Abstract
We performed a case-control association study to evaluate the association between common polymorphisms in MTHFR (C677T and A1298C) and the Arg72Pro polymorphism in the p53 gene and the risk for cervical intraepithelial neoplasia (CIN) or invasive cervical cancer (ICC) in Mexican HPV-infected women. We included 131 women with diagnosis of CIN grade I-II and 78 with CIN III or ICC; as controls we also included 274 women with normal Pap smear and negative HPV test. Genotyping for MTHFR and p53 polymorphisms was performed by PCR-RFPLs. HPV was tested by Hybrid Capture II. Odds ratios and 95% confidence intervals were estimated. Genotype frequencies for the 3 studied polymorphisms were distributed according to the Hardy-Weinberg equilibrium. The A1298C-MTHFR polymorphism showed significant differences for the heterozygous AC genotype and the C allele, whereas the AA genotype and A allele resulted to be genetic risk factors for CIN or ICC (p<0.03). The Arg72Pro-p53 polymorphism showed for the genotypes Arg/Pro and Pro/Pro, and for the Pro allele, a significant association only to the risk for CIN (p<0.03). The MTHFR/p53 interaction showed that the genotype combinations AA/ArgArg and AA/ArgPro were associated, respectively, to the risk of ICC and CIN (p<0.05). This study suggests that the A1298C-MTHFR polymorphism contributes to the genetic risk for both CIN and ICC, whereas the Arg72Pro-p53 polymorphism only contributes to the risk for CIN. The MTHFR/p53 genetic combinations AA/ArgArg and AA/ArgPro are associated genetic risk factors for ICC and CIN in Mexican HPV-infected women.
Introduction
Cervical cancer is the second most frequent cause of cancer among women worldwide and its incidence is disproportionately high (>80%) in the developing world (1). In México, high rates of invasive cervical cancer (ICC) mortality persist in marginalized areas, with the highest rates in the South of the country, where it affects 11.9/100,000 women (2). Cervical intraepithelial neoplasia (CIN), which precedes cervical cancer, has also reached epidemic proportions (3). Cervical infection by human papillomavirus (HPV) has been established as a necessary event in the development of CIN and ICC. The development of ICC is defined through well-known steps: HPV transmission, viral genomic integration, viral persistence, progression of gradually infected cells to neoplasia, and invasion (4). Cervical lesions are classified according to the degree of cellular abnormality. The lowest grade of abnormality is CIN grade I or II, while a CIN grade III describes progressive epithelial dysplasia leading to ICC. Since only a small portion of HPV-infected women develop cervical cancer, genetic factors may play an important role in the persistence of HPV infection and in the malignant conversion of cervical epithelial cells (5).
Polymorphisms in the methylenetetrahydrofolate reductase (MTHFR) gene have been suggested to affect the susceptibility to CIN or ICC (6–8). MTHFR is a critical enzyme in determining the proportions of folate coenzymes for DNA synthesis or DNA methylation, the latter being a major epigenetic modification affecting gene expression and integrity (6). The most common polymorphism, C677T, results in an alanine-to-valine substitution affecting the catalytic domain of the enzyme and leading to a reduction in the enzyme activity (9). Another common variant, A1298C, results in the substitution of glutamate with alanine. Also this polymorphism reduces enzyme activity, although to a lesser extent (10). Association studies of the MTHFR gene with cervical cancer have focused on these 2 functional polymorphisms, revealing inconsistencies on if they confer a risk or a protective effect, due to ethnic variations (7, 8, 11–15). Moreover, the MTHFR variant genotype may increase CIN and cervical cancer risk in women with low folate or vitamin B12 status (16).
p53 is a tumor suppressor gene (located on chromosome 17p13.1) involved in multiple pathways including apoptosis, cellular transcriptional control, and cell cycle regulation (17, 18). HPV-16 and HPV-18-E6 proteins bind p53, promoting its degradation via both the ubiquitin pathway and the E6-associated protein (E6-AP)-dependent mechanism (19, 20). Polymorphisms in the p53 gene modify the conformation of the p53 protein, increasing the ability of HPV to bind and degrade p53 and thus potentially acting early in cervical carcinogenesis (18). A common polymorphism at codon 72 in the p53 gene results in the substitution of an arginine by a proline, conferring different biological and biochemical properties. The arginine form (G allele) is more vulnerable for the binding and degradation by the HPV-E6 oncoprotein than the proline form (C allele) (21). Women with cancer were more likely to be homozygous for arginine (GG genotype), thus suggesting that homozygosity for Arg at codon 72 confers susceptibility to cervical cancer (22). However, association studies of the Arg72Pro-p53 polymorphism and risk of cervical cancer have also shown controversial results (18, 21, 23).
The aim of this work was to evaluate the association of common polymorphisms in both the MTHFR (C677T and A1298C) and p53 (Arg72Pro) genes with the risk for CIN or ICC in HPV-infected women from Southeast México.
Materials and Methods
Study population
We performed a case-control association study in which cases and controls were consecutively recruited between 2006 and 2009 from the external service of the Laboratory of Reproductive Biology at the Centro de Investigaciones Regionales, Universidad Autónoma de Yucatán. The cases' group was made of 209 HPV-positive women with the following distribution in terms of severity of cervical neoplasia: 131 women with histopathologic diagnosis of CIN grade I or II, and 78 women diagnosed with CIN grade III or ICC. The control group was composed of 274 women with normal Pap smear and negative HPV test. For the analysis we used anthropologic and demographic parameters, among which language, birthplace, surnames, genealogy, and history of lifestyle, in order to ethnically match the group of controls with that of cases, as to avoid population stratification. Cases and controls were unrelated women born in Yucatán, México, belonging to the same ethnic group of Mestizos, defined as individuals born in the country, having a Spanish-derived last name with family antecedents of Mexican ancestors back at least to the third generation. In addition, we previously determined the absence of substructure or population stratification (another possible confounder) within the population of Yucatan by using 16 autosomal STR markers (24). We excluded women who had used oral contraceptives or barrier methods of contraception during the last 3 months, as well as women who had received hormonal replacement therapy during the last 6 months.
A sample of 3 mL of blood, a sample of cervicovaginal cells, and an interview with socio-demographic questions were obtained from all women who accepted to participate in the study. The questionnaire was subdivided in: (i) socio-demographic data: age, marital status, and educational level; (ii) data related to sexual and reproductive factors: age of onset of the active sexual life, number of sexual partners, pregnancies, deliveries, caesarean sections and abortions; and (iii) the use of contraceptives.
Ethical considerations
Informed consent was obtained from all participants according to the recommendations of the Helsinki Declaration. This study was approved by the bioethics committee of the institution. Complete information confidentiality of participants was strictly maintained.
HPV detection
HPV was tested with the Hybrid Capture II method, which detects the presence of 13 oncogenic or high-risk HPV types in cervical specimens: 16, 18, 31, 33, 35, 39, 45, 51, 52, 56, 58, 59, and 68. Samples that resulted HPV-positive by this procedure were confirmed by PCR using the universal HPV L1 gene primers, MY09 and MY11 (25), and the PC04 and GH20 primers for β-globin amplification (26) as an internal control. Amplicons were visualized on an 8% acrylamide gel silver-stained.
Genotyping
DNA was isolated from blood using standard techniques in order to determine the presence of the polymorphisms by PCR-RFLPs. The C677T polymorphism was amplified by PCR for a 198-bp fragment of the MTHFR gene and digested with Hinf I, according to the procedures described by Frosst et al in 1995 (9). The A1298C polymorphism was identified by PCR amplification and digestion with MbO II restriction enzyme of a 163 bp DNA fragment of the MTHFR gene according to the method described by Van der Put et al in 1998 (10). The p53 polymorphism was tested using the primers described by Storey et al (27); the method takes advantage of a restriction site for BstUI, which is present in the case of the arginine allele (CGC), and is absent in the case of the proline allele (CCC). A negative control without DNA was included both in the PCR and restriction phases. To ensure a quality control of the genotyping results, 10% of the samples were randomly selected and genotyped by a second investigator, resulting in a 100% concordance. Samples that failed on amplification or digestion were repeated to ensure the correct identification of variants.
Statistical analysis
Genotype frequencies were determined by counting and calculation of the proportions. The frequencies of the genotypes were used to compare the 2 groups of cases (CIN I-II, and CIN III or ICC) with the controls, by means of a standard X2 analysis. Exact methods were preferred whenever the expected values in any cell were <5. The Hardy-Weinberg equilibrium analysis was applied and tested using X2 statistics for goodness of fit (with 1 degree of freedom). Association of MTHFR and p53 genotypes with the risk for developing CIN or ICC in HPV-infected women was estimated considering p values <0.05 as statistically significant. Odds ratios (OR) with 95% confidence intervals (CI) were calculated using STATA case-control odds ratio program with the Woolf method (STATA version 10.2). C677T/A1298C-MTHFR haplotypes were also determined and tested for haplotype disease-association using SNPstats software (28).
Results
Socio-demographic and epidemiological data are described in Table I. The age in the group of cases ranged between 20 and 75 years, with a mean age of 38 years. Control women had a mean age of 37.56 years with a range of 21-62 years. In Tab. II are listed the genotype and allele frequencies of the C677T, A1298C-MTHFR, and Arg72Pro-p53 polymorphisms for controls, CIN I-I, and CINIII-ICC cases, as well as the Hardy-Weinberg expectations, and the comparison of frequencies on an association analysis. Genotype frequencies for the 3 studied polymorphisms were distributed according to the Hardy-Weinberg equilibrium (p>0.05).
Socio-Demographic Features of the Studied Population
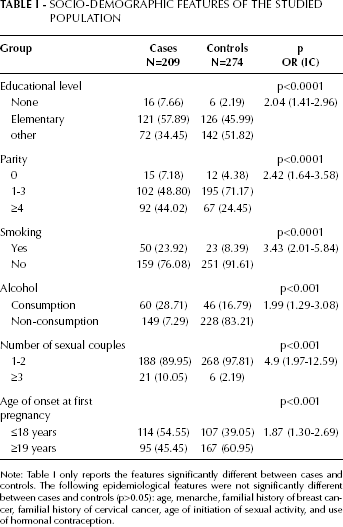
Note: Table I only reports the features significantly different between cases and controls. The following epidemiological features were not significantly different between cases and controls (p>0.05): age, menarche, familial history of breast cancer, familial history of cervical cancer, age of initiation of sexual activity, and use of hormonal contraception.
Genotype and Allele Frequencies of MTHFR and p53 Polymorphisms in Cases (CIN I-II and CIN III-ICC) and Controls from Southeast Mexico
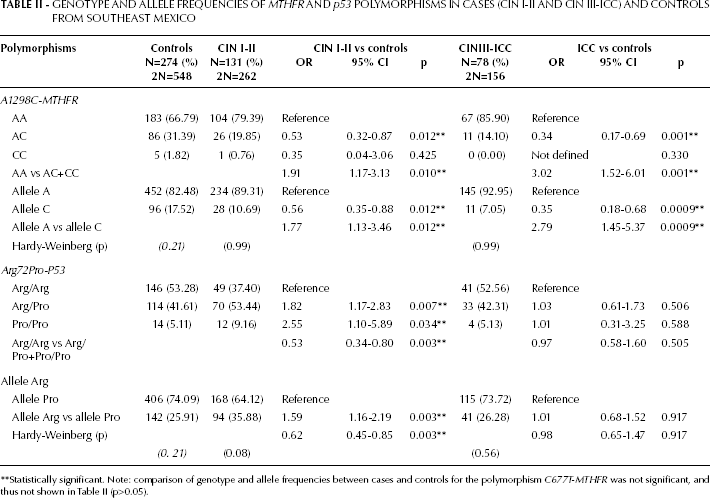
Statistically significant. Note: comparison of genotype and allele frequencies between cases and controls for the polymorphism C677T-MTHFR was not significant, and thus not shown in Table II (p>0.05).
Comparison of the C677T-MTHFR polymorphism between each case group and controls did not show significant differences (p>0.05) for any genotype or allele, suggesting a lack of association with the risk of developing any CIN or ICC. Interestingly, comparisons of the A1298C-MTHFR polymorphism showed significant differences for all alleles and genotypes, except for CC. The heterozygous AC genotype and the C allele were associated with a reduced risk, whereas the homozygous AA genotype (AA vs AC+CC) and the A allele (A vs C) were associated with an increased risk for both CIN I-II and CIN III-ICC (p<0.01). The Arg72Pro-p53 polymorphism showed significant differences for all alleles and genotypes, which were associated only with the risk for developing CIN I-II (p<0.03). The heterozygous Arg/Pro genotype, the homozygous Pro/Pro, and the Pro allele were associated as genetic risk factors (p<0.03), whereas the homozygous Arg/Arg genotype and the Arg allele were associated with a decreased risk only for developing CIN I-II (p=0.005). The Arg72Pro-p53 polymorphism did not show significant differences for any allele or genotype associated with the risk for CIN III-ICC (p>0.05). Our findings suggest that in HPV-infected women from Southeast Mexico A1298C-MTHFR may contribute to the risk of developing CIN I-II, and that this risk increases to develop a severe neoplasia CIN III-ICC. By contrast, the Arg72Pro-p53 polymorphism seems to contribute to the risk of developing CIN I-II only, and not to contribute to the progression to a severe neoplasia CIN III-ICC (Tab. II).
The haplotypes for C677T/A1298C–MTHFR were determined since multiple linked SNPs have the potential to provide significantly more power for genetic analysis than individual SNPs (28). All 4 expected haplotypes were found in the studied population, except for the haplotype T/C, which was not present in any CIN III-ICC case (Tab. III). The overall haplotype distribution was not significantly different between CIN I-II and CIN III-ICC cases versus controls (p>0.05), except for the C/C haplotype, which showed a significant difference associated to a reduced risk only for CIN III-ICC (p=0.039).
MTHFR Haplotype Frequencies in CIN I-II and CIN III-ICC Cases and Controls from Southwest Mexico
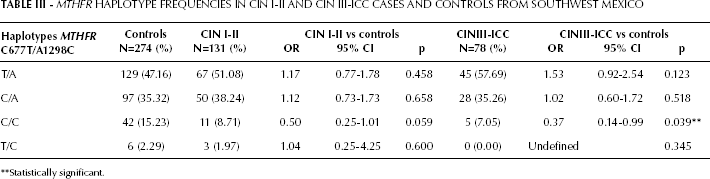
Statistically significant.
The gen-gen interaction MTHFR/p53 was assessed (Tab. IV), showing for the C677T-MTHFR/p53 genotype combinations significant differences only for CT/ArgArg (p=0.047), which was associated with a lower risk of CIN I-II. For the A1298C-MTHFR/p53 interaction, significant differences were obtained for the genotype combination AC/ArgArg, associated with a lower risk of both CIN I-II (p=0.009) and CIN III-ICC (p=0.006). The genotype combination AA/ArgPro was associated with the increased risk of CIN I-II (p=0.003), whereas AA/ArgArg was associated with the increased risk of CIN III-ICC (p=0.048), suggesting that the interaction of A1298C-MTHFR/p53 with both homozygous genotypes AA/ArgArg contributes to the risk of developing a severe cervical neoplasia or cancer (CIN III-ICC). Thus the genotype combination AC/ArgArg contributes to decrease the risk of ICC and CIN of any grade in HPV-infected women.
MTHFR/p53 Genotype Combination Frequencies in CIN I-II and CIN III-ICC Cases and Controls from Southeast Mexico
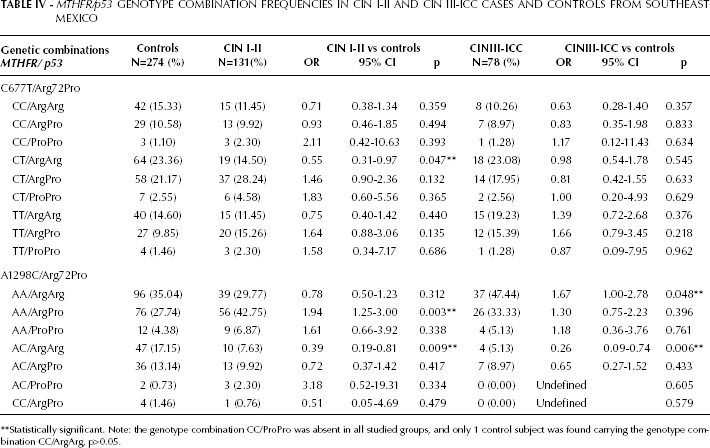
Statistically significant. Note: the genotype combination CC/ProPro was absent in all studied groups, and only 1 control subject was found carrying the genotype combination CC/ArgArg, p>0.05.
Discussion
The findings of this study suggest the potential genetic contribution of the A1298C-MTHFR polymorphism to the risk of developing CIN or ICC, as well as the genetic contribution of the Arg72Pro-p53 polymorphism to the risk of CIN I-II only. Moreover, the interaction between MTHFR and p53 was demonstrated by the fact that the AA/ArgArg increased the risk of developing CIN III-ICC, while the genetic combination AC/ArgArg protected from the development of CIN or ICC in HPV-infected women from Southeast Mexico.
Given that only a fraction of women with an HPV infection eventually develop cervical neoplasia, research has focused on identifying the factors that influence whether an HPV infection will spontaneously clear or progress toward a malignant disease. According to our findings, p53 might play a role as a host factor for the regression process of HPV-infected cells, since p53 genotypes impact only the risk for CIN I-II, and not that for CIN III-ICC, probably due to a regression of the HPV infection. Thus, p53 genotypes might represent biomarkers of a mechanism of protection for developing cervical cancer through HPV clearance or regression.
Previous studies have shown that in HPV-positive women the MTHFR polymorphism, alone or in combination with the MS polymorphism, is associated with a lower risk of CIN II or III, mainly in women who are exposed to folate food fortification (14). Folate fortification might provide elements that contribute with one-carbon metabolism to enhance the regression process. A critical factor of HPV progression is methylation of the HPV genome. Site-specific CpG methylation, partly mediated by adequate methyl donor availability (folate sufficiency), may be sufficient to slow or suppress HPV-driven carcinogenesis. Partial demethylation or hypomethylation from inadequate methyl donor availability (folate insufficiency) is required for HPV-mediated cellular transformation. Sufficient folate dietary consumption has been demonstrated in women from Southeast México according to the frequency of foods and folate erythrocyte concentrations (29). This condition might complete the bioavailability of any folate deficiency and hypomethylation due to MTHFR or p53 polymorphism, and thus support the cellular regression process for suppressing HPV-related transformation.
A controversy exists about the association between MTHFR C677T or A1298C and cervical carcinogenesis (8–13). Although conflicting results might be explained by ethnical variations and by the MTHFR polymorphisms frequency among populations, several studies agreed that the MTHFR variant genotype increases CIN or cervical cancer risk in women with low folate or vitamin B12 status (8, 15, 16). In this study involving HPV-infected women from Southeastern México, the MTHFR 1298AC genotype and C allele showed a significantly lower risk for CIN I-II and CIN III-ICC (Tab. II). Similar findings have been reported by Tong et al, who showed an association of the MTHFR A1298C genotypes with a decreased risk only for CIN II/III in carriers of the C allele (15, 16). The authors also demonstrated the increased risk for cervical cancer in subjects with low serum folate and with the MTHFR 1298AA genotype, and further suggested that the MTHFR genotype modifies the inverse association of serum folate with the risk for CIN and cervical cancer (16).
The MTHFR C677T/A1298C haplotypes did not modify the risk for CIN I-II or CIN III-ICC in our population, with the exception of C/C, which was associated with a lower risk of CIN III-ICC, since these alleles are the low-risk alleles for both SNPs. The haplotypes of 6 MTHFR SNPs did not show any significant association with CIN or cervical cancer risk in Korean women, except for the ACCCAG haplotype, which contains the allelic variants of A1298C (15). The function of MTHFR is essential for providing methyl groups for DNA methylation, thus enzymatic variants due to functional polymorphisms may alter this major epigenetic feature of DNA. In the present study, 1298AA-MTHFR showed to be a genetic factor increasing the overall risk. Lymphocytic DNA from subjects with the MTHFR 1298AA variant showed a reduced genomic DNA methylation compared with carriers of either the 1298AC or 1298CC genotype (30). This condition is evident for subjects with low plasma folate concentrations. Moreover, in the combined 1298AA/677TT genotype the plasma folate concentration and DNA methylation status are decreased, and homocysteine is increased, when folate levels are low (30).
A higher risk of HPV-associated cervical carcinogenesis has been reported in individuals with Arg homozygosity than in those with heterozygous genotype (22). Also, the p53 codon 72Arg allele was found to have a less strong association with cervical abnormalities and HPV infection (23, 31). Other studies found an increased risk for cervical cancer and its earlier stages associated with the Pro allele at codon 72 (18, 32). The present study considering HPV-affected women showed a decreased risk only for CIN I-II associated to the Arg/Arg genotype, and an increased risk associated to the heterozygous Arg/Pro and Pro/Pro genotypes. For the Latin American population, the Arg/Pro and Pro/Pro genotypes respectively conferred a 1.3 and a 1.8fold increased risk for CIN III+/HPV persistence (18). Three haplotypes, including the G to C change in the Arg72Pro polymorphism, showed the highest risk values, which suggested that Pro might be the susceptibility allele or that it may form an haplotype in linkage disequilibrium with the true susceptibility allele (18). Inconsistencies in association studies among populations might be due to the various haplotype structures of the Arg72Pro polymorphism in different study populations with diverse ethnic backgrounds.
We found a decreased risk of CIN I-II associated to the Arg/Arg genotype. Previous meta-analysis supported our findings suggesting that the Arg/Arg genotype may have a somehow protective role for CIN in some populations (23). Given that the Arg72Pro polymorphism has been associated with HPV infection, discrepancies about the risk for HPV-associated cervical carcinogenesis might be explained by the HPV genotype prevalence in each population. For instance, in women from Mexico City, an association between HPV 16 or 18 and Arg72Arg has been found, while other viral types were present in association with the Pro allele (33). Particularly, in our population from Southeastern Mexico the high prevalence of HPV 58 has been reported (34). HPV 58 might be a viral type associated to the presence of the Pro allele in the studied population that modifies the risk for CIN I-II. The E6 on-cogene of HPV 58 has a lower capacity to degrade p53 than the E6 of HPV 18, thus conferring a decreased risk associated to the Arg/Arg genotype. The Arg/Arg genotype might represent a protective factor limiting the progression to CIN III-ICC in the presence of HPV 58 or other viral types, other than HPV 16 and 18.
The AA/ArgArg (A1298C-MTHFR/Arg72Pro-p53) was the sole genotypic combination associated to the increased risk for CIN III-ICC. HPV-infected women with the AA/ArgArg genotype might have been infected by the HPV type 16 or 18, since persistent HPV 16 or 18 infections have been reported to occur in Arg72 homozygote patients with cervical cancer (33); this is probably due to the presence of the E6 of HPV 16 or 18 that contributes to the efficient degradation of p53 (33). The effect of HPV 16 and 18 might be enhanced by the reduced DNA genomic methylation conferred by the risk genotype AA1298-MTHFR in CIN III-ICC patients (30). Other significant MTHFR/p53 genetic combinations were associated with the risk of CIN I-II only, because the Arg-p53 variant induces apoptosis with faster kinetics and suppresses transformation more efficiently than the Pro-p53 variant (35); additionally, prevalent HPV genotypes might modify the risk of CIN I-II in our population.
In this study involving HPV-infected women from Southeastern Mexico, we demonstrated the genetic contribution of A1298C-MTHFR to CIN and ICC, as well as the contribution of Arg72Pro-P53 to the risk of CIN I-II, and the interaction between MTHFR and p53 (AA/ArgArg) to increase the risk of CIN III-ICC.
Footnotes
Acknowledgments
The authors would like to thank Igrid García-González for her technical assistance.
