Abstract
Background
The DNA mismatch repair system is one of the defense mechanisms in the body, and the inactivation of mismatch repair plays a pivotal role in secondary carcinogenesis and progression. However, the significance of mismatch repair in esophageal squamous cell carcinoma (ESCC) has not been established. In this study, we explored the diagnostic and prognostic significance of mismatch repair markers, mutL homologue 1 (MLH1), post-meiotic segregation increased 2 (PMS2), mutS homologue 2 (MSH2), and mutS homologue 6 (MSH6), in patients with ESCC.
Methods
We used a notation based on the proportion of immunoreactivity/expression for immunohistochemistry (PRIME notation), which allows the comparison of mismatch repair expression by assigning a score to PRIME notation. MLH1, PMS2, MSH2, and MSH6 were examined immunohistochemically in 189 surgically resected ESCC specimens.
Results
A total of 100/189 patients with ESCC (53%) received preoperative chemotherapy. The rates of ESCC cases with decreased mismatch repair status were 13.2%, 15.3%, 24.8%, and 12.6% for MLH1, PMS2, MSH2, and MSH6, respectively. The decreased status of individual mismatch repair markers was significantly correlated with worse prognosis in patients with ESCC. Additionally, MSH2, MSH6, and PMS2 were significantly associated with response to preoperative chemotherapy. Multivariate analysis revealed that MLH1, PMS2, and MSH2 are independent prognostic factors.
Conclusion
Our results suggest that mismatch repair is a prognostic biomarker for ESCC and could contribute to the selection of appropriate adjuvant therapy for patients with ESCC.
Keywords
Introduction
Esophageal cancer is one of the most aggressive malignancies of the gastrointestinal tract. Recently, remarkable advances have been reported in surgical techniques and adjuvant management, but their clinical outcomes have remained relatively poor, with 5-year survival rates of 15–25% worldwide. 1 Esophageal squamous cell carcinoma (ESCC) is the most common histological type in Japan and East Asia, accounting for more than 90% of all esophageal cancers. Surgery, chemotherapy, and radiation therapy are the main modalities used for ESCC treatment. Immune checkpoint inhibitors (ICIs) have recently emerged as a new therapeutic strategy for ESCC. For instance, results of the KEYNOTE-181 study, a phase III clinical trial in patients with previously treated advanced/metastatic esophageal cancer, demonstrated the clinical superiority of pembrolizumab, an anti-programmed death 1 (PD-1) immune checkpoint inhibitor, compared to chemotherapy as a second-line treatment for patients with programmed death ligand-1 (PD-L1) combined positive score (CPS) of ≥ 10. 2 Mismatch repair deficiency (dMMR) was also reported to be correlated with PD-1 blockade, 3 and results of the KEYNOTE-158 study demonstrated the clinical benefits of pembrolizumab as a second-line treatment for patients with dMMR/high-level MSI (MSI-H) cancer in 27 different histological types of human malignancies. 4 Therefore, mismatch repair (MMR) markers are becoming increasingly significant in clinical diagnoses of various oncologic areas, but their roles in ESCC remain unclear.
Inactivation of DNA MMR genes is a major cause of microsatellite instability (MSI) and a pivotal factor in cancer development. Dysfunction of MMR genes causes DNA replication errors in tumor suppressor genes harboring microsatellites in their coding regions, and inactivation of key tumor suppressors is also a major cause of initiation and promotion of tumorigenesis. 5 The MMR system usually involves four key genes: mutL homologue 1 (MLH1), post-meiotic segregation increased 2 (PMS2), mutS homologue 2 (MSH2), and mutS homologue 6 (MSH6)—all of which have been reportedly detected using immunohistochemistry (IHC). 6 Loss of MMR proteins is considered a hallmark of dMMR tumors as well as MSI-H. MSI-H/dMMR tumors have a relatively large number of neoantigens owing to increased point mutations, which could serve as potential targets for host immune cells. 7 Anomalies in MMR genes have been reported in various human malignancies, including colorectal, gastric, breast, ovarian, endometrial, and Merkel cell carcinomas.8–13 Hypermethylation of MLH1 and MSH2 promoter regions in ESCC was reported at rates of 43% and 28%, respectively, and found to be associated with a loss of MMR expression. 14 Decreased expression of MLH1 in ESCC was reported to be associated with PD-L1 expression in tumor cells and reduced response to preoperative chemotherapy. 15 However, clinical data are still lacking pertaining to the role of dMMR in the oncologic progression of ESCC and its association with patient prognosis.15–18
In general, IHC in MMR studies the loss of corresponding immunoreactivity compared to internal controls, such as normal epithelium or lymphocytes. When the complete loss of MMR is detected by IHC, dMMR can be easily determined; however, when partial or incomplete immunoreactivity is detected, its interpretation is not necessarily standardized, and its diagnostic significance remains unknown. The College of American Pathologists (CAP) guidelines state that MMR status in gastroesophageal adenocarcinoma, small bowel, colorectal, and endometrial cancers should be presented as MMR-preserved (pMMR) or dMMR. 19 However, an optimal approach for evaluating MMR loss in ESCC is yet to be established; thus, the association between its expression patterns in IHC and patient prognosis remains virtually undetermined. Therefore, in this study, we tentatively devised a method allowing the comparison of MMR status by assigning the score to the proportion of immunoreactivity/expression (PRIME) notation, as previously reported, 20 which could incorporate both staining intensity and proportion in the final result. This study aimed to determine the impact of MMR status obtained by IHC on the clinical outcomes of patients with ESCC.
Methods
Patients and tissue specimens
Surgically resected specimens were obtained from 189 patients who were diagnosed with ESCC at Tohoku University Hospital between 2009 and 2016. One hundred (53%) patients received preoperative chemotherapy according to the Japanese Clinical Oncology Group 9907 (JCOG9907) protocol, 21 which was performed in conjunction with intravenous administration of cisplatin (80 mg/m2 per day on days 1 and 22) and continuous intravenous administration of 5-fluorouracil (800 mg/m2 per day on days 1–5 and 22–26). All patients underwent thoracoscopic subtotal esophagectomy with regional lymph node dissection, and all surgical specimens were pathologically classified according to the 8th Union for International Cancer Control tumor node metastasis staging system for esophageal carcinoma. 22 Pathological criteria for the efficacy of preoperative chemotherapy were defined according to the 11th Japanese Classification of Esophageal Cancer as follows: Grade 0, ineffective (no pathological effect in the lesion); Grade 1a / Grade 1b, slightly effective (Grade 1a, pathological disappearance of tumor cells in less than one-third of the total lesion area; Grade 1b, one-third to two-thirds of the total lesion area); Grade 2, moderately effective (viable tumor cells are histologically detected in less than one-third of the total lesion area); Grade 3, markedly effective (no viable tumor cells). 23 Grade 0–1a were defined as ineffective for preoperative therapy, while Grade 1b–2 were considered effective.24,25 The starting point for patient survival was the date of the surgery. The endpoints for overall survival (OS) rates were patient death and the last censor.
The study protocol was in accordance with the guidelines stated in the Declaration of Helsinki and was approved by the Ethics Committee of the Tohoku University School of Medicine (Accession No. 2020-1-87). Written informed consent was obtained from all patients.
IHC
Tissue specimens were fixed in 10% neutral formalin and embedded in paraffin. Representative tissue blocks containing the invasive front and viable tumor cells were selected for further immunohistochemical analysis. Tissue sections (4 µm thick) were automatically pretreated in the PT-link system (Agilent Technologies, Santa Clara, CA, USA) with heat-induced epitope retrieval (HIER) for 20 min at 97°C. IHC was performed in an automated immunostainer (Dako Autostainer Link 48, Agilent Technologies, Santa Clara, CA, USA) according to the manufacturer's instructions (EnVisionTM FLEX Kit Agilent Technologies, Santa Clara, CA, USA), with ready-to-use monoclonal antibodies. Detailed information regarding immunohistochemical procedures, such as the primary antibodies, control tissues, antigen retrieval methods, and buffers used in this study, are summarized in Supplemental Table 1.
IHC evaluation
We used PRIME notation to evaluate immunoreactivity/expression (IE). In principle, this method is applied only to neoplastic cells, and the subsequent interpretation is performed under an optical microscope without using counting instruments. Each stained tissue section was independently evaluated by two observers (TY and FF) without knowledge of any clinicopathological parameters of the patients, and the entire image of the specimen was carefully evaluated. The basic IE pattern was described with two positive patterns, such as “+,” “(weak) +,” and one negative pattern “−”. Normal epithelium or lymphocytes were used as internal controls during IHC, and the boundary between “+” and “(weak) +” was defined. In addition to the basic patterns, “P/Q” representation was adopted for the compound IE patterns. Approximately equal P and Q values were selected from the three basic patterns and connected with “/” to form a new single unit, resulting in the following three patterns: “+ / (weak) +,” “ + / −,” and “(weak) + / −”. The P/Q patterns were described as a single unit as well as other basic patterns.
According to the flowchart (Figure 1), first (“start”), we evaluated whether the IE pattern was significantly above 50%. The IE pattern was selected from the basic and composite patterns described above (six patterns in total). When IE patterns were > 50%, one of (I), (II), or (III) was selected according to the number of IE patterns. When the IE pattern was approximately 50%, (IV) was selected, and when it was less than 50%, (V) was selected. Therefore, IHC findings were tentatively categorized into any of (I)–(V); (I) contained only IE patterns with 100% reactivity, and (II)–(V) involved the proportion of tumor cells demonstrating various IE patterns. The numbers on the right shoulder of (II)–(V) indicate the notations below them in order from the top. These were described using the range of percentages, inequalities, and symbols as described hereunder, and finally “Finish”.
The proportion of either IE pattern was described using “>,” “/,” or “//.” Inequality of “>” (with the left IE pattern always being greater than the one on the right), single slash “/” (representing adjacent IE patterns are nearly equal) or double slash “//” (used when the notation has three IE patterns and only the left side is around 50%). For “/” we describe a stronger IE pattern on the left side. “[ ]” is used for less than 1%. aX% = around x% = ([X−4] to [X + 4])%
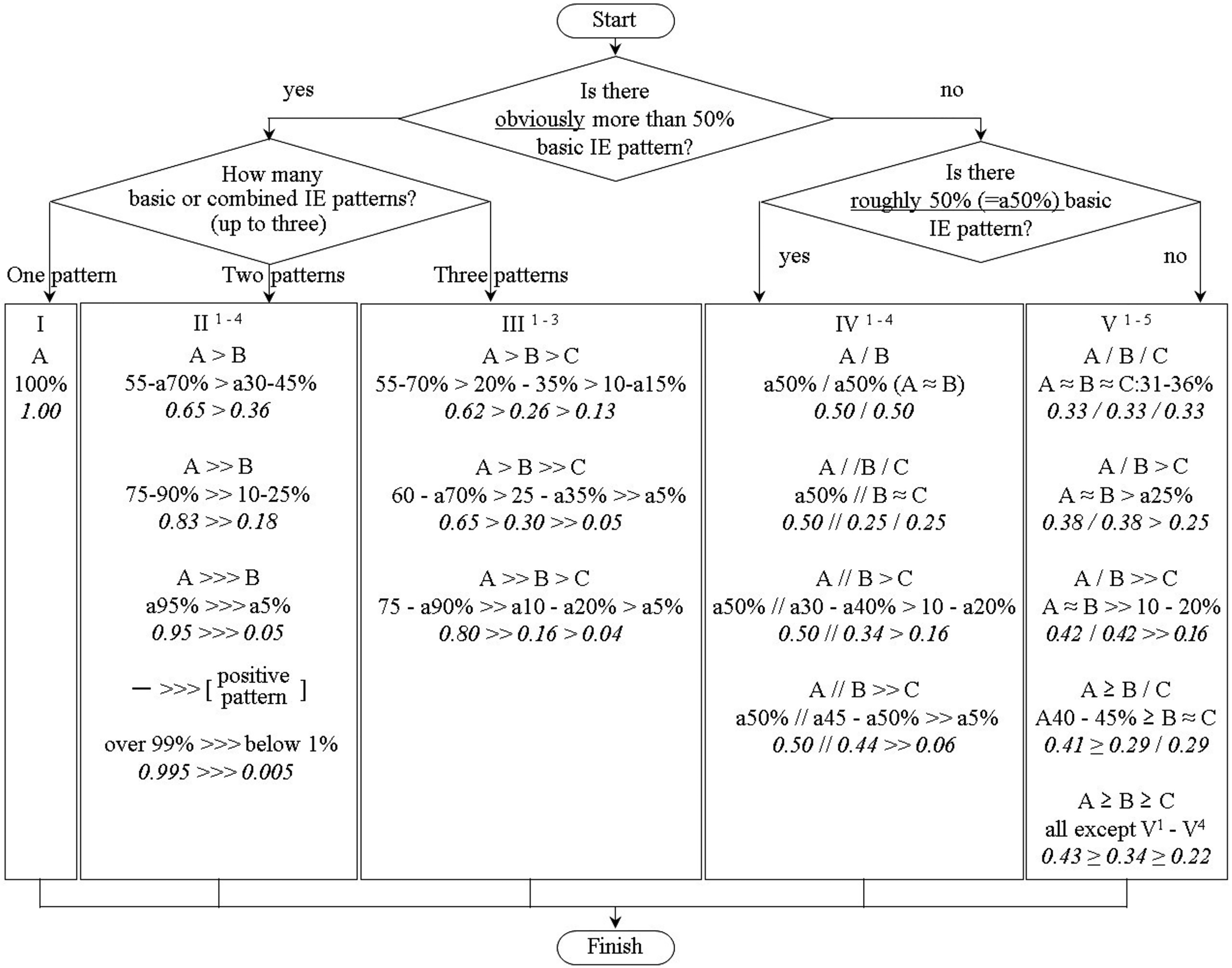
Flowchart of PRIME notation of dividing immunohistochemical findings into a total of 17 basic notations. The italic numbers describe “scoring multipliers” for conversion of PRIME notation to a single score (PRIME score).
A flowchart of the PRIME notation dividing immunohistochemical findings into a total of 17 basic notations. The italic numbers represent the “scoring multipliers” for the conversion of PRIME notation to a single score (PRIME score).
Subsequently, the PRIME notation was converted to a single score (PRIME score) as described below.
1. Basic IE pattern of A, B, or C, namely “+,” “(weak) +” and “−” were assigned scores 10, 6, and 0, respectively.
2. The scores of A, B, and C in each notation pattern were multiplied by “multiplier” described by the italicized numbers (shown in Figure 1) to obtain the product score for each of A, B, or C.
3. The multiplier was obtained by dividing the average population numbers (A, B, or C, respectively) of all 763 notation patterns with all integer values (data not shown) by 100.
4. Then, the total score obtained by summing the product scores of A, B, or C was used as the representative PRIME score, ranging from 0 to 10.
5. Finally, a PRIME score of less than 8 was defined as low expression and greater than 8 as high expression.
Statistical analysis
All statistical analyses were performed using EZR version 1.38. 26 The correlation between clinicopathological variables and immunoreactivity obtained by IHC was analyzed using Fisher's exact test. We subsequently performed rigorous adjustment by propensity score matching for significant differences in baseline characteristics between the retained and decreased expression groups for each MMR protein. Multivariate logistic regression analysis was also used to estimate the propensity score of these patients, and the following five variables were used in each model: age, gender, preoperative treatment, pathological tumor (pT), and pathological lymph node (pN). Subsequently, two-to-one matching was performed using the nearest-neighbor matching method with a standard deviation, and two new groups were obtained for each MMR gene. OS curves were estimated according to the Kaplan–Meier method, and the differences between the two groups were analyzed using the log-rank test. Univariate and multivariate analyses were performed using the Cox proportional hazard model. Statistical significance was set at P < 0.05.
Results
IHC staining patterns of MMR
Representative microscopic images of MMR are illustrated in Figure 2. Details of the PRIME notation are summarized in Supplemental Table 2. A distribution chart and histogram of the PRIME notation scores are presented in Supplemental Figure 1-2. Diffusely positive patterns constituted most of each MMR marker, but weakly positive, incompletely positive, or negative patterns were also detected in some cases. The weak or partial staining patterns of MMR are shown in Figure 3.
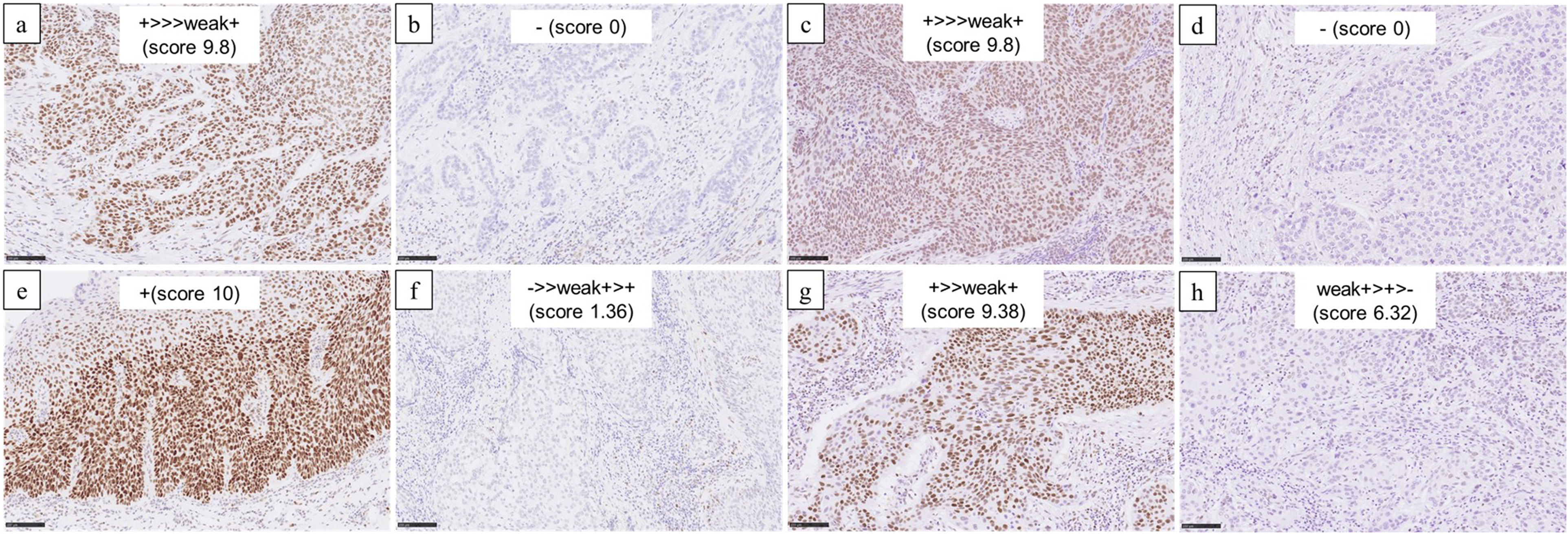
Representative microscopic images of DNA mismatch repair (MMR). (a) High and (b) low immunoreactivity of mutL homologue 1 (MLH1). (c) High and (d) low immunoreactivity of post-meiotic segregation increased 2 (PMS2). (e) High and (f) low immunoreactivity of mutS homologue 2 (MSH2). (g) High and (h) low immunoreactivity of mutS homologue 6 (MSH6).
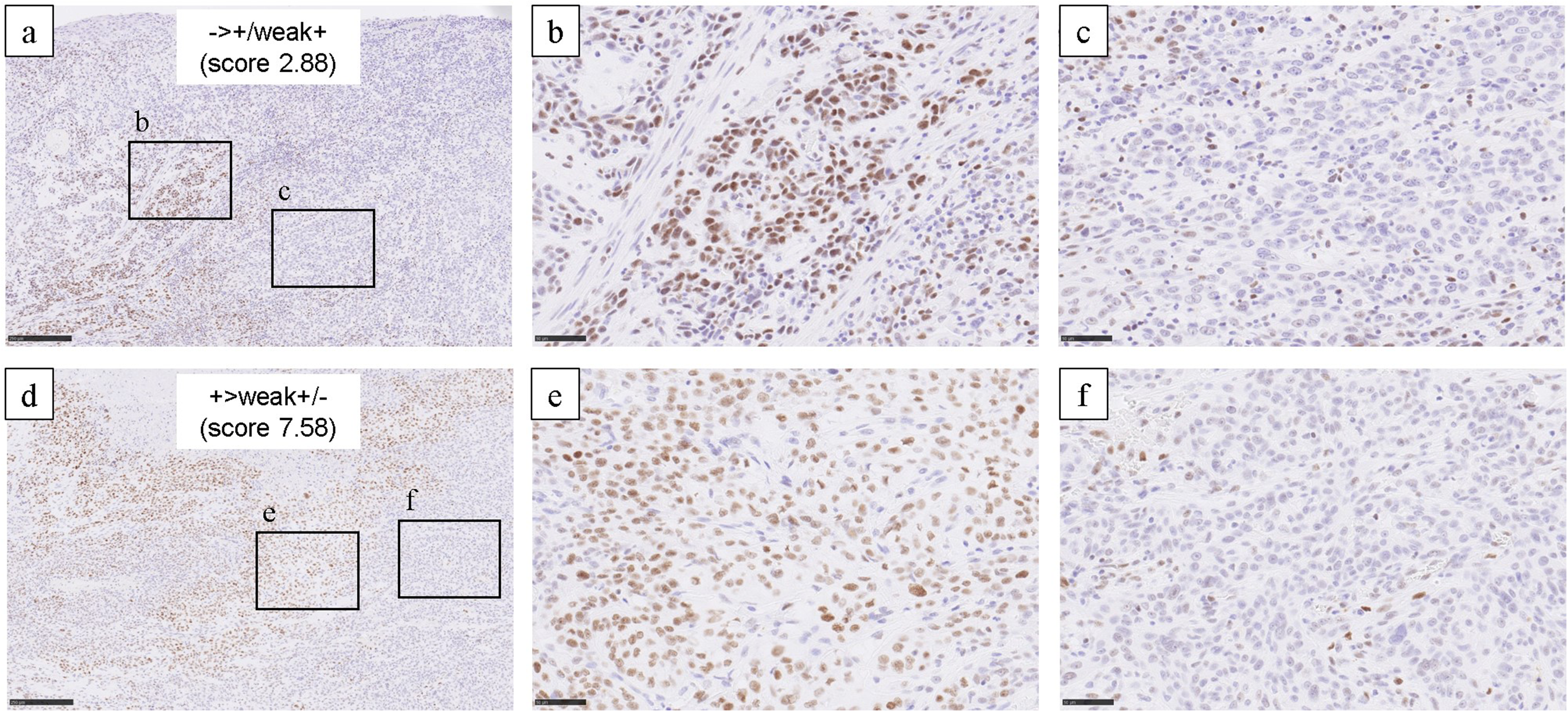
Weak or partial staining pattern of MSH2. (a) (d) Low power field of tumor showing partial positive staining of MSH2. (a) The most dominant staining pattern was “−”, with “+” and “weak+” both present in a single tumor. (b) Tumor cells showed a “+” or “weak+” pattern. (c) Tumor cells mainly showed “−” pattern. (d) The most dominant staining pattern was “+,” with “weak+” and “−” both present in a single tumor. (e) Tumor cells show a “+” or “weak+” pattern. (f) Tumor cells mainly demonstrated “−” pattern.
Representative microscopic images of DNA MMR. (a) High and (b) low immunoreactivity of mutL homologue 1 (MLH1); (c) high and (d) low immunoreactivity of post-meiotic segregation increased 2 (PMS2); (e) high and (f) low immunoreactivity of mutS homologue 2 (MSH2); and (g) high and (h) low immunoreactivity of mutS homologue 6 (MSH6).
Weak or partial staining pattern of MSH2. (a) (d) Low power field of tumor showing partial positive staining of MSH2. (a) The most dominant staining pattern was “−”, with “+” and “weak+” both present in a single tumor. (b) Tumor cells showed a “+” or “weak+” pattern. (c) Tumor cells mainly showed “−” pattern. (d) The most dominant staining pattern was “+,” with “weak+” and “−” both present in a single tumor. (e) Tumor cells show a “+” or “weak+” pattern. (f) Tumor cells mainly demonstrated “−” pattern.
MMR Status and its correlation with clinicopathological variables
We examined the correlations between the immunoreactivity of MMR proteins and clinicopathological variables of the patients (Table 1). Decreased MLH1 expression (score < 8, detected in 13.2% [25/189]) was significantly correlated with pT status (P = 0.048). Decreased PMS2 expression (score < 8, detected in 15.3% [29/189]) was significantly correlated with pT status (P = 0.038) and histopathological response (P = 0.036). Decreased expression of MSH2 (score < 8, 24.8% [47/189]) was significantly correlated with pT status (P < 0.001), pStage (P = 0.006), lymphatic invasion (P = 0.031), and histopathological response (P = 0.001). Decreased expression of MSH6 (score < 8, detected in 12.6% [24/189]) was significantly correlated with gender (P = 0.021), pT status (P = 0.001), pStage (P = 0.032), preoperative therapy (P = 0.028), and histopathological response (P = 0.001).
Status of MMR markers and their correlation with clinicopathological factors of patients with ESCC.
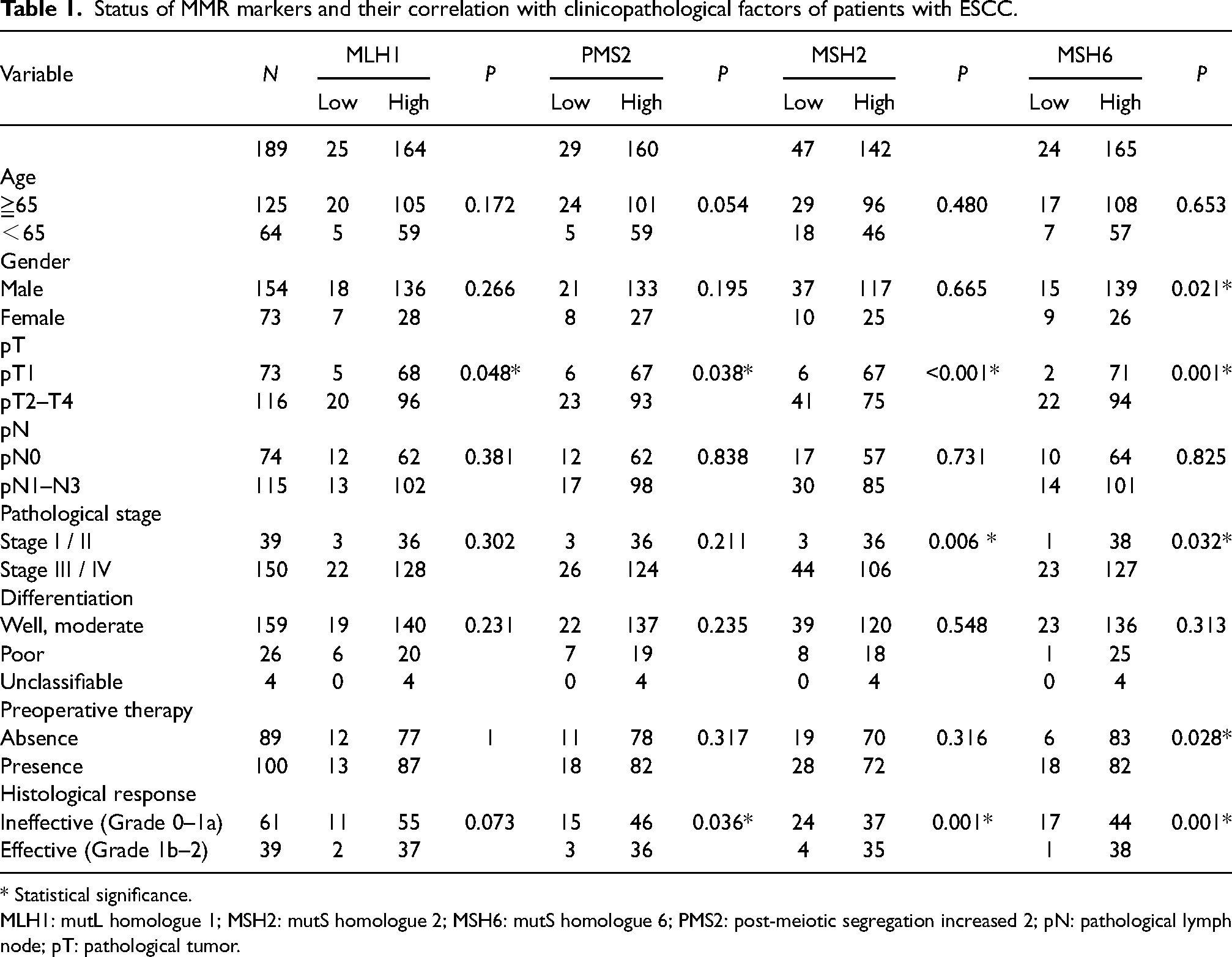
* Statistical significance.
MLH1: mutL homologue 1; MSH2: mutS homologue 2; MSH6: mutS homologue 6; PMS2: post-meiotic segregation increased 2; pN: pathological lymph node; pT: pathological tumor.
MMR status and its correlation with clinical outcomes of patients with ESCC
We performed survival analysis using Kaplan–Meier curves according to MMR status. As mentioned above, 2:1 propensity score matching was performed to reduce the differences in baseline characteristics between the proficient and decreased expression groups, and two new groups were obtained for each MMR marker (Supplemental Table 3). In all four MMR genes, decreased status was significantly correlated with worse 5-year OS (Figure 4). Multivariate analysis also revealed that the OS rate was significantly associated with a decreased MLH1, PMS2, and MSH2 status. These biomarkers were identified as independent prognostic factors, from clinicopathological variables, including “age, gender, pT, pN, and preoperative chemotherapy” (Supplemental Table 4).
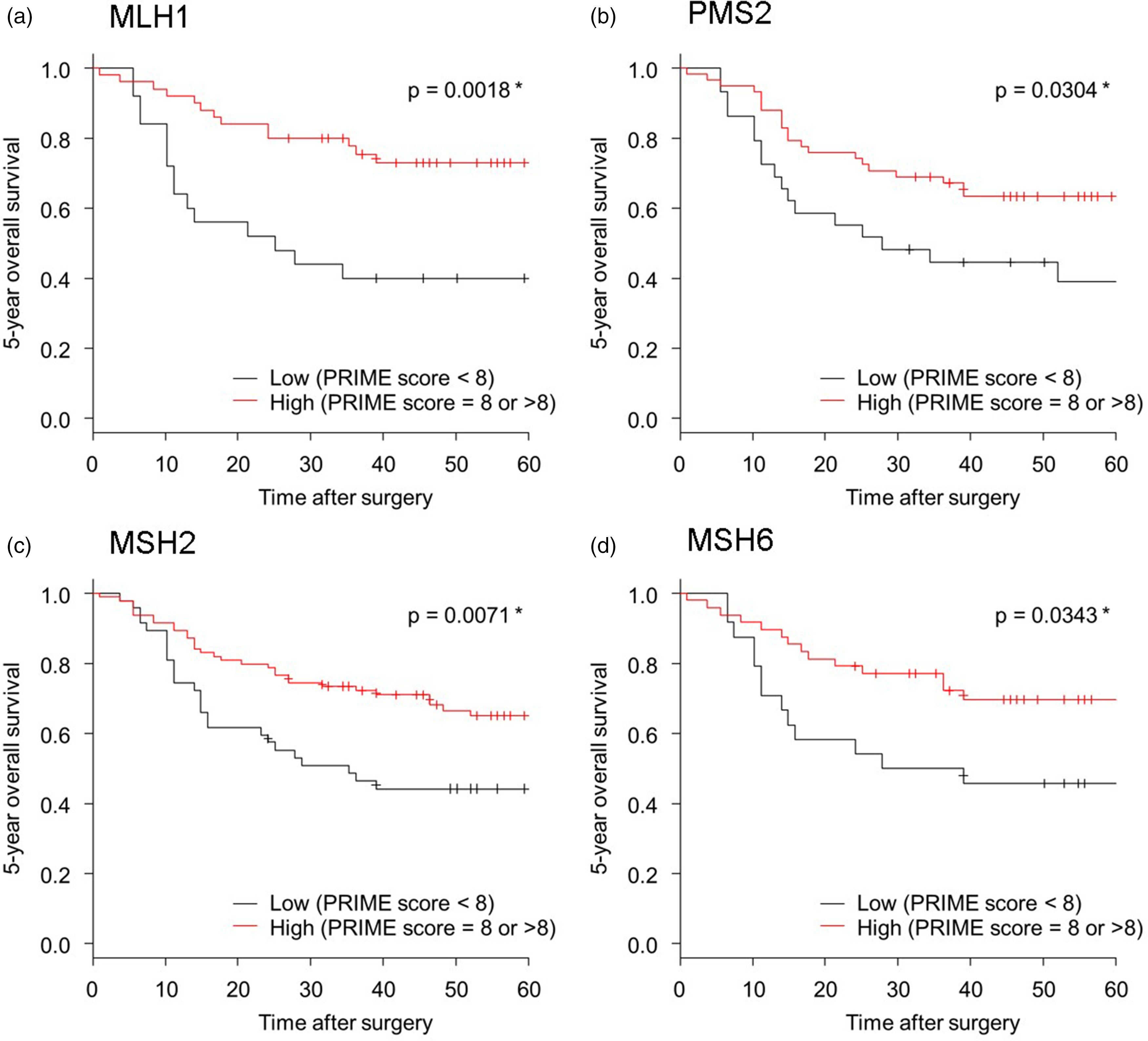
Kaplan–Meier curves according to MMR status of the patients with ESCC. Propensity score matching was performed to reduce the differences in baseline characteristics between the two groups with low and high expression of each MMR marker. The 5-year OS was significantly worse in patients with low expression of any of the MMR markers than in those with proficient MMR.
Kaplan–Meier curves according to MMR status of the patients with ESCC. Propensity score matching was performed to reduce the differences in baseline characteristics between the two groups with low and high expression of each MMR marker. The 5-year OS was significantly worse in patients with low expression of any of the MMR markers than in those with proficient MMR. MMR, mismatch repair; OS, overall survival; DFS, disease-free survival.
Discussion
This is the first study to comprehensively evaluate the status of the four representative MMR proteins in ESCC. Notably, low expression of MLH1, PMS2, MSH2, and MSH6 was significantly associated with poor prognosis of patients with ESCC. Consistent results have not been reported regarding the association between MMR status and the clinical course of patients with ESCC in previously reported studies; therefore, in this study, we comprehensively evaluated the immunoreactivity of each marker based on PRIME notation, which incorporated both intensity and the proportion of immunostained tumor cells as evaluation criteria. Some researchers defined “MLH1 Low” as cases showing weaker staining or loss of staining compared to those in the endogenous controls, and the frequency of “MLH1 Low” was 30–36% according to this definition.15,17 In this study, MMR was evaluated using the PRIME notation, and 13% of all cases were defined as MLH1 low. It is difficult to compare these results with previously reported findings; however, we may have set a stricter threshold in the current study than in previous studies. While previous studies evaluated tumor cells without distinguishing between weak and negative staining, our method assigned separate scores to weak and negative staining and incorporated them into the overall evaluation. Recent genomic analyses have revealed that ESCC has complex heterogeneity at the genomic and epigenomic levels. 27 These heterogeneities have a significant impact on tumor progression, metastasis, and resistance to therapy, but they also complicate the findings of IHC in the intertumoral or intratumoral forms, confounding their interpretation. The method used in this study to assign a score to PRIME notation is compatible with a wide variety of IHC findings and could be useful in the evaluation of potentially heterogeneous tumors.
In general, loss of immunoreactivity of MMR proteins was evaluated in IHC analysis by comparing the findings to those of internal controls, such as the normal epithelium of lymphocytes. However, in this study, weak or partial immunoreactivity of MMR proteins was occasionally detected, and the frequency of ESCC cases in which weak + was the most common immunolocalization pattern was as follows: MLH1, 3.7% (7/189); PMS2, 4.7% (9/189); MSH2, 2.6% (5/189); and MSH6, 4.7% (9/189). In colorectal cancer, some carriers of genetic mutations of MLH1 yielded weak or partial immunoreactivity in IHC studies 28 ; however, the significance of their incomplete immunoreactivity remains virtually unknown. In previous studies on esophageal cancer, the methodologies of discerning or interpreting weak or partial immunoreactivity were not necessarily consistent. This could have contributed to the discrepancy in reported findings, but further investigations are required to confirm this.
MMR is a defense mechanism that repairs base sequence mismatches during DNA replication. In addition, the inactivation of MMR genes is one of the causes of secondary genetic mutations leading to tumor development and progression. The MMR system also functions as a checkpoint for DNA-damaged cells and acts as an initiator of apoptotic signaling in response to DNA damage induced by specific types of chemotherapeutic drugs, such as cisplatin. 29 If the MMR system is disrupted, effective apoptosis could be compromised and response to chemotherapy is likely to be diminished. 30 Therefore, MMR loss is considered one of the causes of chemotherapeutic resistance. 5 Dysfunction of the MMR system also allows cancer cells with damaged DNA to proliferate, and damaged DNA can be inherited as genetic mutations that contribute to further malignant transformation. 17 In this study, we demonstrated that a decreased MMR protein status was significantly correlated with poor prognosis and response to preoperative chemotherapy in patients with ESCC. These findings also suggest that dysfunction of the MMR system may contribute to the progression of ESCC and help cancer cells resist chemotherapy. In summary, IHC of MMR markers could be clinically useful as a prognostic biomarker and contribute to better clinical outcomes by identifying patients who are at high risk for recurrence, guiding appropriate adjuvant therapy and genetic testing.
This work has certain limitations in the study design. We evaluated the expression status of MMR with a novel method termed PRIME notation and its score. However, we did not perform molecular biological analysis, such as for MSI. Therefore, further investigations are required to clarify the functional correspondence between IHC findings and MMR. This is a single-center, retrospective study with a relatively small sample size; further investigation is needed to confirm whether the assessment of IHC findings by PRIME notation can be performed with high reproducibility among multiple centers and researchers.
In conclusion, our study demonstrated that decreased MMR expression is substantially associated with clinical outcomes of patients with ESCC. MMR evaluated by PRIME notation could serve as a potential prognostic biomarker and allow us to select the appropriate adjuvant therapy for patients with ESCC.
Supplemental Material
sj-docx-1-jbm-10.1177_03936155231165068 - Supplemental material for MMR markers correlate with clinical outcome in patients with esophageal squamous cell carcinoma
Supplemental material, sj-docx-1-jbm-10.1177_03936155231165068 for MMR markers correlate with clinical outcome in patients with esophageal squamous cell carcinoma by Takuro Yamauchi, Fumiyoshi Fujishima, Junichi Tsunokake, Atsushi Kunimitsu, Ryujiro Akaishi, Yohei Ozawa, Toshiaki Fukutomi, Hiroshi Okamoto, Chiaki Sato, Yusuke Taniyama, Takashi Kamei, Ryo Ichinohasama and Hironobu Sasano in The International Journal of Biological Markers
Supplemental Material
sj-docx-2-jbm-10.1177_03936155231165068 - Supplemental material for MMR markers correlate with clinical outcome in patients with esophageal squamous cell carcinoma
Supplemental material, sj-docx-2-jbm-10.1177_03936155231165068 for MMR markers correlate with clinical outcome in patients with esophageal squamous cell carcinoma by Takuro Yamauchi, Fumiyoshi Fujishima, Junichi Tsunokake, Atsushi Kunimitsu, Ryujiro Akaishi, Yohei Ozawa, Toshiaki Fukutomi, Hiroshi Okamoto, Chiaki Sato, Yusuke Taniyama, Takashi Kamei, Ryo Ichinohasama and Hironobu Sasano in The International Journal of Biological Markers
Footnotes
Acknowledgements
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
