Abstract
Background
Methods
Results
Conclusions
Keywords
Introduction
Colorectal cancer (CRC) is a leading cause of mortality in developed countries (1). About 20% of CRC patients are initially diagnosed with metastases, which represent the main cause of death (1). The evolution of CRC from normal colonic epithelium to the malignant phenotype starts from polyps, progressing to adenomas and carcinomas. During the progression of the adenoma, a degree of dysplasia and of villous histology occurs along with genetic changes such as the induction of the k-ras oncogene followed by a loss of the p53 gene at the late stages of the disease (2). Early detection of CRC represents the best chance of overcoming the disease. At present, invasive tests such as flexible sigmoidoscopy and colonoscopy are used for early detection. Colonoscopy, which is performed in developed countries for screening populations over the age of 50 years, is the most common form of early diagnosis, despite its miss rates have been estimated to be 22% for adenomas and 2%-6% for CRC (3). Regarding the non-invasive tests, the most common method recommended for CRC screening, is the guaiac fecal occult blood test (gFOBT), characterized by highly variable and brand-dependent sensitivities and specificities (4). In the last decade, a number of potential tumor markers have been characterized in CRC. Lower soluble serum CD26 levels were detected in CRC patients as compared to healthy donors (5). Additionally, higher levels of kininogen-1, also known as alpha-2-thiol proteinase inhibitor, have been found in CRC patients compared to adenoma patients (6). Further differences in terms of plasmatic characteristics between patients with adenomas and with carcinomas (but not healthy controls) were described for apolipoproteins' regulation patterns and for the upregulation of the cytokines interleukin-8, and tumor necrosis factor-α (TNF-α) (7).
Glucose-regulated protein 78 (GRP78) is a well-characterized protein that belongs to the family of the heat shock protein 70 (HSP70). GRP78 is primarily resident in the ER, where it serves as a regulator of the UPR. Disturbance of ER homeostasis, by adverse physiological stress or pharmacological conditions, triggers a protective UPR, which induces the activation of GRP78. Activation of the intracellular GRP78 was found to result in protein expression on the cell surface (8). Furthermore, overexpression of GRP78 is correlated with an aggressive phenotype and a poor prognosis in prostate, breast, hepatocellular, and gastric cancers (9). The cell surface-associated form of GRP78 was identified in some tumors (prostate cancer, ovarian cancer, and melanoma), where it functions as a signaling receptor that promotes proliferation and survival (9). Cell surface GRP78 in cancer cells triggers the production of anti-GRP78 autoantibodies. An increase of anti-GRP78 antibodies in the serum of cancer patients correlated positively with prostate cancer progression and shorter overall survival. The effect of anti-GRP78 IgGs was evaluated on 1-LN, PC-3, DU145, and LnCap prostate cancer cells. Anti-GRP78 antibodies from prostate cancer patients bind to and stimulate the proliferation of tumor cells expressing GRP78 on their surface, and protect them from apoptosis induced by TNF-α. However, other effects of these autoantibodies, such as antibody-dependent cellular cytotoxicity and complement-dependent cytotoxicity, could also play a role in the biology of tumors in these patients (10, 11).
In this study we screened 85 patients, diagnosed by colonoscopy, for anti-GRP78 antibodies in serum, and correlated these results to those obtained by colonoscopy. This was aimed at searching a new biomarker for the early detection of CRC.
Methods
Patients
The study protocol was approved by the Helsinki Human Research Ethics Committee of the Rabin Medical Center, Beilinson Hospital, Petah Tikva, Israel. Participants gave written consent for participation and for the investigators to obtain their medical records. Patients' serum was collected before colonoscopy and kept at -80ºC. Clinical data were collected from medical charts, as well as colonoscopy and pathology reports. The patients' cohort included 85 cases diagnosed during 2006 at the Department of Gastroenterology by laparoscopy. Thirty patients were diagnosed as healthy (without signs of disease or polyps), 28 patients were diagnosed with polyps, and 27 patients with colorectal carcinoma. The latter condition was confirmed by pathology evaluation. The cohort was composed of 43 males and 42 females, aged 35-91 years (mean, 64.4±3.6 years). No significant differences in either gender or age were observed among the groups. The data for each group is summarized in Table I.
PATIENTS' GROUPS BASED ON COLONOSCOPY ANALYSIS
Determination of Anti-GRP78 Antibody in Patients' Serum by ELISA
We coated 96-well high-protein-binding ELISA plates (Corning, NY, USA) with 5 µg/mL recombinant GRP78 diluted in 0.1 mol/L Na2CO3 and 0.01% NaN3 (pH 9.3), one day before the assay. Plates were blocked with PBS-10% BSA for 1 hour at room temperature. A total of 200 µL of serum was defrosted immediately before assay. Serum was diluted in PBS (1:10, 1:100, and 1:1000) before use and maintained on ice. A total of 50 µL/well of the diluted serum were placed in triplicates in the blocked, coated plates for 2 hours at 37ºC, followed by 2 washes with PBS. A secondary anti-human IgG biotinylated antibody (1 µg/well) (BethylLaboratories, Texas, USA) was added for 2 hours at room temperature. After intensive washes with PBS, streptavidin HRP (R&D Systems, Minneapolis, USA) was added to the plates for 1 hour at room temperature. One-step TMB substrate (eBioscience, California, USA) was added at 100 µL/well until color developed (approximately 15 minutes). In order to stop the reaction, 50 µL/well of HCl 1M were added. The plates were read at 450 nm in an ELISA reader, and the results are here presented in arbitrary numbers ± SEM. The correlation between clinical parameters and titer of anti-GRP78 antibodies are presented as mean ± SEM.
Statistical Analysis
All values are expressed as mean ± SEM. Differences and cutoff determination were compared using the ANOVA mutivariate test. Correlations between the levels of anti-GRP78 antibodies and clinical parameters were analyzed by the Spearman correlation coefficient. P values <0.05 were considered statistically significant.
Results
Anti-GRP78 Antibody in Serum
We first detected the anti-GRP78 IgG antibody titer in the serum of patients in the different study groups: compared to healthy subjects we found an increase in the mean titer in polyp-bearing patients (4.4±0.3 vs 6.1±0.4) and cancer-bearing patients (6.2±0.5) (Fig. 1A). These results demonstrated a statistically significant difference between polyp and CRC patients and healthy subjects (p<0.001). By contrast, the difference between polyp and cancer patients was not statistically significant (Fig. 1B).
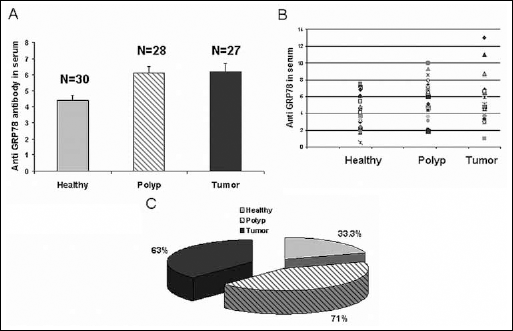
Anti-GRP78 antibodies in the patients' serum
We then compared the percentages of patients in the different groups with high serum titers of anti-GRP78 antibodies using a cutoff point of 5.0±00; this resulted in a sensitivity of 86% in healthy patients and of 80% in polyp and CRC patients. We found high serum titers of anti-GRP78 antibodies in 71% of polyp patients and 63% of tumor patients. In contrast, high serum levels of anti-GRP78 antibodies were found only in 33% of the healthy subjects (Fig. 1C). Follow-up of this 33% of subjects with high levels of anti-GRP78 antibodies in serum showed no change in their health status.
Correlation between Anti-GRP78 antibody Titers and Clinical Evaluations in Polyp Patients
The levels of anti-GRP78 antibodies in the serum of polyp patients correlated with age, gender, as well as polyp number and size, which were determined by colonoscopy and polyp biopsy and then verified by pathology evaluation.
The pathological analysis of the polyps diagnosed 7 patients with hyperplasia and 21 with tubular polyps. No correlation was found between the polyp pathology results and the levels of anti-GRP78 antibodies in the patients' serum. We observed that 70% of the patients with tubular polyps and 80% of those with hyperplastic polyps, presented >5.0 anti-GRP78 antibody titer.
We then analyzed the correlation between anti-GRP78 titer and age, sorting out patients >65 years old (6.0±0.4) and <65 years old (6.27±0.6), and we found no significant differences. The serum anti-GRP78 antibody titer in females was 5.8±0.7, similar to that in males, 6.23±0.4. Similarly, we found no significant differences in antibody titers between females and males with regard to the number of polyps (cutoff >3.0). The same was true for the size of the polyps: the group of patients with a polyp of less than 5-mm in size presented a mean serum antibody titer of 5.8±0.9, while patients with a polyp of more than 5 mm in size showed a mean serum antibody titer of 6.71±0.5.
Colonoscopies performed for patients' follow-up after 7 years showed recurrent polyps in 9 out of 28 patients. Seven out of these 9 patients had high levels of anti-GRP78 antibodies in serum.
Correlation of Anti-GRP78 Antibody Titer and CRC Tumor Classification
The total number of CRC patients studied was 27. Of these, 17 had a Duke's B cancer, 5 patients had a Duke's C, and the remaining 5 had Duke's D cancer (12).
The serum titer of anti-GRP78 antibodies in patients bearing colon carcinoma was compared after grouping the patients according to the Duke's classification (12): anti-GRP78 values >5.0 were found in 80% of CRC patients with Duke's C, in 65% of those with Duke's B, and in 40% of those with Duke's D. No statistically significant correlations were found between the anti-GRP78 levels and the grouping according to the Duke's classification, although a marked trend was observed. The highest titer of anti-GRP78 antibodies was found in Duke's C patients (7.9±1.8) whereas, surprisingly, Duke's D patients presented the lowest titer (5.0±0.7), and Duke's B patients had values that were in between those of Dukes' C and D patients.
The serum titers of the anti-GRP78 antibodies were correlated with age, gender, and carcinoembryonic antigen (CEA) levels. Statistical analysis revealed no correlation between clinical parameters and anti-GRP78 antibody titer, even though patients younger than 65 years old (14/27 patients) showed lower values than patients over 65 years old (13/27 patients) (5.2±0.5 vs 7.3±0.9). The same tendency was observed when comparing females to males (5.3±0.6 vs 7.1±0.8). No differences were found between CRC patients with high levels of anti-GRP78 antibodies and patients with low levels.
Discussion
An immune response against cell-surface GRP78 was observed in prostate as well as hepatocellular carcinoma and correlated with a poor prognosis of the disease (13). In this study we screened patients diagnosed by colonoscopy for serum anti-GRP78 antibodies. The results were analyzed dividing the patients into the groups of healthy subjects, polyp patients, and CRC patients as diagnosed by colonoscopy; the diagnosis was then correlated with the levels of anti-GRP78 antibodies in their serum. We found a highly significant increase in the levels of anti-GRP78 antibodies in the serum of CRC and polyp patients compared to healthy subjects. Our data shows that 71% of the polyp patients and 63% of the CRC patients had high levels of anti-GRP78 antibodies in their serum. Similar results were also reported for autoantibodies against GRP78 in the sera of patients with gastric cancer (28.3%), in 26.7% of esophageal cancer patients, and in 20% (3/15) of colon cancer patients, in contrast with the 0% value of healthy individuals (14). Moreover, in a metastatic mouse model, an increase in circulating anti-GRP78 autoantibodies was found to be associated with the detection of primary tumor and metastases (15). Several studies demonstrated that anti-GRP78 antibodies were identified in the presence of cancer (13, 14). We herein present the first report demonstrating the presence of anti-GRP78 antibodies in the serum of patients at the early stage of the disease, which is initially manifesting only with polyps, in the absence of malignancy; therefore, the anti-GRP78 titer could be used for the early detection of patients prior to cancer development.
No additional correlation was observed between age, gender, polyps' number and size, and levels of anti-GRP78 antibodies for polyp patients.
Similarly, no correlation was found in CRC patients between the anti-GRP78 antibody titer in serum and age or gender. In addition, anti-GRP78 antibody titer did not correlate with CEA levels. It was suggested that the CEA blood test is not a reliable screening test for the early detection of cancer (16). A tendency towards lower levels of serum anti-GRP78 were found in females, compared to males, and in patients <65 years, compared to those >65 years; however, these differences were not statistically significant. Similar results were obtained when grouping CRC patients according to the Duke's classification: also in this case we found a trend but no statistically significant difference. We thus suggest that possible statistically significant differences in terms of anti-GRP78 antibodies in the serum of CRC patients with different clinical parameters may be found when increasing the number of CRC samples included. Interestingly, 65% of patients with Duke's B cancer and 80% of patients with Duke's C cancer had high levels of anti-GRP78 antibodies, whereas only 40% of patients with Duke's D cancer showed high levels of anti-GRP78. In a recent study we found that tumorigenicity was reduced in tumor cells expressing cell-surface GRP78, and that tumor cells lacking cell-surface GRP78 showed increased proliferation, growth, and metastasis (17). It is possible that cells from metastatic Duke's D CRC represent tumor cells with reduced cell-surface GRP78 and, as a consequence, are tumor aggressive and metastatic; however, the reduced cell-surface GRP78 also resulted in a lower anti-GRP78 humoral response.
Considering the number of patients tested for serum anti-GRP78 in the first cohort presented in this study, we propose to use this type of screening together with other tests, such as FOBT (18), and with a further confirmation by colonoscopy, in order to improve the accuracy of the currently used tests.
At present, this is the first study showing a significant difference in the serum levels of anti-GRP78 antibodies between healthy subjects and polyp-bearing patients. The 7 years of follow-up for healthy subjects and polyp patients showed no differences between patients with higher or lower values of anti-GRP78 antibodies. This observation suggests that this test may only be valid for diagnosis, without having a prognostic value. Further studies with a larger number of patients might add information to the assessment of anti-GRP78 serum titers as a new biomarker for early detection of CRC.
