Abstract
Purpose
Tissue regeneration can be improved by local application of autologous bone marrow derived progenitor cells (BMSC) and platelet rich plasma (PRP). However, there is a lack of standardized application procedures for clinical use. Therefore, a technique in accordance with the guidelines for advanced therapies medical products of the European Medicine Agency was developed and established.
Methods
In detail, a process for the isolation and formulation of autologous bone marrow cells (BMC) and PRP in a clinical setting was validated. To investigate the influence of storage time and temperature on gel formation and gel stability, different concentrations of BMC were stored with and without additional platelets, thrombin and fibrinogen and analyzed over a period of 28 days. In addition, cell vitality using a live-dead staining and migration ability of human mesenchymal stem cells (hMSC) in the gel clot was investigated.
Results
For an optimized stable gel clot, human BMC and PRP should be combined with 10% to 20% fibrinogen (9 mg/mL to 18 mg/mL) and 5% to 20% thrombin (25 I.E. to 100 I.E.). Both freshly prepared and stored cells for 1 to 7 days had a stable consistence over 28 days at 37°C. Different platelet concentrations did not influence gel clot formation. The ratio of living cells did not decrease significantly over the observation period of 5 days in the live-dead staining.
Conclusions
The study identified an optimal gel texture for local application of BMC and PRP. Seeded hMSC could migrate therein and were able to survive to initiate a healing cascade.
Introduction
In the light of a constantly aging population, degenerative, atraumatic and traumatic diseases of the musculoskeletal system are major socioeconomic issues (1). One possibility to improve the healing of damaged tissue is the use of cellular-based therapies. Especially bone marrow has been identified as an excellent source of mesenchymal stem cells (MSC), which can generate multiple cell lines including osteo-, teno-, fibro- and chondrocytes (2-3-4-5). The great potential of MSC for tissue regeneration has been evaluated in many animal and a few clinical studies, and it has been suggested that cellular-based therapies should further be evaluated in humans (6-7-8-9). Classically, MSC are grown on plastic dishes from the mononuclear cell fraction after Ficoll gradient centrifugation (10). This procedure takes 2 to 4 weeks and for patient use “good-clinical practice” facilities are needed, which are costly. In contrast, “point-of-care procedures” might have several advantages: the fraction of the nucleated cells can be generated from bone marrow by use of volume reduction protocols, which can be done on the same day of the procedure in closed systems without a major laboratory facility. There are preclinical and clinical studies that prove the effectiveness of this technique (11), e.g. in avascular necrosis of the femoral head (12), nonunion (13-14-15), myocardial infarction (16, 17) and critical limb ischemia (18).
In recent years, platelet rich plasma (PRP) has been studied extensively in orthopedics, sports medicine, cardiology and neurology (19, 20). PRP is a concentrate of human platelets in a small volume and has a high concentration of growth factors like platelet-derived growth factors (PDGF), transforming growth factors (TGF), insulin-like growth factors (IGF), vascular endothelial growth factor (VEGF) and cytokines, which are necessary for cell differentiation and tissue healing (21, 22). There is evidence that PRP has several potential advantages, including faster recovery, e.g. from joint replacement (23, 24), and improvement in tissue healing (25), although other studies could not demonstrate improvement (26). However, the first results are promising, although clinical trials with high evidence level remain to be seen.
A critical issue for the translation of cell therapies in humans is an application which ensures local stable and viable cells. In several studies fibrin has been used as a natural biopolymer scaffold in tissue engineering (27-28-29-30). Currently, there is a lack of standardized application systems for clinical use. Therefore, a novel and patient-orientated technique was developed and established by the use of a double-chamber syringe with autologous bone-marrow concentrate (BMC) and allogen PRP, in accordance with the guidelines for advanced-therapies medicinal products of the European Medicine Agency.
Methods
Evaluation of the ratio between simulated bone-marrow concentrate, PRP, thrombin and fibrinogen solution.
The composite should contain the desired cells for tissue regeneration and provide a gel stable solution to ensure immobilization. Therefore, the optimal concentration ratio between bone-marrow concentrate, PRP, thrombin and a glue protein solution containing fibrinogen had to be evaluated. For the first investigations a simulated BMC (sBMC) was used to save human bone-marrow concentrate: a buffy coat (BC) contains most of the white blood cells and platelets of a whole blood donation after centrifugation of human blood. In brief, a BC was prepared according to standard blood-bank procedures. The BC was diluted with leukocyte-free red cell concentrate and plasma to a concentration of mononucleated cells (MNC) of approximately 20* 109 cells/L. The ratio of platelets versus the total of MNC and erythrocytes was calculated from the blood counts of the used PRP and sBMC. This number should represent the platelet supplement to a BM needed for formation of a stable gel. In a second series, the BC was depleted of platelets prior to dilution. This was done by low-spin centrifugation, discarding the supernatant, washing with 100 mL of PAGGS-M (conservation medium for packed red blood cell concentrate composed of phosphate, adenine, glucose, guanosine and mannitol) solution and repeating the first two steps. PRP was obtained by centrifugation of a regular pooled platelet concentrate, discarding an aliquot of the cell-free supernatant and re-suspending the platelets in the solution in varying concentrations.
For the assessment of the individual cell configuration, we analyzed the gel stability with an ‘Eppendorf test’, a standardized clot-forming test and documented the results photographically. For preparation of the glue protein, solution Tissucol Duo S Immuno (Baxter, Germany) was used. It contains fibrinogen (70-110 mg/mL, mean: 90 mg/mL), blood coagulation factor XIII (10-50 IU/mL), plasma fibronectin (2-9 mg/mL) and aprotinin (bovine; 3000 KI.E/mL). For activation, a thrombin solution with 500 IU/mL thrombin and CaCl2 × 2 H2O (5.88 mg/mL) was used. Before utilization, the kit components (stored at -20°C prior to use) were defrosted in a water bath at 37°C. To evaluate the influence of different concentrations of sBMC and PRP (Tab. I), all samples were pipetted in different autoclaved Eppendorf tubes (ET) and combined with fibrinogen and thrombin in various concentrations in a final volume of 0.3 mL (Tab. II). After preparation, storage at 37°C in an incubator and time ranges of 1, 2 and 5 days, the ET were turned upside down and documented photographically to evaluate the gel stability.
Ratio of sBMC/platelet rich plasma (PRP) and ratio of PRP to other cells like erythrocytes (Ery) and mononuclear cells (MNC)
Samples of glue protein solution (initial concentration fibrinogen: 90 mg/mL) and thrombin (initial concentration thrombin: 500 I.E.)
Influence of storage time, temperature of gel forming and stability
To investigate the influence of storage time and temperature of the gel formation and stability, the sBMC of varying compositions with and without additional PRP was stored at room temperature or 4°C for 24 h before preparation. After storage, the sBMC was mixed with 20% ‘glue protein solution’ and 9% thrombin in 0.5 mL ET with an end volume of 0.3 mL. Gel stability was evaluated after incubation at 37°C for 24 h, 48 h and 5 days as described before. As reference, fresh sBMC was tested in ET for gel forming and gel stability.
Influence of volume and PRP concentration
Different total-end volumes (0.3 and 0.5 mL) were investigated in the previously described way with end concentration of 20% glue protein solution and 9% thrombin. The stability was investigated with the ET test over a storage period of 5 days at 37°C.
To reflect subsequent donor PRP concentration variance, we evaluated the influence of platelet concentration in the sBMC samples by combining with a high (ratio platelets per ∑(Ery + MNC) = 5.7:1), middle (ratio platelets per ∑(Ery + MNC) = 0.9:1 and low (ratio platelets per ∑(Ery + MNC) = 0.3:1) concentration of platelets. Furthermore, the influence of different total volumes (0.3 mL and 0.5 mL) for the ET test was investigated. The gel stability was observed over the period of 24 h, 48 h and 5 days storage at 37°C.
Implementation of a double-chamber syringe
The optimal concentration for local application was prepared by using the double-chamber syringe FibriJet® with a large chamber (CatalogNumber: SA-1001, Vmax: 11 mL) and a small chamber (CatalogNumber: SA-1001, Vmax: 1 mL). The large chamber was filled with sBMC (ratio platelets per ∑(Ery + MNC) = 0.9:1) and glue protein solution (concentration: 90 mg/mL fibrinogen; final concentration in the application: 20%) and the small chamber with thrombin (concentration: 500 IU; final concentration in the application: 9% Table III). After placement of the syringe tip (Applicator Tip Dual Cannula, Malleable [10.2 cm]; FibriJet®; CatalogNumber: SA-3600) the components were uniformly emitted in: 0.5 mL Eppendorf tubes, 2 mL Eppendorf tubes, 2 × 25 mL centrifuge tubes, 3 × 6 well cell culture plates. The gel stability was documented photographically after 24 h, 48 h and 5 d storage at 37°C.
Preparation of double-chamber syringe
Gelation and gel stability with human bone-marrow concentrate (hBMC)
After finding the suitable concentrations of sBMC, platelets, ‘glue protein solution’ and thrombin, the tests were repeated with hBMC of three different donors. The hBMC was provided by Medical Clinic I of the local University Hospital after patients’ informed consent and approval by the local ethic committee (EK263122004, 10.12.2004). The process has been optimized to yield high recoveries of bone marrow progenitor cells using an automated volume reduction protocol on a Sepax 2 machine (Biosafe, Switzerland). Human bone marrow was connected via sterile tube welding (TSCD II; Terumo BCT, Japan) to a single use sterile separation kit (CS-490.1; Biosafe S.A.). The kit was installed into the Sepax 2 and the Generic Volume Reduction Protocol (GVR) Version 300 was selected. The parameter for a good yield of hMSC along with a low contamination of erythrocytes was determined in preliminary tests with sBMC. CD34-positive populations were taken as a substitute for MSC. Finally, the method providing a relative proportion of the BC, setting the parameter to 15%, was chosen. With this method the yield of CD34+ cells was 40-60% related to the unprocessed bone marrow, reducing the hematocrit to a value below 20%.
Live/dead assay
For the viability test, a live/dead viability/cytotoxicity kit from Invitrogen (Cat. # L-3224) was used. At 10 min after gel formation (250 µL gel per well of a 48-well plate; 20% ‘glue protein solution’ and 9% thrombin) a 0.2 mL staining solution (2.4 µM and 2.4 µM Calcein AM Ethidium homodimer-1 in phosphate buffered saline [PBS]) was supplied on the gel surface. After 30 min incubation in the dark the straining solution was replaced by phosphate-buffered saline (PBS) and the gels were cut exact centrally and were observed by using a fluorescence microscope (Keyence, Japan) in cross section in a 4-fold magnification. The photo-documented samples were blinded, so no observer was able to deduce the time of observation. The measurement was performed semi-quantitatively by three independent observers.
Migration
To test the ability of cell migration through the gel (250 µL gel per well of a 48-well plate; 20% ‘glue protein solution’ and 9% thrombin), the surface of the gels was seeded with stained (CellTracker™ CM-DiI, Molecular Probes® Invitrogen, 1 µg/mL) hMSC from three different donors with a cell number of 20,000 cells/gel in 0.1 mL α-MEM + GlutaMAX™ (Life Technologies GmbH, Darmstadt) containing 10% fetal calf serum (Sigma, Taufkirchen) and 1% penicillin-streptomycin (Biochrom AG, Berlin) and incubated in a humidified atmosphere with 5% CO2 at 37°C for 24 h, 2 and 4 days. In order to avoid dehydration, the gels were covered with additional 250 µL cell culture medium after 24 h. The hMSC were obtained from bone-marrow aspirates as mentioned above. After migration the cells were fixated in 4% formalin and the gels stored at 4°C. Cell migration and morphology was observed by fluorescence microscopy (Keyence, Japan) in cross section in a 4-fold and 20-fold magnification.
Results
Ideal ratio of simulated bone marrow concentrate, PRP, thrombin and glue protein solution
A ratio of 3:1, 1:1 and 1:3 (sample 1, 3 and 6 of Tab. I) regarding sBMC to PRP yielded no sufficient gel stability over the tested incubation time. Best results were obtained with a ratio of 2:1, 1:2 and 2:3 (sample 2, 4 and 5 of Tab. I). As a negative control, a sample without glue protein solution and thrombin (sampling 0_0 of Tab. II) was used for which no sufficient gel stability could be found. The addition of 10% glue protein solution (fibrinogen 9 mg/mL) and 20% (fibrinogen 18 mg/mL) allowed for an optimal stable gel texture. Regarding thrombin a concentration of 5% (25 IU), 15% (75 IU) and 20% (100 IU) showed a stable gel texture in the ET test (Fig. 1).
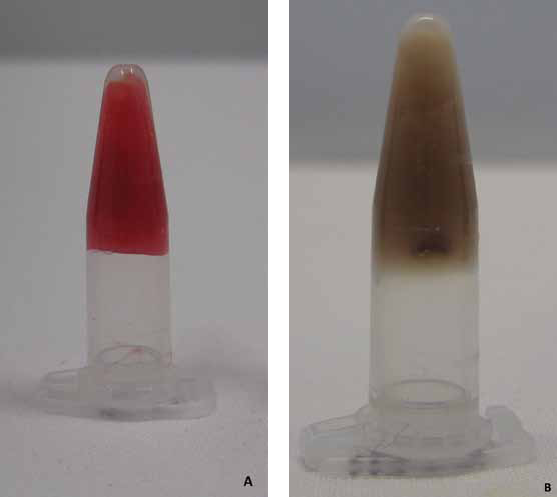
Gel stability of real bone marrow concentrate over 5 days (
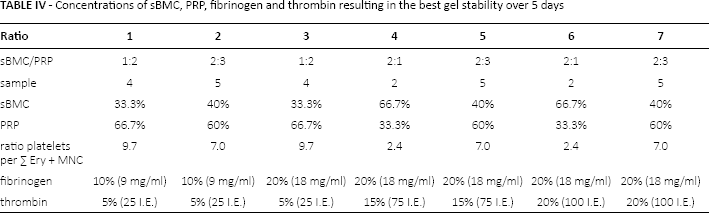
The ratios of an optimal gel stability of BMC, PRP, fibrinogen and thrombin are shown in Table IV.
Concentrations of sBMC, PRP, fibrinogen and thrombin resulting in the best gel stability over 5 days
Influence of storage time and temperature on gel forming and stability
Both fresh and sBMC, which were stored at room temperature for 24 h, allowed for a stable gel formation over 5 days. Addition of PRP did not influence gel formation and gel stability neither positively nor negatively. A storage temperature of 4°C did not cause stable composites (data not shown).
Influence of volume and platelet concentration
The volume up-scaling and the influence of platelet concentration in sBMC to simulate donor variance on gel formation and gel stability were repeated with a final concentration of 9% thrombin and 20% fibrinogen. It could be shown that sBMC in a volume of 0.3 and 0.5 mL with low (ratio platelets per ∑(Ery + MNC) = 0.3:1), medium (ratio platelets per ∑(Ery + MNC) = 0.9:1) and high (ratio platelets per ∑(Ery + MNC) = 5.7:1) platelet concentration were stable over 5 days. Further addition of platelets did not affect gel formation and stability, either negatively or positively. An up-scaling of volume from 0.3 mL to 0.5 mL ET and 0.5 mL to 2 was successful.
Implementation into clinical practice with a double-chamber syringe
After successful volume up-scaling, the application of the cells by use of a double-chamber syringe (final concentration of 9% thrombin and 20% fibrinogen; ratio platelets per ∑(Ery + MNC) = 0.9: 1) was tested. A stable gel formation was achieved with sBMC and a total volume of 1 mL in 2 mL ET. The gels were stable over the observation period of 5 days at 37°C.
Gelation and gel stability with human bone marrow concentrate (hBMC)
To evaluate the clinical translation and application, hBMC were used and the tests were repeated in regard of the influence of storage options, gel formation and stability. Both with freshly prepared and BMC, which was stored for 1 and 7 days, a stable gel could be achieved over 28 days (Fig. 2). These results confirm the feasibility of cell based therapy of hBMC and PRP in a glue protein solution and can serve as a preclinical base for further good medical practice (GMP) treatments.

Gel stability of real bone marrow concentrate over 28 day at 37°C. (
Live/dead assay
It could be shown that both fresh and stored BMC and PRP with the addition of 20% fibrinogen and 9% thrombin could form stable gels at room temperature. To document the viability of the cells in the gel, a viability assay was performed. Figure 3 illustrates that in fluorescence microscopically living (green) and dead cells (red) are detectable in the gel over the observation time of 5 days. As shown in Figure 3A, after initial gel formation there were already dead cells in the fresh samples. In the semi-quantitative measurement, a decrement of living and dead cells was reproducible. In the viability assay there were a mean 352 dead cells (n = 3, min: 345; max: 367; SD: 12.7) on day 0 and on day 5, 271 (n = 3; min: 252; max: 286; SD: 17.5) were observed. The living cells also decrease with a mean of 794 at day 0 (n = 3, min: 756, max: 844, SD: 45.1) to 616 on day 5 (n = 3, min: 577, max: 644, SD 34.8).

Live/dead staining of bone marrow and fibrin-gels (
Migration
We could demonstrate that hMSC were able to migrate into the gels. Figure 4 shows migrated cells.
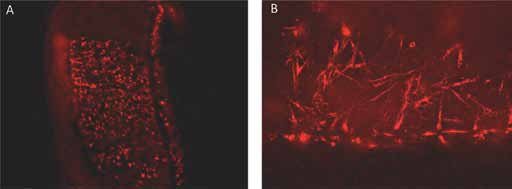
Migration of hMSC into bone marrow fibrin gels after 2 d incubation time at 37°C and 5% CO2; fluorescence micrographs taken with different magnifications (
Discussion
The present study aims at the application of cell-based therapies in clinical practice. The study demonstrates that there is an optimal ratio of BMC and PRP that can be applied in a clinical setting to enforce tissue regeneration. The process has been optimized to yield high recoveries of bone marrow progenitor cells using an automated, closed system volume reduction protocol on a Sepax 2 machine (Biosafe, CH). By adapting the volume reduction protocol, the yield of CD34+ cells was 40-60% related to the unprocessed bone marrow, reducing the hematocrit to below 20%. The prepared cells can be easily applied through a dual-chamber syringe in clinical use, and injected, e.g. into the site of injury.
There are several studies suggesting that BMC as well as PRP enhances tissue repair (31-32-33). Wang et al reported that treatment with PRP accelerate the extent of cell proliferation and collagen production by human tenocytes in vitro (34). Clinical studies confirm that local application of BMC can be utilized safely (35, 36). The cell composite of PRP and BMC improved pain, range of motion and strength, e.g. in rotator cuff injuries (36). One main issue in clinical practice is the extraction of bone marrow. As known from other indications, in the present investigation the iliac crest was used. Campbell et al used a system for bone marrow extraction (OnControl Automated Bone Marrow Harvest System by Vidacare, San Antonio, USA) that concentrated the bone marrow and injected the BMC into the hip joint hip in case of capsular injury in a professional soccer player by using a simple syringe (35). This technique does not guarantee an accurate retention of the cells at the site of interest. Therefore, the immobilization of the cells in a gel might be a good option. Nevertheless, the ideal matrix for delivery of BMC has not yet been identified. Some studies use a collagen gel matrix for colonization by bone marrow-derived mesenchymal stem cells (37). Awad et al achieved good results in terms of the biomechanical stability in rabbit patellar tendon. What is more, fibrin can be used as a carrier substance which creates a homogenous cell-matrix construct (37, 38).
The present study could show that a combination of 10% to 20% fibrinogen (9 mg/mL to 18 mg/mL) and 5% to 20% thrombin (25 I.E. to 100 I.E.) is optimal to achieve a stable gel texture for immobilization of BMC and PRP. To our knowledge, there are only few similar studies. Ho et al found the best proliferation of hMSCs within 5 mg/mL of fibrinogen (39). This is slightly less as compared to our study. However, the focus of this study was only the primary stability. In the present study, MSC also proliferate in the gel with a fibrinogen concentration between 9-18 mg/mL. Hankemeier et al performed a study with a mixture of hMSC and liquid fibrin glue by use of the same fibrin glue (40). The group found histological in an immunodeficient rat model, a more mature tissue formation and more regular patterns of cell distribution in the human BMSC group with a fibrin glue matrix, compared to a control group without BMSC and fibrin glue only after injection in a patellar tendon defect. The group did not investigate different concentrations of fibrin. Ahmed et al examined the influence of fresh fibrin and platelet-rich fibrin in an in vitro study; they found that fresh fibrin is more promising than platelet-rich fibrin as a tissue-engineered cartilage texture for chondrogenic differentiation of hMSCs (41). In the latter study, a commercial fibrin glue system (CryoSeal® FS System) was utilized (fibrinogen concentration: 20.1 mg/mL; thrombin component: 38 U/mL). Yasuda et al could show that by using an injectable fibrin matrix survival, migration and differentiation of bone marrow stromal cells is possible (42). To our knowledge no study reported the influence of the storage time and temperature on the stability and viability of BMC. The current study demonstrates that BMC, which was stored at room temperature for 24 hours, formed a stable gel clot over the period of 5 days at a temperature of 37°C.
Several studies demonstrate long-term vitality and proliferation of BMSC in fibrin glue in vivo (9, 37). In the present study, the number for both living and dead cells decreases slightly from day 0 to day 5. Nevertheless, this study confirmed that to any investigate point of time living cells (approximately 80% of initial value) are capable of bioactive interactions.
However, the current study has two limitations: on the one hand, it is a semi-quantitative measurement for living and dead cells, which allow an inter-individual bias. On the other hand, the living cells were stained with calcein, which stained both bone marrow cells and platelets. A differentiation and quantification between the cells was not performed and a final conclusion about the value of bioactive BMC was not possible. Nevertheless, the method confirms that cells are able to survive in the gel. The present study also demonstrates that MSC that were applied on the top of a fibrinogen clot are able to migrate into the clot and survive for at least 5 days.
Conclusion
The present study confirms that BMC in a composite of 20% fibrinogen and 9% thrombin could form a stable gel for 28 days at a temperature of 37°C. By using a double-chamber syringe a stable gel clot consisting of BMC and PRP could be applied in a translational approach. The advantage of this tissue engineering approach is the potential minimal invasive application through an injection. Therefore, the current study presents a novel application technique and makes a contribution for future in vivo studies.
Footnotes
Financial support: The authors have not received any financial support for this study.
conflict of interests: The authors have no conflict of interest for this study.
