Abstract
BACKGROUND:
Expressions of Caspase-8 and Caspase-3 have been identified as important markers in many malignant tumors, but their roles in colorectal cancer (CRC) have not been confirmed. The purpose of this study was to investigate the role of Caspase-8 and Caspase-3 in CRC.
METHODS:
We enrolled 470 CRC patients in this study. Archival paraffin-embedded CRC tissue samples were used to construct tissue microarray (TMA), expressions of Caspase-8 and Caspase-3 that were stained by immunohistochemistry. Prognostic and predictive role of Caspase-8 and Caspase-3 expressions, alone or united, were evaluated by univariate and multivariate analysis respectively.
RESULTS:
In comparison with adjacent normal tissues, Caspase-8 and Caspase-3 protein levels were upregulated in CRC tissues significantly, furthermore, high expressions of Caspase-8 and Caspase-3 were correlated with decreased overall survival (OS) (
CONCLUSION:
Caspase-8 and Caspase-3 expressions in tumor tissues are novel candidate prognostic markers for CRC patients. It was the first time to be identified that Caspase-8 and Caspase-3 expressions had synergistic role as efficient prognostic indicators for CRC patients.
Introduction
Colorectal cancer (CRC) is the fourth most commonly cancer in Asia [1]. The CRC incidence trend in some regions, for example Beijing in China, is increasing especially in older groups of patients, while in other regions such as New York, it is decreasing in older age groups [2]. However, among younger adults in the whole world, the incidence trend of CRC is Increasing [3, 4]. Most people are diagnosed with CRC at the advanced stage usually with local lymph node or distant metastasis in the whole world. Despite the application of multidisciplinary treatment strategies including surgical operation, perioperative chemotherapy, radiotherapy and targeted therapy, the actual 5-year survival rate for advanced CRC is 60% or less, because of its invasion and metastasis [5, 6, 7]. In the past recent years, some genes have been identified as significant biomarkers such as septin9, which is a good diagnostic biomarker complementary to fecal occult blood test (FOBT) as a screening tool for CRC [8], the sensitivity for CRC detection was 73.3% (95% CI 63.9–80.9%) and 68.0% (95% CI 58.2–76.5%) for Septin9 and fecal immunochemical test (FIT), respectively. Specificity of the Epi proColon test was 81.5% (95% CI 75.5–86.3%) compared with 97.4% (95% CI 94.1–98.9%) for FIT [9]. Nevertheless, there is still no definite prognostic factors for CRC to guide therapy. Therefore, we need to find some molecular markers associated with CRC to guide clinical therapy urgently.
Cell apoptosis or named programmed cell death (PCD), is a process of cell physiological self-destruc-tion, which plays a very important role in embryonic development, biological homeostasis and defense against of the external and internal injury of multicellular organisms [10, 11]. Proteases of the caspase family, including Caspase-2, 3, 6, 7, 8, 9, 10 et al. are the initiators (2, 8, 9, 10) and executioners (3, 6, 7) of proteolysis [12, 13, 14]. Caspase-8 is one of initiators of proteolysis that not only can cut PARP (poly ADP-ribose polymerase) but also is involved in the activation of other proteases such as Caspase-3 (32 KD) [15, 16]. Executioner Caspase-3 is the key implementation protease of PCD, in which it functions in many ways in apoptotic signal transduction [17, 18, 19, 20]. Caspase-3 exists in the form of zymogen in cytoplasm, which is activated in the early stage of apoptosis, eventually lead to cell apoptosis. But in the late stage of apoptosis and death cells, the activity of Caspase-3 significantly decreased. It was reported that brewers’ rice can induce apoptosis and inhibit the proliferation of HT-29 cells via activation of Caspase-8 and Caspase-3 and down regulation the Wnt/
Our previous studies have shown that cullin1, MMP-2 and c-Myc expressions are novel diagnostic and prognostic markers for CRC. CRC patients with low expression of both cullin1 and c-Myc, or both cullin1 and MMP-2 had a favorable survival outcome [24, 25]. In this study, we aimed to elucidate the expression patterns of Caspase-8 and Caspase-3 in a CRC patients cohort, and to examine the possibility of Caspase-8 and Caspase-3 alone or together as a prognostic and predictive biomarkers of CRC.
Materials and methods
Patients and samples
The study was approved by Institutional Review Boards of Yixing Hospital Affiliated to Medical College of Yangzhou University. All 470 CRC patients with histopathologically confirmed CRC were retrospectively recruited to this study in our hospital from January 2000 to December 2006 in our hospital .
Finally, Caspase-8 and Caspase-3 expressions were assessed in 464 and 462 patient tissues, respectively, because of some missing samples. All the patients were followed up for at least 5 years. Overall survival (OS) was the primary endpoint of this study, and the OS was calculated from the date of surgery to the date of death or to the last follow-up. The clinicopathological features of the CRC patients including age, gender, pathological classification, tumor-node-metastasis (TNM) stage et al. were summarized in Table S1. The database of patients’ death was obtained from follow-up by telephone calls, and was verified by local civil affairs department.
Tissue microarray (TMA) construction and immunohistochemistry
The CRC TMAs were constructed by contract service at the National Engineering Center for Biochip, Shanghai, China. Briefly, duplicate 1.0-mm diameter cores of tissue from each sample were punched from paraffin tumor block and corresponding non-tumor tissues. As a tissue control, the biopsies of normal colorectal epithelium tissues were inserted in the 4 angles and the center of each slide. A standard protocol used for immunostaining was provided in our previous study [26, 27]. The omission of the primary antibody served as the negative control.
Assessment of immunohistochemistry
Staining of Caspase-8 and Caspase-3 in the tissues were scored by two independent pathologists who were blinded to the clinical data. Semi-quantitative immunoreactivity score (IRS) was applied as was previously reported [26, 27]. The intensity of immunostaining was shown in Figs S1 and S2. Category A documented the intensity of immunostaining as 0–3 (0, negative; 1, weak; 2, moderate; 3, strong). Category B documented the percentage of immunoreactive cells as 1 (0%–25%), 2 (26%–50%), 3 (51%–75%), and 4 (76%–100%). The concordance for IRS staining score of Caspase-8 and Caspase-3 between the 2 pathologists was 450 (95.7%) and 446(94.9%) in 470 samples respectively and the discrepancies were resolved by consensus using a multihead microscope.
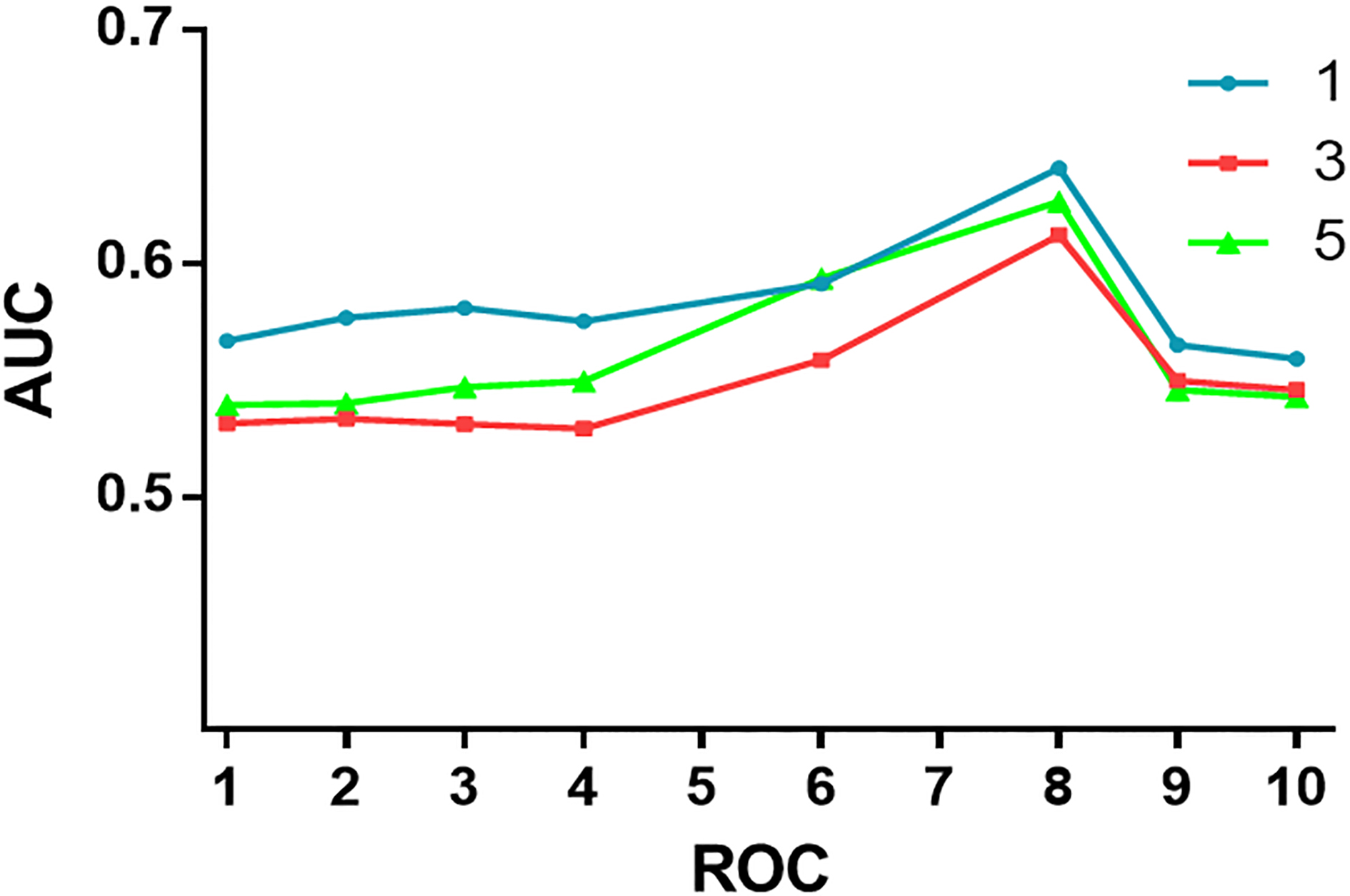
The optimum cutoff value of IRS is obtained by receiver operator characteristic (ROC) analysis, the area under the curve (AUC) at different cutoff values of Caspase-8 and Caspase-3 IRS for 1, 3, and 5 years of OS time was calculated. The optimal value of the cutoff point for IRS of the Caspase-8 was 4 and for Caspase-3 was 6 and these cutoff points were the best for evaluation of relation of IRS with death date (Supplementary Figs S3 and S4). Under these conditions, samples with IRS 0–3 and IRS 4–12 as well as IRS 0–5 and IRS 6–12 were classified as low and high expression of Caspase-8 or Caspase-3 in CRC tumors, respectively.
Western blotting
Western blot analyses were performed as previously described [24, 27]. The monoclonal rabbit anti-Caspase-8 antibody (1:1,000 dilution, Epitomics, California, USA), the rabbit polyclonal anti-Caspase-3 antibody (1:1,000, dilution; Abcam), and monoclonal mouse anti-
Statistical analysis
The associations between Caspase-8/Caspase-3 expressions and clinicopathologic data were evaluated by Fisher’s exact test. The statistical processing was executed using SPSS 20.0 software (SPSS, Inc, Chicago, IL,USA). The significance of correlations of Caspase-8 and Caspase-3 staining in primary CRC tumors and their corresponding non-tumors counterparts were assessed by paired Wilcoxon test (grouped) and Spearman rank-order correlation (raw scores). The correlation of Caspase-8 and Caspase-3 expressions was established by Spearman rank-order correlation (raw scores) and Fisher’s exact test (grouped). Probability of differences in OS as a function of time was ascertained by use of the Kaplan–Meier method, with a log-rank test probe for significance. Univariate or multivariate Cox regression analysis was used to estimate the crude hazard ratios (HRs), adjusted HRs and their 95% confidence intervals (CIs), with adjustment for potential confounders. Then we analyzed the predictive value of the parameters using time-dependent ROC curve analysis for censored data and calculated AUC of the ROC curves. We evaluated the performances of different scores by plotting (t, AUC[t]) for different values of follow-up time (t). All the statistical analyses were conducted by STATA statistical software (version 10.1; StataCorp College Station, TX, USA). P-value of
Results
Association of Caspase-8 and Caspase-3 expressions with clinicopathologic characteristics in CRC patients
The expression of Caspase-8 protein was significantly associated with clinicopathologic characteristics including depth of invasion (T-category) and lymph node metastasis (N-category) in the CRC tissues (Table 1), the significant association was also seen in protein expression of Caspase-3 with depth of invasion (T-category) and lymph node metastasis (N-category) in the CRC tissues (Table 2). Nevertheless, the expressions of Caspase-8 and Caspase-3 had no correlation with other clinicopathologic characteristics of CRC patients such as age, gender, pathological classification of CRC, tumor diameter and distant metastases.
Relationship between expression levels of Caspase-8 and clinicopathological features in CRC patients
Relationship between expression levels of Caspase-8 and clinicopathological features in CRC patients
Relationship between expression levels of Caspase-3 and clinicopathological features in CRC patients
Eight pairs of human CRC samples, including tumor tissues and adjacent normal tissues were used to test Caspase-8 and Caspase-3 proteins expressions by Western blot. Tested by Western blot, Caspase-8 and Caspase-3 proteins expressions, compared with the paired adjacent normal tissues, significantly increased in all of CRC tissues (Fig. 1A). Caspase-8 and Caspase-3 proteins expressions were also detected by immunohistochemical staining of the colorectal TMA in 450 and 446 CRC patients respectively as well as matched normal mucosa (Fig. 1B and C). Tested by immunohistochemical staining, Caspase-8 and Caspase-3 expressions were both upregulated in tumor tissues compared with that noted in the paired adjacent noncancer tissues (
Correlation of Caspase-8, Caspase-3 expressions with OS in CRC patients
Our data revealed that significantly higher Caspase-8 and Caspase-3 expression in colorectal cancer tissues was correlated with shorter OS in CRC patients using Kaplan-Meier survival assay (
Univariate Cox regression analysis of Caspase-8 or Caspase-3 expression and clinicopathological variables predicting survival in patients with CRC patients
Univariate Cox regression analysis of Caspase-8 or Caspase-3 expression and clinicopathological variables predicting survival in patients with CRC patients

Multivariate Cox regression analysis of Caspase-8, Caspase-3, Caspase-8/Caspase-3 expression and clinicopathological variables predicting survival in patients with CRC
Kaplan-Meier curves depicting OS according to expression patterns of Caspase-8, Caspase-3, and combined with Caspase-8/Caspase-3 expression in training cohort 
We conducted a time-dependent ROC analysis for the censored data in order to further evaluate whether Caspase-8 and Caspase-3 expressions have a synergetic effect on the prognosis of CRC patients. The results of above ROR analysis indicated that combination of clinical risk score (TNM stage, histological type and tumor diameter) and Caspase-8 or Caspase-3 as well as Caspase-8 plus Caspase-3 expressions contributed much more to prognosis CRC patients than clinical parameters alone (Fig. 3). For instance, the AUC at year 5 was 0.633 (95% CI: 1.249–1.841) for clinical risk score, whereas it was significantly increased to 0.717 (95% CI: 1.434–2.778) when combination of the clinical risk score with Caspase-8 plus Caspase-3 risk score.
Time-dependent ROC analyses for clinical risk score (TNM stage, histologic type, and tumor diameter), or the combination of Caspase-8, Caspase-3, or Caspase-8 plus Caspase-3. AUC 
The stratified analysis showed that CRC patients with both high expressions of Caspase-8 and Caspase-3 had a more unfavorable outcome of survival (
CRC was the third most commonly diagnosed cancers (1.36 million) worldwide in 2012 reported by GLOBOCAN [28, 29, 30]. Nowadays, TNM staging system for CRC is the main prognostic system [31]. However, it is not sufficiently reliable because of the cell heterogeneity of CRC tumors. Therefore, novel prognostic and predictive biomarkers are demanded to predict outcomes and determine personalized treatment plan.
Caspases family, especially Caspase-8 and Caspase-3, plays an important role in tumors apoptosis [21, 32, 33]. The activation of Caspase-8 could form a metastasis and invasion signaling complex, resulting in activation of ERK and JNK/AP-1 signaling pathway that mediate the elevation and activation of matrix metalloproteinase-1 (MMP1) and eventual promotion of cancer invasion and metastasis [32]. Caspase-8 activation is the proapoptotic pathway in human colon cancer cell lines in contact with proanthocyanidins extracted from various berries [34]. In the same time, Caspase-3 activation is confirmed to be related to the NF-
In present study, our results showed that Caspase-8 expression is higher in CRC tumor tissues versus matched adjacent normal tissues. High expression of Caspase-8 correlated significantly with depth of invasion, lymph node metastasis and TNM stage in CRC tumor tissues. Furthermore, the patients with high Caspase-8 expression in tumor tissues had poor OS in CRC patients using Kaplan-Meier survival analysis. Univariate and multivariate Cox proportional hazards regression analyses showed that Caspase-8 expression is an independent negative prognostic factor of CRC.
Caspase-3 expression was also upregulated in tumor tissues compared with that noted in the paired adjacent normal tissues in both CRC fresh tissues and a TMA cohort. Our results demonstrated that high Caspase-3 expression in CRC cancer tissues was correlated significantly with depth of invasion, lymph node metastasis and TNM stage. High Caspase-3 expression was also correlated with worse OS and was found to be an independent negative prognostic factor in CRC patients.
In our cohort of CRC patients, significantly increased expressions of Caspase-8 and Caspase3 correlated significantly with unfavorable clinicopathologic parameters and worse OS. It was the first time to be found that significantly increased Caspase-8 expression combined with significantly increased Caspase-3 expression had synergistic potential role in predicting the unfavorable prognosis of CRC patients and may be more effective than Caspase-8 or Caspase-3 expressions alone
In conclusion, our findings clearly demonstrated that high Caspase-8 and Caspase-3 expressions in CRC tumor tissues are associated with poor prognosis in CRC patients. We identified a combined value of Caspase-8 with Caspase-3 expressions as potential prognostic and predictive biomarkers for CRC for the first time.
Footnotes
Acknowledgments
The present study was supported in part by Young Medicine Focus Talent Foundation of Jiangsu Province (NO. QNRC2016206).
Conflict of interest
None.
Supplementary data
The patients’ clinicopathological features in CRC
Variables
n
All patients
470
(%)
Age (years)
267
56.8
203
43.2
Gender
Males
281
59.8
Females
189
40.2
Pathological classification
I
5
1.1
II
423
91.2
III
36
7.7
Depth of invasion
T1
9
2.0
T2
94
20.2
T3
347
74.6
T4
15
3.2
Lymph node metastasis
N0
276
59.2
N1
126
27.0
N2
64
13.8
TNM stage
I
88
18.9
II
179
38.6
III
180
38.8
IV
17
3.7
Tumor diameter
378
80.6
91
19.4
Distant metastasis
M0
451
95.9
M1
19
4.1
Representative images of Caspase-8 immunohistochemical staining in normal colorectal tissue and colorectal cancer tissue. A, B, C, D is adjacent normal tissue; E, F, G, H is cancer tissue. A, E, Negative staining. B, F, Weak staining. C, G, Moderate staining. D, H, Strong staining. All panels: original magnification, 40
Representative images of Caspase-3 immunohistochemical staining in normal colorectal tissue and colorectal cancer tissue. A,B,C,D is adjacent normal tissue; E,F,G,H is cancer tissue. A, E, Negative staining. B, F, Weak staining. C, G, Moderate staining. D, H, Strong staining. All panels: original magnification, 40
Receiver operating characteristic (ROC) curves were obtained to show the relation between area under the curve (AUC) at different cutoff values of Caspase-8 immunoreactivity score (IRS) for 1, 3 and 5 years of overall survival time.
Receiver operating characteristic (ROC) curves were obtained to show the relation between area under the curve (AUC) at different cutoff values of Caspase-3 immunoreactivity score (IRS) for 1, 3 and 5 years of overall survival time.