Abstract
Purpose
The purpose of this in vitro study was to evaluate the effect of propolis on bond strength to dentin of a self-etch adhesive when used as a final irrigant.
Methods
126 human third molars were sectioned 3 mm below the occlusal level and then randomly divided into 7 groups (n = 18). The exposed dentin surfaces were treated with irrigation solutions as follows: Group 1: 5.25% NaOCl; Group 2: 5.25% NaOCl followed by 17% EDTA; Group 3: 5.25% NaOCl followed by Tubulicid Plus (Temrex, Freeport, NY, USA); Group 4: 5.25% NaOCl followed by MTAD; Group 5: 5.25% NaOCl followed by 20% propolis; Group 6: 5.25% NaOCl followed by 2% CHX; Group 7: distilled water (control). Irrigated specimens were bonded using Clearfil SE Bond (Kuraray, Osaka, Japan) with Tygon (Saint-Gobain, Saint-Gobain Performance Plastics, France), tubes for microshear bond strength testing. Data was analyzed using a one-way ANOVA and Tukey test.
Results
Among the tested groups, although the lowest bond strength values were detected in Group 1 (11.25 ± 1.97), a significant statistical difference was detected only between this group and Group 4 (18.70 ± 1.82) and Group 5 (16.75 ± 3.70) (p = 0.0012).
Conclusions
20% propolis solution as a final irrigant was found to have a favorable effect on the dentin bond strength of the tested self-etch adhesive to coronal dentin when compared with other irrigation solutions.
Introduction
The effective cleaning, shaping, and creation of a fluid-tight seal in a root canal are essential aspects of a successful endodontic treatment (1). Also, coronal sealing after root canal treatment is an important factor in the prognosis of endodontic treatment (2, 3). A lack of sealing and adhesion in a coronal restoration and tooth structure can cause reinfection of the root canal system due to the penetration of microorganisms (4).
The use of adhesive systems has been suggested for the restoration of endodontically treated teeth (5). Adhesive systems have many advantages over nonadhesive materials, such as reducing microleakage and reinforcing weakened tooth structure (6). However, many factors can potentially affect the adhesion of bonding systems to dentin. Many irrigants used in endodontics, such as sodium hypochlorite (NaOCl), ethylenediaminetetraacetic acid (EDTA), chlorhexidine gluconade (CHX), and BioPure MTAD (a mixture of citric acid, doxcycline hydrochloride, and a detergent; Dentsply, Tulsa, OK, USA) (7) can change dentinal properties and effect the adhesion of materials used for coronal sealing (8).
In recent years, propolis has been investigated as an irrigation agent as a new alternative to conventional materials because of its antibacterial activity (9, 10). In dentistry, the use of propolis has been proposed in different areas, including cariology (11), oral surgery (12), endodontics (9), and periodontology (13). Propolis is a natural, resinous hive substance that honeybees produce from plants (14). The composition of propolis is very complex, consisting of organic compounds such as phenolic compounds and esters, all type of flavonoids (flavonols, flavones, flavonones, dihydroflavonols, and chalcones), terpenes, beta-steroids, aromatic aldehydes and alcohols, sesquiterpenes, and stilbene terpenes (15). The chemical composition of propolis is also highly variable and depends on the local flora at the site of collection. Propolis has been shown to possess antibacterial, antifungal, antiviral, anti-inflammatory, hepatoprotective, antioxidant, antitumor, and immunomodulatory effects (15).
Although the effects of NaOCl, EDTA, chlorhexidine, and MTAD on the bond strength of root canal dentin have been studied previously (3, 16, 17), the effects of propolis used as a final irrigant have yet to be studied. Currently, there has been no study in the literature that compares the effects of propolis with other endodontic irrigants. The purpose of this in vitro study was to evaluate the effect of propolis on bond strength to dentin when used as a final irrigant. The null hypothesis was that the effect of propolis used as final irrigant would have no difference from the other tested endodontic irrigants on bond strength to dentin.
Materials and Methods
The present study was approved by the Ondokuz Mayis University Ethics Committee (OMU.KAEK.2014/657). In this study, 126 intact human third molars were used within 3 months following extraction. The teeth were stored in a 0.2% thymol solution at 4°C before use. The teeth were sectioned approximately 3 mm below the occlusal level with a low speed diamond saw to expose their deep-coronal dentin surface. All specimens were mounted in auto-polymerizing polymethyl methacrylate resin using a Teflon mold, leaving the flat surface exposed. The sides with the flat bonding surfaces were polished with 320-grit abrasive papers and then randomly divided into 7 groups (n = 18) according to the final irrigant used:
Irrigated with 5.25% NaOCl for 10 min, flushed with 5.25% NaOCl for 5 min.
Irrigated with 5.25% NaOCl for 10 min, flushed with 17% EDTA (Henry Schein, Melville, NY, USA) for 5 min.
Irrigated with 5.25% NaOCl for 10 min, flushed with Tubulicid plus (Temrex, Freeport, NY, USA) for 40 s according to the manufacturer's instructions.
Irrigated with 5.25% NaOCl for 10 min, flushed with MTAD (Dentsply, Tulsa, OK, USA) for 5 min according to the manufacturer's instructions.
Irrigated with 5.25% NaOCl for 10 min, flushed with 20% propolis for 5 min.
Propolis was collected from Rize in Turkey and kept desiccated in the dark until processing. Twenty grams of crude propolis was dissolved in 80% ethanol for 5 days. This mixture was then incubated at 37°C for 10 days. The aqueous ethanol extract was filtered twice with Whatman (Whatman International, Maidstone, UK) no. 4 and 1 filter papers. The filtered solution was labeled ethanol extract of propolis.
Irrigated with 5.25% NaOCl for 10 min, flushed with 2% CHX (Drogsan, Ankara, Turkey) for 5 min.
Irrigated with distilled water for 10 min, flushed with distilled water for 5 min (control group).
After treatment, all prepared dentin surfaces were irrigated with distilled water for 2 min and gently dried using an air syringe. Then all prepared surfaces were treated with Clearfil SE Bond (Kuraray, Osaka, Japan) according to the manufacturer's recommendations. Prior to adhesive resin polymerization, a silicone tube with an internal diameter of 0.8 mm and a height of 2 mm was placed on the center of the bonded area, light cured, and filled with Filtek Z550 composite resin (3M ESPE, Minneapolis, MN, USA), and then polymerized (Fig. 1). Specimens were stored in distilled water at 37°C for 1 week before the microshear bond test was performed.
Experimental design (
Microshear bond strengths and failure analysis
The bond strength was tested with an LRX Universal Testing Machine (Ametec, Leicester, UK) at a crosshead speed of 1.0 mm/min. Maximum loads at bond failure were recorded in newtons (N) and bond strengths were then calculated in mega pascals (MPa) by dividing this value by the bonded area (mm2). The debonded area was examined with a stereo microscope (Nikon, SMZ 1500, Tokyo, Japan) at 40 x magnification for a failure mode analysis and classified as adhesive, cohesive, or mixed. In adhesive failure, 80% to 100% of the failure occurred between the resin and the dentin. In cohesive failure, 80% to 100% of the failure occurred in the underlying dentin or overlying composite. In mixed failure, 80% to 100% of the failure occurred in adhesive failure between the resin and the dentin, and in cohesive failure in the bonding agent and/or dentin.
Statistical Analysis
Data was analyzed using a 1-way ANOVA and Tukey HSD tests. Failure modes were analyzed for statistically significant differences by a nonparametric Pearson's chi-square test. All the computational work was performed using Minitab Statistical Software analysis program (Minitab, State College, PA, USA).
Results
Shapiro-Wilk's test showed a normal distribution of variables (P>.05), and Levene's test indicated homogeneity of variances. There were significant differences in the bond strengths of the tested groups (P = .0012).
Among all groups, the lowest bond strength values were found in group 1 (11.25 ± 1.97 MPa) and the highest values were found in group 4 (18.70 ± 1.82 MPa) and the difference was statistically significant (P = .0012). However, there was no statistically significant difference between group 4 (18.70 ± 1.82 MPa) group 5 (16.75 ± 3.70 MPa), group 2 (14.89 ± 1.91 MPa), group 6 (14.40 ± 2.30 MPa), and group 7 (15.88 ± 3.54 MPa) (P>0.05). Among the tested groups, group 1 (11.25 ± 1.97) which has the lowest bond strength values showed significant differences only from group 4 (18.70 ± 1.02) and group 5 (16.75 ± 3.70 MPa) (P=.0012). Also, there was a statistically significant difference between group 3 (12.80 ± 2.01 MPa) and group 4 (18.70 ± 1.82 MPa) (P = .0012).
The results of failure modes are summarized in Table I. Pearson's chi-square test showed that the failures were mostly adhesive. The groups with the highest bond strength values (the MTAD and propolis groups) exhibited more cohesive failures than the other groups (P<.05).
Percentage of failure modes of specimens (%)
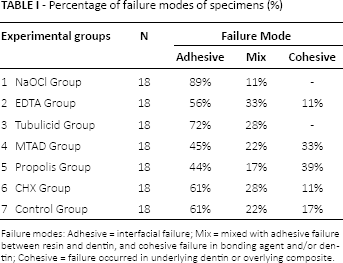
Failure modes: Adhesive = interfacial failure; Mix = mixed with adhesive failure between resin and dentin, and cohesive failure in bonding agent and/or dentin; Cohesive = failure occurred in underlying dentin or overlying composite.
Discussion
The null hypothesis of this study was partly rejected, because the propolis group 5 exhibited higher dentin bond strength than the NaOCl group 1.
There are several methods for assessing the bond strength of dental materials to tooth structures (18). Compared to conventional tensile and shear tests, microtensile and microshear tests allow standard tooth regions to be selected and thus preserve uniformity of the testing area. The simple test protocol of the microshear test allows for straightforward specimen preparation (19). Therefore the microshear bond strength test was preferred to allow for the testing of small areas in the present study.
There are some advantages of self-etch adhesive systems compared to conventional total-etch adhesive systems. For one, they have weak acids, resulting in less change in the dentinal wall structure than total-etch systems. In addition, the collapse of collagen fibrils is avoided and technique sensitivity is reduced (3). In the present study, Clearfil SE Bond (Kuraray, Osaka, Japan) was used because it is considered to be a benchmark for all other self-etch adhesives when it comes to dentin bond strength (20).
Various results have been reported regarding the effect of irrigation solutions on the bond strength of adhesive systems to dentin (3, 4, 6, 21). The majority of these results report decreased bond strength values after NaOCl treatment (4, 22). Similarly, in this study the lowest bond strength values were obtained in specimens irrigated only with NaOCl. It has been claimed that remnant superoxide radicals generated by NaOCl within the dentin surface serve to inhibit polymerization of resin monomers (23).
The results of the present study indicated that the highest bond strength values were detected in the MTAD group (18.70 ± 1.82 MPa). The presence of calcium ions (Ca2+) plays an important role in the bonding of dental adhesives to dentin (24). Sayın et al (25) found that MTAD extracted the least Ca2+ when it was used in root dentin. Machnick, et al (26) applied MTAD as an irrigant (20 min 1.3% NaOCl/5 min MTAD) during endodontic therapy and found that it may not need any additional dentin conditioning before the application of dental adhesives.
In endodontics, propolis has been investigated as an intracanal medicament in root canal treatment, a promoter of bone regeneration, an inducer of hard tissue bridge formation in pulp capping and pulpotomy, a cavity disinfectant before adhesive system, and a storage medium for avulsed teeth (9, 14, 27, 28). One of the chemical compounds in propolis is flavonoid. Flavonoids are responsible for antioxidant activity (24). Yonar et al (24) stated that the antioxidant activity of these components is principally based on their radical scavenging effect. In this study, propolis exhibited the second highest bond strength values among all irrigation solutions when applied as a final irrigant after NaOCl treatment. This can be attributed to the antioxidant capacity of flavonoids in propolis that eliminates the adverse effect of NaOCl inhibiting polymerization of resin monomers.
A study by Arslan et al (23) represents the only study found in literature regarding the efficiency of propolis on bond strength. The study compared the effects of different cavity disinfectants with a 30% propolis solution on the shear bond strength of a silorane-based composite resin. In contrast to our study, they did not find any impact of propolis on bond strength. This is probably due to differences between concentrations of propolis solutions and the percentage of ethanol extract, as the latter was remarkably higher in Arslan's study than in our study.
CHX is a synthetic cationic bis-guanide. It is the most frequently used disinfectant and shows effective antimicrobial activity (29). Numerous studies have investigated the effects of CHX application on tooth structure, but the results have been inconsistent. Therefore, more research on the interaction of CHX with adhesive systems is warranted (3, 20). Erdemir et al (21) found that CHX significantly increased bond strength to dentin, whereas Santos et al (3) reported that CHX irrigation had no effect on adhesion. Similar to Santos's study, in this study CHX did not compromise the bond strength of a self-etching adhesive system, possibly due to its non-oxidizing structure.
The specimens treated with Tubulicid Plus showed lower bond strength values than the other groups except for the NaOCl treated group. Similar to our findings, Fawzi et al (16) found that Tubulicid Plus might have left behind some residual chemical constituents (benzalkonium chloride) that hindered resin monomer penetration and permeation.
Higher bond strength normally produces more cohesive and fewer adhesive failures (11). Kahveci and Belli (30) found that higher bond strength values produce more cohesion and fewer adhesive failures. Similar to this finding, in this study the propolis group 5 and MTAD group 4, which had the highest bond strength values, exhibited more cohesive failures.
This study is limited by the fact that propolis from only 1 origin was used. Propolis from different origins might cause different effects due to different concentrations of the ingredients. Therefore, further studies should be performed to compare propolis from different origins on dentinal adhesion.
Conclusions
Despite the limitation of the present study, it was determined that the different irrigation solutions used as final irrigants affected bond strength. Propolis was shown to have a positive effect on bond strength to dentin. Although the present study was performed under in vitro conditions, its findings should prove beneficial to clinicians. Furthermore, long-term studies are needed to evaluate the effectiveness of propolis on bond strength.
Footnotes
Financial support: There is no financial support.
Conflict of interest: The authors deny any financial affiliations related to this study or its sponsors.
