Abstract
The aim of the study is to evaluate the effect of fluoride varnishes on both the color stability and surface microhardness of different fluoride-releasing restorative materials exposed to beverages commonly consumed by children . This study was conducted as an in vitro experimental study design. Three different restorative materials (Dyract XP, Beautifil II, and Cention N) and a colorless fluoride varnish were used. The solutions in which the samples were immersed were selected as beverages commonly preferred by children: cherry juice, chocolate milk, and cola. Color changes of the samples were measured using a spectrophotometer at 1, 7, 14, and 28 days and calculated according to the CIEDE2000 total color difference formula. Surface microhardness values were measured using a digital microhardness testing device at 1 and 28 days. To assess the effects of variables and their interactions in repeated observation designs, generalized linear models and generalized linear mixed models were fitted. Following model estimation, Tukey’s-adjusted estimated marginal means were computed to identify significant pairwise differences. Fluoride-releasing restorative materials exhibited varying degrees of color change and decrease in surface microhardness when immersed in different staining solutions. Among the tested materials, Cention N demonstrated the greatest color stability (ΔE=1.01 for fluoride applied group, ΔE=1.45 for the group without fluoride application) and least reduction in surface microhardness. Fluoride application initially caused a slight discoloration immediately after application. However, following immersion in different solutions, fluoride application appeared to enhance color stability (ΔE=1.59 in the varnish-applied group, ΔE=2.21 in the group without varnish application), but did not have a statistically significant effect on the surface microhardness. A negative correlation was observed between color change and surface microhardness; as the surface microhardness decreased, the extent of color change increased. This study highlights the prominent role of intraoral environmental factors in influencing the durability and aesthetic properties of fluoride-releasing restorative materials. Topical fluoride application has a beneficial effect on the color stability with a more pronounced effect observed with Cention N.
Introduction
In recent years, advancements in material science and the emergence of new technologies have led to considerable improvements in the quality of restorative materials. However, discoloration and wear of resin-based restorative materials continue to be major concerns in long-term clinical studies. 1 The composition of restorative materials used in pediatric dentistry plays a crucial role in maintaining pediatric oral health long term, while also affecting the aesthetics of restorations. Composite resins, glass ionomer cements, and compomers are widely used restorative materials that provide satisfactory outcomes in the treatment of pediatric patients.2,3 Although glass ionomer cement is commonly used in pediatric dentistry, modifications of these materials by the addition of reinforcing particles has become increasingly popular to overcome their inherent disadvantages. 4
Polyacid-modified composite resins (compomers) combine the technologies of composite resins and glass ionomers, offering a formulation that incorporates the advantageous properties of both materials. Due to their fluoride-releasing ability and subsequent caries-preventive effect, compomers are frequently preferred for use in primary teeth. 5 Giomers are a newer class of resin-based glass ionomer materials that contain pre-reacted glass ionomer (PRG) fillers. Giomers have the fluoride release and recharge capabilities of glass ionomers along with the aesthetic properties, polishability, and wear resistance of composites. 6 Cention N is classified as an alkasite restorative material, capable of releasing acid-neutralizing ions from alkaline fillers within its resin matrix during acid attacks in the oral environment. 7 The simultaneous release of fluoride and hydroxide ions from Cention N enhances its potential for caries prevention. 8
Color stability, surface roughness, and microhardness are key properties that determine the functionality, fracture resistance, wear resistance, and aesthetic success of dental restorative materials. The cost-effectiveness of any material is directly related to the longevity of the restoration.9,10 Although recent technological advancements have improved the reliability of resin-based materials, concerns still remain regarding their physical durability in relation to discoloration. 11
Color stability refers to the ability of a material to resist changes in color and can be influenced by various factors such as environmental conditions, material composition, and technique-sensitive variables. 11 Foods and beverages frequently consumed in modern societies can lead to adsorption on the surfaces of resin restorations. In particular, acidic beverages can degrade the surface of restorative materials, causing breakdown of the organic matrix structure. 12 As a result, the wear resistance, surface hardness, and roughness of restorative materials are adversely affected. Furthermore, discoloration might occur due to the penetration of color pigments from foods and beverages into porous areas created by surface degradation. Discoloration of resin-based restorations is a leading causes of restoration replacement. 13
Adequate surface hardness values are also crucial for the long-term success of restorative materials used in dentistry. These materials are exposed reside in the mouth and are subject to various factors, such as pH fluctuations, occlusal forces, and temperature changes. The ability of restorative materials to withstand these challenges depends on maintaining high surface hardness values. Hardness contributes to a material’s mechanical durability, scratch resistance, and wear resistance, and their ability to retain shape under load. Therefore, surface hardness assessment is an effective and valid method for predicting a material’s clinical performance. 14 Thermal fluctuations in the oral cavity, combined with mechanical and chemical interactions, such as contact with acidic and pigmented foods can have deleterious effects on the color stability and structural integrity of dental restorative materials. Acidic foods have been reported to increase surface roughness and reduce microhardness of restorative materials, further compromising their performance and aesthetics. 15
Fluoride varnishes are commonly used for caries prevention in young children, patients with a sensitive gag reflex, and individuals with physical or mental disabilities due to their advantages, such as simple and safe application, low toxicity and side effects, ease of use, and short clinical application time. 16 After fluoride varnish is applied, a sticky layer or film forms on the tooth surface. This film enhances the penetration and absorption of fluoride ions into the enamel. Although the sticky and sometimes colored nature of certain fluoride varnishes raises concerns about potential discoloration of tooth-colored restorations, studies have shown that fluoride varnishes can be used without causing significant color changes in aesthetic restorative materials.17,18 However, a limited number of studies have investigated the protective effects of fluoride-containing agents against the adverse effects of consumed foods and beverages on the surface microhardness and discoloration of restorative materials. Furthermore, in many studies evaluating the effects of beverages on restorative materials, the focus has primarily been on drinks frequently consumed by adults -such as coffee, tea, and red wine-while beverages frequently consumed by children receive little attention. Therefore, the aim of this study was to evaluate the protective effect of fluoride varnishes against the negative effects of beverages with differing pH levels—commonly consumed by children—on the surface microhardness and discoloration of fluoride-releasing restorative materials. The null hypothesis of this study is as follows;
Materials and methods
This in vitro experimental study was conducted at the Department of Pediatric Dentistry, Faculty of Dentistry, Karabük University, and the Department of Restorative Dental Treatment, Faculty of Dentistry, Okan University. Three different restorative materials and a fluoride varnish were used. The solutions for sample immersion were selected as beverages commonly preferred by children: sour cherry juice (pH = 3.4), chocolate milk (pH = 6.2), and cola (pH = 2.4). Distilled water (pH = 7.2) was used as the control. The properties of the restorative materials are presented in Table 1.
The restorative materials and fluoride varnish used in the study.

Sample size estimation
A power analysis was performed to establish the minimum sample size required to detect statistical significance with 80% power (power = 0.80) and a 5% significance level (α = 0.05). Based on an effect size (
Specimen preparation
Stainless steel molds measuring 7 mm in diameter and 2 mm in height,
20
fabricated from auto-matrix bands, were used to prepare the specimens of the restorative materials. To obtain a smooth surface, the metal molds were placed onto a glass plate covered with a mylar strip (Universal strip, EDD Dental, Hofheim, Germany),
3
and the restorative material was inserted into the molds. Pressure was applied to the top surface using a cellulose acetate matrix band (Mylar band), ensuring complete condensation of the material between the glass plate and the band.
21
Beautifil II and Dyract XP specimens were prepared according to the manufacturers’ instructions and light-cured for 40 seconds on one surface using a Valo Cordless curing unit (Ultradent, South Jordan, UT, USA). For Cention N, the powder and liquid components were mixed according to the manufacturer’s guidelines and placed into the molds, where they were allowed to self-cure. A total of 144 discs were prepared, with 48 discs for each restorative material. All samples were stored in distilled water for 24 hours to complete polymerization.3,22 The specimens of each material group were randomly divided into two equal subgroups: one to be treated with fluoride varnish (

Flowchart of the study.
Immersion cycle
The samples were immersed twice daily (morning and afternoon) for 15 minutes in the beverages, and this procedure was repeated daily for 28 consecutive days. 23 When not immersed in the beverages, the specimens were stored in artificial saliva at 37°C.
Measurement of color change
Initial color measurements of all specimens were performed using a spectrophotometer (VITA Easy shade V, Zahnfabrik, Bad Säckingen, Germany) before applying the fluoride varnish and immersing in the tested beverages. Before each measurement, the spectrophotometer was calibrated according to the manufacturer’s instructions. Measurements were taken under standardized lighting conditions using a white background, with the device tip positioned parallel to the surface and perpendicular to the specimens. The

After the first measurement, fluoride varnish was applied to the fluoride group. Fluoride varnish was applied to each specimen in a standardized amount of 0.1 mL using a dropper and evenly distrubuted over the entire surface with a brush. The varnish was left on the surface for 1 minute. Subsequently, all specimens were placed in artificial saliva and stored at 37°C for 24 hours, after which the second color measurement was performed after 24 hours. Using the first color measurement values before applying fluoride varnish and the second measurement after application, the effect of the fluoride varnish on color change (Δ
Measurement of surface microhardness
The surface microhardness of the prepared specimens was measured before immersion in the beverages (baseline) and on 28th day. Surface microhardness values were established using a digital microhardness tester (Shimadzu HMVG, Shimadzu Corp., Japan). For each specimen, three indentations were made with a pyramidalshaped indenter with a conical tip and a square base. The microhardness indenter performed three linear indentations starting from the center of the specimen in four main directions, with a 4 mm distance between each indentation. 27 The Vickers hardness number (VHN) was calculated by averaging the three values from the indentations. A load of 100 g was applied for 15 seconds during each indentation. 28
Statistical analysis
All statistical analyses were performed using IBM SPSS Statistics (version 30.0.0.0) and R (version 4.4.3) software. Due to the variety of research questions and factor levels, several methods were applied. For comparisons between independent groups, independent samples
To assess the effects of categorical variables and their interactions for repeated observation designs, generalized linear models (GLMs) and generalized linear mixed models (GLMMs) were fitted. Following model estimation, Tukey’s-adjusted estimated marginal means (EMMs) were calculated to identify significant pairwise differences.
Results
Color stability
The application of fluoride varnish prior to the immercion cycle (at 24 hour) had a significant effect on color change of the restorative materials (Table 2). For the group comparisons of color changes for each restorative material, fluoride application caused the greatest color change in the Cention N group (Figure 2).
Color change values according to the fluoride varnish application.


Effect of fluoride varnish based on restorative materials (24 hours after varnish application-before immersion cycle).
Comparisons of color changes for each restorative material are presented in Table 3. When color changes were calculated for the groups with and without fluoride varnish application, the mean color change (Δ
Comparisons of color changes for each restorative material.

Uppercase letters indicate comparisons among time points within each fluoride varnish–material–solution combination. Values sharing the same letter are not significantly different, whereas values with different letters are significantly different (Tukey-adjusted,
Lowercase letters indicate comparisons among solution groups within each fluoride varnish–material–time combination. Values sharing the same letter are not significantly different, whereas values with different letters are significantly different (Tukey-adjusted,
The restorative material with the most color stability was Cention N. In the fluoride applied group, Cention N exhibited the lowest median color change (Md = 1.01, interquartile range [IQR] = 1.16), followed by Beautifil II (Md = 1.19), and Dyract XP (Md = 1.35). In the group without fluoride application, Cention N again showed the lowest median color change (Md = 1.45, IQR = 1.59), followed by Beautifil II (Md = 1.49), and Dyract XP (Md = 1.79; Figure 3).

Effect of fluoride varnish on color stability over time based on restorative materials.
Paravina et al. 29 reported a perceptibility threshold for color change (ΔE₀₀) of 0.8 and the acceptability threshold of 1.8. When ΔE₀₀ values were evaluated accordingly, cherry juice caused clinically unacceptable color changes above the acceptability threshold at all time intervals in both fluoride-applied and non-applied groups of the Dyract XP. For the Beautifil II group with fluoride application, cherry juice caused clinically unacceptable color changes at all time intervals except for day 7. For the Cention N group, both fluoride-applied and non-applied samples showed clinically unacceptable color changes at days 14 and 28. Furthermore, samples immersed in cola also exhibited clinically unacceptable color changes at most time intervals. For the control group samples immersed in distilled water, clinically unacceptable color changes above the acceptability threshold were observed on day 28 in the Dyract XP group without fluoride application, and on days 14 and 28 in the Cention N group with fluoride application. Unlike the other groups, in the Cention N group, the color change in samples immersed in cola was higher in the fluoride-applied group compared with the non-applied group (Table 3).
In conclusion, the type of immersion beverages used had a significant effect on the color stability (ΔE₀₀) of the restorative materials (for the fluoride-applied group: H(3) = 28.3,

Effect of fluoride varnish on color stability over time based on solution groups.
Surface microhardness
A statistically significant difference was found between the material groups in terms of initial surface microhardness (χ²(2) = 101.42,
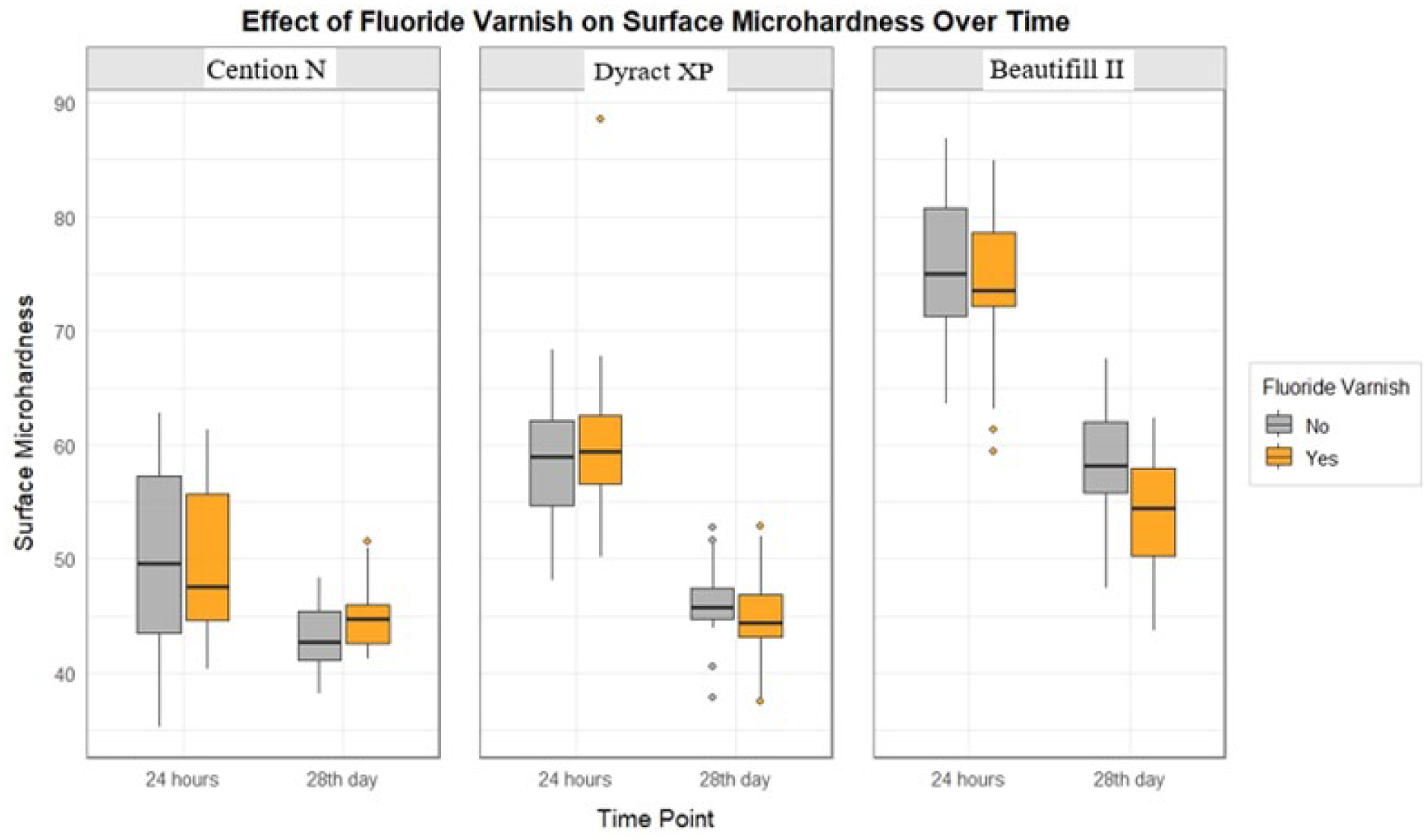
Effect of fluoride varnish on surface microhardness over time.
Relationship between surface microhardness and color stability
Both color change (ΔE) and time had significant effects on surface microhardness (

Relationship between surface microhardness and color stability.
Discussion
In line with increasing aesthetic expectations and demands for minimally invasive treatments, various restorative materials with contents properties have been introduced in dentistry. A thorough understanding of the mechanical and physical properties of restorative materials enables the development of more effective treatment approaches. 30 A significant factor affecting the clinical success and longevity of direct restorations is the consumption of colored and acidic beverages. The color changes caused by these beverages can cause concern among parents and negatively affect social interactions among preschool children. 31 This may require replacement of stained restorations in some clinical scenarios. 32
The effects of frequently consumed beverages by children on the surface microhardness and color stability of newly developed fluoride-releasing restorative materials—and when fluoride-varnished—used in pediatric dentistry, have not been comprehensively investigated. Therefore, in our study, the color stability and surface microhardness of newly developed materials, Cention N and Giomer, were compared with those of compomer, a conventional restorative material commonly used in pediatric dentistry.
With in vitro studies, the exposure time to beverages is considerably longer compared with daily consumption. In real life, beverages encounter restorative materials only for short periods. In most in vitro studies, materials are immersed in solutions for days or weeks, without considering the remineralization and buffering capacity of saliva. 33 In contrast, in this study, to better simulate daily consumption, the materials were immersed in the beverages twice a day for 15 minutes each time. During the remaining time, the samples were stored in artificial saliva to benefit from the buffering capacity of saliva.
In this study, beverages with different colors caused a significant decrease in the color stability and microhardness values of restorative materials. Additionally, the application of fluoride varnish improved the color stability of restorative materials but had no significant effect on surface microhardness. Accordingly, the null hypothesis of this study was rejected.
A common method for evaluating color stability is to assess color changes using either a spectrophotometer, spectroradiometer, or colorimeter. 34 Instrumental techniques such as spectrophotometry have the advantage of eliminating subjective differences in color perception and can detect color changes that are imperceptible to the human eye.35,36 Although the CIELAB formula is commonly used to evaluate color changes after dental materials are immersed in staining solutions, Herrera et al. 37 noted that the CIEDE2000 formula developed by the International Commission on Illumination corrects small color differences and improves the visual perception of color differences compared with the CIELAB formula. 38 Therefore, the CIEDE2000 formula was used in this study to measure color changes.
In this study, the color change of restorative materials statistically significant increased after the application of fluoride varnish. It is possible that the formation of a sticky film layer on the tooth surface after application might have made the restorative material more susceptible to color change. Similar to our findings, Prabhakar et al. 39 reported that the application of fluoride varnish affects the surface roughness of restorative materials, and changes in surface roughness alters the refractive index of reflected light, leading to color changes. In contrast, Autio-Gold and Barrett 17 reported that the use of fluoride varnish did not significantly affect the color change of restorative materials.
All restorative materials evaluated in this study showed color change after exposure to the beverages. On the 28th day, Cention N stood out as the material with the highest color stability, followed by Beautifil II and then Dyract XP. Factors that might influence color change include the properties of the resin matrix, degree of polymerization and hydrophilicity, the size and amount of filler particles, the mode and depth of polymerization, and the surface characteristics of the material.40–43 Some common monomers found in the resin matrix of resin-based restorative materials have hydrophilic properties, which can lead to color changes over time due to water absorption. The higher proportion of hydrophobic urethane dimethacrylate in Cention N’s composition is thought to contribute to its color stability.44–46 Unlike our study, Güler and Ünal 47 reported that a Beautifil II, which contains hydrophilic structures, such as Bis-GMA and TEGDMA, exhibited greater color change compared with a Dyract XP. The higher color stability observed in the Beautifil II in our study might be attributed to its lower resin content and smaller particle sizes that allow for better surface polishing, resulting in improved color stability.
In the literature, it has been reported that the degree of color change in resin-based restorative materials is affected by the duration of the testing periods.5,48 Consistent with other studies, our study also found that time is a crucial factor for the color stability of restorative materials, and that longer immersion times result in more pronounced color changes. 5
Tüzüner et al. 49 evaluated the effects of various pediatric medications on the color difference of polyacid-modified composite resin, composite resin, and glass ionomer cements. Their results showed that only the type of restorative material had a significant effect on the color difference values. In contrast, this study found that the restorative material, immersion solution, and fluoride application all had significant effects on the color difference values. These differing results might be due to variations in the formulations, pH values, and colors of the tested immersion solutions. 32 In our study, the pH values of cherry juice and cola immersion solutions were lower than the crucial pH value of 5.5, causing greater discoloration compared with samples immersed in chocolate milk and distilled water, which had higher pH values. Additionally, the highest color change observed in the red-colored cherry juice highlights the influence of the immersion solution’s color. Dark-colored immersion solutions, such as cherry juice, negatively affected the color stability of all restorative materials, although Cention N was the least affected material. With proper polymerization, finishing, and polishing, Cention N is expected to show more aesthetic durability.
In samples immersed in distilled water, the group treated with fluoride showed greater color change compared with the untreated group, whereas in samples immersed in other solutions, the addition of fluoride reduced color change. This effect might be due to the protective layer formed by fluoride varnishes on the surface of the restorative material. This layer acts as a barrier to the penetration of the immersion solutions into the materials. Similarly, Çevik and Hazar Bodrumlu 50 reported that fluoride varnishes could be beneficial against color changes caused by pediatric medications on the surface of compomer, which aligns with the findings of our study.
An important factor to consider when selecting dental restorative materials is their mechanical properties. Materials used for dental restorations should withstand masticatory forces. 51 Surface hardness is accepted as a key test parameter because it is an important property for the long-term durability of restorative materials in the oral environment. 52 Therefore, a decrease in the hardness of a material might lead to early failure of the restoration and the need for its replacement. The surface hardness of restorative materials can be affected by the characteristics of the filler particles (e.g., filler content, particle size, and resin formulation),53,54 water absorption, and chemical reactions occurring on the surface. 51 Microhardness tests are used to evaluate these mechanical properties in vitro. To assess surface hardness, various in vitro studies have been conducted using Vickers and Knoop hardness tests. The Vickers hardness test, also considered a microhardness test, is based on an optical measurement system. This test is commonly used for small samples and thin sections. When test specimens are carefully prepared, it is highly useful for evaluating various materials.53,54 In our study, the Vickers hardness test was used to evaluate surface hardness.
In this study, a significant decrease in microhardness was observed in all three materials after 28 days. While the type of restorative material had a significant effect on this change, fluoride application and the type of immersion solution did not have a significant effect on microhardness. Consistent with this study, most previous studies have reported that prolonged exposure of restorative materials to aqueous environments can reduce surface microhardness.44,55,56
Fluoride varnishes or other protective coatings can influence the microhardness and color stability of restorative materials; however, these effects are not always consistent in direction or magnitude. Fluoride varnishes, particularly on ion-releasing materials (e.g., Cention N, Giomer, Plyacid-modified
A statistically significant difference was found between the material groups in terms of surface microhardness at baseline and 28th day, Beautifil II had the highest microhardness, followed by Dyract XP and then Cention N. The lower surface microhardness of Cention N compared with compomer and Beautifil II could be due to its powder and liquid components being mixed, which might cause the formation of pores and air bubbles during this process. A review of the literature revealed no studies directly comparing Cention N with Dyract XP or Beautifil II. However, in a study by Adsul et al., 27 Cention N was compared with glass ionomer and zirconomer, and it was reported that Cention N had the highest surface microhardness. This finding was attributed to Cention N’s thick polymer network and high degree of polymerization. Nevertheless, the lower mechanical strength of glass ionomer and zirconomer compared with compomer and Giomer might explain the observed differences between the two studies.
The study found a negative correlation between color change and surface microhardness; as surface microhardness decreased, color change increased. As the surface microhardness of restorative materials decreases, the adhesion of pigments and coloring agents to the surface becomes easier, thereby increasing the risk of discoloration. However, microhardness alone is not the sole determinant of color stability. In this study, Cention N, which exhibited the lowest surface hardness, demonstrated the highest color and surface stability. The resin/monomer matrix of Cention N, present in the “LIQUID” phase, consists of four different dimethacrylates, accounting for 21.6% by weight of the final material. The combination of UDMA, DCP (dimethanol dimethacrylate), aromatic aliphatic-UDMA, and PEG-400 DMA (polyethylene glycol 400 dimethacrylate) cross-links during polymerization, providing strong mechanical properties and long-term stability. 58 Furthermore, Cention N does not contain Bis-GMA, HEMA, or TEGDMA and possesses a UDMA-dominant hydrophobic matrix. This composition reduces water absorption and limits the penetration of colored pigments into the material, thereby contributing significantly to its color stability. 59
The fact that our study was conducted under in vitro conditions may only partially reflect the biological variables that could be observed in a clinical setting. Factors such as the buffering capacity of saliva, oral hygiene practices, and individual dietary habits play a decisive role in the color stability and surface roughness of restorative materials. Nevertheless, in vitro studies are important because they allow for a controlled comparison of materials independently of clinical variables. Therefore, this research provides a foundational insight into the interactions between fluoride varnishes and bioactive restorative materials, serving as a precursor for future clinical investigations.
Conclusion
Fluoride-releasing restorative materials exhibited varying degrees of color change when immersed in different staining solutions. Among the tested materials, Cention N provided superior color stability and surface durability compared with Beautifil II and Dyract XP. Fluoride varnish application significantly reduced discoloration, supporting its potential benefit for routine clinical use, without affecting surface microhardness.
Footnotes
Ethical considerations
Since this study is based on material testing, ethical approval was not required (not applicable).
Author contributions
Conceptualization: MGT and MÇ, Data curation: MGT and MÇ, Investigation: MGT, Methodology: MGT and MÇ, Project administration: MGT and MÇ, Supervision; MGT, Writing: MGT and MÇ, Reviewing and editing: MÇ.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data generated in this study are available upon request from the authors.
