Abstract
Introduction
Nasopharyngeal carcinoma (NPC) is a malignant tumor derived from nasopharyngeal mucosal epithelial cells. It is especially common in Guangdong, Guangxi, and other regions in China, and its incidence rate is as high as 20-30/100,000 per year. 1 Epidemiological and experimental studies have shown that genetic susceptibility, Epstein–Barr virus (EBV) infection, and environmental factors are specific and multifactorial causes of NPC. 2 Although many people are exposed to EBV infection and ecological risk factors, including heavy smoking and alcohol consumption, only a small proportion of the population has developed NPC, indicating that individual genetic susceptibility may play a more important role in the carcinogenic mechanism of NPC. However, an increasing number of studies have shown that the main cause of individual susceptibility differences is gene polymorphism, 3 and its relationship with the occurrence of NPC has gradually attracted many researchers.
Single nucleotide polymorphism (SNP) is a widely used biomarker in molecular genetic analysis, which refers to the impact of the conversion, inversion, insertion, and deletion of individual nucleotides in the genome on the occurrence of tumors. Studying the SNP of genes can provide a theoretical basis for further exploring the corresponding genes of mutation sites and even the impact of functional chromosome changes on tumor occurrence. 4 The role of SNPs in DNA repair-related genes in tumor occurrence and development and related mechanisms has gradually received attention.5,6
DNA repair is one of the important events in human cells responsible for maintaining the integrity of the human genome. The normal cellular metabolism process in the body produces endogenous carcinogens, such as hydroxyl radicals, which can cause oxidative damage to DNA. 7 If the accumulation of DNA damage is not repaired, it can lead to tumor transformation of normal cells. 8 Base excision repair (BER) and nucleotide excision repair are the main DNA repair pathways responsible for removing carcinogenic metabolites.9,10 Human 8-oxoguanine DNA glycosylase 1 (hOGG1) is a major DNA transglucosidase in the BER pathway, located in the 3p25-26 region of the human chromosome and composed of seven exons. 11 In addition to acting as a glycosidase, the hOGG1 protein also functions as an apurinic/apyrimidinic (AP) lyase, removing damaged bases from DNA. 12 The hOGG1 gene has the characteristic of SNP, and the base of exon 7 can undergo a C→G transition, resulting in the amino acid being replaced from Serine (C) to Cysteine (G) at codon 326 (rs1052133), 13 indicating genetic polymorphism differences. At present, the hOGG1 rs1052133 polymorphism has been proven to have a higher cancer risk, such as hepatocellular carcinoma, 14 breast cancer, 15 and laryngeal cancer. 16
However, there is no unified conclusion on whether the hOGG1 rs1052133 polymorphism increases the risk of NPC because different studies have given conflicting results. A study from Taiwan showed that the Cys/Cys (GG) and Ser/Cys (CG) genotypes of hOGG1 rs1052133 significantly increased the risk of NPC. 17 However, studies from Morocco 18 and Malaysia 19 have shown that the hOGG1 rs1052133 polymorphism may not affect the susceptibility to NPC. To address the controversial topic, we conducted a systematic review and meta-analysis by combining all relevant studies to evaluate the relationship between hOGG1 rs1052133 polymorphism and susceptibility to NPC.
Materials and Methods
Data Sources
We aimed to search for all studies related to this topic without limitations on methods and language. The search date was limited to July 1, 2023. We searched for studies through PubMed, Web of Science, Scopus, CNKI, and VIP. The search terms included “hOGG1,” “OGG1,” “8-oxoguanine DNA glycosylase,” “C > G,” “Ser326Cys,” “rs1052133,” “nasopharyngeal carcinoma,” “nasopharyngeal cancer,” “NPC,” and “cancer of nasopharynx.”
Inclusion and Exclusion Criteria
All included studies should meet the following inclusion criteria: (1) The patients included in this study were diagnosed with NPC through biopsy and were at least 18 years old. (2) The studies were case–control trials, mainly focusing on the relationship between hOGG1 rs1052133 and the occurrence of NPC. (3) The studies provided general clinical characteristics of all populations, analysis results of the study and control groups, and their OR values. (4) The studies could provide full texts.
Exclusion criteria: (1) Animal experiments, reviews, case reports, and other meta-analyses. (2) The studies were not case–control trials. (3) The studies did not describe population characteristics and related OR values. (4) Unable to obtain full text or only able to get abstract. (5) Low relevance to the theme of this meta-analysis.
Data Extraction
According to the PRISMA guidelines, 20 the selected studies ultimately entered the systematic review stage. We used the Newcastle–Ottawa Scale (NOS) evaluation scale 21 for systematic review, and the studies with a score ≥ 7 were ultimately included in further meta-analysis. 22 Two researchers (Li and Yao) extracted relevant data from the texts, tables, and figures of the studies. If there were any differences, we consulted the third party (Wang) to discuss and resolve the problems. For information lacking in studies, we contacted the authors for supplementation.
Trial Sequence Analysis
Trial sequence analysis (TSA) is used to estimate the sample size of the meta-analysis and overcome the shortcomings of classical meta-analysis. Meta-analysis typically requires accumulating multiple experimental results to increase random errors, and some “positive” analysis results may be attributed to the random errors. When the number of trials and patient sample size in the meta-analysis are small, random errors may lead to erroneous results.23–25 Therefore, it is important to apply TSA in meta-analysis to reduce the risk of false positive or false negative results. In this study, we used TSA 0.9.5.10 Beta software for analysis to calculate the required information size (RIS), which referred to the number of cases required to achieve statistically significant differences in meta-analysis. At the same time, the alpha expenditure function, continuous monitoring boundaries, and evaluation of ineffective boundary areas were estimated.26,27
Meta-Analysis
We used Stata version 17.0 software to analyze the results. We grouped all studies based on different genotypes of the rs1052133 locus, first integrated the OR values of each study in every group using the random-effects model, and plotted a forest map. For the consistency evaluation of the studies, the traditional statistical test (Cochrane's Q test) was first used for evaluation, and then the I2 test was further validated. If I2 ≤ 50%, we changed to the fixed-effects model for all OR values. If I2 > 50%, it indicated significant heterogeneity, we continued to use the random effects model and conducted further sensitivity analysis and subgroup analysis to evaluate the sources of heterogeneity. 28 We determined whether the rs1052133 polymorphism was related to the occurrence of NPC based on the cross between the diamond in the forest plot and the invalid line. If the diamond in the forest plot intersected with the invalid line, it was considered that the integrated OR value was not statistically significant, indicating that the polymorphism of rs1052133 was unrelated to the occurrence of NPC. If it did not intersect with the invalid line and was on the right side, it was considered that the polymorphism of rs1052133 was indeed a risk factor for NPC. It was considered a protective factor if it was on the left side. 29 We validated the evaluation of publication bias using Begg's and Egger's tests. 30 P < .05 indicated that the results were statistically significant.
In Silico Analysis
We investigated the relationship between different genotypes of the rs1052133 locus and the hOGG1 mRNA expression using eQTL (expression quantitative trait loci) analysis. We used the genotype-tissue expression (GTEx) database (https://www.gtexportal.org) to investigate the effects of different genotypes of the rs1052133 locus on the expression of hOGG1 mRNA in different normal human tissues. 31 To explore the relationship between hOGG1 expression and head and neck squamous cell carcinoma (including NPC), as well as its impact on the prognosis, we used the latest developed interactive web server and mobile application GEPIA (http://gepia.cancer-pku.cn/) and GE-mini (http://gemini.cocancer-pku.cn/) which provided expression data of tumor and normal samples from TCGA and GTEx projects.32–36
Results
Studies Screening
We searched for 1126 articles from all databases and excluded 23 duplicate articles. We screened the remaining 1103 articles based on their titles or abstracts and excluded 1068. The remaining 35 articles were screened, and 29 articles were removed based on the inclusion and exclusion criteria. After screening, we conducted a systematic review of six studies using the NOS evaluation scale. All studies were scored ≥ 7 and included in the final meta-analysis (Table 1). The process of study selection was shown in Figure 1.

The flow chart of the study selection.
NOS Evaluation Scale for Studies.

Characteristics of Studies
The meta-analysis ultimately included six studies, and their specific clinical characteristics were shown in Table 2. Researchers obtained peripheral blood samples from NPC patients or healthy individuals and extracted their genomic DNA. Subsequently, polymerase chain reaction was used to detect and statistically analyze different genotypes of the hOGG1 rs1052133 locus in NPC patients and healthy individuals. All studies belonged to case–control trials and the main researchers in these studies were from different countries. These studies had been published from 2003 to 2020, with five studies mainly involving Asians and one study including Africans.
The Clinical Characteristics of All Studies.

NA: Not Applicable.
This meta-analysis included a total of 2021 nasopharyngeal cancer patients and 2375 healthy individuals. In five studies, the P values of Hardy–Weinberg Equilibrium (HWE) test results between the study group and the control group were >0.05, while the result of one study was not available. From the results in Table 3, the study from Taiwan demonstrated the correlation between the hOGG1 rs1052133 polymorphism and the occurrence of NPC, while the results of other studies suggested no clear correlation.
The Conclusions Drawn From All Studies About the Relationship Between the Different Genotype Groups of hOGG1 rs1052133 and the Occurrence of NPC.

To further clarify the relationship between the hOGG1 rs1052133 polymorphism and the occurrence of NPC under smoking history conditions, we screened and ultimately included three studies Given the history of smoking, the studies of Li et al 37 and Ban et al 19 did not demonstrate the correlation between the hOGG1 rs1052133 polymorphism and the occurrence of NPC. However, the study of Keppen et al 39 found a correlation between the CG genotype and the occurrence of NPC, while GG was not. Under the condition of no smoking history, the studies of Li et al 37 and Ban et al 19 indicated that neither genotype was associated with the occurrence of NPC. Moreover, the study of Keppen et al 39 demonstrated that both genotypes were associated with the occurrence of NPC (Table 4).
The Conclusions Drawn from All Studies About the Relationship Between the Different Genotype Groups of hOGG1 rs1052133 and the Occurrence of NPC According to the Smoking History.

Trial Sequence Analysis
We used TSA 0.9.5.10 Beta software for the sequential analysis of this meta-analysis. The probability of Type I error was set to 0.05 and the probability of Type II error was set to 0.2, with the sample size as the RIS. As shown in Figure 2, the RIS for conducting this meta-analysis was 2223 for nasopharyngeal cancer patients and normal healthy individuals, and the sample size we have included exceeded the expected size. Therefore, this meta-analysis could continue. In addition, the Z-curve did not cross the traditional threshold and the TSA threshold, but intersected with the invalid line, indicating that there might be no difference in the frequency of hOGG1 rs1052133 polymorphism between NPC patients and healthy populations.

TSA for hOGG1 rs1052133 polymorphism.
Meta-Analysis
We grouped all studies based on different genotypes, with seven studies included in each group (two of which were from the same study). We first used the random effects model to integrate all OR values for each group. After integrating, it was found that the I2 values of the “CG vs CC” and “GG vs CC” groups were less than 50%, indicating little heterogeneity between the studies in the two groups. Therefore, the fixed effects model was used to integrate all OR values for each group, as shown in Figure 3. It showed that the I2 value of the “CG vs CC” group was 45.4%, P = .089 > .05, and the total OR value of this group was 1.00 (95% CI: 0.85-1.18). The I2 value of the “GG vs CC” group was 26.5%, P = .226 > .05, and the total OR value of this group was 1.06 (95% CI: 0.87-1.28). The intersection of the diamond and invalid line (black vertical solid line) for each group in the forest plot indicated that these results were not statistically significant (P > .05). Therefore, it indicated that the polymorphism of hOGG1 rs1052133 was not significantly correlated with the occurrence of NPC. The funnel plot results for each group were shown in Figure 4.

Forest plot of the relationship between the different genotype groups of hOGG1 rs1052133 and the occurrence of NPC.
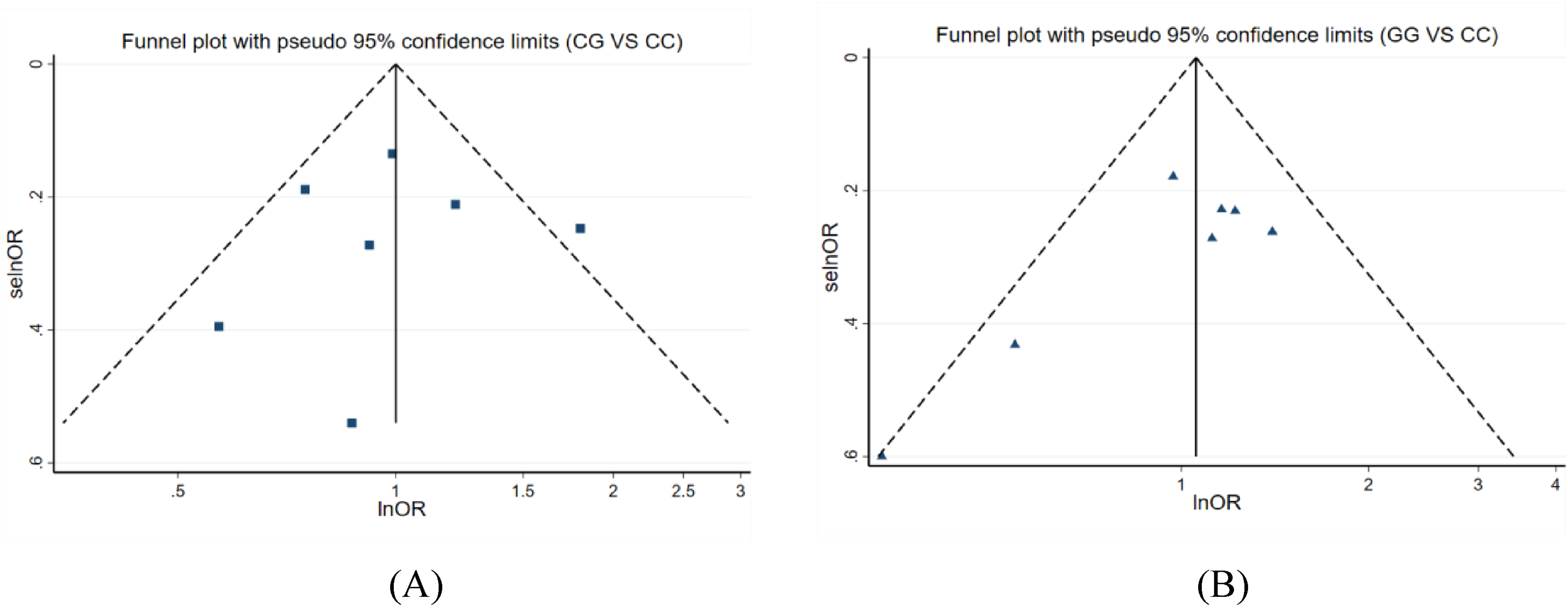
Funnel plots of the relationship between the different genotype groups of hOGG1 rs1052133 and the occurrence of NPC. (A) The funnel plot of the “C
We further analyzed the correlation between the polymorphism of hOGG1 rs1052133 and the occurrence of NPC under different smoking conditions. Figure 5A showed that the I2 value of the “CG vs CC” group was 60.9%>50%, while the I2 value of the “GG vs CC” group was 0%<50%. Therefore, the random effects model continued to be used to integrate OR values. After analysis, it was found that the total OR value of the “CG vs CC” group was 1.52 (95% CI: 0.74-3.15), while the total OR value of the “GG vs CC” group was 1.15 (95% CI: 0.73-1.81). The diamonds and the invalid line (the black vertical solid line) in the forest plot of these two groups intersected, indicating that these results were not statistically significant (P > .05), further indicating that the polymorphism of hOGG1 rs1052133 did not increase the risk of NPC with the smoking history. We continued to analyze the correlation between the polymorphism of hOGG1 rs1052133 and the occurrence of NPC without a smoking history. Figure 5B showed that the I2 values of both groups were greater than 50%, then the random effects model was used to integrate the OR values. After integration, it was found that the total OR value of the “CG vs CC” group was 0.76 (95% CI: 0.33-1.74), while the total OR value of the “GG vs CC” group was 0.66 (95% CI: 0.26-1.71). Similarly, the diamonds and the invalid line (the black vertical solid line) intersected in the forest plot, indicating that the polymorphism of hOGG1 rs1052133 did not increase the risk of NPC without the smoking history.

The forest plot of the relationship between the different genotypes of hOGG1 rs1052133 and the occurrence of NPC under different smoking conditions. (A) The forest plot of the relationship between the different genotypes of hOGG1 rs1052133 and the occurrence of NPC with the smoking history. (B) The forest plot of the relationship between the different genotypes of hOGG1 rs1052133 and the occurrence of NPC without the smoking history.
We analyzed the relationship between hOGG1 rs1052133 polymorphism and the occurrence of NPC in other models (allele, dominant, recessive, and additive). The results of OR and P-value of each study in different models were shown in Table 5. We integrated all studies from different models for analysis, and the OR values were shown in Figure 6. Because the I2 value in the Allele model was greater than 50%, while the I2 values in other models were less than 50%, we ultimately used the random effects model for analysis. From the forest plot in Figure 6, it could be seen that the diamonds and the invalid lines (the black vertical solid lines) intersected in each model, indicating that the results of OR values from each model had no statistical significance (P > .05), indicating that there were no clear correlations between the hOGG1 rs1052133 polymorphism and the NPC in other genotype models.

Forest plot of the relationship between the different genotype groups of hOGG1 rs1052133 and the occurrence of NPC in other genotype models.
The Conclusions Drawn From All Studies About the Relationship Between the Other Genotype Groups of hOGG1 rs1052133 and the Occurrence of NPC in Other Models.

NA: Not Applicable.
In Silico Analysis
Based on the data from the GTEx portal website, we found that the CG or GG of the rs1052133 locus increased the expression of hOGG1 mRNA in muscle-skeletal (P = 7.03 × 10−8) and cultured-fibroblast cells (P = 2.16 × 10−5) but decreased the expression of hOGG1 mRNA in sun-exposed skin (P = 4.02 × 10−5). In addition, the expression of hOGG1 in head and neck squamous cell carcinoma (including NPC) tissues was higher than that in normal tissues (Transcripts Per Kilobase Million [TPM] = 7.08 vs 5.17 significantly, P < .01; Figure 7). In addition, we also analyzed whether the expression of hOGG1 affected the overall survival (OS) and disease-free survival (DFS) of the head and neck squamous cell carcinoma. Kaplan–Meier survival results showed no significant difference in OS and DFS between the low TPM and high TPM groups of hOGG1 in NPC (OS: log-rank P = .39, DFS: log-rank P = .095; Figure 8).

The effects of different genotypes of rs1052133 (CG or GG) on the expression of hOGG1 mRNA in human tissues. (A) In muscle-skeletal. (B) In cultured fibroblasts cells. (C) In sun-exposed skin (lower leg).
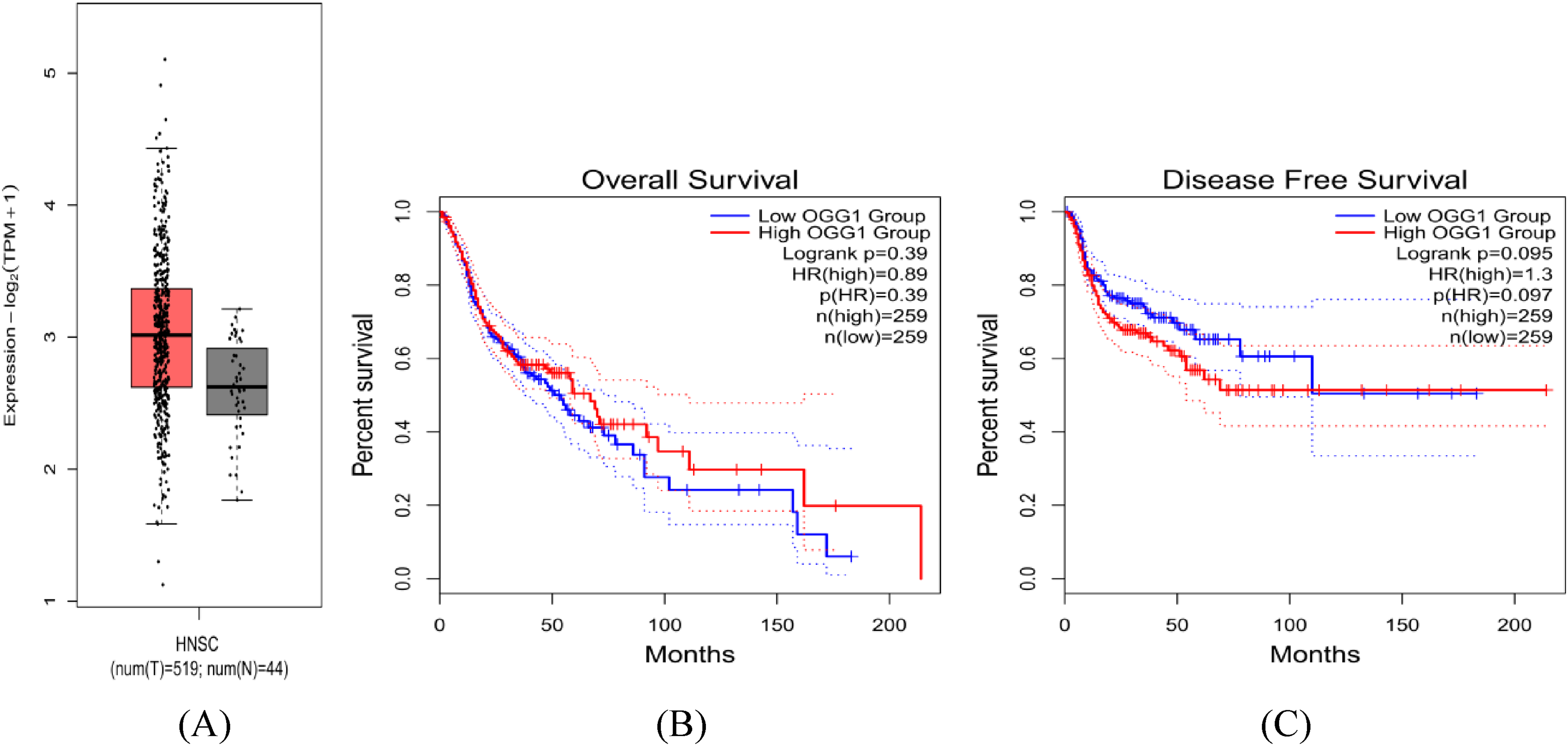
(A) The expression of hOGG1 in normal tissues and head and neck squamous cell carcinoma tissues. (B) The effect of hOGG1 on OS in patients with head and neck squamous cell carcinoma. (C) The effect of hOGG1 on DFS in patients with head and neck squamous cell carcinoma.
Publication Bias
We evaluated publication bias for all included studies using Begg's and Egger's tests. The results of Begg's test for all studies were P = .457 > .05, while the results of Egger's test were P = .074 > .05. These results suggested no significant publication bias from all studies.
Discussion
NPC is a malignant tumor originating from the epithelium of the nasopharynx mucosa. According to reports, its onset results from genetic factors, EBV, environmental factors, and trace elements. 40 Cho et al 17 first published a study about the association between hOGG1 rs1052133 (Ser326Cys) polymorphism and susceptibility to NPC and found that CG and GG genotypes could increase the risk of NPC. The author combined previous research 41 in their study and elucidated potential mechanisms, suggesting that CG and GG genotypes might reduce the ability of hOGG1 to repair 8-OH-GUA in damaged DNA, leading to the accumulation of a large amount of erroneous DNA and increasing the likelihood of cancer.
It was known that hOGG1 was an important transglucosidase in the DNA repair system, mainly responsible for removing damaged bases in DNA. 42 If the base C at codon 326 of hOGG1 was converted to G, non-synonymous coding would occur, causing hOGG1 to lose its original DNA repair function, and normal nasopharyngeal mucosal cells could not be repaired during replication, ultimately leading to carcinogenesis. According to the above mechanism, the conversion from C→G should increase the incidence of NPC. However, many subsequent studies reached negative conclusions, and most researchers believed that there was no significant correlation between the polymorphism of hOGG1 rs1052133 and the occurrence of NPC. This controversial and contradictory conclusion has yet to be resolved, and no systematic evaluation or meta-analysis has clarified the conclusion further.
In order to address the controversial topic, we integrated these studies for analysis in our meta-analysis. We strictly followed the inclusion and exclusion criteria and ultimately included six studies. Among them, one study showed a correlation between the polymorphism of hOGG1 rs1052133 and the occurrence of NPC, while the other five studies showed no correlation. Further analysis and forest mapping were conducted by integrating these studies, and it was found that there was no correlation between the polymorphism of hOGG1 rs1052133 and the occurrence of NPC. The CG and GG genotypes did not increase the incidence of NPC. Previous studies have shown that smokers with hOGG1 rs1052133 CG and GG genotypes have a higher risk of prostate cancer. 43 We further analyzed the correlation between hOGG1 rs1052133 polymorphism and the occurrence of NPC under different smoking conditions. Under the condition of the smoking history, we included three studies, two of which found no correlation between CG and GG in the occurrence of NPC and one study found the association between CG and NPC, but not GG. In addition, three studies were included without the smoking history. Two studies found that both CG and GG were not associated with the occurrence of NPC, while one study found that both CG and GG were associated with the occurrence of NPC. To address the controversial conclusion, after integrating the effect values from each study and conducting a comprehensive analysis, it was found that there was no clear correlation between the polymorphism of hOGG1 rs105213 and the occurrence of NPC, regardless of smoking history. Moreover, no correlation was found between the polymorphism of hOGG1 rs105213 and the occurrence of NPC in other models. Based on the research findings of Cho et al and the current experimental mechanism, the transformation of C→G should increase the risk of NPC, but our meta-analysis did not draw a positive conclusion. The reason might be: (1) The sample size was insufficient. (2) There were significant individual differences between each study. (3) There were other protective mechanisms, such as when hOGG1 lost its original repair function, it might induce other mechanisms to compensate for this function. This conclusion still needs further validation in future research.
Although our meta-analysis did not find a correlation between the polymorphism of hOGG1 rs105213 and the occurrence of NPC, there were clear correlations in the occurrence of other malignant tumors, which had important clinical significance for early screening and prevention. It could screen high-risk populations for early intervention and instruct us to develop new treatment methods, such as gene therapy. Our study had some limitations: (1) The relationship between hOGG1 rs105213 polymorphism and the occurrence of NPC was controversial, but the number of published studies was too small. Although the total sample size of the experimental and control groups included in this meta-analysis exceeded the expected size in TSA, the sample size was not very large. Moreover, it was recommended that at least 10 studies be conducted for meta-analysis. However, the number of studies in this meta-analysis did not meet the requirements, and the accuracy of its conclusions was not very clear. Further validation is needed in subsequent large-scale studies. (2) When analyzing whether there was a relationship between hOGG1 rs1052133 and the occurrence of NPC based on the smoking conditions, there was significant heterogeneity due to the small number of studies in each group, leading to increased errors in the results. (3) In some studies, the definition and specific information of the control group were unclear and only described as the healthy population, resulting in lower scores in NOS assessment.
Conclusion
At present, exploring the relationship between different gene polymorphisms and the risk of NPC is a hot topic that has gradually attracted the attention of many researchers, which is of great significance for clarifying its etiology and mechanism. The relationship between the hOGG1 rs105213 polymorphism and the occurrence of NPC is controversial, and different studies have yielded different results. We conducted a meta-analysis based on the current controversy and found no clear correlation between the two. Due to the limited number of relevant studies, the accuracy of their conclusions still needs to be further proven in future large sample data.
Footnotes
Abbreviations
Acknowledgments
We appreciate all the authors of the studies who provided data for this study.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This article does not contain any studies with human participants or animals performed by any of the authors. This Meta-analysis has been registered with PROSPERO and INPLASY, with PROSPERO registration number: CRD42023440290 and INPLASY registration number: INPLASY202370016.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
