Abstract
Background:
Immunoglobulin binding protein 1 (IGBP1) is an important signal transduction regulator that mediates various functions. However, its expression profile, role, and clinical significance in cancers are uncertain. The purpose of this study was to determine the expression profile and the prognostic significance of IGBP1 in esophageal squamous cell carcinoma (ESCC).
Methods:
Polymerase chain reaction assay, western blotting, and immunohistochemistry (IHC) assay were performed to examine IGBP1 expression in ESCC tissues and matched adjacent non-cancerous tissues. Moreover, IHC was used to evaluate IGBP1 expression in archived 190 paraffin-embedded ESCC specimens. Statistical analyses were applied to evaluate the prognostic value and the correlations between IGBP1 expression and the clinical parameters.
Results:
We found that the messenger RNA and protein levels of IGBP1 were up-regulated in the ESCC tissues compared with their adjacent non-cancerous tissues. High expression of IGBP1 in ESCC patients was positively associated with T classification (P=0.013) and vital status (P=0.03). The ESCC patients with higher IGBP1expression had a shorter survival time than those with lower IGBP1 expression. Importantly, multivariate analysis demonstrated that the expression of IGBP1 was an independent prognostic factor for ESCC (P< 0.05).
Conclusions:
We provide the first evidence that increased IGBP1 expression correlates with poor prognosis of ESCC, and that IGBP1 may be a tumor promoter of ESCC, which provide a promising prognostic biomarker and therapeutic target for ESCC.
Introduction
Esophageal cancer remains one of the most virulent malignancies characterized by a high incidence and poor prognosis. Each year, an estimated 455,800 new esophageal cancer cases occur worldwide, with 400,200 expected deaths.1,2 The global incidence burden and mortality are expected to continue to increase over time. Esophageal squamous cell carcinoma (ESCC) is the predominant pathological type of esophageal cancer in East Asia and belongs to the most frequent aggressive malignant cancers, which ranks as the fourth leading cause of cancer-related death in China.3,4 Despite considerable advances achieved in diagnosis and multimodality therapies, the overall 5-year survival rate for esophageal cancer remains approximately 17%.2,5 Previous reports have shown that genetic variations frequently associated with the development of esophageal cancer include the p53 mutation, the inactivation of p16, the amplification of cyclin D1, and the amplification of c-Myc or the epidermal growth factor receptor.3,6,7 The carcinogenesis and development of esophageal cancer is a complex process that involves cumulative mutations or amplification in multiple genes. Therefore, its exact molecular pathogenesis is still unknown. It is as advantage to identify potential molecular biomarkers for predicting prognosis in the progression of ESCC at an early stage.
Immunoglobulin binding protein 1 (IGBP1; CD79a), also known as alpha4, was initially identified as a signal transduction molecule coprecipitating with MB1 (Igα) of the B cell antigen receptor complex and was later found to be broadly expressed in various tissues. Previous studies have indicated that IGBP1 could bind to the protein phosphatase 2A (PP2A), which is the most abundant phosphatase in mammalian cells.8,9 IGBP1 is an essential regulator of PP2A phosphatase activity; it plays important roles in anti-apoptotic effect, DNA damage, and cell cycle control; and it possesses an oncogenic function.10-12 IGBP1 is ubiquitously highly expressed in human cancers including hepatocellular carcinomas, primary lung cancers, primary breast cancers. 13 Moreover, it is expressed universally in advanced lung adenocarcinomas, and its overexpression is significantly correlated with their poor prognosis.14,15 However, the expression and role of IGBP1 in ESCC is still unknown.
In the present study, we investigated the expression of IGBP1 in ESCC and further assessed its clinical significance. Our results showed that elevated IGBP1 expression appeared in clinical ESCC tissues compared with adjacent non-cancerous tissues, and a higher IGBP1 level predicted a poor outcome of ESCC. Importantly, the expression of IGBP1 was positively associated with the T classification of ESCC patients. As an aggressive malignant tumor, ESCC commonly presents in a locally advanced stage with a poor survival. The depths of invasion indicated as T classification is crucial to the progression to a locally advanced stage of ESCC. The treatment of locally advanced esophageal cancer is controversial and still evolving. Our results (a) suggest that the ESCC patients with an elevated IGBP1 level could be assessed as high-risk tumor phenotypes; and (b) finally provide a molecular clue to establish the standard treatment regimen for locally advanced esophageal cancer in the future.
Materials and methods
Patient information and tissue specimens
The study was approved by the Ethics Committee of Cancer Hospital of Jiangxi Province according to the International Ethical Guidelines for Biomedical Research Involving Human Subjects (CIOMS). Patients involved in this study were diagnosed with ESCC from 2005 to 2013 at the Cancer Hospital of Jiangxi Province, and underwent esophageal cancer resection. Clinicopathologic features of the 190 patients are shown in Table 1. ESCC specimens were staged in accordance with American Joint Cancer Committee/Union for International Cancer Control (AJCC/UICC) classification guidelines. The grading and histopathology subtyping of ESCC specimens was based on WHO criteria. ESCC biopsy samples and their adjacent matching non-cancerous esophageal tissues were frozen in liquid nitrogen and stored.
Correlation between IGBP1 expression and clinicopathologic characteristics of esophageal cancer patients.
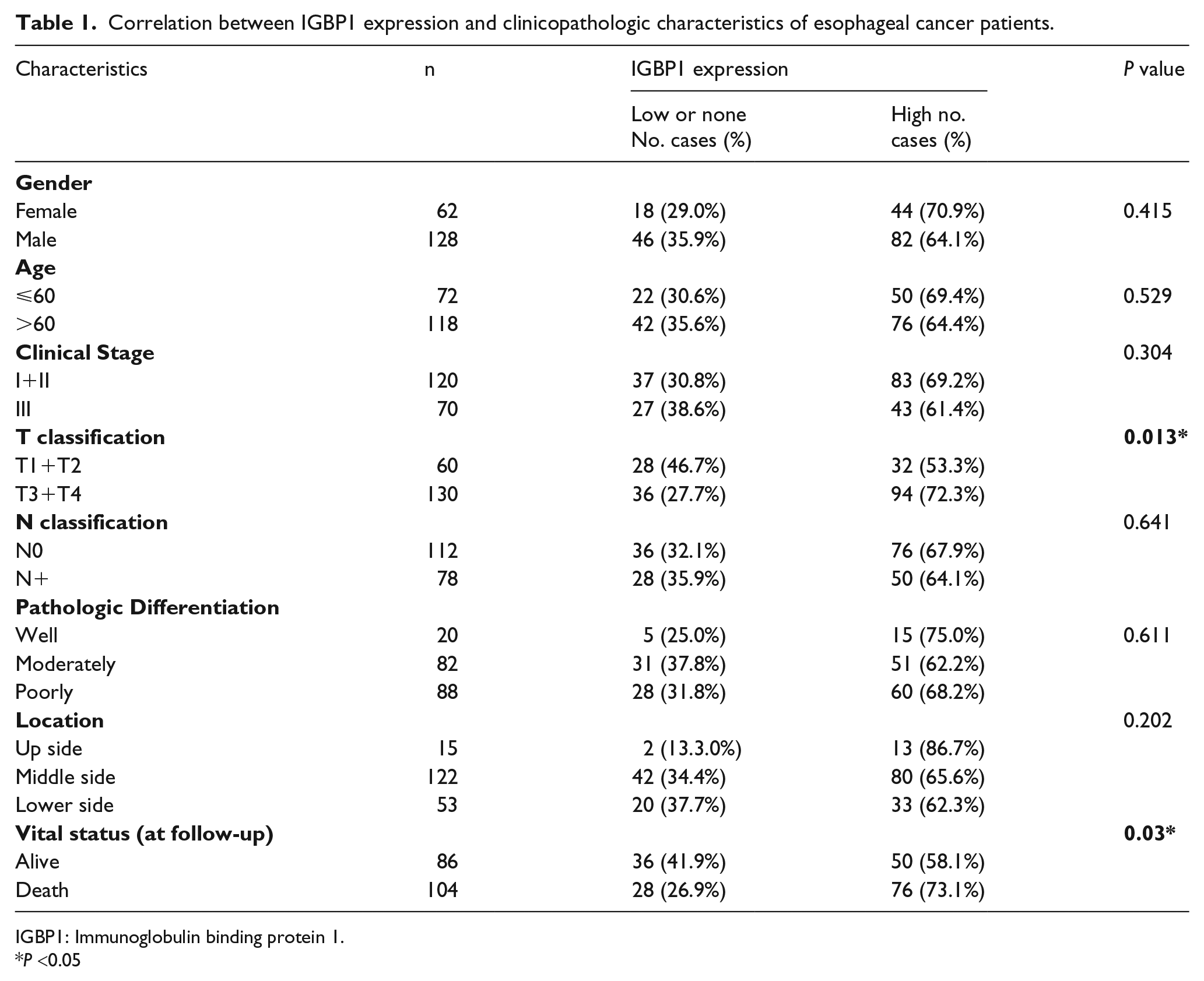
IGBP1: Immunoglobulin binding protein 1.
P <0.05
Reverse transcription-polymerase chain reaction
Reverse transcription-polymerase chain reaction (RT-PCR) was performed as described previously.16-18 The total RNA of tissue specimens was isolated using TRIzol (Invitrogen) according to the manufacturer’s protocol. First-strand cDNA was synthesized using the PrimeScript RT reagent Kit with gDNA Eraser. And PCR was performed for the detection of IGBP1 messenger RNA (mRNA) using ProFlex™ PCR system (Thermofisher Scientific). The sequences of primers were as follows: for IGBP1: 5′TACTGGACGAAGTAGAAGTGGC3′ (forward), 5′AGCTGCGATAACATTTCGGCA3′ (reverse); for GAPDH: 5′ACAGTCAGCCGCA TCTTCTT3′ (forward), 5′GACAAGCTTCCCGTTCTCAG3′ (reverse). The PCR condition was:95oC for 10 min, followed by 35 cycles of 95oC for 5 s, 60oC for 15 s and 72oC for 30 s.
Western blotting analysis
Western blotting was performed as previously described.16-18 Briefly, cells were lysed in MCLB (50 mM Tris-HCl (pH 8.0), 2 mM DTT, 5 mM EDTA, 0.5 % Nonidet P-40, 100 mM NaCl, 1 μM microcystin,1 mM sodium orthovanadate, 2 μM PMSF, protease (Sigma Chemical Co.) and phosphatase inhibitor cocktail (Calbiochem)), and the clarified lysates were resolved by SDS-PAGE and transferred to PVDF membranes for western blotting using ECL detection reagents (Beyotime Co.). Antibodies against IGBP1 and GAPDH were from Sigma Aldrich and Cell Signaling Technology, respectively.
Immunohistochemistry
Immunohistochemical staining for IGBP1 and immunohistochemistry (IHC) evaluation in human ESCC tissues was performed as previously described.16-18 Briefly, sections of paraffin-embedded specimens were baked and deparaffinized. Sections were stained with an anti-IGBP1 antibody (Sigma Aldrich), and the antigen was detected using a secondary anti-rabbit HRP-conjugated antibody and a DAB chromogen kit (Genetech) followed by counterstaining with hematoxylin. IGBP1 expression was evaluated by two independent pathologists. The immunohistochemical staining score was calculated according to the percentage of positive tumor cells and the staining intensity. The proportion of positive tumor cell was scored as: “0” (0–10%), “1” (11–25%), “2” (26–50%), “3” (51–75%), and “4” (76–100%). Staining intensity was scored as: “0” (negative), “1” (weak), “2” (moderate), and “3” (strong). The final scores were calculated by multiplying the two scores derived from the percentage of positive tumor cells and the staining intensity. A score of 7 was defined as the cutoff value according to receiver operating characteristic curve analysis.
Statistical analysis
The SPSS software (version 16.0, SPSS Inc., Chicago, IL, USA) was used for statistical analysis. The relationship between IGBP1 expression and the clinicopathological parameters was examined by the Pearson chi-square and Kruskal–Wallis tests.
The correlations between IGBP1 expression and overall survival curves were plotted by the Kaplan–Meier method and compared with the log-rank test. Univariate and multivariate Cox regression analyses were used to evaluate survival data. Differences were considered statistically significant when P values were < 0.05.
Results
IGBP1 was up-regulated in primary human ESCC tissues
To explore the potential roles of IGBP1 in ESCC, we collected several paired fresh tissues, including ESCC tumor specimens and the corresponding adjacent non-tumor tissue specimens. RT-PCR, western blot, and IHC methods were performed to measure IGBP1 mRNA and protein levels in these tissues. As shown in Figure 1(a), compared with their non-tumor counterparts, the expression level of IGBP1 mRNA was significantly elevated in ESCC tumor tissues. Similarly, the expression level of IGBP1 protein was also up-regulated in ESCC compared with that in the adjacent nonmalignant esophageal tissues (Figure 1(b)). This finding was further verified by IHC assay in the six paired ESCC and non-cancerous tissue samples. These results indicated that IGBP1 is overexpressed in ESCC.
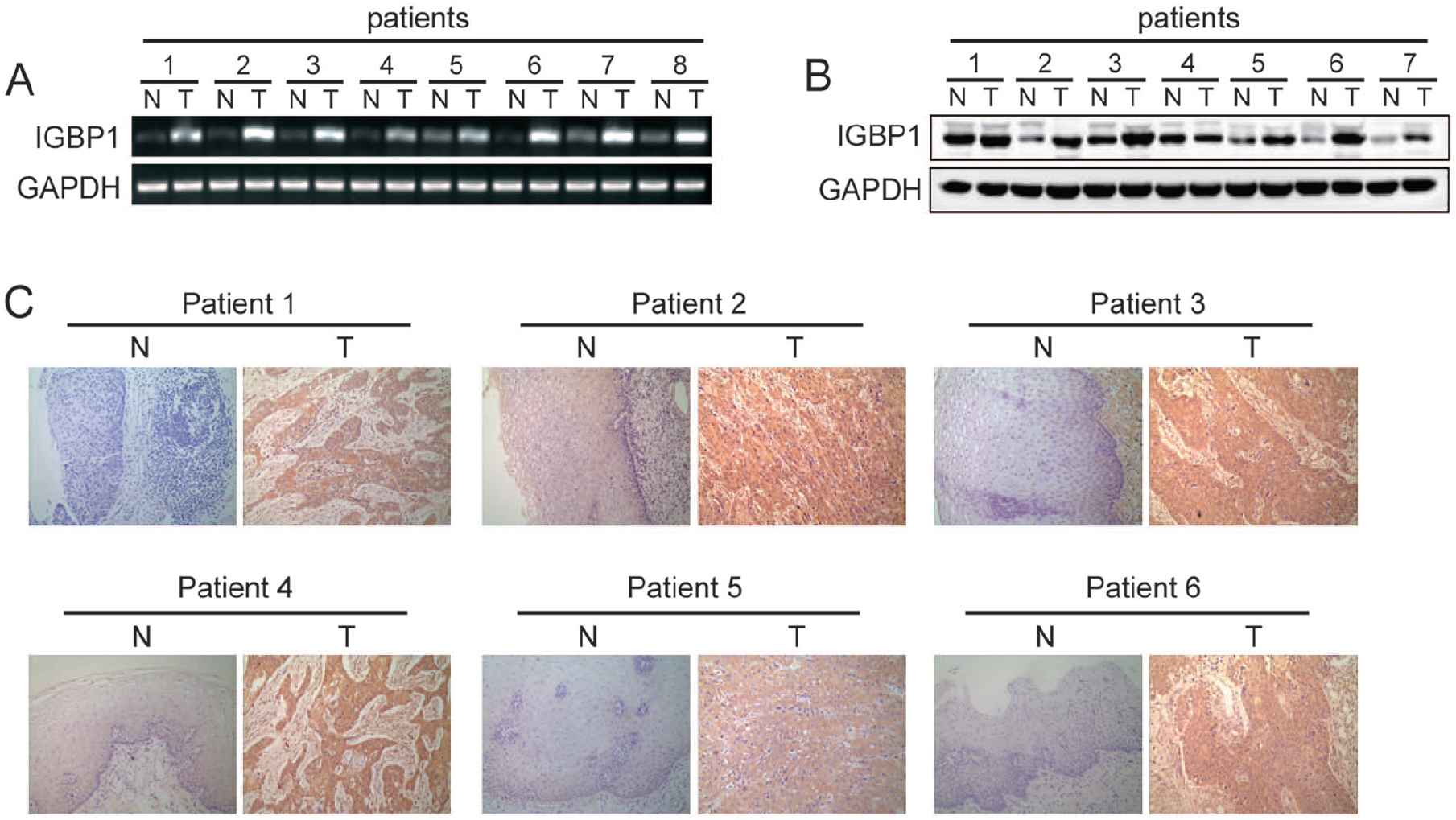
IGBP1 was up-regulated in primary human ESCC tissues.
Association between increased expression of IGBP1 and progression of ESCC
To further define the relationship between IGBP1 protein expression and the clinicopathologic parameters of ESCC, 190 paraffin-embedded, archival primary ESCC tissues were detected by IHC, which included 120 cases of clinical stage I and stage II (63.2%), and 70 cases of stage III (36.8%). According to the results of immunohistochemical staining and statistical analysis, the IGBP1 protein level was significantly associated with T classification (P = 0.013), representing the depths of invasion, as shown in Table 1. The ESCC patients with higher IGBP1 levels tended to show the increased depths of invasion. However, there was no significant correlation between the expression level of IGBP1 protein and the other clinicopathologic characteristics of ESCC including age (P = 0.529), gender (P =0.415), tumor location (P =0.202), pathologic differentiation (P = 0.611), and lymph node metastasis (P = 0.641), as shown in Table 1.
Association between IGBP1 expression and ESCC patient survival
To determine the association of IGBP1 expression with overall survival of patients with ESCC, the Kaplan–Meier analysis and the log-rank test were used. According to the immunohistochemical staining score (see Materials and methods section), the190 cases of ESCC tissues were divided into 128 cases in the high-expression group and 64 cases in the low-expression group. As shown in Figure 2(b), the ESCC patients with a high expression of IGBP1 had a lower overall survival rate than those with low IGBP1 expression (P =0.03). Furthermore, to avoid the potential biases, the Propensity Score Matching method was used to match the high-expression group and the low-expression group. The factors for matching included gender, age, clinical stage, pathologic differentiation, and location (as shown in Table 2). The matched 64 pairs of high expression and low expression were selected for survival analysis. The result was consistent with the previous results (P =0.034; as shown in Figure 2(c)). These results revealed that IGBP1 expression is significantly associated with the prognosis of ESCC patients.
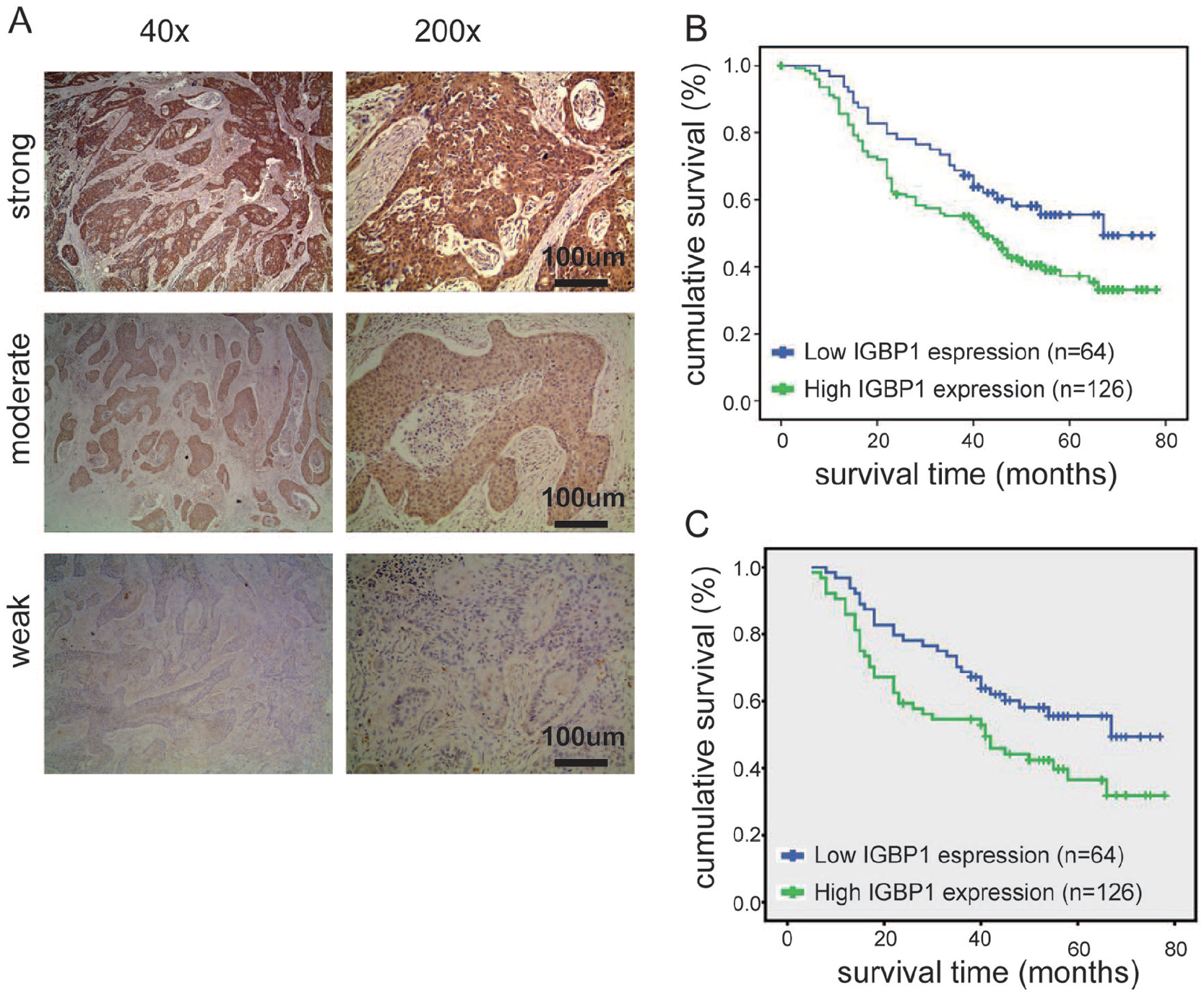
IGBP1 expression correlated with overall survival.
Clinicopathologic factors used for Propensity Score Matching.
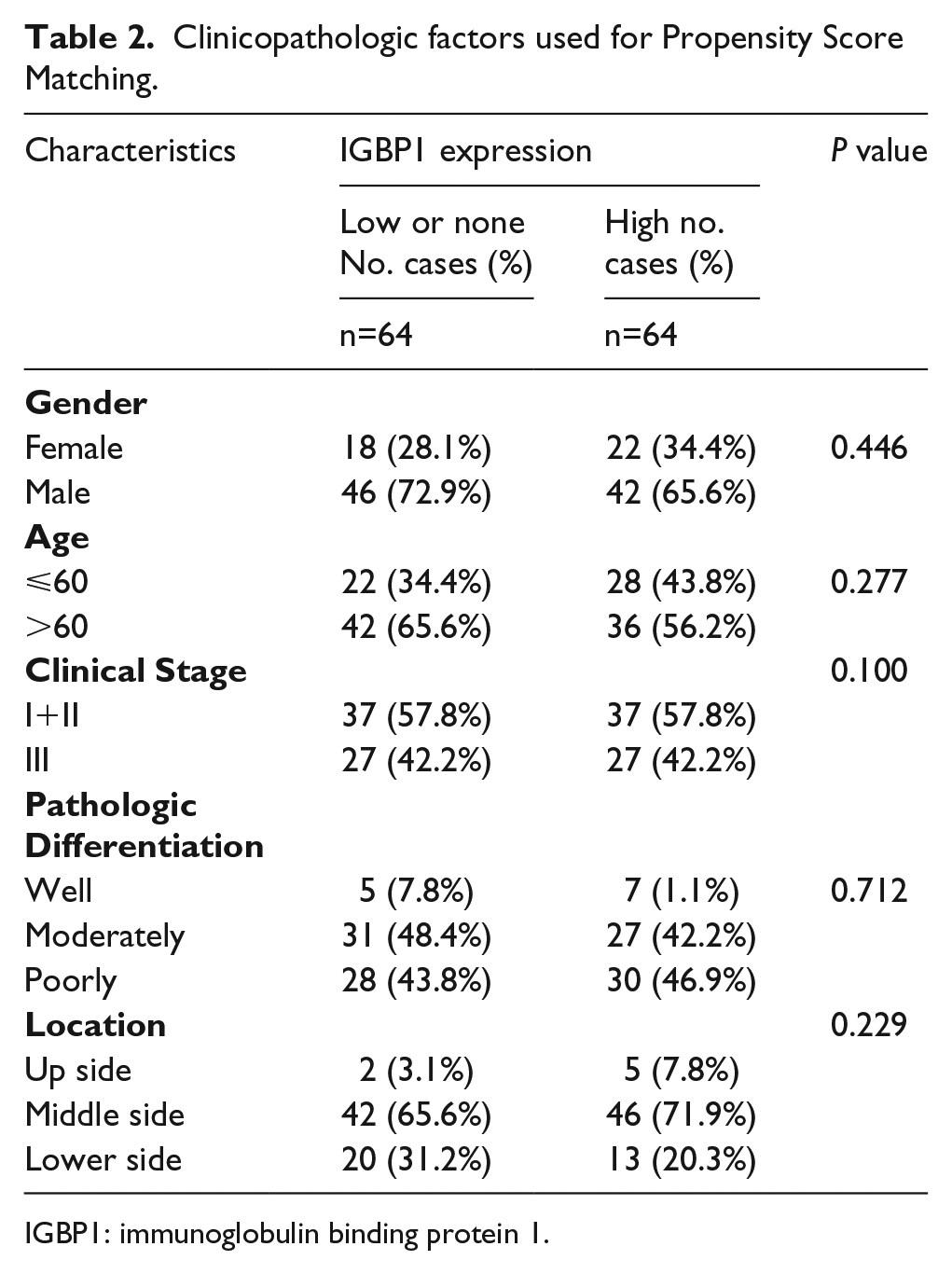
IGBP1: immunoglobulin binding protein 1.
As shown in Table 3, univariate analysis demonstrated that the factors significantly associated with overall survival of ESCC patients include IGBP1expression (P<0.05), T classification (P<0.01), N classification (P<0.01), clinical stage (P<0.01), and tumor differentiation (P<0.01). Importantly, IGBP1 expression, T classification, N classification, and tumor differentiation were independent prognostic factors for the overall survival of ESCC patients (P<0.05, respectively) by multivariate analysis, indicating that IGBP1 may be a useful biomarker for the prognosis of ESCC patients.
Univariate and multivariate analysis for overall survival (Cox proportional hazards regression model).
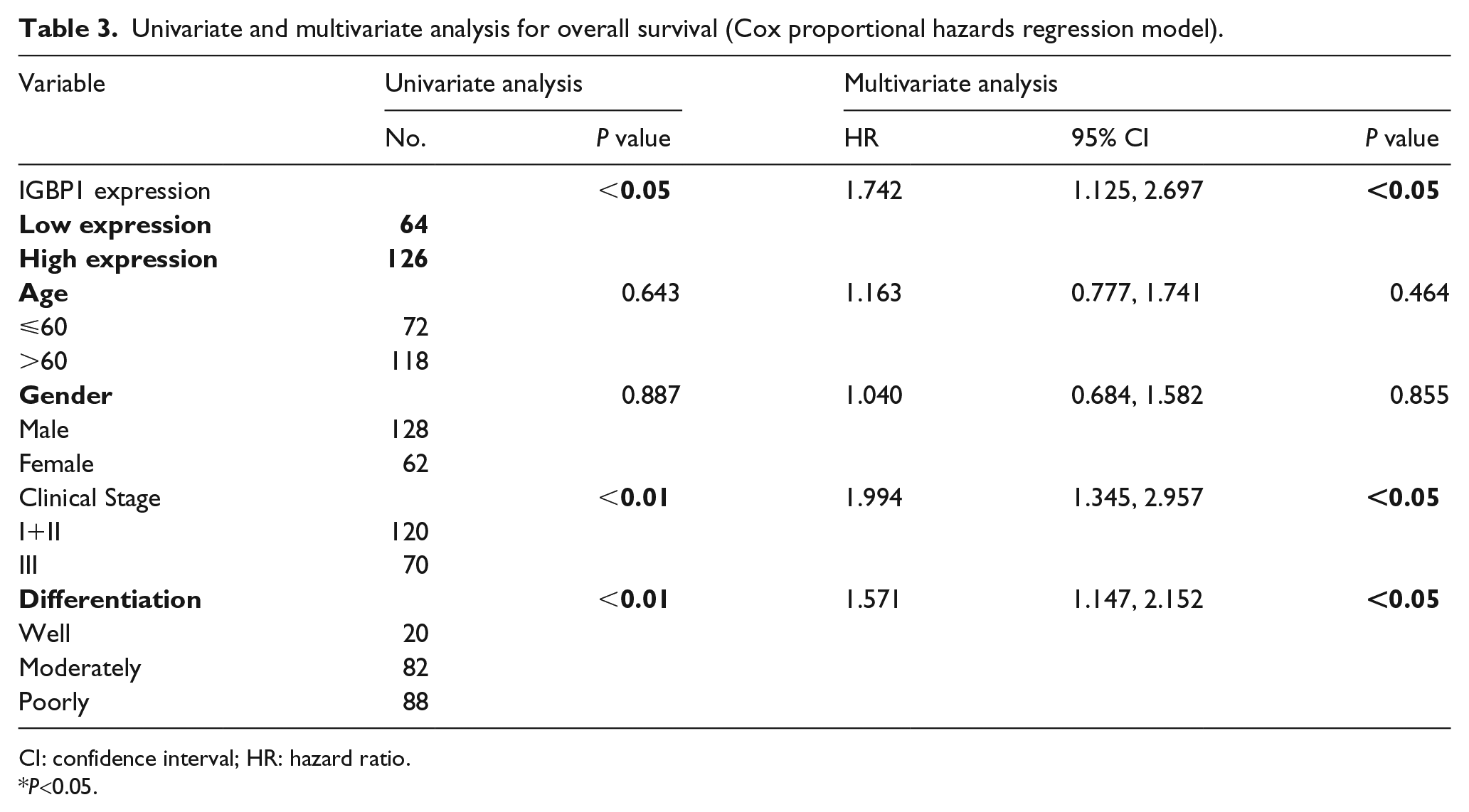
CI: confidence interval; HR: hazard ratio.
P<0.05.
Discussion
Esophageal cancer is one of the most common malignant cancers in the world, especially in China, where it is the fourth most common cause of cancer-related deaths, with an approximately 5-year survival rate of 17%.1,2 As the main pathological type of esophageal cancer, ESCC accounts for about 90% of cases of esophageal cancer worldwide and distributes a generally poor prognosis due to lack of a singular effective clinical method for early diagnosis.5,19 The still poor clinical outcome implies an urgent requirement to improve our recognition of the molecular mechanism underlying carcinogenesis of esophageal cancer, which may contribute to the development of novel prognostic methods. Accumulating evidence indicates that dysregulation of specific proteins is crucial for ESCC progression.6,20-23 Therefore, identification of novel biomarkers that can be utilized in early diagnosis and prognostic assessment to improve personalized therapy will help to decrease the mortality of ESCC patients and to improve clinical outcomes.
In this report, we investigated the potential for the IGBP1 expression level to be a clinical prognostic indicator for disease progression and patient survival in ESCC. We found that IGBP1 was highly expressed in ESCC tissue samples at the levels of both mRNA and protein, and that IGBP1 protein overexpression correlated with the T classification, vital status, and prognosis. Furthermore, the cumulative 5-year overall survival rate of patients with high IGBP1 expression was lower than those with low or undetectable IGBP1 expression. Therefore, patients with high IGBP1 expression have a poorer prognosis than those with low or absent IGBP1 expression, revealing IGBP1 as a potential independent prognostic factor for ESCC.
IGBP1, also known as alpha4, was identified as a non-redundant regulator of the dephosphorylation of the transcription factors c-Jun and p53, and is considered an essential inhibitor of apoptosis. Deletion of IGBP1 induced multiple proapoptotic gene transcriptions and promotes the process of apoptosis. An enhanced expression of IGBP1 protects cells from a variety of stress stimuli, such as DNA damage and nutrient limitation.8,13,24 The up-regulation of IGBP1 expression has been detected in human cancer including hepatocellular carcinomas, primary lung cancers, primary breast cancers and advanced lung adenocarcinomas. 13 Specially, its overexpression is significantly correlated with a poor prognosis of lung adenocarcinomas patients. It also has been reported that IGBP1 was downregulated by miR-3941 cell proliferation and apoptosis of lung adenocarcinoma at an early stage.14,15
In summary, the previous reports indicated that IGBP1 was essential for the growth of tumor cells; the increased expression of IGBP1 promoted the proliferation of, and inhibited the apoptosis of, cancer cells. The IGBP1 regulated a multiple variety of oncogenes and tumor suppressor genes. As we know, cancer patients are characteristic of heterogeneity and individual difference, and, at the same time, IGBP1 is expressed by other factors at the transcription and translation levels. Therefore, the expression of IGBP1 may be different among patients, which is the molecular basis for its prognostic value. Until now, the expression profile and prognostic significance of IGBP1 in other cancers remain largely unknown. In addition, more investigations are needed to explore the underlying molecular mechanism of the overexpression of IGBP1 in malignancies.
Our results have demonstrated that IGBP1 was highly expressed in ESCC tissues compared with the corresponding adjacent non-tumor tissue. Aberrant IGBP1 expressions were found to be significantly associated with ESCC survival. The ESCC patients with increased IGBP1 expression had a higher T stage, and those with higher IGBP1 expression had a shorter survival time than those with lower IGBP1 expression. These results were consistent with the hypothesis that IGBP1 is an oncogene previously reported by other groups. Esophageal cancers are highly aggressive tumors, which are commonly presented in a locally advanced stage with a poor prognosis and survival.25-27 The depths of invasion indicated as T classification is crucial to progression to the locally advanced stage in esophageal cancer patients. Thus, the patients with the characteristics of an elevated IGBP1 level can be defined as high-risk tumor phenotypes that require more aggressive primary surgery or adjuvant treatment following surgery. Assessments of illness should be given more closely in the postoperative follow-up in order to achieve early detection of recurrence and prompt treatment to improve therapeutic effects. However, while our studies offer some insight into the function of IGBP1 in ESCC, the underlying mechanism of IGBP1-mediated ESCC progression, and the functions of IGBP1 in malignant transformation, proliferation, and sensitivity of radiochemotherapy remain to be defined. We further propose that targeting IGBP1 may be a useful strategy for developing novel therapeutic techniques. Although ESCC has a poor prognosis, targeting patients’ specific molecular characteristics may improve the clinical outcome in the near future. As such, careful analysis of each patient’s specific tumor may lead to a personalized medicine approach.
Conclusions
In this study, we first showed that increased IGBP1 expression is markedly associated with the T classification and poorer clinical outcomes of patients with ESCC. IGBP1expression was revealed to be an independent prognostic factor for ESCC patients, suggesting that it may serve as a valuable and promising predictor of survival. Thus, testing the IGBP1 protein level may be useful for formulating the prognosis and for guiding the follow-up schedule in patients with ESCC. However, the detailed molecular mechanisms by which IGBP1 promotes tumor progression in ESCC requires further studies.
Footnotes
Acknowledgements
We thank the members of the laboratory for their helpful comments on the manuscript. This work was supported by the National Nature Science Foundation of China (NSFC) (81560403 to JianJun Tang) and the Natural Science Foundation of Jiangxi Province (20161BAB215254 to JianJun Tang).
Author contributions
Sicong Jiang and Daojing Li contributed equally to this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the National Nature Science Foundation of China (NSFC) (81560403 to JianJun Tang) and the Natural Science Foundation of Jiangxi Province (20161BAB215254 to JianJun Tang).
