Abstract
Purpose
Gastrointestinal (GI) malignancies are among the 5 most common cancers in Iran, and their high associated mortality rates are attributable to late diagnosis and poor treatment options. SOX2, a transcription factor necessary for maintenance and induction of pluripotency and self-renewal, has been identified as a lineage-survival oncogene in several cancers. In the present study, we examined SOX2 expression in esophageal squamous cell carcinoma (ESCC), gastric adenocarcinoma and colon squamous cell carcinoma (SCC), as well as normal GI tissues, in Iranian patients.
Methods
To elucidate the role of SOX2 in GI carcinogenesis, formalin-fixed tissues were analyzed using immunohistochemistry (IHC), while frozen ESCC samples were studied by quantitative reverse transcription polymerase chain reaction (qRT-PCR).
Results
IHC studies indicated presence of SOX2+ cells in a subset of cancerous and normal tissues of stomach and colon, while no significant difference was observed between groups, and no correlation was found between SOX2 expression and tumors grades. Nevertheless, studying ESCC samples with IHC and qRT-PCR revealed overexpression of SOX2 in comparison with normal adjacent tissues.
Conclusions
The present results are in line with other studies and indicate SOX2 up-regulation in ESCC; however, due to our small sample size and contradictory reports, more research is needed to determine the importance of SOX2 in GI cancers.
Introduction
Besides its high and increasing rates of incidence, cancer is known as one of the most challenging diseases for management and therapy, especially in developing countries. In Iran, for instance, cancer is the third most frequent cause of death, and according to published studies, malignancies of esophagus, stomach and colon-rectum are among the 5 most common cancers in the Iranian population (1).
Sex-determining region Y (SRY)–box 2, also known as SOX2, is one of the crucial transcription factors involved in embryonic development, morphogenesis and cell fate determination. The actions of SOX2 are linked to the emergence and maintenance of stem cell phenotype; and in cooperation with other factors, SOX2 controls self-renewal and pluripotency in stem cells, reprograms differentiated cells and maintains stem cell–like features in cancer cells (2).
The contribution of SOX2 to later events of carcinogenesis, such as tumor progression and metastasis, has been demonstrated in various kinds of malignancies. There are also several reports on the importance of SOX2 in gastrointestinal (GI) cancers; for instance, it was demonstrated that SOX2 is the amplification target at chromosome subband 3q26.3 in esophageal squamous cell carcinoma (ESCC) (3, 4), and its elevated expression was reported in patients with esophageal adenocarcinoma (EAC) and ESCC (5-12). Furthermore, studies of gastric adenocarcinoma (GA) (13-16) and colorectal cancer (CRC) (17-20) have also indicated overexpression of SOX2 in such malignancies. Meanwhile, a number of studies have revealed a significant correlation between SOX2 expression and clinicopathological features of GI cancers such as recurrence, invasion depth, lymph node metastasis and disease-free survival of patients (13, 14, 17, 21-23). Nevertheless, contradictory data have led to questions regarding the role of SOX2 in GI malignancies, as loss of SOX2 expression was related to poor prognosis and survival in EAC (24) and GA (25-27), and no correlation was found between SOX2 expression and clinicopathological features in ESCC and EAC (7). In vitro studies have also fueled the controversy, because the repressed transcriptional activity of β-catenin/TCF complex in colon cancer cells has been attributed to SOX2 activity (28). Furthermore, SOX2 was responsible for growth inhibition of gastric cancer cells through cell cycle arrest and apoptosis (25), and also mediated autophagy and cellular senescence in colon cancer cells (29).
Although the role of SOX2 in GI cancers has been reported by several groups, little is known about its expression in Iranian patients with ESCC, GA and colon squamous cell carcinoma (CSCC). Therefore, the objective of present work was to study SOX2 expression in normal and tumoral tissues of esophagus, stomach and colon, and evaluate the role of SOX2 as a prognostic marker for GI cancers in Iranian patients. To do so, formalin-fixed samples were examined by immunohistochemistry (IHC), while ESCC frozen specimens were also analyzed by quantitative reverse transcription polymerase chain reaction (qRT-PCR).
Materials and Methods
IHC studies
To study SOX2 expression in tumoral and normal tissues of esophagus, stomach and colon, 3- to 5-μm-thick sections from formalin-fixed paraffin-embedded tissues were provided on poly L-lysine (Sigma) precoated slides by Dr. Moayyed Pathology Laboratory (Mashhad, Iran). A list of tissues and clinicopathological information of patients are presented in Table I. After dewaxing sections in xylene (Merck) and rehydrating by decreasing percentages of ethanol (absolute, 96%, 75% and 50%; Merck), slides were boiled in sodium citrate solution (10 mM, pH 6.5; Merck) for 30 minutes to retrieve antigens. Samples were then incubated with 3% H2O2 (Merck) in tris-buffered solution (TBS, pH 6.5; Merck) for 15 minutes, washed 3 times with TBS and covered by blocking buffer containing 2% bovine serum albumin (BSA; Sigma) in TBS, for 60 minutes. Then, blocking buffer was replaced with primary IgG1 SOX2 antibody (clone 10F10, diluted 1:200; Novus Biologicals), and slides were incubated at 4°C overnight. After washing 3 times in TBS, sections were subjected to the secondary IgG1 horseradish peroxidase (HRP)-conjugated antibody (diluted 1:800; Abcam) for 60 minutes, followed by washing in TBS and staining with 3,3’-diaminobenzidine (DAB; Sigma) working solution. Finally, slides were washed in CuSO4 solution (5 mg/mL; Merck), stained with hematoxylin (Novocastara), dehydrated with ethanol (96% and absolute), rinsed in xylene, dried and fixed with enthalan glue (Merck). To better evaluate results, positive and negative control tissues were also included: human lung adenocarcinoma (30) was considered as positive control, while human seminoma (31) and also specimens only treated with secondary antibody were used as negative controls.
List of tissues analyzed for SOX2 expression, including clinicopathological features of patients
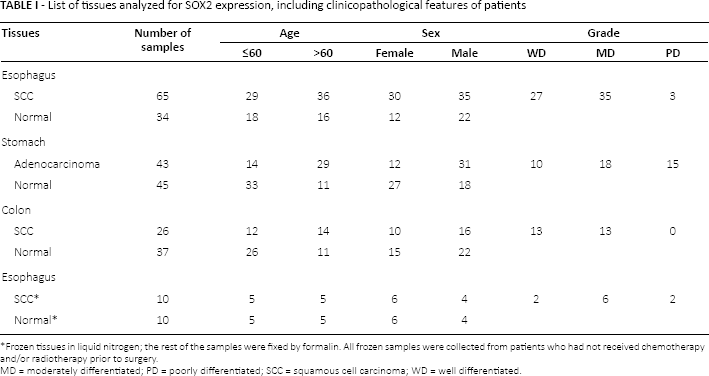
Frozen tissues in liquid nitrogen; the rest of the samples were fixed by formalin. All frozen samples were collected from patients who had not received chemotherapy and/or radiotherapy prior to surgery.
MD = moderately differentiated; PD = poorly differentiated; SCC = squamous cell carcinoma; WD = well differentiated.
qRT-PCR Analysis
To better examine SOX2 expression in normal and tumoral tissues of esophagus, total RNA was extracted from frozen tissues purchased from the Iran National Tumor Bank (founded by the Cancer Institute of Tehran University of Medical Sciences for cancer research, Tehran, Iran; Tab. I), by TRIzol reagent (Invitrogen) using the manufacturer's protocol. To avoid DNA contamination, extracted RNAs were treated with RNase-free DNase I (Genet Bio), and for cDNA synthesis, M-MuLV reverse transcriptase (Fermentas) was used according to the manufacturer's instruction. The fidelity of amplified cDNAs was then confirmed by semiquantitative RT-PCR using GAPDH primers, and final products were loaded on 1% agarose gel (Invitrogen) for electrophoresis.
qRT-PCR was performed using SYBR green master mix (Pars Toos) in an iQ5 real-time PCR detection system (Bio-Rad). To compare the level of SOX2 expression between ESCC specimens and their normal counterparts, GAPDH transcripts were used as internal control, and normalized values were plotted as relative fold change over normal samples. For GAPDH and SOX2 primers (Tab. II), thermal cycling conditions were as follows: 95°C for 4 minutes followed by 40 cycles of denaturation at 95°C for 30 seconds, annealing at 60°C for 30 seconds, and extension at 72°C for 30 seconds. PCR efficiencies were calculated for both primers from the slopes of standard curves, generated from serial dilutions of a positive control, and fold change expression was calculated as 2(-ΔΔCT) (32).
Primer sequences used in current study for qRT-PCR

qRT-PCR = quantitative reverse transcription polymerase chain reaction.
Statistical Analysis
Statistical analyses were carried out using SPSS software; normal distribution of data was determined by Kolmogorov-Smirnov normality test (KS-test), and according to each experiment, Pearson correlation, one-way ANOVA or Wilcoxon nonparametric analysis was applied. Results were reported as means ± SD, and statistically significant differences were calculated as p<0.05.
Results
Immunostaining of SOX2
To study SOX2 expression in Iranian patients with GI cancers, IHC was performed on ESCC, GA and CSCC specimens, as well as normal GI tissues. As shown in Figure 1A-F, SOX2+ cells were detected in a subset of both tumoral and normal tissues. However, statistical analyses indicated no significant difference in SOX2 expression between ESCC, GA and CSCC samples (92%, 89% and 82%, respectively) and normal tissues (67%, 85% and 92%, for normal esophagus, stomach and colon, respectively; Fig. 2A). Furthermore, our results indicated no correlation between SOX2 expression and clinicopathological characteristics of patients, including tumor grade and patient's sex and age. As presented in Table I, considering ESCC, GA and CSCC patients, 41%, 23% and 50% were diagnosed with well-differentiated cancers, 53%, 72% and 61% were male, and 55%, 67% and 53% were more than 60 years of age, respectively.
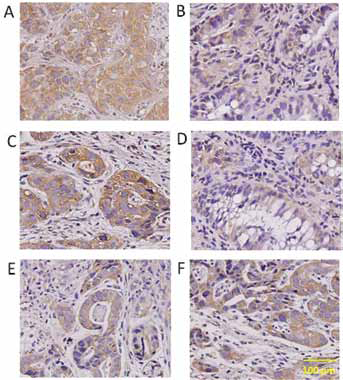
Immunohistochemical detection of SOX2 in tissue samples of esophagus, stomach and colon. Representative images indicate SOX2+ cells in tumoral (A, C, E) and normal (B, D, F) tissues of esophagus (A, B), stomach (C, D) and colon (E, F).
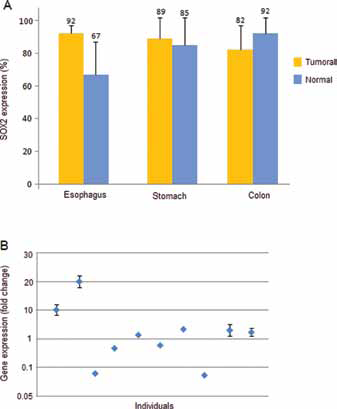
SOX2 expression in gastrointestinal (GI) tissues. Histogram indicates percentage of SOX2+ cells in tumoral and normal tissues of esophagus, stomach and colon as detected by immunohistochemistry (IHC). Statistical analysis revealed no significant difference in SOX2 expression between malignant and normal tissues (A). Scatter plot demonstrates SOX2 expression in esophageal squamous cell carcinoma (ESCC) samples and their normal counterparts as analyzed by quantitative reverse transcription polymerase chain reaction (qRT-PCR). SOX2 overexpression and down-regulation were detected in 6 and 4 ESCC patients, respectively (B). Data are expressed as means ± SD.
SOX2 mRNA Expression
Further studying ESCC samples for SOX2 expression at the mRNA level, tumoral and normal specimens of 10 ESCC patients were examined by qRT-PCR (Fig. 2B). Expression of SOX2 in ESCC samples was very variable, showing a wide range of fold changes from 0.023 to 20.1; SOX2 overexpression and down-regulation were detected in 6 and 4 ESCC patients, respectively. Comparing SOX2 expression between tumoral samples and their normal counterparts indicated a significant up-regulation of SOX2 (p<0.05) in ESCC patients. However, statistical analyses revealed no correlation between SOX2 expression and clinicopathological features of patients, including tumor grade and patient's sex and age. In particular, 20% of patients were diagnosed with well-differentiated ESCC, 40% were male, and 50% were more than 60 years of age (Tab. I).
Discussion
SOX2 is a member of the high mobility group-domain DNA-binding protein family, which is implicated in the regulation of transcription and chromatin architecture. It is known as a master regulator of pluripotency and self-renewal in normal and cancer stem cells, and as an important factor in establishing induced pluripotent stem cells (2). In the upper GI tract, SOX2 is expressed in developing foregut endoderm and is thought to play important roles in establishing the boundary between keratinized, squamous-lined esophagus and glandular hind stomach (33). In human ESCC, frequently occuring amplifications at the chromosomal band 3q26 result in SOX2 overexpression, as the SOX2 gene is located on the 3q26 amplicon (3, 4). Besides, elevated expression of SOX2 has been reported in ESCC, EAC, GA and CRC (8-12, 14-16, 19, 20). However, there is a controversy around the role of SOX2 in GI carcinogenesis, as a number of studies have reported SOX2 down-regulation in patient samples and cancerous cell lines (24-29).
Due to limited information on SOX2 expression in GI malignancies of Iranian patients, we aimed to study expression of SOX2 in ESCC, GA and CSCC patients and assess its validity as a prognostic marker. Our results indicated the presence of SOX2+ cells in normal and tumoral tissues of stomach and colon, while no significant difference was observed between normal and malignant samples, and no correlation between percentage of SOX2+ cells and tumors grades. Due to discrepant reports on overexpression (14-16, 19, 20) and down-regulation (25-27) of SOX2 in GA and CRC, its importance and correlation with the diseases features still remain undetermined. Even though there is no exact explanation to address these contradictions, they reflect to some extent the genetic heterogeneity of such abnormalities. Furthermore, it seems that studying patients of different sex and age with tumors of different stages/grades, employing different techniques and materials, and interpreting results inaccurately are among the reasons for these inconsistencies.
The present results also revealed that in ESCC specimens, the number of SOX2+ cells was higher than that in normal esophageal samples, although the difference was not significant. Therefore, to better evaluate the role of SOX2 in esophagus carcinogenesis, its expression was quantitatively assessed at mRNA level, and the results demonstrated a significant up-regulation in SOX2 expression. These current results are in agreement with previous reports that indicated overexpression of SOX2 in ESCC (5-9, 11, 12). However, our IHC and qRT-PCR results could be best explained by a recent meta-analysis demonstrating that high incidences of SOX2 expression in ESCC were associated with significant heterogeneities (10). In the current study, no correlation was found between SOX2 expression and ESCC grade, which is in line with one published study (7), but in contrast to others (5, 8, 11).
In the present study, detection of SOX2+ cells in normal esophagus, stomach and colon tissues indicates the presence of stem cells that guarantee constant epithelial cell turnover. In the basal layer of esophagus, for instance, it has been demonstrated that stem cells and transient amplifying cells gradually differentiate into early and late differentiated cells in the suprabasal layer (34). As dysregulated apoptosis and uncontrolled growth of cells are responsible for malignant transformations, stem cells resident in epithelia of esophagus, stomach and colon have been introduced as potential tumor-initiating cells in these tissues (35). In this regard, it was shown that cooperation of SOX2 with STAT3 is responsible for malignant transformation of foregut basal progenitor cells (36), and also, activation of the AKT signaling pathway by SOX2 promotes esophageal tumor growth (9, 15). Moreover, a number of reports have shown that SOX2 might be related to GI cancer stem cells, as SOX2+ stem-like cancer cells have been detected in esophageal (37, 38), gastric (39) and colorectal (40) cancer cell lines.
In conclusion, the current results indicate the presence of SOX2+ cells in normal tissues of esophagus, stomach and colon, which confirms the unquestioned role of stem cells in tissue turnover. On the other hand, detection of SOX2-expressing cells in ESCC, GA and CSCC specimens presumably explains the malignant transformation of stem cells that are involved in carcinogenesis. However, our results should be interpreted with some caution, as the major limitation was the small number of patients, and therefore, a larger study population will allow us to validate our conclusions.
Footnotes
Acknowledgement
Authors would like to thank Mr. Farshchian for his advice and technical assistance.
Financial support: This work was supported by a grant from Ferdowsi University of Mashhad (No. 100314).
Conflict of interest: The authors have no conflict of interest.
