Abstract
Osteoarthritis (OA) is the most prevalent musculoskeletal disease in humans and domestic animals. It causes significant clinical problems and substantial health care costs. In the absence of disease-modifying medical intervention, therapy is currently restricted to palliative measures prior to surgical intervention.
We review the pathogenesis, as well as conservative and emerging restorative therapeutic approaches, including cytokines, stem cells, and platelets. The various methods of platelet concentrate preparations and their reported outcomes are discussed. Data collected from the use of intra-articular platelet therapy (IAPT) in dogs are reviewed, which suggest that this approach may delay or in some cases even obviate the need for surgical intervention.
Introduction
Musculoskeletal diseases are a significant healthcare expense,1–3 and osteoarthritis (OA) is the most prevalent among them.4,5 As the population ages, the incidence of OA increases. 6 The pathophysiology of OA is incompletely understood and conventional drugs do not modify the disease. Palliative therapies include analgesics such as acetaminophen;7–9 nonsteroidal antiinflammatories such as cox-2 inhibitors;10–13 topical applications such as diclofenac and capsaicin;14,15 intra-articular medications such as hyaluronic acid (HA),16,17 corticosteroids,18,19 and prolotherapy compounds.20,21 Dietary supplements such as glucosamine and chondroitin22,23 are used in poorly documented attempts to rebuild cartilage. Acupuncture,24,25 transcutaneous electrical nerve stimulation,26–28 and orthotic devices29–31 are used on occasion as well. Therapy is further complicated by a variety of presentations, structural and symptomatic stages, co-morbidities, as well as individual factors including gender, age,32,33 and genetics.34,35
OA is the result of cartilage degradation with loss of tensile and compressive strength, bone-on-bone contact, and inflammation. Cartilage preservation, together with regenerative therapies directed toward subchondral bone and synovia,36–41 is a logical treatment goal. The immune system and a host of cytokines are operative in the disease process and ultimately must be considered in any therapeutic approach. 42
Bone Cartilage and Synovium
Figure 1A displays the knee, a synovial or diarthrodial joint. Apposing ends of the femur and tibia are covered by hyaline cartilage with intervening meniscal tissue. The joint is bathed in synovial fluid contained within the capsule (Fig. 1B) consisting of an inner synovial membrane and an outer collagenous fibrous layer, ie, the synovium (Fig. 1C). Blood vessels in the fibrous layer supply fluid and nutrients to synovium and cartilage. 43 Four major ligaments, anterior and posterior cruciate and medial and lateral collateral ligaments, hold the bones together (Fig. 1A).
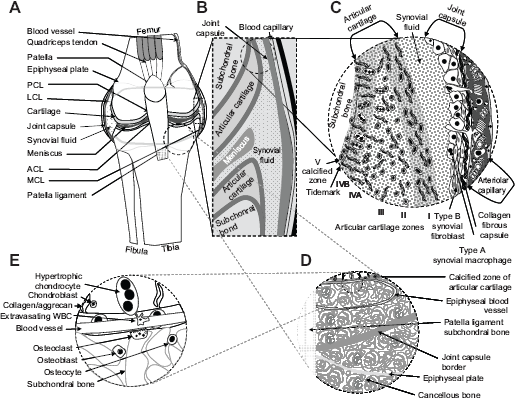
Conceptual drawing of a diarthrodial (knee) joint depicting varying levels of detail.
The synovial blood vessels (Fig. 1C) provide most of the oxygen found within the synovial fluid bathing the chondrocytes, the cells largely responsible for the production of cartilaginous tissue. 44 Chondrocytes thrive in a medium that is avascular, hyperosmotic, and relatively hypoxic. 45 Gene transcription factors including Sox9 and hypoxia-inducible factor 1-alpha (HIF-1α) 46 facilitate the hypoxic adaption.47,48
The synovium comprises two layers, the intima and subintima (Fig. 1C). The subintima is composed of collagenous fibers and contains macrophages, fibroblasts, adipocytes, and tissue molecules like elastin (Fig. 1C). 49 The synovium when challenged by disease responds with an increase in the intimal thickness and an influx of macrophages, fibroblasts, and blood-derived leukocytes. Damage to all of these joint structures is a feature of OA.50–54
Bone comprises an external compact surface, cortical bone, and an inner porous structure, trabecular or cancellous bone. Dense cortical bone provides strength and offers a rich vascular supply. It is an important source of mesenchymal and hematopoietic stem cells 55 for bone remodeling.
Both the osteocytes and osteoblasts function in the modulation of mineral deposition. 56 Calcium crystal deposition is a common feature of OA, which is of interest in the study of the articular cartilage mineralization and a therapeutic target for OA. 57
Osteoclasts demineralize bone through the action of acid proteases, like cathepsin K. Remodeling consists of both resorption by osteoclasts and bone formation by osteoblasts and osteocytes. 58 In addition, remodeling is, in part, governed by bone morphogenic protein (BMP)-2 59 and transforming growth factor (TGF)-β, which also play a role in the pathogenesis of OA. 60
Articular and fibrous cartilages afford tensile strength and resistance to shear forces; however, their chemical compositions differ. Type II collagen (CII) normally present in the articular cartilage is often replaced by other types of collagen in joints affected by OA. This alteration decreases tensile strength.61,62 The meniscus and joint capsule contain type I collagen, which together with fibrochondrocytes is arranged in layers that allows for a distribution of radial stress. 63 Collagen types provide a platform for HA polymers to which aggrecan molecules are affixed through a linker protein.64–66
Aggrecan itself comprises a core protein, chondroitin sulfate (CS), and keratin sulfate (KS) molecules (Fig. 2). CS and KS are negatively charged polysulfated glycosaminoglycans (GAG) 67 and provide for water retention within the extracellular matrix (ECM). HA or hyaluranon serves as a thread to which aggrecans attach.
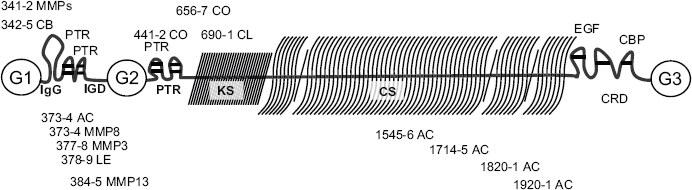
Aggrecan and sites of proteolytic cleavage.
Cartilage also contains other non-collagenous proteins including cartilage oligomeric matrix protein (COMP), 68 often used as a marker of disease progression.69–71
HA complexes with a glycoprotein, lubricin, which provides lubrication.
72
The recent data that support HA therapy in OA are not compelling,52,73–75 however, lubricin therapy shows promise.76,77 CS with or without glucosamine or
The degradation of collagen and aggrecan is largely the result of proteolytic enzymes, metzincins.
80
Catabolism of collagen and ECM proteins is mediated by matrix metalloproteinases (MMP). Aggrecan breakdown is caused by enzymes known as
The MMP and ADAMTS not only cleave ECM proteins but also activate other MMP, establishing an interdependence in function.86–89 Statistical data combined with protein microarray analyses reveal that MMP-7 is a good biomarker for the early detection of OA, preceding symptoms by as much as 10 years. 90
The action of aggrecanases may be required prior to collagen cleavage by MMPs, making anti-aggrecanase strategies an area of therapeutic interest.91,92 Of particular interest is MMP-13 as it appears to be the most prominent MMP in the breakdown of aggrecans and CII.93,94
Joint and tissue inflammation is the result of a cascade of proteolytic enzymes and cytokines. Cytokines are categorized as interleukins (IL), lymphokines, and chemokines. 95 Among the IL are IL-1 to IL-6 and granulocyte-macrophage colony stimulating factor (GM-CSF). Lymphokines include GM-CSF and interferon-gamma (IFN-γ). Chemokines such as IL-8 are cytokines that are chemoattractants, which cause leukocytes to migrate toward areas in which they eventually concentrate. Synovial tissue contains a variety of cells that produce pro-inflammatory cytokines present in patients with OA, although the precise source of these cytokines is incompletely understood. 48
Pro-inflammatory cytokines are recognized as an important part of the pathophysiologic process of OA resulting in the receptor-mediated stimulation of downstream biological response modifiers.42,96,97 TNFα appears to drive the inflammatory cascade that suppresses synthesis of proteoglycan, link protein, and CII. IL-1β effects cartilage destruction by inhibiting collagen and aggrecan levels and stimulates the release of MMP-1, -3, and -13 as well as IL-6, IL-8, monocyte chemoattractant protein (MCP)-1, and regulated on activation, normal T cell expressed and secreted (RANTES). Major research regarding the role of cytokines in OA and potential therapeutic intervention has been focused on the pro-inflammatory cytokines, tumor necrosis factor-alpha (TNFα), and IL-1β.98–100
Cytokines modulate the expression of MMP and ADAMTS as well as alter cellular mechanisms of inducible nitric oxide synthase (iNOS), soluble phospholipase A2, cyclooxygenase-2 (COX-2), and membrane prostaglandin E synthase (mPGES) leading to PGE2 expression. Cyclooxygenases are selective for the COX-2 isomer in chondrocytes and synovial fibroblasts. 101 Stimulation by IL-1β and TNFα results in the upregulation of mPGES. 102 While prostaglandin E has been targeted as an anti-inflammatory strategy, the approach is limited by gastrointestinal irritation and myocardial damage. Future studies targeting one of the three isoforms of mPGES have been suggested as a refinement in prostaglandin therapies for OA.103,104
Chondrocyte exposure to pro-inflammatory cytokines increases gene expression of iNOS. This in turn facilitates the secretion of nitric oxide (NO), and mPGES increasing the release of PGE2. PGE2 inhibits collagen and proteoglycan formation and also the production of the natural antagonist to IL-1β, the IL-1 receptor antagonist (IL-1Ra). The latter has been shown to positively affect joint inflammation and OA
105
although resulting in marginal clinical improvement.
106
The recent observation of three single nucleotide polymorphisms in the
Both IL-1β and TNFα stimulate the production of reactive oxygen species (ROS) including a cascade of oxidants such as NO, peroxynitrites, the superoxide anion, the hydroxyl radical, hydrogen peroxide, hypochlorous acid, and chloramines. The ROS are highly destructive to living cells unless contained or reacted with neutralizing molecules, 108 which are known to be down-regulated in OA. 109 The result is cell damage including programmed cell death or apoptosis of chondrocytes.110,111
The molecular mechanism by which several cytokines initiate inflammation in the OA joint has been studied through nuclear transcription.35,96 IL-1P and TNFα appear to mediate chondrocyte hypertrophy by modulating several transcription pathways including c-Jun N terminal kinase, p38 mitogen-activated kinase, and nuclear factor kappa B (NF-κB).96,112 Collagen, MMP, and growth factors like vascular endothelial growth factor (VEGF) mediate effects of the runt-related transcription factor 2 (Runx2), parathyroid hormone-related protein (PTHrP)/Indian Hedgehog, Wingless/Int (Wnt)/β-catenin, and TGF-β/Sma and Mad Related Family (Smad) pathways, 113 and Sox9.114,115 Interfering with transcription factor control then becomes another path to develop disease-modifying treatments for OA.
The complexity of approaches to develop a disease-modifying therapy for OA is diagrammatically illustrated in Figure 3. 35 Given the diversity of the paths and the requirements for proof of safety and clinical efficacy, it will take considerable time for such treatments to become popular alternative to conventional therapy.
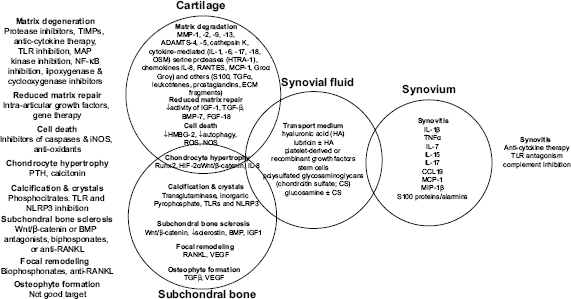
Treatment strategies for osteoarthritis.
Stem Cells, Growth Factors, and Platelet Therapy
OA is characterized by intra-articular cartilage loss, osteophyte formation, and capsular thickening. Regenerative injection therapies are thought to promote inflammation, proliferation, and remodeling. 116 Tissue repair by recruitment of endogenous stem cells has been demonstrated in humans and animal models.117-120 Regenerative therapy is complicated by the selection of cell type, stage of differentiation, cell number, pre-conditioning regimens, route of administration, time of intervention, and many other factors. 121 Much of the early data regarding clinical efficacy are derived from level IV evidence; large prospective randomized trials have yet to be completed.122,123 Nevertheless, stem cells are currently being considered for a wide range of reparative applications including OA.124–127 The tissues targeted in OA include the inflamed synovium, 128 the meniscal cartilage, 129 and subchondral bone.130,131 Mesenchymal stem cells (MSC) are procured from a variety of autologous and/or allogeneic tissues and have been used in several OA trials.132,133 The effect of aging on autologous stem cell properties and their ability to synthesize Type I collagen has yet to be defined. 126 The stage of inflammation at which treatment is initiated is of importance, because the inflammatory component of joint disease can influence stem cell activity.134,135 While stem cell therapy will be a subject of great interest and study for quite some time, the mechanism of action of stem cells remains to be elucidated. Clinical application of stem cell treatment in OA has shown some success. Both subjective and MRI measurements confirm improvement over 12 months of observation.136–138 Osteochondral lesions of the knee have also been successfully treated with MSC.139–141
Studies of fat-derived MSC in dogs revealed significant improvement in outcomes when treating hip 142 or elbow OA.143,144
Growth factors, perhaps as surrogates for direct application of stem cells, also may provide a path for regenerative healing. 145 Although the use of isolated growth factors has not revealed consistent results, use of conditioned plasma or platelet therapy, as a source of cytokines and growth factors, has shown more promise.106,146 Recognition of the potential interactions between growth factors such as BMP-7, IGF-1, bFgF, and FGF-2 has prompted the use of platelet concentrates due to their recognized association with healing as they are a repository for multiple growth factors.
Platelet Therapy
A recent review of the use of platelet concentrate therapy in tennis elbow and rotator cuff repair shows promise, 147 however, other applications such as long bone healing remain to be proved. 148 Platelet concentrate proves to facilitate stem cell mediated chondrogenesis in animals with artificially induced OA. 149 In addition, tendon,150–154 ligament,155,156 and disease injury including OA 157 have all been reported to be successfully treated in humans with platelet therapy.
Much remains to be detailed concerning the use of platelet therapy, including the dose-response effect, the duration of efficacy, the manner of administration, the platelet concentration required, and the synergistic role of white cells in the preparation. Although observational studies suggest a favorable influence of platelet therapy in OA, the mechanism of action remains unknown. Of interest is evidence that an increase in growth factor blood levels occurs following IA injection of platelet concentrates. 158
Platelet therapy for OA of the knee has been reported as beneficial in a number of studies.159–162 Most investigators conclude that platelet therapy alone or in combination with stem cells deserves further study in large randomized trials. 106
Types of Platelet Therapy
Platelet therapy is often referred to as platelet-rich plasma (PRP), a term used in blood banking; however, in regenerative medicine it is a misnomer. The product is actually platelet concentrate and is prepared using either a centrifuge or a filter. Some methods are standard centrifugation steps that are reminiscent of blood banking protocols and are conducted in a biological safety cabinet to maintain an aseptic environment. Automated centrifugal methods and filtration employ disposable components with aseptic fluid paths. The resultant products have characteristics that can be used to distinguish the method of preparation and might affect their clinical effects.
The preparations can be characterized using a 2 × 2 matrix defining them as either WBC rich or poor and containing or not containing fibrin. 163 Fibrin is produced by the addition of an agonist such as bovine thrombin and/or calcium salts. The presence of fibrin is thought to provide a matrix from which growth factors released from platelets will remain entrapped. Additionally, the agonist produces a more immediate release of growth factors from platelets allowing high levels to be achieved over short periods of time.
Some investigators express concern over the presence of WBC and their potential to cause inflammation 164 while others argue that they serve a valuable purpose in augmenting the levels of TGF-β 165 and may decrease the risk of infection following intra-articular injection.166–171 Yet another classification characterizes the platelet preparation based on its platelet count, activation status, and WBC content (PAW). 172
Little is known concerning the role of red cells present in the platelet product intended for IA injection. Data from horses at 24 hours after the injection of 10 mL of autologous blood into the metacarpophalangeal joint show no untoward effects. In fact computerized force measurements comparing the injected limb with the un-injected limb remained unchanged. 173
Variables in Platelet Therapy Clinical Trials
Pharmacologic evaluation of platelet therapy is desirable but has yet to be done. Some animal model data on platelet-derived growth factor (PDGF-BB) in rat Achilles collagenase-induced tendinopathy exist, 174 but a full characterization of platelet therapy has not been forthcoming. In part, this is due to the variable composition of the preparation. Platelet therapy has been used in a variety of indications with varying degrees of success. Data collected to date used to support or refute the selection of a given preparation have been largely absent, and what is available comes from in vitro or explant cultures.
Other variables include the route of administration, intra-lesional injection for tendon and ligament, intra-articularly for joints, and option of single or multiple therapy sessions. Standardization and characterization of the platelet therapy is a reasonable step toward understanding the limitations of the product in any particular application.
The following studies are summarized to exemplify the divergence of reports in the literature. Seventy eight patients with bilateral OA of the knee received either a single or double intra-articular injections spaced three weeks apart. Both platelet therapy groups improved significantly when compared to saline controls, however, no differences were observed between those receiving a single or double injection. 175
One hundred patients with OA of the knee and chondropathy received either intra-articular platelet or HA therapy. Assessment was performed at 12 months using the Likert scale measurements and range of motion. 176 Those with low-grade articular degeneration appeared to respond more favorably but statistical significance was not achieved. Of note, when more than one injection was used in this study, frozen and thawed platelets were administered, which could have affected the results.
A group of 50 patients with OA of the knee, previously treated with cartilage shaving or microfracture procedures, received two doses of platelet therapy with a 12-month period. 177 All showed significant improvement from baseline without significant differences between the previous treatment subgroups.
Half of the 120 patients with OA of the knee were treated with three weekly intra-articular injections of either platelet therapy or HA. The latter being a brand specifically chosen to have a molecular weight shown to possess enhanced viscoelasticity. 178 The platelet product contained a 4.5-fold increase in platelet concentration and a 3.6-fold increase in WBC compared to native blood. Both treatments improved outcome over the six-month follow-up period as determined using subjective score measurements, however, the improvement with platelet therapy was superior.
The impact of age on success with platelet therapy was revealed in a study of 150 patients with OA of the knee. Patients were treated with either low- or high-molecular-weight HA or with platelet therapy. 179 Results revealed significant clinical improvement over the six-month trial with platelet therapy outlasting either form of HA; however, all treatments were less effective in older patients or in patients who had more progressive disease.
A study of 140 individuals with severe OA of the hip entailed treatment with intra-articular administration of calcium chloride treated platelets, designed to produce fibrin formation. The majority experienced clinical improvement for at least six months. Many experienced early responses including reduced pain and improved mobility within 6–7 weeks. 180
An additional 91 patients with OA of the knee treated with platelet therapy achieved significant improvement at one-year post-treatment. 181 After two years, patients continued to show improvement from baseline although the extent of the improvement declined thereafter. 182
Some remain critical of the paucity of high quality randomized controlled trials for platelet therapy,116,183 whereas others recognize the potential value of platelet therapy as a treatment option. The complexities and lack of standardization of the platelet therapy constituents make controlled studies difficult to design and evaluate. 184
Animal studies are a logical approach to more concisely document the benefits of platelet therapy in degenerative joint disease. In a recent publication, the effect of platelet therapy on canine OA was studied using force plate kinetics, a visual analog score for lameness, and the canine brief pain inventory (CBPI) for pain measures. 185 Platelet therapy was prepared by closed system filtration producing a product that was 3-fold platelet and 2-fold WBC enriched over whole blood with an hematocrit of 27%, ie, leukocyte rich, fibrin poor, and consequently a non-activated platelet product. Dogs with naturally occurring OA of the hip were treated with either a single dose of saline or platelet therapy by IA injection using volumes from 3 to 6 mL in the affected joint. Three months after treatment the platelet therapy group significantly improved compared with the saline controls with respect to all response measures. The magnitude of change of peak vertical force among platelet therapy treated animals was 8%, statistically significant from baseline, and with a magnitude that rivals that found with NSAIDs. 186 The magnitude of change measured using subjective measurements of clinical efficacy suggested a 35% improvement in the Hudson visual analog score (HVAS) for lameness as well as the CBPI; comparable to those seen for treatment of canine OA with accepted therapies. 187 Recent data confirm that subjective measures such as the CBPI and HVAS correlate well with the more objective measurement, peak vertical force. 188
This paper describes the pilot studies that justified the recently published prospective randomized control trial described above. 185 In a yet to be published study sponsored by the manufacturers of the Pall platelet filtration system, 20 veterinarians enrolled 100 dogs with confirmed lameness based on clinical judgment. Analgesics were discontinued for one week prior to assessing the animals with the HVAS for lameness. The filter based autologous platelet concentrate contained a 3-fold increase in platelets, a 2-fold increase in leukocytes, and a hematocrit approximating 27%. It was administered as a single injection and repeat HVAS scores were obtained at three months for each dog.185,189
HVAS data were analyzed using non-parametric analyses. The Wilcoxon matched pairs test was used to compare the median of pre- vs. post-treatment values in a paired test. A test of the hypothesis that slope was equal to zero was employed to determine if the regression coefficient should be considered important enough to consider its magnitude.
Results of the platelet therapy were previously reported.
185
Interestingly, animals for which data were available regarding platelet concentration, white cell concentration, and/or normalized values for both platelets and white cells failed to show a statistically significant difference in HVAS scores (Fig. 4). Support for a dose-dependent or component-dependent effect could not be obtained. HVAS baseline and three-month post-treatment responses are depicted in Figure 5. From these data, we see that all responses show significant (
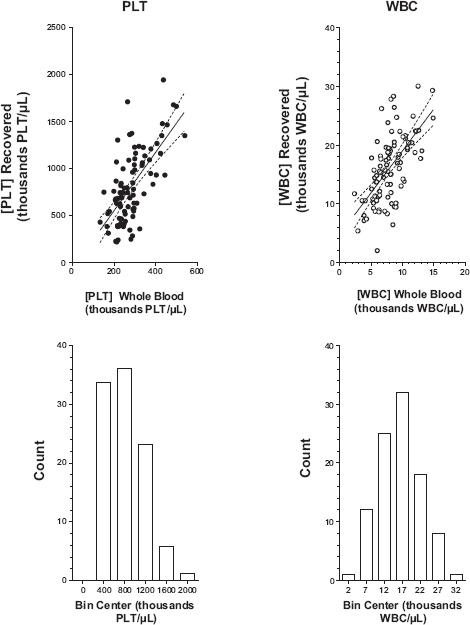
Platelets and white cells in whole blood compared with levels found in the platelet therapy produced using the Pall C-PET filter system.
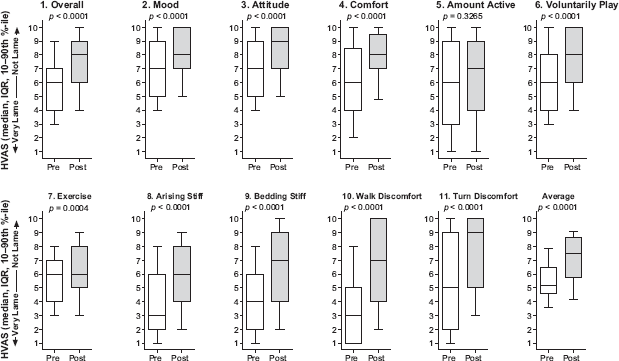
Responses to individual questions of the Hudson visual analog score before and three months after intra-articular platelet therapy for lameness and pain.
The condition responsible for lameness allowed for the disease to be sub-classified as either OA, tendon or ligament damage (T/L)-associated lameness, and a less descriptive category involving bone-related lameness other than OA. All animals showed improvement (Fig. 6). The magnitude of the improvement for OA, tendon or ligament damage and bone appear to be approximately 30% by weighted average (OA = 28.1%, T/L 35.6% and bone 54.5% improvement).
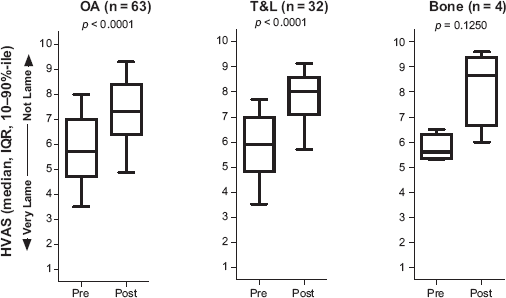
Response to platelet therapy by indication.
Data suggest that older subjects do not improve as well as younger ones. Segmenting the dog population by age (Fig. 7) illustrates that most animals receive benefit through the age of 10. Animals appear to be less responsive to platelet therapy beyond this age when analyzed by the one sample Wilcoxon sign test.
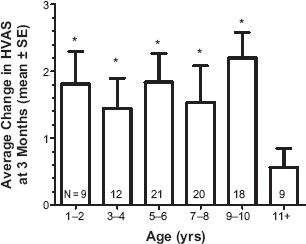
Response to platelet therapy by age. • Denotes statistically significant difference from the value zero as determined by application of either
Dose-response data were examined as a difference in HVAS pre- vs. post-treatment at three months as a separate function of individualized platelet concentration, WBC concentration, normalized platelet concentration, WBC concentration, and as combined normalized values for platelets and WBC. None resulted in a correlation coefficient greater than 10%, yet all showed statistically significant non-zero slopes. A trend was observed in a subset of animals treated for OA who had one- and three-month follow-up data, suggesting that a longer duration of improvement may correlate with higher concentrations of platelets, although there was no statistically significant difference in HVAS scores at three months with platelet concentration (Fig. 8).
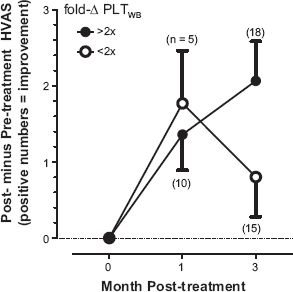
Dose-response of platelet therapy. Dogs less than 11 years of age with diagnosis of OA and for whom both one- and three-month follow-up data exist along with identifiable fold-change in platelet concentrations relative to whole blood (fold-δ PLTWB). A Mann–Whitney
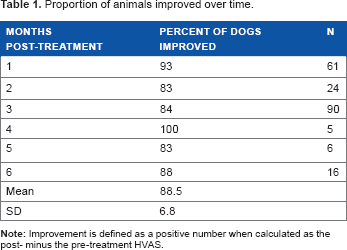
Additional data were derived from studies conducted by veterinarians who followed treated animals beyond three months (Table 1). Difference in pre- and post-treatment HVAS values suggest that some degree of improvement persists for at least six months following platelet therapy. Three animals have sustained improvement at one-year post-treatment.
Proportion of animals improved over time.
Discussion
OA is a complex disease process in which the biochemistry and structure of cartilage, subchondral bone, and synovium are altered. Current medical therapies are strictly palliative and newer approaches, although encouraging, are poorly supported by clinical trials.
Among the emerging therapeutic approaches is the use of platelet concentrate, a product which generates a host of cytokines known to be involved in tissue repair. The observations reported here for canine OA are consistent with studies found in the literature. The subjective assessment employed correlates with force plate kinetics data 189 and is consistent with a prospective randomized, saline control trial. 185 The magnitude of the response is the same, approximating 30% improvement.
These observations uniquely analyze the duration of effect of a single injection. Reported human studies employed multiple injections over 2–4 weeks using an initial freshly prepared platelet preparation followed by thawed frozen aliquots of the originally procured product. The current data suggest multiple injections may not be required. The single injection provides rapid onset and a sustained duration lasting at least up to six months. Limited anecdotal data suggest effects can last a year or more. Eighty four percent of the animals treated in these studies experienced some degree of improvement. Human data show that the effect of treatment regimens can peak at about a year and persist for two years although the magnitude of the effect is lower than at one year but still higher than baseline. 190
The majority of the published studies involve platelet preparations obtained with varying methods, a small number of patients, the absence of a suitable control group, or subjective measurements. Although attempts at systematic review or meta-analysis have been made,191–193 the majority of data are currently not suited for such an approach. Large, randomized control trials are required in the final analysis.
This review is consistent with the majority of publications regarding platelet therapy. It is intended to provide the reader with resources to identify the weaknesses in the body of literature, understand the rationale, purported mechanisms of action, and limitations of platelet therapy for OA.
The platelet product used in the canine studies proves to be an effective adjunctive treatment. Its role as a primary therapy remains to be elucidated. The technique offers the advantage of being self-contained, provides a sterile fluid path, and is easily incorporated into a clinical practice.
Author Contributions
Conceived the concepts: GAO and BW. Analyzed the data: GAO and BW. Wrote the first draft of the manuscript: GAO and BW. Contributed to the writing of the manuscript: GAO and BW. Agree with manuscript results and conclusions: GAO and BW. Jointly developed the structure and arguments for the paper: GAO and BW. Made critical revisions and approved final version: GAO and BW. All authors reviewed and approved of the final manuscript.
