Abstract
Background
Compomers are mostly used in primary dentition. The thermal conductivity properties of traditional or colored compomers have not been investigated in detail so far. The aim of this in vitro study was to assess and compare the thermal conductivities of traditional and colored compomers.
Method
Two sets of compomers – namely, Twinky Star (available in berry, lemon, green, silver, blue, pink, gold and orange shades) and Dyract Extra (available in B1, A3 and A2 shades) –were included in this study. All of the traditional and colored compomers were applied to standard molds and polymerized according to the manufacturers’ instructions. Three samples were prepared from each compomer. Measurements were conducted using a heat conduction test setup, and the coefficient of heat conductivity was calculated for each material. The heat conductivity coefficients were statistically analyzed using Kruskal-Wallis and Duncan tests. Uncertainty analysis was also performed on the calculated coefficients of heat conductivity.
Results
Statistically significant differences were found (p<0.05) between the thermal conductivity properties of the traditional and colored compomers examined. Among all of the tested compomers, the silver shade compomer exhibited the highest coefficient of heat conductivity (p<0.05), while the berry shade exhibited the lowest coefficient (p<0.05). Uncertainty analyses revealed that 6 out of 11 samples showed significant differences.
Conclusions
The silver shade compomer should be avoided in deep cavities. The material properties could be improved for colored compomers.
Introduction
Compomers are light-cured restorative materials used in dentition (1). They are designed to offer the benefits of traditional composite resins and glass ionomer cements (2). Compomers provide good mechanical and esthetic properties and are biocompatible. They release fluoride and are easy and safe to apply, which makes them a useful alternative to amalgam used in pediatric dentistry (3). In addition, the applications of compomers have been expanded to the anterior and posterior restorations of primary teeth since their first use (4, 5).
Colored compomer materials were developed in 2003 to attract the attention of child patients. MagicFil (Zenith/DMG, Englewood, NJ, USA) was the first colored compomer introduced in the market. This material is available in 4 colors (blue, purple, yellow and universal) with a dual-cure hardening mechanism. Twinky Star (Voco, Cuxhaven, Germany) is another colored compomer brand, and it is available in 8 different colors (berry, lemon, green, silver, blue, pink, gold, and orange shades). It hardens only by photopolymerization. The formulation of this material is comparable to that of traditional compomers, except for the presence of colored pigments and glitter particles (5, 6). It was noted that colored compomers can be motivational tools particularly for young patients. Allowing the patients to choose the color of a restoration gives them the opportunity to take part in the dentition procedure and increases the treatment efficiency (7).
The polymerization of resin materials, on the other hand, has been reported to cause a temperature rise (8). Plant and Jones reported that there was a correlation between the histological response of a material and the increase in temperature (9). Studies reported that the temperature increases to as high as 40°C during resin composite photo polymerization (7, 10, 11). This is far above the critical temperature at which adverse pulpal tissue reaction occurs. It was shown that a 5.6°C rise in temperature in healthy pulp resulted in necrosis in “small teeth” by 15% (12). In addition, a 10°C increase in normal body temperature will cause damage to the tissues surrounding the teeth (13). Therefore, the thermal conductivity of restorative materials is an important factor for protecting pulp health and tissues surrounding the teeth.
Heat conduction in materials is expressed by Fourier's law of heat conduction as Q = -k A(ΔT/Δx) (14). This equation is particularly used to estimate the coefficient of heat conductivity of solids and fluids (15). It expresses a direct proportion between the units of heat, which are transferred in time (Q), the cross-section of the material used in the experiment (A), and the heat difference between the 2 points (AT), and an indirect proportion of these parameters with the thickness of the material in which the direction of the heat produced is transferred (Ax). The parameter k is referred to as the coefficient of heat conductivity.
It has been previously reported that darker-colored resin materials have lower light transmission and less depth of polymerization than lighter-colored resin materials. Furthermore, darker-colored resins were found to absorb light, and rendering polymerization in these resins was more difficult than in the case of lighter-colored resins (16, 17). Çoğulu et al reported that lighter-colored compomers may be preferred over darker ones for deep cavities (18). They reported that the microhardness of darker compomers (Compoglass F shade A4 and Twinky Star shades green and orange) at the bottom surface was statistically lower than that of the lighter compomers (Compoglass F shade A1 and Twinky Star shade silver). In addition, they found that curing time should be increased for adequate polymerization of colored compomers in deep cavities. Vandenbulcke et al reported that the curing depth was affected by the use of organic or inorganic color pigments in colored compomers (19). Therefore, it would be interesting to determine the effects of these color pigments and glitter particles on the thermal conductivity in colored compomers.
Curing and polishing processes in the restoration of teeth result in increased temperatures (8, 10, 11). When a temperature difference occurs inside a solid material, heat is transferred from the higher temperature zone to the lower temperature zone by means of conduction (14). Methods used to measure the thermal conductivity fall into 2 categories: steady state and non-steady state. In steady-state methods, the specimen is subjected to a temperature profile that does not vary with time. After equilibrium is reached, thermal conductivity is determined directly by measuring both the rate of heat flow per unit area and the temperature gradient (20).
Only a few studies in the literature have investigated colored compomers, and these studies primarily focused on clinical performance (6, 21, 22), microhardness (18), microtensile bond strength (23), curing depth (19), surface roughness (24) and conversion degrees (25). Another study on posterior restorative material selection revealed that colored compomers were of interest for use in dentition for children (26). To the best of the authors’ knowledge, there is no dedicated study evaluating the thermal conductivity of traditional and colored compomers, even though these are very popular restorative materials used in dentition for children. Therefore, the aim of this study was to evaluate the thermal conductivities of traditional and colored compomers. The tested null hypothesis was that there would be no difference between the thermal conductivity of traditional and colored compomers, since only the color pigments and glitter particles are added to the colored compomers compared with the traditional compomers.
Materials and methods
Sample preparation
The materials investigated in the present study are listed in Table I. Twinky Star's (Voco, Cuxhaven, Germany) berry, lemon, green, silver, blue, pink, gold and orange shades were the colored compomers used in this investigation. The traditional compomer Dyract Extra's (Dentsply DeTrey, Konstanz, Germany) B1, A3 and A2 shades were also included in the study as reference materials.
Traditional and colored compomers used in this study
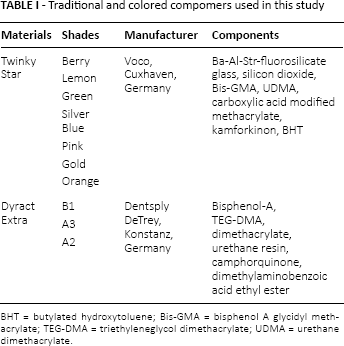
BHT = butylated hydroxytoluene; Bis-GMA = bisphenol A glycidyl methacrylate; TEG-DMA = triethyleneglycol dimethacrylate; UDMA = urethane dimethacrylate.
Polyurethane, an insulating material, was used to prepare molds that were 25 mm in diameter and 1.5 mm in thickness (Fig. 1A and B). All of the Twinky Star and Dyract Extra compomers were placed into these molds. All compomers were cured using an LED light source (3M ESPE, St. Paul, MN, USA). Three samples were prepared from each shade. First, both sides of the sample were polished with grainy sandpaper to remove the excess compomer. A smooth polished surface was obtained and polymerized according to the manufacturer's instructions (Fig. 1C). The thickness of the sample was then measured again with the help of a digital compass to verify the 1.5-mm thickness. In addition, the weights of the samples were measured using an electronic balance (AUW-220D; Shimadzu, Kyoto, Japan) to verify that the volumes of the tested samples were equal.

Preparation of test samples. (A) Sample disc being set in Teflon mold. (B) Polyurethane mold. (C) Sample discs removed from Teflon mold.
Heat conduction experiments
A heat conduction unit's (PA Hilton Ltd., Stockbridge, UK) linear heat conduction module was used to determine the thermal conductivities of the traditional and colored compomers (Fig. 2). Heat conduction experiments were carried out based on the procedure suggested by Keles et al (27).
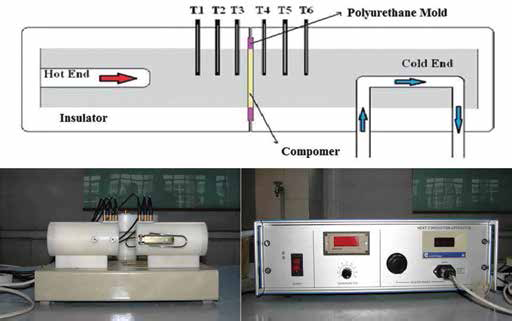
Schematic representation and photos of the heat conduction unit.
Thermal-conducting paste was applied as a thin layer on each side of the samples. The sample to be tested was placed in the sample slot of the conduction equipment's linear module between the heating and cooling compartments. The linear module's pieces were then locked in a suitable form. For each sample tested, the module's hot end was heated with 10 W of energy while the other end was cooled with water. Each test sample was held to reach the steady-state temperature prior to experiments. Even though the time required for the system to attain stability varied among the samples, the average time needed to reach the steady-state conditions was 40–60 minutes. When the system reached the steady state, the temperature from the temperature sensors placed on both sides of the tested samples was recorded using the digital heat reader.
Three temperature sensors were placed in both the heating and cooling sections, starting at a distance of 5 mm from the sample and with 10 mm of space allotted between each one. Thus, the temperature values, with the sensors remaining at a designated distance away from the test sample, could be recorded. Regression curve analysis was conducted with these recorded temperature values and the temperature of the test samples’ heated (Ta) and cooled surface (Tb) using Microsoft Excel 2007 (Tab. II). Hence, temperature was measured at 8 points from each sample and recorded. Then, using Fourier's equation, the coefficient of heat conductivity value of k was calculated for each sample, in W/m°C units. Data were analyzed statistically by Kruskal-Wallis and Duncan tests (α = 0.05; SPSS 16.0; SPSS, Chicago, IL, USA). Uncertainty analyses were also performed on the results per the Joint Committee for Guides in Metrology document. Type B evaluation was found to be more appropriate for the current study, as the uncertainty was based on temperature measurements (28). After calculating the standard deviations, estimated standard uncertainty was calculated using the following equation:
Mean values for Ta and Tb temperatures (°C) obtained for tested compomers
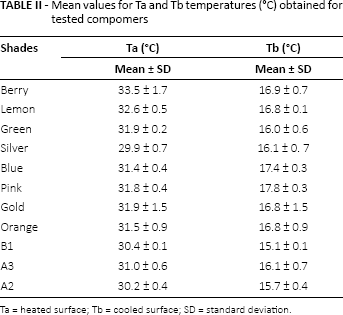
Ta = heated surface; Tb = cooled surface; SD = standard deviation.

where SD is the standard deviation, and n is the number of measurements. The values obtained were multiplied with the coverage factor of

Results
Table II shows the mean temperature values of the test samples’ heated surfaces (Ta) and cooled surfaces (Tb). Table III shows the mean values for coefficients of heat conductivity for the tested compomers. Kruskal-Wallis tests showed significant differences between the thermal conductivity of traditional and colored compomers (p<0.05; Tab. III). The highest mean coefficient of heat conductivity was obtained for the silver shade samples among all of the tested compomers, whereas the lowest value was obtained for the berry shade samples (p<0.05; Tab. III).
Coefficients of heat conductivity (W/m°C) for the tested compomers
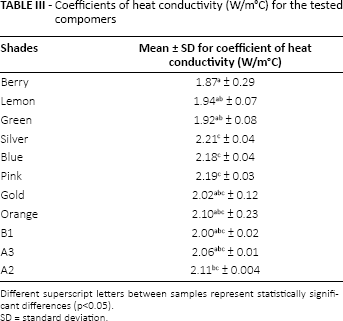
Different superscript letters between samples represent statistically significant differences (p<0.05).
SD = standard deviation.
Apart from the Kruskal-Wallis test, the coefficients of heat conductivity values were examined in terms of uncertainty (again for 95% confidence intervals). Table IV shows the expanded uncertainty results for the coefficients of heat conductivity of the tested compomers. Changes in the coefficients of heat conductivity for 6 out of 11 samples (for silver, blue, pink, B1, A3 and A2) were still found to be significant. Since the coefficients of heat conductivity calculations are based on temperature measurements, the Type B method was applied in the uncertainty analyses.
Expanded uncertainty values for tested compomers’ coefficients of heat conductivity (W/m°C)

Coverage factor kc = 2 for 95% confidence interval.
Discussion
Traditional and colored compomers were evaluated in terms of their thermal conductivities in the current study. Statistically significant differences were found between the thermal conductivities of the traditional and colored compomers. Therefore, the null hypothesis was rejected.
Heat conduction occurs in teeth during dental procedures as well as in daily life. The thermal environment of the teeth is highly variable, and the temperature range is 0–70°C, in daily life (29). Even if the pulp is protected from harmful thermal agitations, the dentin layer is insufficient to provide effective thermal isolation in deep cavities, since dentin is a poor heat conductor (30). However, the temperature variations generated during the curing and polishing processes in dental restoration procedures may cause pulpal damage (31, 32). There is also a potential risk of heat-induced pulpal injury during photo polymerization. As the extent of tolerable thermal trauma by pulp tissues is unknown, care and consideration should be given to the choice of light source and the exposure time when curing resin-based composites, particularly during bonding (33). Therefore, the same light source (LED) was preferred in curing of all samples so that the thermal conductivity of the compomers would not be affected by that factor. In addition, all tested compomers were cured according to the manufacturers’ instructions.
Clinical studies showed that increasing the intrapulpal temperature by 5.6°C is considered unacceptable, because 15% of healthy dental pulp fails to heal after such a temperature increase (12). Increasing the intrapulpal temperature by about 11.1°C and 16.7°C was showed to result in 60% and 100% pulp necrosis, respectively. Hannig and Bott reported that the polymerization of light-curing resin composites can result in a temperature increase caused by the exothermic reaction process (34). It has previously been reported that polymerization of darker resin materials is more difficult than that of light-shade resins (16, 17). Çoğulu et al reported that lighter compomers may be preferred over darker compomers in deep cavities and that the curing time should be increased to ensure adequate polymerization in deep cavities (18). The present study showed that statistically significant differences were found between the thermal conductivities of traditional and colored compomers. The difference in the thermal conductivity of colored compomers can cause pulp necrosis in deep cavities. Therefore, it is concluded that the use of colored compomers for deep cavities should be limited.
Different methods can be used to examine heat conduction of dental tissues and materials. Fourier's heat conduction law was used in this study because of its ease of application. The heat conduction properties of the test samples were examined by placing them between metal pieces equipped with sensors that could be heated and/or cooled. In this system, heat is transferred from the hot end to the cold end by means of conduction. If the heat conductivity of tested material is high, heat produced from the hot end can easily be transferred to the cold end. Hence, the system can reach the steady state without an increase in heat at the hot end. If the heat conduction feature of the tested material is not good, less heat is transferred to the cold end. Therefore, it might be necessary to increase the heat at the hot end. Once the rate at which heat is generated is equal to the rate at which heat is removed, a steady-state condition is assumed to be satisfied, and temperatures will be fairly constant, allowing readings to be taken.
In the test procedure followed, thermal-conducting paste is applied on both surfaces of each test sample. The function of thermal-conducting paste is to convey the heat produced at the hot end to the test sample. The thermal-conducting paste does not affect the thermal conductivity properties of the material. When thermal-conducting paste is not used, there may not be full contact between the heat bars and test samples. Consequently, the heat produced cannot be fully transferred to the test sample.
Keles et al (27) examined the thermal conductive properties of 5 different root canal sealers in vitro. They found that the root canal sealers functioned as thermal insulators and had different heat conductivity properties depending on their composition. Brantley and Kerby (35) reported that the thermal diffusivity of metal-reinforced glass ionomers was higher than that of other glass ionomers, owing to the presence of metal ions. However, the study did not evaluate the thermal conductivity characteristics of traditional or colored compomers. Al-Qudah et al (36) reported that there was a quantifiable amount of heat generated in resin-containing materials during light curing. In addition, they noted that dentine sections were good thermal insulators and significantly reduced temperature increases associated with resin composite photo curing. Nevertheless, the dentine section was not used in the present study. Thermal conductivity of colored compomers might be different in an oral environment.
For traditional compomers and composites, darker shades are known to reduce the curing depth compared with the lighter shades (16, 17). Vandenbulcke et al reported that the curing depth of colored compomers could be affected by the amount and type of pigment (19). They found that the relatively darker shades (blue and green) had the greatest curing depths, which could be caused by wavelength distribution and the glittering effect. This confirms the important role of pigments and their effect on curing depth. Atabek et al stated that the conversion degree of different colors is variable (25). They found that the silver- and green-colored samples could not adequately achieve an acceptable degree of conversion. According to the manufacturer, the composition of the Twinky Star material used in this study is similar to that of the traditional compomer, except for the addition of color pigments and glitter particles. Statistically significant differences (p<0.05) were found between the thermal conductivities of traditional and colored compomers based on their shades. The differences in the coefficient of heat conduction could be affected by the use of color pigments and glitter particles in colored compomers.
From a clinical point of view, there are limitations concerning the correlation between these in vitro tests, on the one hand, and in vivo tests and clinical usage, on the other. Firstly, only 2 different compomer materials were used in this study. The thermal conductivity may change when a different compomer material is used. The characteristics of filler particles, such as their composition, shape and size, as well as the entanglement of the resin and inorganic matrices play an important role in the behavior of restorative materials. Secondly, only one light source was used in this study. Thermal conductivity may change when using different light sources. A third limitation was that the thermal conductivities were recorded using compomer discs of the same thicknesses in an in vitro model. Thermal conductivity might also be affected by the thickness of the compomer discs. In addition, thermal conductivity can be different when the dentin section of the teeth is included. In a clinical setting, lower thermal conductivity is expected with the remaining dentine thicknesses, dentinal fluids and dentinal tubules, as well as lower heat dissipation by pulpal, periodontal and osseous circulations. The fourth limitation was that the color match and color analysis of the traditional or colored compomers were not evaluated in this study. There are several studies on color match and color analysis of composite resins in the literature (37–39). Nevertheless, there is no study dedicated to the color match and color analysis of colored compomers.
Clinicians must be careful to protect pulp vitality. Temperature rises, adequate polymerization, shade, color match and thickness of restorative materials are important factors in clinical practice. The literature is limited in terms of studies concerning colored compomers. Therefore, further in vitro and in vivo studies are required to investigate the effect of different factors on the thermal conductivity of colored compomers.
Conclusion
Within the limitations of the present study, the following conclusions can be drawn:
Some of the traditional and colored compomers tested showed statistically significant thermal conductivity properties based on their shades.
The highest coefficient of heat conductivity was noted for the silver shade.
The lowest coefficient of heat conductivity was obtained for the berry shade.
Footnotes
Acknowledgments
The authors wish to thank Dr. Silvia Jarchow, Scientific Service Manager, Voco GmbH, Cuxhaven, Germany, for providing the Twinky Star compomers used in this study.
Financial support: No grants or funding have been received for this study.
Conflict of interest: None of the authors has any financial interest related to this study to disclose.
