Abstract
Background
Prostate cancer (Pca) is one of the most frequently encountered multifactorial malignant diseases worldwide. The human oxoguanine glycosylase 1 (hOGG1) C1245G polymorphism (rs1052133) has been found to be associated with Pca. However, the conclusions have been controversial.
Methods
Based on the PubMed, Embase, HuGENet and Chinese National Knowledge Infrastructure (CNKI) databases, this meta-analysis was conducted with 4 models. Eleven qualified studies were included.
Results
Although no positive relation was discovered in the pooled analysis, significant associations between rs1052133 and Pca were found in the Asian population (recessive: odds ratio [OR] = 1.580, 95% confidence interval [95% CI], 1.189-2.098; GG vs. GC: OR = 1.504, 95% CI, 1.114-2.030; GG vs. CC: OR = 1.677, 95% CI, 1.201-2.342; allele analysis: OR = 1.249, 95% CI, 1.077-1.449), whites (dominant: OR = 2.138, 95% CI, 1.483-3.083; recessive: OR = 3.143, 95% CI, 1.171-8.437; GG vs. CC: OR = 3.992, 95% CI, 1.891-8.431; allele analysis: OR = 1.947, 95% CI, 1.467-2.586) and mixed populations (recessive: OR = 0.636, 95% CI, 0.484-0.834; GG vs. GC: OR = 0.654, 95% CI, 0.492-0.871; GG vs. CC: OR = 0.624, 95% CI, 0.473-0.823; allele analysis: OR = 0.857, 95% CI, 0.771-0.954). After excluding studies deviating from the Hardy-Weinberg equilibrium, a significant association was also found in the same ethnic groups. In addition, a new positive relation was identified in the “other country” subgroup (with China, South Korea and Australia included) (dominant: OR = 1.622, 95% CI, 1.163-2.261; recessive: OR = 1.773, 95% CI, 1.308-2.404; GG vs. GC: OR = 1.614, 95% CI, 1.169-2.230; GG vs. CC: OR = 2.108, 95% CI, 1.456-3.051; allele analysis: OR = 1.494, 95% CI, 1.235-1.808) and among the Chinese-Korean population.
Conclusions
In conclusion, we suggest that the hOGG1 C1245G polymorphism might be potentially associated with Pca risk in different ethnicities and countries, especially among Asians. Further studies are needed to confirm these relations.
Introduction
Prostate cancer (Pca) is one of the most frequently encountered malignant diseases worldwide. It is the second leading cause of cancer mortality in Western countries (1). Among Asian countries, the incidence of Pca has also increased rapidly in recent years (2). It was estimated that 219,000 new cases of Pca and 27,000 deaths occurred in 2007 in the United States (3). The morbidity and death associated with this disease have also increased in recent years (4, 5). As a multifactorial disease, many factors are known to play a key role in Pca development, such as age, ethnicity, diet and geographic factors (6). Recently, hereditary factors have also been identified as being significantly associated with Pca (7), including the human oxoguanine glycosylase 1 (hOGG1) C1245G polymorphism (rs1052133) (8).
hOGG1 is located on 3p25 from 9749944 bp to 9787790 bp. It encodes for a glycosylase/AP lyase belonging to the DNA repair enzymes, and is hypothesized to play an important role in preventing carcinogenesis by repairing oxidative damage to DNA (9, 10). It is mainly responsible for recognizing and repairing 8-oxoguanine (8-oxoG) damage, which is a mutagenic base byproduct that occurs as a result of exposure to reactive oxygen species (ROS) (11, 12). There are several variants in this gene (http://www.ncbi.nlm.nih.gov/SNP/snp_ref.cgi?locusId=4968), among which the hOGG1 C1245G (rs1052133) gene polymorphism has been studied frequently. The rs1052133 polymorphism is situated in exon 7 of the hOGG1 gene; this polymorphism induces an amino acid substitution in codon 326 from serine to cysteine (13). It has been estimated that a nearly sevenfold greater DNA repair activity is carried out by the protein encoded by the 326Ser allele compared with 326Cys (13). Recently, this variant was found to be associated with many diseases, such as breast cancer (14), lung cancer (15) and urothelial carcinoma (16). Therefore, the status of this polymorphism might be important in oxidative DNA damage and the development and progression of many diseases. In addition, studies have suggested that rs1052133 might also play a key role in Pca risk. In 2002, Xu et al conducted a study of about 18 variants of the hOGG1 gene and Pca risk, in which 2 polymorphisms, including rs1052133, were shown to be significantly related to Pca risk (17). As a follow-up, Chen et al (18) showed that hOGG1 might have a role in the repair of 8-OH-dG adducts in prostate tissue and that the hOGG1 C1245G polymorphism is associated with Pca risk. However, the converse opinion has also been presented (19). Starting from these controversial conclusions, this meta-analysis was conducted. The present study was focused on this vital variant of the hOGG1 gene to provide new information on the relationship between the hOGG1 C1245G polymorphism and Pca risk, by analyzing different subgroups.
Methods and Materials
Search and Collection
Our eligible studies were mainly retrieved from the PubMed, Embase, HuGENet and Chinese National Knowledge Infrastructure (CNKI) databases. The following key words were used: “human 8-oxoguanine glycosylase 1”, “hOGG12”, “OGG12”, “OGG”, “hOGG”, “8-oxoguanine DNA glycosylase”, “8-oxoguanine glycosylase”, “Pca”, “prostate cancer”, “prostatic carcinoma” and “prostate carcinoma.” All the studies were published in English or Chinese, were primary research articles and were not replicated in other studies. Additional searches were collected from the references lists of eligible studies. It was estimated that we required about 2-3 weeks ensuring the veracity and comprehensiveness of this analysis. The latest search was performed in July 2014.
When selecting eligible studies, the inclusion criteria were as follows: (i) the essential contents were about the hOGG1 C1245G polymorphism and Pca risk; (ii) the study was designed as a case-control or cohort study, which distinguished the case group from controls; and (iii) the number of cases and controls for separate genotypes was provided. As for the exclusion criteria, the following studies were removed: (i) studies not focused on the association of Pca risk and the hOGG1 C1245G polymorphism but which were rather gene expression and functional studies; (ii) studies that were mainly conducted on Pca cells; (iii) studies that did not distinguish between cases and controls; (iv) studies with subjects who all had Pca; and (v) studies not providing raw data on the frequency of every genotype used in our analysis. A flowchart of the selection process is shown in Figure 1.
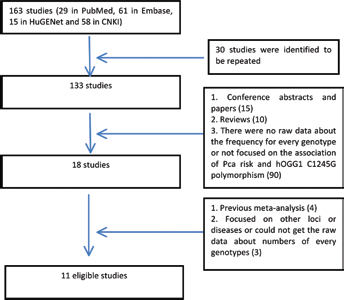
Flowchart for study selection in this analysis.
When considering the studies included, we recoded the following characteristics: names of the authors; year of publication; country; ethnicity of the samples (categorized as white, Asian, African and mixed); methods of detection; the definition of cases and controls; the number of cases and controls for each studies. All of this information is shown in Table I.
Characteristics of studies included in analysis
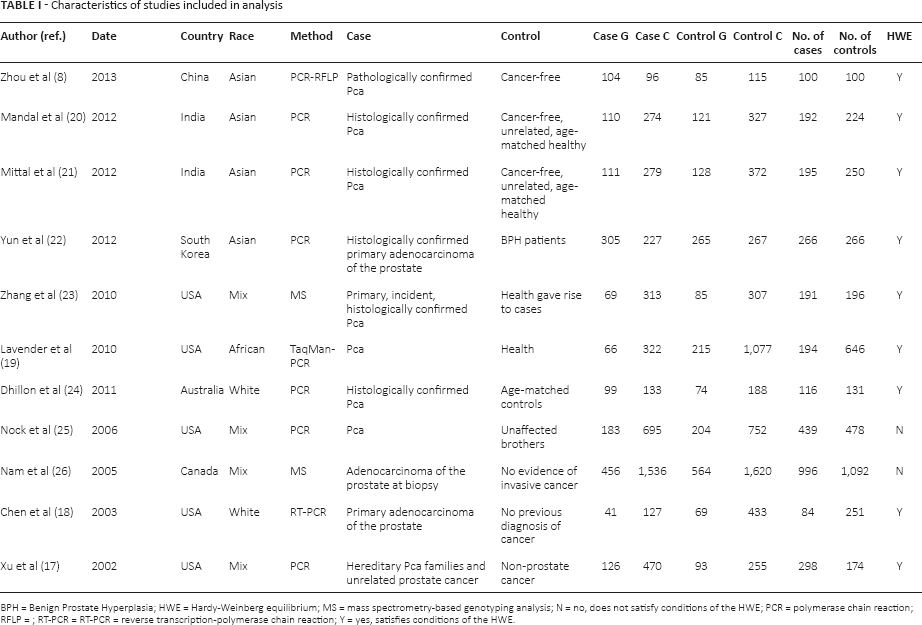
BPH = Benign Prostate Hyperplasia; HWE = Hardy-Weinberg equilibrium; MS = mass spectrometry-based genotyping analysis; N = no, does not satisfy conditions of the HWE; PCR = polymerase chain reaction; RFLP =; RT-PCR = RT-PCR = reverse transcription-polymerase chain reaction; Y = yes, satisfies conditions of the HWE.
Statistical Analysis
The frequencies of every genotype were included in the subsequent analysis. To describe the association between the hOGG1 C1245G polymorphism and Pca risk, we selected 4 common genotype models: the dominant (GG + GC vs. CC), recessive (GG vs. GC + CC) and codominant models (GG vs. GC; GG vs. CC) as well as per allele analysis (G vs. C). As for the effect models, I2 was treated as standard (when I2<50%, the fixed effect was applied; otherwise, the random effect was selected). The effects incorporated an estimate of the interstudy variance and provided wider 95% confidence intervals (95% CI) if the results of the constituent studies differed among themselves. In all analyses, the odds ratio (OR) was used to evaluate the effect of the association with the corresponding 95% CI. Pooled ORs were estimated according to the individual ORs. Significant results were defined by the 95% CI of the OR values. Meanwhile, the chi-square-based Q statistic (p<0.10 as the standard) was applied to estimate heterogeneity, which represented the weighted sum of the squared difference in the overall effect sizes from each study (27).
In this study, subgroup analysis was conducted by collecting similar characteristics from eligible studies, such as country and ethnicity. Among the eligible studies, 6 countries were included: China, India, South Korea, United States, Australia and Canada, which included subjects of different ethnicities including Asian, white, African and mixed. On the basis of the countries, 2 different categories were applied: (i) India, United States and other; (ii) China-Korea, Indian Subcontinent, North America and Oceania. As for ethnicities, Asian, white and mixed were taken into consideration. In addition, to confirm our results and discover new positive signals, the Hardy-Weinberg equilibrium (HWE; p>0.05) in the control group was tested using Stata software (28). We excluded the divergent studies which did not achieve HWE. Then the meta-analysis was conducted with the remaining data.
Publication bias was evaluated using funnel plots and Begg's unweighted regression tests for every genotype model. All p values were 2-tailed, and the data were analyzed using Stata 9.0 (Stata Corporation, College Station, TX, USA).
Results
Characteristics of the Retrieval and Eligible Studies
In the retrieval, 4 databases (PubMed, HuGENet, Embase and CNKI) were searched after combining the key words. Finally, 163 studies (29 in PubMed, 61 in Embase, 15 in HuGENet and 58 in CNKI) were obtained. In the next stage, all of the abstracts were scanned. First, repeated studies in the 4 databases were removed. Then, according to our inclusion and exclusion criteria, 18 articles were left, which were found to investigate the association between polymorphisms in the hOGG1 gene and Pca risk. After a careful reading of all of the texts, 8 studies were excluded for the following reasons: (i) 1 study mainly discussed the relationship between variations in the hOGG1 gene and Pca recurrence, in which the samples were obtained from patients with Pca (29); (ii) although 1 study was focused on the hOGG1 gene and Pca, the specific frequencies of every genotype in the cases and controls were undefined (30); (iii) 1 study concentrated on rs3218997 and Pca risk (31); (iv) 4 studies were meta-analyses regarding hOGG1 gene polymorphisms and Pca risk (32–35); and (v) 1 study did not provide raw data on the numbers of each genotype (36). In addition, to ensure that as many eligible studies were included as possible, the reference lists in the articles were checked; 1 study with accessible data was discovered (19). Finally, 11 qualifying studies were included in this meta-analysis (8, 17–26) (Tab. I). The funnel plot for allele analysis (G vs. C) as an example is presented in Figure 2. All of the p values for publication bias were also evaluated with Begg's test.
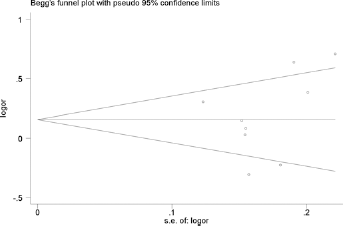
Begg's funnel plot for the allele analysis (G vs. C).
Among these eligible studies, the samples all came from local medical institutions or the general population. Cases were mainly defined as Pca patients who had been confirmed by histopathological examination. Patients who were cancer-free or who had other diseases (such as benign prostate hyperplasia) were identified as the controls, and were matched to the cases by age or other features. The participants in 4 of the eligible studies were Asian, mainly coming from China and India (8, 20–22). The other 4 studies from the United States and Canada included mixed ethnicities from Europe, Africa and other regions (17, 23, 25, 26). Two studies included whites from Australia and the United States (18, 24). The last study included subjects of African descent in the United States (19).
Results Assessment
In this meta-analysis, to ensure our that conclusions were more believable and valuable, we adopted many methods to avoid study bias and improve the power of effect sizes as much as possible: (i) The meta-analysis was mainly based on 4 authoritative databases (PubMed, HuGENet, Embase and CNKI) with comprehensive key words, which should help to include as many eligible studies as possible. The strict retrieval process should also have helped to ensure the veracity of the whole statistical analysis. (ii) Samples were mainly humans. And cases were defined as Pca patients with a diagnosis confirmed by histopathological examination. Cancer-free patients, those with other diseases (such as benign prostate hyperplasia) and healthy participants were identified as controls, who were matched with the cases by age or other features. The explicit definitions reduce the bias to some extent. (iii) In the processes of analysis, 4 common genotype models – the dominant (GG + GC vs. CC), recessive (GG vs. GC + CC) and codominant model (GG vs. GC; GG vs. CC) and per allele analysis (G vs. C) – and different subgroup analyses were applied in this study. This method may also reduce the study bias and improve the power of effect sizes. And (iv) last but not least, the HWE was applied. Studies not meeting the HWE were excluded in order to ensure the stability of the samples, which reduces the study and sample biases vastly. Thus, using these criteria and methods, we can propose that the results of our meta-analysis should be significant for further research.
hOGG1 C1245G Polymorphism Associated with Pca Risk in Different Ethnicities
In the first analysis, all 11 studies were included. The subgroups were mainly classified by country and ethnicity. For the ethnicities, Asian, white and mixed populations were identified. Although we did not find a positive relation in the pooled analysis, significant associations between the hOGG1 C1245G polymorphism and Pca risk were found in the Asian population (dominant model: OR = 1.216, 95% CI, 0.984-1.503, I2 = 0%; recessive model: OR = 1.580, 95% CI, 1.189-2.098, I2 = 0%; codominant model [GG vs. GC]: OR = 1.504, 95% CI, 1.114-2.030, I2 = 0%; codominant model [GG vs. CC]: OR = 1.677, 95% CI, 1.201-2.342, I2 = 0%; allele analysis: OR = 1.249, 95% CI, 1.077-1.449, I2 = 0%), the white population (dominant model: OR = 2.138, 95% CI, 1.483-3.083, I2 = 0%; recessive model: OR = 3.143, 95% CI, 1.171-8.437, I2 = 41.2%; codominant model [GG vs. GC]: OR = 2.150, 95% CI, 0.828-5.581, I2 = 33.6%; codominant model [GG vs. CC]: OR = 3.992, 95% CI, 1.891-8.431, I2 = 6.9%; allele analysis: OR = 1.947, 95% CI, 1.467-2.586, I2 = 0%) and mixed populations (dominant model: OR = 0.886, 95% CI, 0.778-1.008, I2 = 0%; recessive model: OR = 0.636, 95% CI, 0.484-0.834, I2 = 0%; codominant model [GG vs. GC]: OR = 0.654, 95% CI, 0.492-0.871, I2 = 0%; codominant model [GG vs. CC]: OR = 0.624, 95% CI, 0.473-0.823, I2 = 0%; allele analysis: OR = 0.857, 95% CI, 0.771-0.954, I2 = 0%) (Tab. II). The forest plot for the recessive model is shown in Figure 3. For the country subgroups, there were 2 different analytical methods. As the first one, countries were divided into three groups (India, United States and other countries), in which we could not find any potentially significant signal for any country (Suppl Tab. I. Available online at www.biological-markers.com). Then, when the countries were split into China-Korea, Indian Subcontinent, North America and Oceania, it was found that, in the Chinese and Korean population, rs1052133 might play a key role in Pca risk (dominant model: OR = 1.376, 95% CI, 0.976-1.940, I2 = 0%; recessive model: OR = 1.702, 95% CI, 1.221-2.373, I2 = 0%; codominant model [GG vs. GC]: OR = 1.628, 95% CI, 1.144-2.318, I2 = 0%; codominant model [GG vs. CC]: OR = 1.893, 95% CI, 1.249-2.868, I2 = 0%; allele analysis: OR = 1.383, 95% CI, 1.126-1.700, I2 = 0%) (Suppl Tab. II. Available online at www.biological-markers.com).
Results of meta-analysis with dominant model (GG+GC vs. CC), recessive model (GG vs. GC+CC), codominant model (GG vs. GC; GG vs. CC) and allele analysis (G vs. C)
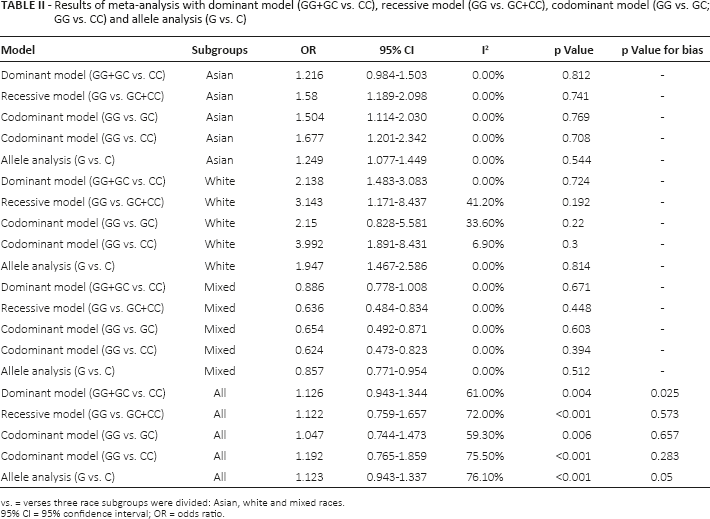
vs. = verses three race subgroups were divided: Asian, white and mixed races.
95% CI = 95% confidence interval; OR = odds ratio.
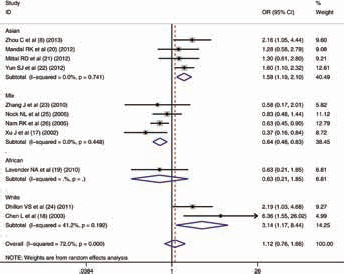
Meta-analysis with random effects and recessive model (GG vs. GC+CC) for the association between hOGG1 C1245G polymorphism and prostate cancer. The first author and year of publication for each study are shown. In this analysis, 3 race subgroups are shown: Asian, white and mixed races. Odds ratio (OR) and accompanying 95% confidence interval (95% CI) are also presented for the association of the hOGG1 C1245G polymorphism and prostate cancer.
hOGG1 C1245G Polymorphism Associated with Pca Risk in the Country Subgroups
To ensure the correctness of our results, we filtered our included studies again by testing the HWE in every article. After excluding studies that did not satisfy the HWE, all analyses were conducted. In the ethnicities group, the same association between the hOGG1 C1245G polymorphism and Pca risk was also discovered for the same genotype models (Suppl Tab. III. Available online at www.biological-markers.com). Meanwhile, to our surprise, in the “other country” subgroup (with China, South Korea and Australia included), a latent relationship between this polymorphism and Pca risk was also identified for the 4 different genotype models (dominant model: OR = 1.622, 95% CI, 1.163-2.261, I2 = 22.8%; recessive model: OR = 1.773, 95% CI, 1.308-2.404, I2 = 0%; codominant model [GG vs. GC]: OR = 1.614, 95% CI, 1.169-2.230, I2 = 0%; codominant model [GG vs. CC]: OR = 2.108, 95% CI, 1.456-3.051, I2 = 0%; allele analysis: OR = 1.494, 95% CI, 1.235-1.808, I2 = 7.8%). The forest plot for the recessive model is shown in Figure 4. Even so, a positive association was not detected in India and the United States (Suppl Tab. IV. Available online at www.biological-markers.com). In addition, there were no new distinct associations discovered in the “other country” subgroup analysis (Suppl Tab. V. Available online at www.biological-markers.com).
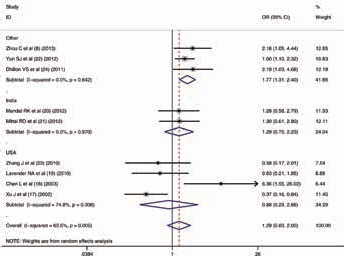
Meta-analysis with random effects and recessive model (GG vs. GC+CC) for the association between hOGG1 C1245G polymorphism and prostate cancer after removing the studies that did not satisfy the criteria for the Hardy-Weinberg equilibrium. The first author and year of publication for each study are shown. In this analysis, 3 country subgroups are shown: India, United States and other countries. Odds ratio (OR) and accompanying 95% confidence interval (95% CI) are also presented for the association of the hOGG1 C1245G polymorphism and prostate cancer.
Discussion
Pca is a serious cancer and influences a large number of populations. As one aspect of a multifactorial disease, genetic mutation is known to play a key role in Pca development and progression. Recently, the hOGG1 C1245G polymorphism was found to be associated with Pca risk (8). However, the conclusion was inconclusive, so this meta-analysis was conducted. Our results suggested that the hOGG1 C1245G polymorphism might be a hereditary factor regulating Pca.
The hOGG1 gene encodes for a DNA glycosylase/AP lyase, which is mainly responsible for repairing oxidative damage to DNA (9). The mutation of this gene could lead to an alteration in heredity. Correspondingly, the transcription and translation of DNA could be influenced, which could result in many diseases, especially cancers. In 2012, Ma et al (37) suggested that the hOGG1 C1245G polymorphism may contribute to susceptibility to bladder cancer in a Chinese population. Meanwhile, in the same population, this polymorphism was found to be associated with susceptibility to childhood acute lymphoblastic leukemia (38). In 2009, Okasaka et al (39) also showed that the hOGG1 C1245G polymorphism might contribute to the risk of lung adenocarcinoma. In addition, other cancers such as urothelial carcinoma (16) were also presented. On the basis of these studies and the function of the hOGG1 gene, we believe that the hOGG1 C1245G polymorphism is important in regulating the development and progression of cancer.
As a common cancer, Pca is thought to be influenced by the hOGG1 C1245G polymorphism. In 2002, Xu et al (17) investigated hOGG1 sequence variants and Pca susceptibility, and suggested that hOGG1 C1245G is important in determining Pca risk. Then, in 2011, Dhillon et al (24) showed that Pca risk is significantly increased only in carriers of the G allele of the C1245G polymorphism of the hOGG1 gene. Moreover, Yun et al (22) confirmed a significant relationship in the Korean population. On the basis of these studies, this meta-analysis was conducted. In the first stage of analysis, 4 genotype models were applied in the ethnicity and country subgroups. Although there was no significant association discovered in the pooled analysis, the hOGG1 C1245G polymorphism seemed to be positively related to Pca risk in the Asian, white and mixed populations. In 2013, Zhou et al (8) investigated hOGG1 genetic variants and Pca risk, and found that the hOGG1 C1245G gene polymorphism might increase susceptibility to Pca in the Chinese population. This conclusion was also confirmed in the Korean population (23). As for the white population, Dhillon et al (24) and Chen et al (18) discovered a significant relationship between the hOGG1 C1245G polymorphism and Pca risk in Australia and United States, respectively. In addition, the association was also found in a mixed population, identified by Xu et al (17) in the white and African American populations. Thus, considering these studies, we can understand the potential function of the hOGG1 C1245G polymorphism in regulating Pca risk. To confirm our results further, the second stage of analysis was conducted, in which studies divergent from the HWE were excluded. Fortunately, our conclusion was confirmed.
In addition, we also conducted a comprehensive analysis in country subgroups with 2 different analytical methods: (i) the countries were divided into India, United States and other country; and (ii) countries were divided into China-Korea, Indian Subcontient, North America and Oceania. In the first category, we could not identify a possible association between the hOGG1 C1245G polymorphism and Pca risk in any of the country groups (India, United States and other). However, when 1 study was removed according to the HWE analysis (26), a positive relationship in the “other country” group was discovered, which suggests that, in different countries, the hOGG1 C1245G polymorphism might also contribute to the development and progression of Pca. We then tried to describe the features of this study. Firstly, this study diverged from the HWE, which suggests that its results cant not represent the general population. Secondly, the control samples in this study were with no evidence of invasive cancer. The definition seemed to be ambiguous which might result in bias. Meanwhile, the population in this study was mixed, which also reduced the function of representativeness by increasing deviation in the results. As for the second category (countries were divided into China-Korea, Indian Subcontinent, North America and Oceania), the results confirmed that, among the Asian population, rs1052133 might play a key role in the development and progression of Pca.
Combining the first- and second-stage analyses, we suggest that the potential association between the hOGG1 C1245G polymorphism and Pca risk cannot be ignored. More studies are needed to confirm our conclusions and the real associations in different countries and ethnicities. However, this study provides a new direction for genetically targeted therapy in the future.
Limitations
After a comprehensive meta-analysis, a relationship between Pca and the hOGG1 C1245G polymorphism was found. However, there were still some limitations to this study. Firstly, the number of studies on the association between the hOGG1 C1245G polymorphism and Pca is limited, especially for different ethnicities and countries, and may not illustrate the real function of this polymorphism in the development and progression of Pca. Secondly, the populations in the studies included were from different areas, especially for the mixed ethnicities subgroup, which resulted in some heterogeneity in the results. Finally, the definitions of cases and controls had some differences, which could also influence the results.
Conclusion
Pca is a global cancer. The hOGG1 C1245G polymorphism has been found to be associated with Pca risk. To make the relationship clear, this study was conducted. Our results suggest that the hOGG1 C1245G polymorphism is significantly associated with Pca, especially in the Asian population. However, the function of this locus for the risk of Pca in India and the United States remains uncertain. Further studies are needed to confirm these correlations.
Footnotes
Financial support: This study was supported by grants from the Guangxi science and technology project (no. 1355005-3-17).
Conflict of interest: None of the authors has any financial interest related to this study to disclose.
