar grade was the only independent prognostic factor for DSS or PFS in the multivariate analysis. Membranous and cytoplasmic expression of Talin-1 were not significant risk factors for prognosis in the multivariate analysis.
Based on Kaplan-Meier survival analysis, no statistically significant association was found between membranous, cytoplasmic, and nuclear Talin-1 protein expression and patients’ survival outcomes in pRCC (type I & II) and chRCC patients. Additionally, univariate and multivariate analyses demonstrated that the listed clinicopathologic variables are not significant factors affecting the DSS or PFS of patients with papillary and chRCC.
Co-expression of Talin-1/B7-H3 markers with clinicopathological parameters in ccRCC
The results showed a statistically significant correlation between the cytoplasmic expression of Talin-1 protein and cytoplasmic B7-H3 protein expression (Spearman’s rho, 0.318;
Co-expression of Talin-1/B7-H3 markers with survival outcomes in ccRCC
The survival rate of patients with the co-expression of cytoplasmic Talin-1/B7-H3 indicated higher expression of Talin-1
Univariate and multivariate cox regression analyses of potential prognostic factors of Talin-1/B7-H3 for disease- specific survival (DSS) and progression-free survival (PFS) in patients with clear cell RCC (ccRCC)
Univariate and multivariate cox regression analyses of potential prognostic factors of Talin-1/B7-H3 for disease- specific survival (DSS) and progression-free survival (PFS) in patients with clear cell RCC (ccRCC)
HR; hazard ratio, CI; confidence intervalValues in bold are statistically significant.
shorter DSS in comparison to other phenotypes (
Kaplan-Meier curves for disease-specific survival (DSS) and progression-free survival (PFS) based on co-expression of Talin-1/B7-H3 and stratified analysis curves of DSS for patients with distant metastasis (positive and negative) in clear cell renal cell carcinoma (ccRCC). (A) The results showed that there were significant differences between DSS (Log-rank test; 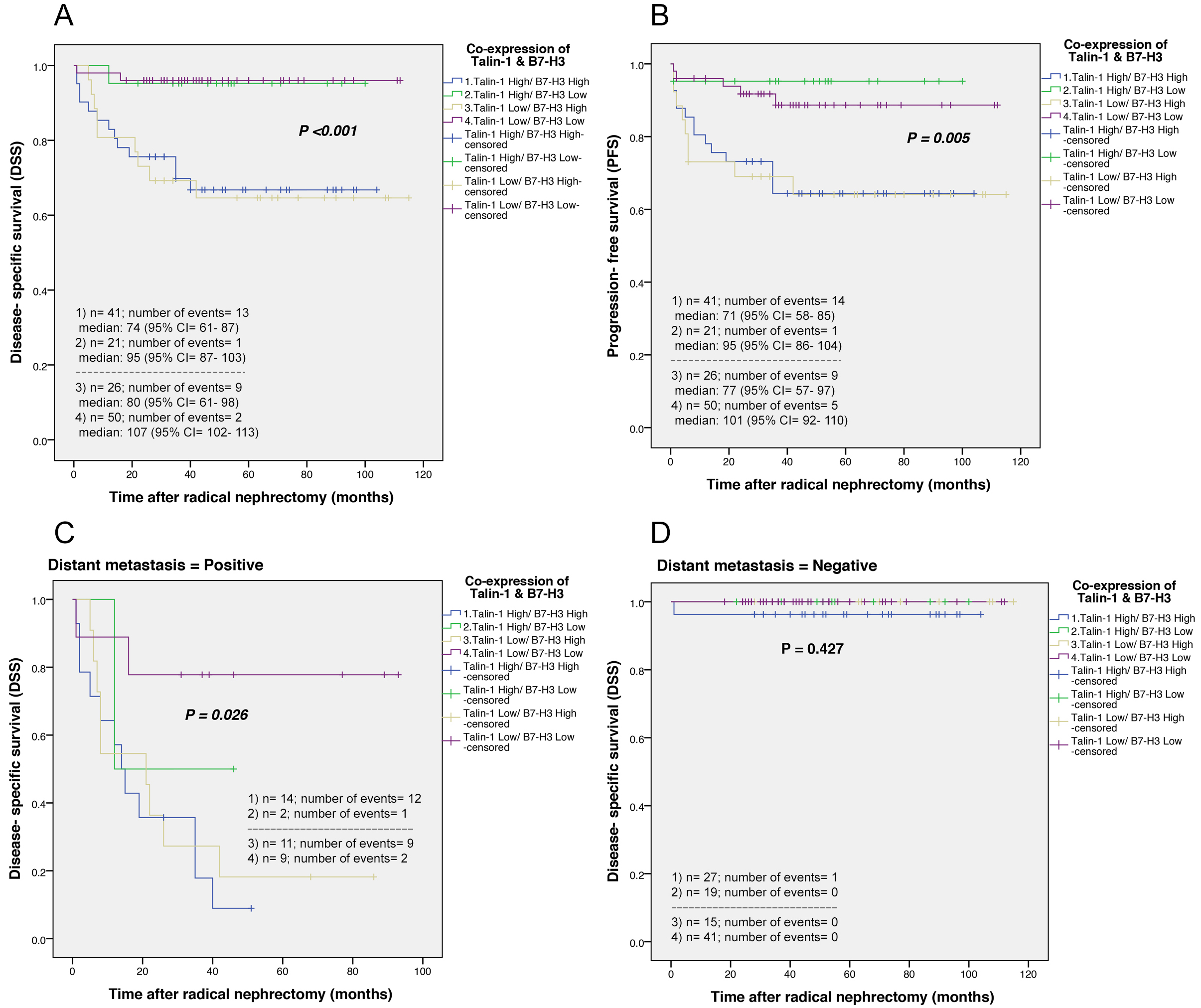
Despite new improvements in the treatment of RCC, drug resistance to targeted therapies has caused more failure in advanced RCC patients’ treatment [7]. Thus, it is urgently needed to explore novel and practical clinical prognostic markers of RCC in the patient’s early diagnosis in future renal cancer therapy.
The bioinformatics techniques might be helpful in identifying molecular markers from omics data to use in clinical RCC research. In this regard, we focused on PPI network analysis to obtain common differentially expressed proteins from other studies related to 3 major subtypes of RCC [12, 13, 14]. The proteins with the highest degree of connectivity (highest confidence) based on PPI network analysis were screened on Enrichr online tool for cancer. Then enrichment analysis was performed for determining the pathways of the screened genes involved. These analyses indicated several proteins with the highest confidence in PPI network related to cancer. Among them, we selected Talin-1 which warranted further investigation. Therefore, to validate the Talin-1 protein as a prognostic marker for RCC, for the first time, expression levels of Talin-1 protein were investigated in a well-characterized series of 269 tissues specimen from patients treated with radical nephrectomy in three main subtypes of RCC including ccRCC, pRCC type I & II, and chRCC.
The evaluation of the staining pattern in each subtype of RCC displayed differential expression of Talin-1 protein in the membrane, cytoplasm, and nucleus with a range of intensities from weak to strong. Furthermore, there was a statistically significant association between the membranous and cytoplasmic expression of Talin-1 protein but not in nuclear expression and the RCC subtypes. The results indicated that the expression of Talin-1 protein in the membrane and cytoplasm versus the nucleus is more important in subtypes of RCC. It also showed that these expression patterns vary among the histological RCC subtypes, thus this may affect their prognostic value and treatment options.
IHC analysis of human RCCs compared to adjacent normal tissue samples demonstrated that Talin-1 protein expression is upregulated in RCCs. The present study also considered the expression of Talin-1 on UALCAN database, containing a large amount of proteomics data which showed increased expression of Talin-1 in ccRCCs rather than in normal samples. Thus, these results are in line with our findings of Talin-1 protein expression using IHC on ccRCCs.
Our finding of analyzing the subcellular distribution of Talin-1 in RCCs showed that membranous and cytoplasmic expression of Talin-1 protein is positively associated with major clinicopathological parameters, including advanced nucleolar grade, MVI, histological TN, invasion to Gerota’s fascia, and renal pelvis, while no association was found in nuclear expression. Importantly, our results revealed higher expression of the median membranous and cytoplasmic of Talin-1 protein in the high grade of RCC compared with the low grade of RCC, and increased expression in patients with MVI present rather than MVI absent. The data showed the association of membranous and cytoplasmic Talin-1 protein expression with the aggressiveness of ccRCC. In addition, in this study, the nucleolar grade was found as an independent prognostic factor for DSS and PFS in membranous and cytoplasmic Talin-1 protein expression. After the tumor stage, one of the strongest prognosticators of survival in patients with RCC is the nucleolar grade, and tumors with a high nucleolar grade display a more aggressive phenotype; thus, they are associated with local invasion and distant metastasis [53]. Previous studies demonstrated that MVI is related to cancer progression and survival in RCC and is the most significant prognostic variable for the prognostication of metastatic spread and survival outcome independent of macrovascular invasion [54, 55]. Histological TN has also been proposed to be a sign of tumor aggressiveness that generally leads to poor survival outcomes [56]. A recently systematic review and meta-analysis study suggested that histological TN is associated with DSS, OS, RFS (recurrence-free survival), and PFS of RCC patients and may serve as a predictor of poor prognosis in RCC patients [57]. Moreover, in this study, tumor size and tumor stage were found as prognostic variables in univariate analysis that depicted the associations between these parameters and more aggressive tumor behaviors. The tumor stage is a major factor for predicting tumor progression and recurrence in RCC [58], and tumor size was shown to be significantly associated with the risk of metastasis [59]. Therefore, these results indicated that membranous and cytoplasmic Talin-1 protein expression compared with nuclear expression are related to the degree of malignancy and progression of disease in ccRCCs. The main location for activation of Talin-1 has been shown in the cytoplasm which is an indispensable component of the integrin-cytoskeleton link [60]. Besides, the role of Talin-1 in the cancer cell nucleus must be determined accurately. Our findings demonstrated that cytoplasmic and membranous Talin-1 protein expression is associated with more aggressive behavior which added prognostic value in ccRCC. The biological function of Talin-1 protein, based on its site of expression as well as subcellular distribution can be changed [61]. Therefore, further studies are required to understand the Talin-1 protein and underlying mechanisms in the different organelle of cells.
Our results from Kaplan-Meier survival curves and 5-year survival rate in ccRCC cases showed that higher membranous and cytoplasmic Talin-1 protein expression is associated with significantly worsened DSS as well as worsened DSS or PFS compared to low Talin-1 protein expression, respectively. However, the pattern of Talin-1 protein expression was not a predictor of survival in multivariate analysis.
The results of co-expression of cytoplasmic Talin-1
Talin-1 binds to adhesion molecules, consists of integrins, vinculin, and actin as well, [18] and might be important for the regulation of FAK by integrins activation and crosstalk [49, 62]. FAK as a part of focal adhesion signaling provides scaffolding functions at sites of integrin adhesion that is crucial in cell migration control [63]. FAK which interacts with Src and is related to Src signaling and downstream of cell-ECM interactions can regulate cell-cell adhesions. Also, the FAK can affect PI3K-AKT signaling [49, 62, 64], and activated FAK can promote processes, including cancer cell survival, growth, angiogenesis, migration, and invasion. Inhibition of FAK function can decrease RCC metastasis both in in-vitro and in-vivo [65, 66, 67] because it potentially regulates gene expression to affect cancer progression [68]. The intracellular domain of the integrin
However, Talin-1 has a different expression in other cancers such as HCC. A previous study by Kanamori et al. [16] showed high-level expression of Talin-1 in HCC tissues while another study demonstrated that Talin-1 is downregulated in HCC [72]. Others demonstrated significantly higher expression levels of Talin-1 in serum [73]. In addition, Talin was found to be completely absent in endometriosis and endometrioid carcinomas [74]. Therefore, it seems that expression levels of Talin-1 are different in various cancers, and more investigations are needed to explore the exact mechanism and function of Talin-1 to find new ways for therapeutic targeting.
Previous studies indicated that Talin affects chemoresistance such as cisplatin, a common chemotherapy drug, which can be considered a failure in cancer treatments [75]. In oral carcinoma, Talin regulates nuclear NF-kB activity, and resistance to cisplatin can lead to proliferation induction. In other words, overexpression of Talin is sufficient to induce nuclear NF-kB activity which is dependent on focal adhesion kinase (FAK) [76]. Singel et al. showed that Talin-1 plays a role in response to cytotoxic chemotherapy, and decreased expression of Talin-1 can be a prognostic marker for a better outcome after cytotoxic chemotherapy. Therefore, low-level expression of Talin-1 in a variety of breast cancer cell lines is associated with higher chemosensitivity to docetaxel in triple-negative breast cancer treatment [77]. To sum up, Talin-1 can be introduced as an effective marker in cancer drug resistance. However, further research needs to be done on the role of Talin-1 and drug resistance in RCCs.
In the present study, for the first time, we showed Talin-1 protein expression in other important subtypes of RCC. We found no significant association between Talin-1 protein and clinicopathological features and patients’ outcomes in pRCC cases. In chRCCs, a significant association was observed between the increased membranous expression of Talin-1 protein and advancement of tumor stage. More investigations need to be carried out to support these findings using a larger number of cases of each pRCC and chRCC subtype.
Conclusions
In summary, bioinformatics analysis indicated Talin-1 protein as a potential prognostic marker in RCC. Our finding also demonstrated that the Talin-1 protein is upregulated in human RCC tissues compared to adjacent normal tissues. Furthermore, our results for the first time revealed statistically significant differences between membranous and cytoplasmic Talin-1 expression in various histological subtypes of RCC. A better understanding of the histological subtypes in RCC is urgently needed for further development of treatment options. Moreover, the current study indicates that membranous and cytoplasmic Talin-1 protein expression, particularly cytoplasmic expression plays an important role in more aggressive tumor behaviors, metastasis, and progression of ccRCC. In addition, high membranous and cytoplasmic expression of Talin-1 protein was identified as a worse prognostic variable affecting DSS or PFS in univariate analysis, indicating that Talin-1 may serve as a novel prognostic biomarker in ccRCC if the follow-up time is extended. Furthermore, a combined analysis of Talin-1/B7-H3 indicated an effective biomarker to predict the progression of disease and prognosis in ccRCC. However, further studies on the function and mechanism of action of Talin-1 are required to provide new opportunities for therapeutic targeting of RCC.
Authors’ contributions
LS designed and supervised the work in the original and revised manuscript, gathered the paraffin-embedded tissues, collected the patient data, prepared the information on patient survival outcomes, analyzed and interpreted the data as well as wrote and prepared the original and revised manuscript; SV performed the immunohistochemistry examinations and contributed to writing some sections of the manuscript; MA examined hematoxylin and eosin slides, marked the most representative areas in different parts of the tumor for preparing the TMAs blocks, and scored TMAs slides after Immunohistochemical staining; FF performed the bioinformatics analysis, wrote the bioinformatics sections, and contributed to preparing the revised manuscript in the bioinformatics sections. ZM provided us with the laboratory equipment and some materials for this project. All authors read and approved the final and revised manuscript.
Funding
This work was supported by a grant from Iran University of Medical Sciences (Ref no: 16289).
Data availability
The analyzed data during the current study are available from the corresponding author on reasonable request.
Supplementary data
The supplementary files are available to download from http://dx.doi.org/10.3233/CBM-220018.
sj-xlsx-1-cbm-10.3233_CBM-220018.xlsx - Supplemental material
Supplemental material, sj-xlsx-1-cbm-10.3233_CBM-220018.xlsx
Footnotes
Acknowledgments
The authors acknowledge the Kanehisa Laboratories for giving permission the use the KEGG pathway map image in our article (Focal adhesion – Homo sapiens (human) (hsa04510).
Conflict of interest
The authors declare that they have no conflict of interest.
