Abstract
Objective:
Brushite cement (BrC) has been extensively utilized in hard tissue engineering implementations due to their osteoconductive and bioresorbable nature. The key limitations associated with the clinical application of brushite cement are complex handling procedures, short working time, and inadequate mechanical strength. This study investigates the development of MXene-incorporated brushite cement with improved properties for various clinical applications. The objective was to evaluate the influence of MXene concentration on the mechanical, physical and biological properties of BrC.
Method:
The incorporated MXene at 1% and 2% concentrations were evaluated for characterization by SEM, EDX, XRD, FTIR, mechanical strength by UTM, physical properties by degradation and setting time, and biologically by DDT, DCT, cytotoxicity and gene expression.
Results:
MXene inclusion increased the crystallinity of BrC, mechanical strength (SBS, CS and BFS), significantly (p < 0.05) particularly at 2 wt.%. The prolonged setting times, enhanced antibacterial property (ZOI 20.5 mm in DDT; 1.20 OD prior and 0.05 OD after the test in DCT), and acceptable cytocompatibility around 75% cell viability. Significant upregulation of RUNX2, ALP and OCN in the 2% group.
Conclusion:
Inclusion of MXene into brushite cement generated a composite biomaterial with improved mechanical strength, controlled degradation, strengthened antibacterial activity, while maintained biocompatibility. Exhibits potential for clinical applications requiring enhanced regenerative ability and antibacterial performance.
Keywords
Introduction
Bone damage created by traumatic injury, pathology, diseases (cancer and osteoporosis), or surgical removal needs bone substitution to restore the gap, stimulate bone repair and osseointegration.1,2 Autogenous graft has been recognized the benchmark of all grafting options available clinically because of low immunogenicity and potent osteoinductive, and osteogenic abilities.3,4 But, the use of autogenous graft has remained restricted because of insufficient reserves and donor area morbidity. 3 Allogenic graft has been a choice to autogenous grafts because of being accessible from various sources such as cadaveric and living donors, including the dimensions of the allograft bone moldable to fit bone gaps.2,5 These commercial bone grafts have remained imperfect regenerating option since being costly and poor osteoinductive capacity, however additionally poses a risk of cross infection and immune rejection. 6 Thus, finding a suitable biologically active material is important to regenerate bone lesions.
In the recent decades in biological engineering for instance polymeric materials, bioceramics, and alloys have become progressively acknowledged as an economical choice, biocompatible, and osteoinductive substitute to bone having prospects for resolving the shortcomings of autografts and allograft.7–9 Hence, synthetic bone substitutes, due to their limitless accessibility, reliability, and absence of repeated operations, have captivated further consideration throughout the previous eras.10,11
Among them, calcium phosphate cements (CPCs) are frequently employed as bone substitute materials, owing to their superior biocompatibility, osteoconductivity. These cements chemically resemble the mineral component of bone and demonstrate osteoconductive properties. 12 CPCs are categorized into basic CPCs (such as apatite) and acidic CPCs (including brushite and monetite). 13 Brushite cements are produced by an acid-base reaction 14 and are usually produced by a mixture of basic calcium phosphate (β-tricalcium phosphate; β-TCP; – Ca3(PO4)2) with an acidic calcium phosphate (monocalcium phosphate monohydrate; MCPM; Ca(H2PO4)2·H2O), incorporating water or an aqueous solution to produce a paste. This paste can subsequently be injected into bone defects, where it gradually hardens on-site through the entanglement of brushite crystals. 15 The favorable injectability helps to reduce the need for invasive surgical techniques. Acidic CPCs have garnered interest due to their faster resorption rates compared to their basic counterparts. While apatite cement maintains relative chemical stability under physiological conditions and may persist for years within the body,16,17 brushite and monetite can be resorbed in a matter of months.18,19 This rapid turnover potentially allows for quicker replacement by natural bone tissue, as evidenced in recent observations involving cranial applications. These quickly resorbing CPCs have been clinically promising for big skull defects, where inert implant materials frequently show failure because of lack in biological interaction. In skull repair applications it has been observed that BrC and monetite-based composites exhibits regulated turnover rate, permitting combined osteogenesis and regeneration with host skull tissue. 20
Nonetheless, traditional acidic CPCs face challenges such as complex handling procedures, short working times, and inadequate mechanical strength that hinder their broader clinical use. Previous research has sought to mitigate these issues by slowing down the setting reaction and enhancing mechanical properties through various means such as incorporating retardants like pyrophosphate and citrate, 21 modifying reactant ratios, or adjusting particle size distributions of the starting powders.22,23
To solve these complications, MXene were introduced and are characterized by unique 2D buildings, metal atomic layers, excellent conductivity, biodegradability and low toxicity. MXene are currently employed widely in a variety of applications particularly ceramic-matrix composites. 24 New forms have metal carbides, nitrides, and carbonitrides, and due to similarity in structure to graphene, the suffix “ene” is derived from “graphene,” while the letters “M” and “X” stand in for the transition metal atomic unit, carbon, or nitrogen, respectively. 25 Ti3C2 MXene, has an excellent promise for bone regeneration, strong surface functionalization, great hydrophilicity, and antibacterial characteristics in addition to Ca+2 binding sites. The super thin 2D configuration and large surface area of MXene Ti3C2Tx enabling a firm bonding at the interface and an efficient reinforcement. The presence of numerous surface hydroxyl and oxygen based molecular groups, creates a negative surface with large attraction for ionic interaction. This would result in potent activity at interface and biological interaction, creating an electrostatic interaction to Ca+2, thus backing increased bioactivity. 26
Brushite cement has low mechanical, physical and biological properties. Bone cement with different modifications have been used to improve its property of adhesion, regeneration and antimicrobial properties. In this regard MXene (bone regeneration, high mechanical strength, antibacterial properties), have been added into brushite bone cement. Moreover, MXene based brushite cement have never been used in dentistry for bone regeneration. The modified brushite cement ought to enhance bone regeneration, adhesion, and control post operative infection. We hypothesized that the incorporation of MXene into the brushite cement would modulate properties of BrC. The aim of this research study was to optimize the inclusion of MXene into brushite cement and to evaluate their effect on its mechanical, physical, and biological properties. Accordingly, this study aimed to include MXene into brushite cement at 1 wt.% and 2 wt.% concentrations. This aim was achieved by: Comparing mechanical, physical and biological properties of MXene based brushite cement with control. Comparing characterization of MXene based brushite cement with control. Utilizing SEM, EDX, XRD, FTIR, UTM, antibacterial tests, biocompatibility and gene expression analysis.27,28
Methodology
Cement preparation
Monocalcium phosphate monohydrate (MCPM) and β-tricalcium phosphate (β-TCP) were manually mixed for 10 s in proportions of 1.00 and 1.23 g respectively, prior to incorporating the liquid, continuing till achieving homogenous consistency to prepare brushite cement, resulting in a total mass of 2.23 g for the control formulation. To obtain 1 wt.% and 2 wt.% of MXene concentrations, 0.023 and 0.046 g of MXene were added and mixed with 1 mL of 800 mM solution of citric acid to form a paste. The resultant cements were allowed to set at RTP as dry samples and at 37°C as wet samples. 29 The β-TCP was used in a little surplus amount to provide enhanced mechanical strength. 23 The manual mixing was carried on a clean glass slab with a clean mixing spatula. MXene Ti3AlC2 (molecular weight 194.605 g/mol), sourced from MACKLIN Shanghai Biochemical Industry Limited, China, with lot number C12954660 was used. 30
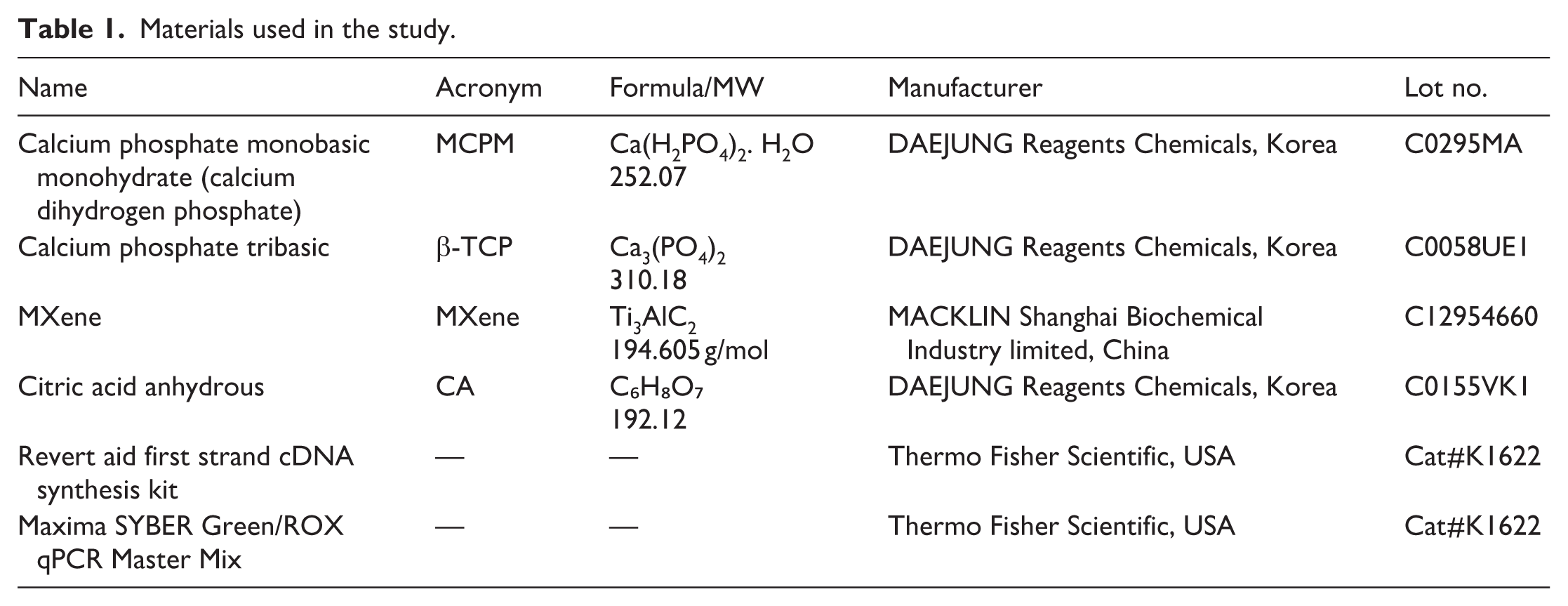
Materials
The list of resources used in study is provided in Table 1. All chemicals were used as received without any further purification.
Materials used in the study.
Characterization techniques
The SEM (JSM5910, JEOL, Japan) was utilized to study the surface morphology of prepared samples 24 h after mixing, under high vacuum conditions. The SEM utilized an energy beam of (30 KV) and resolving power maximum (2.3 nm). The magnification started from 200× to 30,000×.
Scanning Electron Microscopy (SEM) with Energy Dispersive X-ray (EDX) (JEOL JSM-IT100; 20 kV) analysis was performed to evaluate the elemental composition of dry samples 24 h after mixing. The cylindrical mold with dimensions of D = 10 mm and length = 3 mm was used as per the study by Cui et al. 31
X-ray Diffraction (XRD) (JDX-3532, JEOL, Japan) (20–40 kV) was used to study the crystalline phases of MXene cement and were analyzed by XRD origin software. The samples were ground and scanned for the phase compositions at a range of 5°–60° with a step size 0.01°/min (2θ).
Fourier Transform Infrared (FTIR) Spectroscopy (FTIR-990 LABOR China) was used to analyze functional groups which exist in the cement. Spectra were recorded in the 400–4000 cm−1 range for analysis.
A total of three samples per group were used for characterization (Table 2).
EDX comparison between control and experimental samples.

Mechanical properties
The mechanical properties of the prepared materials were evaluated using a Universal Testing Machine (UTM) (AG-IS Autograph Shemadzu Japan) equipped with a 5 kN load cell and crosshead speed of 1 mm/min until failure. The shear bond strength (SBS) testing of five prepared samples was performed in accordance with ISO 29022, using 20 orthodontically extracted human teeth. Each tooth was prepared and sectioned, retro-cavities were created in the roots using ultrasonic retro tips. The MXene modified brushite paste was injected into the cavities, and the samples were stored in saline at 37°C for 1 week. The SBS was performed, and a constant force to release the cement was recorded and was calculated according to the equation (1):

Where SBS = Force/surface area of the experimental sample. N was force in Newtons, p was constant (3.14), r was the radius of root canals (2 mm) and h was the thickness of radicular dentine sliced segment (3 mm) in mm.32,33
Compressive strength (CS) was measured as per ASTM F451, utilizing three cylindrical specimens (6 mm × 13 mm), 34 utilizing the formula (2):

While, P is applied force, and d = 6 mm.
The Biaxial flexural strength (BFS) of three specimens was calculated by utilizing a (UTM) with a piston-on-3-ball test. The discs were supported on three balls, and a central load was applied (ASTM Standard F394-78). 35 The load at fracture was employed to calculate the BFS based on Timoshenko’s equation (3). The strength was determined using the formula:

While, P is load (Newtons), a = 4 mm support radius, t = 2 mm average sample thickness, Ω Poisson’s ratio = 0.3. 29 Three cylindrical dry samples (13 mm diameter × 3 mm height) stored at room temperature (RTP) and wet samples stored for 1 day at 37°C.29,34
Physical properties
The physical properties of the three prepared material were tested using the degradation study. MXene samples were submerged in distilled water at 37°C for the duration of 2 days to mimic physiological conditions. Each sample was removed at predetermined intervals (1, 3, 6, 12, 24, and 48 h) from the distilled water, dried with a blotting paper, re-weighed. The distilled water was replenished at each time point to maintain consistent conditions throughout the 48-hour degradation period. A change in the mass was calculated as:

Where
For setting time evaluation was done following the cement preparation and mixing procedure described in section “Cement preparation.” The paste was being molded in a metal mold of 10 mm in diameter × 2 mm in height. 37 Cement was mixed and allowed to set at 37°C. 38 Setting time of MXene cement was measured using Gillmore needle technique. The initial and final set times were determined using 113 g load (2.12 mm tip diameter) and 453.5 g (1.06 mm tip diameter) according to ASTM C266-99. The time at which there was no indentation on the surface was considered the setting time. Three samples were tested per group and average vales noted.39,40 The results were reported as mean values.
Antibacterial properties
The antibacterial properties were tested for the three prepared material using both the disc diffusion test DDT and direct contact test (DCT). For the DDT, Staphylococcus aureus ATCC 6538 (106 CFU/mL) was inoculated onto nutrient agar plates. Cement discs were placed on the Agar surface; the plates were incubated at 37°C for 24 h. The zones of inhibition were measured. The antimicrobial effectiveness of MXene was compared against BrC (Control) and positive controls (antibiotics; Azithromycin/ciprofloxacin). 41 Additionally, the antimicrobial effect was also evaluated by direct contact test (DCT). Inhibition of Staphylococcus aureus growth was assessed in MXene samples after 1 h of exposure at 600 nm using spectrophotometry. 42
Biocompatibility
The cytotoxicity of MXene modified brushite cement was evaluated using the Alamar Blue assay in NH3T3 mouse fibroblast cell line. In a 24-well plate, the cells were seeded at a density of 2 × 104 cells per well and treated with different concentrations of MXene. After 24 h, cells were treated with Alamar blue reagent and fluorescence was measured to determine cell viability and cytotoxicity. All experiments were performed in triplicate to ensure reproducibility.43,44
RNA extraction, cDNA synthesis, and real-time quantitative reverse transcriptase polymerase chain reaction (qRT-PCR)
Mouse (MC3T3-E1 pre-osteoblasts; ATCC#CRL-2593 Manassas, VA, USA) were generated to extract RNA using triazole reagent and quantified using a Nanodrop spectrophotometer. RNA was reversed transcribed to cDNA with the revert aid cDNA synthesis kit. Gene expression was studied using syber green-based qPCR. The expression levels of selected osteogenesis-related genes (RUNX2, ALP, OCN) were determined, and the relative expression of target genes was computed by the ΔΔCT method. The concluding amount of 0.3 µM of the Primers (Primer 3 and blast) were utilized for the forward and reverse primers. Data were normalized to GAPDH expression and triplicate samples were used to conduct the experiment. 45
Statistics
The data were displayed as mean ± standard deviation. Results were analyzed utilizing one-way, two-way ANOVA and unpaired T test (parametric; two tailed variance), followed by Tukey’s test (multiple comparison). Statistical comparison of data at the same time point between two groups, the Mann–Whitney U-test (Multiple T tests and non-parametric tests) has been used. The normality of data distribution was evaluated using Shapiro–Wilk test, and the homogeneity of variance was confirmed using Levene’s test (GraphPad Prism 10.6.1). Differences between groups stood statistically significant at p < 0.05.
Results
Characterization
SEM images of MCPM, TCP, Brushite, MXene, 1% and 2% MXene with brushite cement have been revealed in Figure 1. Figure 1(a) of MCPM depicted particles of planer with irregular morphology with the range of 5 to 10 µm. Figure 1(b) of TCP shown spherical particles of agglomerate nature revealing particle size of around 40 nm. The brushite cement displays porous texture along with void spaces in Figure 1(c). The MXene powder nanoparticle display porous, irregular, and sharp-edged shape with particles range from 1 to 20 µm (Figure 1(d)). Whereas, incorporating 1% and 2% of MXene in brushite cement transforms its morphology to dense packed like structure as illustrated in Figure 1(e) and (f) with particle size range from 0.3 to 3 µm.

SEM images of: (a) MCPM ×1000, (b) TCP ×2500, (c) BrC ×1000, (d) MXene ×1000, (e) MXene 1% + BrC ×1000, (f) MXene 2% + BrC ×1000.
EDX analysis (Table 1) verified the elemental existence of O, P, Ca, corresponding to brushite cement (CaHPO4·2H2O). MXene 1% and 2 wt.% additionally revealed the presence of C, Cl, Na, Al, and Ti signifying effective incorporation. 26
XRD pattern (Figure 2(a)), regarding brushite displayed major diffraction peaks corresponding to brushite cement at 2θ ≈ 29.96°, and 30.79°, 46 verifying the creation of the DCPD structure. The major diffraction peaks of MXene at 2θ ≈ 9.44°, 38.66° and minor peak at 41.75°, corresponding to Ti3AlC2 MXene at 2θ ≈ 9.17°, 38.97°, and 41.96°.47,48 MXene incorporated brushite cement displayed similar main reflections, indicating that MXene inclusion did not change the basic phase composition of BrC. Although, an obvious amplification in peak intensity (44.37° and 77.98°), attenuated peak intensity (9.4°–33.28°), and minor peak shifts (leftward – 2θ ≈ 30.34°–31.72° and rightward – 27.86°) towards higher 2θ angles were noticed in MXene 1 and 2 wt.% concentrations. This implied increased crystallinity and enhanced lattice organized structure because of MXene addition.

XRD (a) and FTIR (b) comparison between BrC and experimental group.
FTIR comparative spectra (Figure 2(b)) additionally supported the production of DCPD, displayed characteristic absorption band P–O (phosphate group) and O–H (hydroxyl group), at 1000–1200 cm−1 confirms BrC. 47 Distinctive bond vibrations at 700 cm−1, and 1400 cm−1, related to Ti–O (titanium–oxygen) stretching and oxygen–hydrogen (O–H) bending respectively, confirms MXene. 49 Addition of 1 and 2 wt.% MXene revealed numerous peak shifts towards higher angles (leftward shift; at 563–1625 cm−1), (rightward shift; at 563–856 cm−1), (attenuated; at 657–3496 cm−1), (amplified; at 553–3496 cm−1), verifying chemical interaction and incorporation of MXene into brushite cement matrix.
Mechanical properties
Comparative SBS value in Figure 3(a) enhanced significantly with MXene inclusion. For 2 wt.% SBS enhanced maximums from 3.46 ± 0.08 MPa to 8.68 ± 2.00 MPa (p = 0.0184), and nonsignificant for 1 wt.% to 3.88 ± 0.70 MPa.

Shear bond strength (a), compressive strength (b), and biaxial flexural strength (c) between MXene and BrC-control (*p < 0.05) is significant.
Comparative CS values in Figure 3(b) increased notably. The control (BrC) exhibited 14.83 ± 0.92 MPa (dry) and 2.05 ± 0.26 MPa (wet), whereas 2 wt.% MXene enhanced strength maximum and non-significantly to 19.21 ± 3.6 MPa (dry) and 2.23 ± 0.35 MPa (wet). The 1 wt.% concentration displayed a nonsignificant increase to 16 ± 0.00 MPa (dry) and 2.25 ± 0.35 MPa (wet).
Comparative BFS values in Figure 3(c) enhanced significantly and maximum for 2 wt.% from 0.79 ± 0.30 MPa (dry) and 1.83 ± 0.16 MPa (wet) to 3.92 ± 1.13 MPa (dry) and 4.25 ± 1.05 MPa (wet) (p = 0.0070 dry, and p = 0.0428 wet). The 1 wt.% concentration displayed non-significant increase to 2.50 ± 0.00 MPa (dry) and 2.61 ± 0.27 MPa (wet).
Physical properties
The degradation activity in Figure 4 exhibited a concentration dependent phenomenon. BrC exhibited a steady mass loss over time with minimal mass gain (maximum degradation) – 0.630 g after 48 h. The 2 wt.% MXene concentration displayed significant (p = 0.0381) and maximum gain (least degradation) – 3.490 g, while 1 wt.% MXene revealed moderate gain (nonsignificant) – 1.664 g.
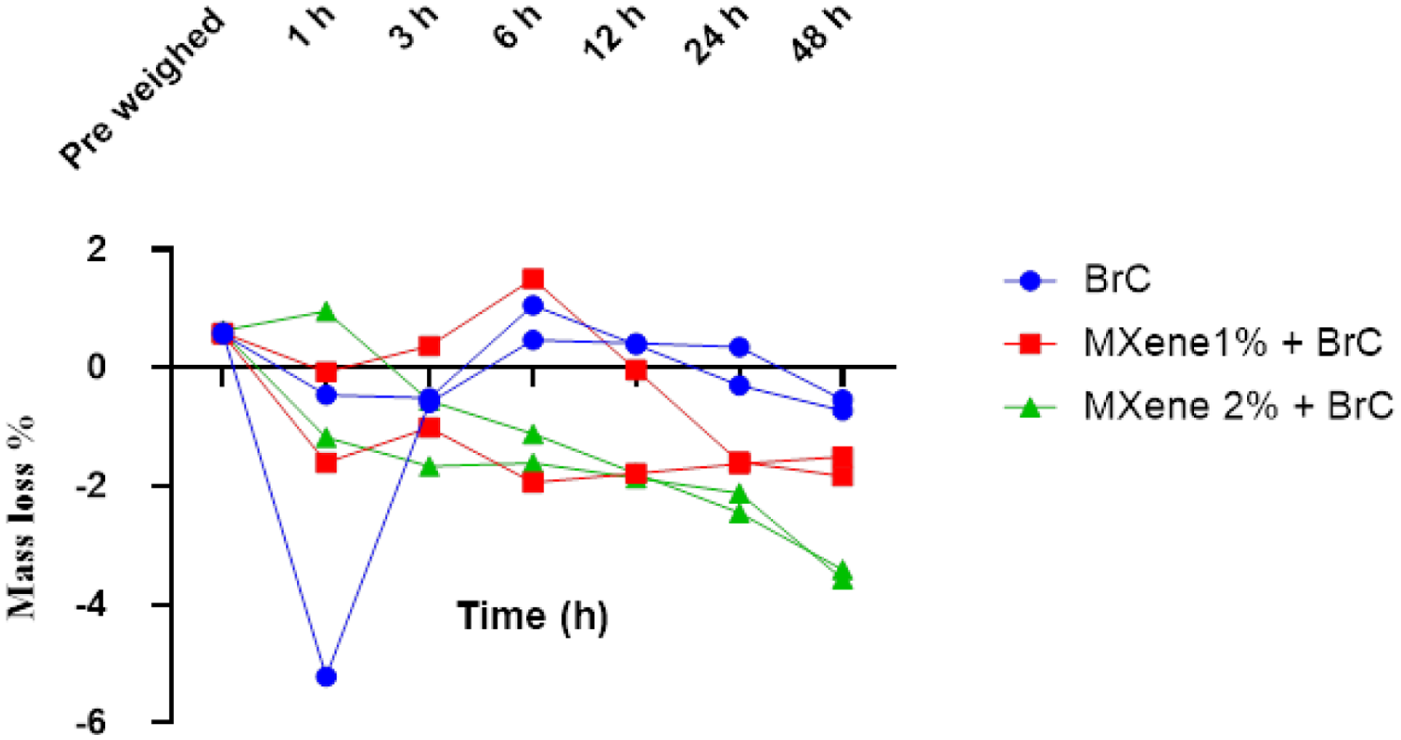
Degradation study comparison between BrC and MXene modified brushite cement.
Setting times (Initial I and final F) in Figure 5, were extended by MXene addition in comparison to BrC. For 1 wt.% MXene, setting times (Initial and final) significantly increased from 14.5 ± 0.7071 to 21.5 ± 0.7071 min (p = 0.0005) and from 24.5 ± 0.7071 to 35.5 ± 0.7071 min (p < 0.0001), respectively. For 2 wt.% MXene, additionally improved to 22.5 ± 0.7071 min (p = 0.0002) and 46.5 ± 0.7071 min (p < 0.0001).

(I) Initial and (F) Final setting time comparison between BrC and MXene modified brushite cement (*p < 0.05) is significant.
Anti-bacterial activity
Disc diffusion test (DDT) displayed in Figure 6, showed 2 wt.% MXene concentration with maximum and significant zone of inhibition (ZOI) (20.5 ± 0.70 mm; p < 0.0001), followed by 1 wt.% (16.5 ± 2.12 mm; p < 0.0001). MXene exhibited 17 ± 1.41 mm, while control revealed none zone of inhibition.

DDT pictures and graph comparison between Brushite, MXene modified brushite cement for Stap aureus. Measured Zone of inhibition (*p < 0.05) is significant.
Direct contact test (DCT) displayed in Figure 7 presents optical density (OD) value changes pre and post incubation. MXene 1 wt.% revealed a nonsignificant decrease in OD (0.13 to 0.04 = 0.09), and a nonsignificant decrease in OD for MXene 2 wt.% (1.20 to 0.05 = 1.14) (Collectively p = 0.33; and nd on Mann–Whitney U; negative values of Mean rank difference)), while BrC kept unchanged.
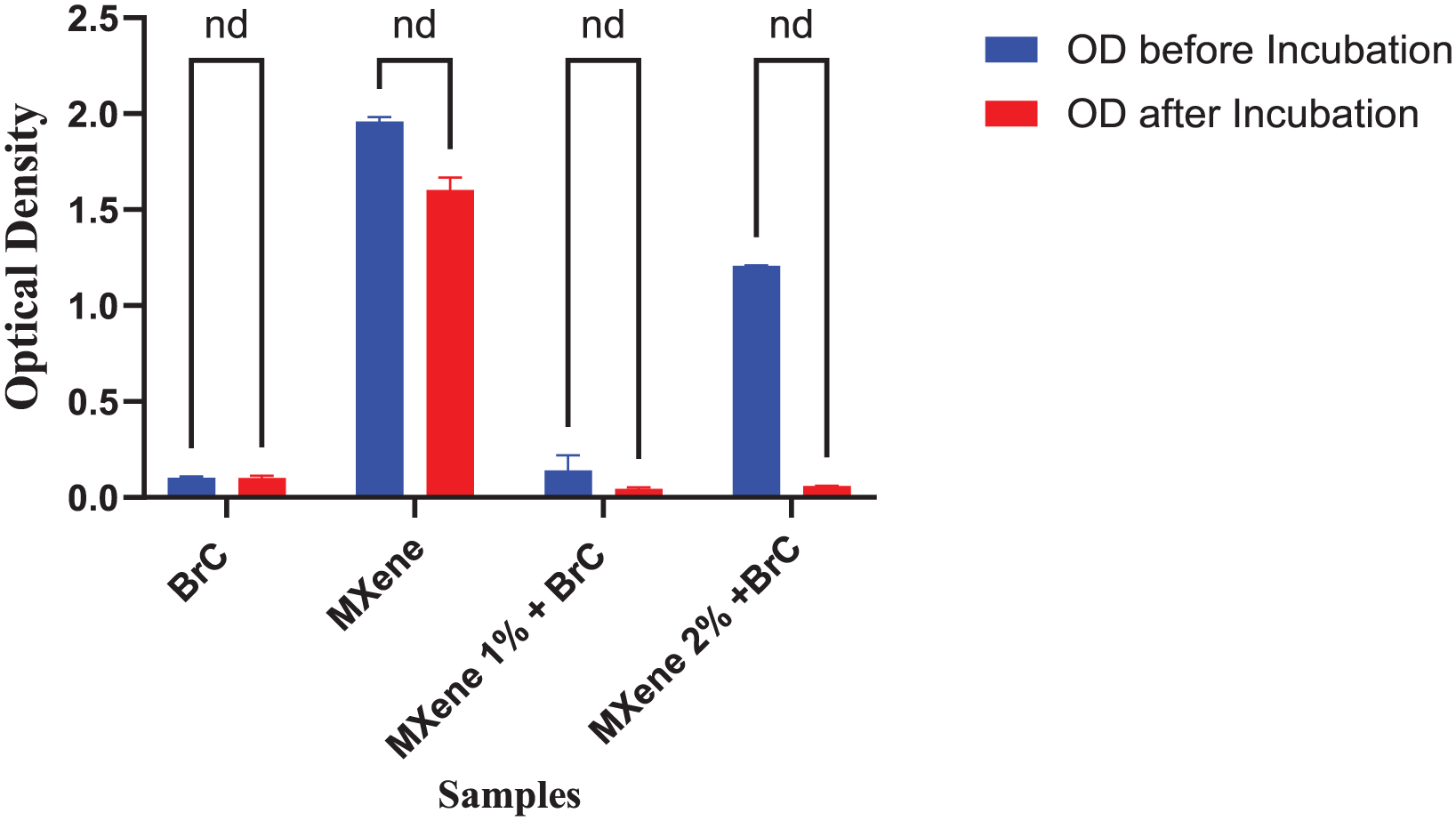
DCT comparison between Brushite, MXene modified brushite cement (p > 0.05) is nonsignificant (ns), and (nd) not a discovery on Mann–Whitney U.
Biocompatibility
Cytotoxic assessment in Figure 8, displayed lower cell viability after MXene incorporation in comparison to BrC. Percentage cell viability lowered for 1 wt.% (from 0.852 ± 0.008 to 0.705 ± 0.027; p = 0.0001), and for 2 wt.% (to 0.743 ± 0.029; p = 0.0009).
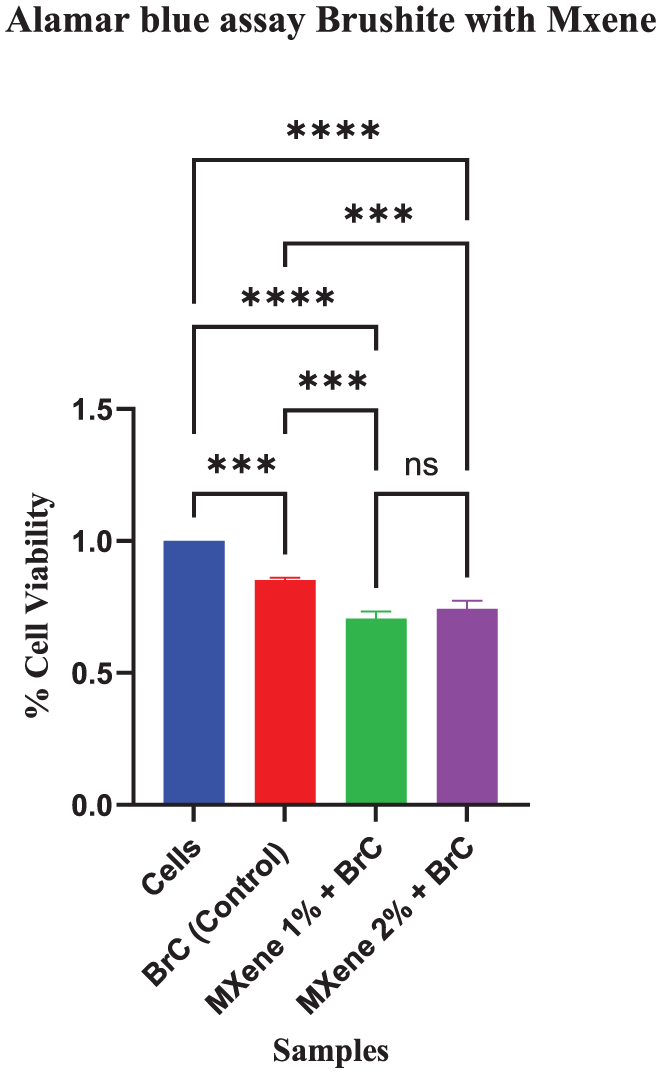
Cytotoxicity comparison between Brushite and MXene modified brushite cement (*p < 0.05) is significant.
Gene expression (RT-qPCR)
Gene expression assessment (Figure 9) revealed significant variation for bone gene markers. RUNX2, ALP, and OCN genes showed significant upregulation and greater relative expression, (1.69 ± 0.03/1.01 ± 0.24; p = 0.0092), (5.44 ± 0.42/1.17 ± 0.25; p = 0.0067), and (751.28 ± 111.9/8.94 ± 14.60; p = 0.0003) respectively, at MXene 2 wt.% in comparison to BrC.

Expression of specific genes comparison for RUNX2, ALP and OCN (*p < 0.05) is significant.
Discussion
This research paper explored the effects of MXene nanoparticle inclusion into brushite cement to reduce its basic drawbacks in mechanical behavior, stability, and antibacterial activity. The data revealed MXene inclusion significantly improved the physical, mechanical and biological properties of brushite cement, emphasizing its capability as a next generation bone cement for bone repair.
MXene inclusion refined the crystalline lattice of brushite matrix, encouraging dense and irregular crystalline morphology obvious from SEM, and in accordance with Zhang 50 that mentioned MXene topography as uneven, disorganized, and appropriate for osteogenic maturation. 51 Ti3AlC2 MXene exhibits sheet like topography helpful for electron conduction, 52 and a flake like topography which enhanced the surface area and contact leading to an improved ion exchange/passage for electron transportation along the planes for rapid electron conduction. 48
The corresponding peaks for BrC crystals are more diffuse and less intense than MXene, indicate brushite with lower crystallinity. The BrC pattern matched the basic peak intensities of modified cement, but showed differences indicating enhanced microcrystalline matrix with sharper peaks and higher intensity consistent with previous studies.53–55 Increased crystallinity explained previously is due to effective merging of intermediate layer and the enhancement of Ti3C2Tx that will lead to stability. Existence of strong MXene diffraction peaks and increased intensities signify the fine crystalline structure, and absence relate to impurity and pure phase. 48
The Ti-O bend vibration observed is in accordance with literature but with slight peak difference from 700 cm−1 to 638 cm−1.56,57 The distinctive O–H bending bond vibrations shows similarities with literature but with slight peak intensity difference from 1400 cm−1 to 1488 cm−1. 58 The sharper and well-defined MXene peaks indicate well-defined crystal structure, transferred into BrC. Peak positions and intensity changes indicate, MXene interacts with brushite to form more stable crystalline composite, and improve strength and life span. FTIR results are comparable to Ji et al., 59 and adding MXene nanomaterials stabilized mechanical degradation because of enhanced surface hydrophilicity.
Mechanical properties improved with MXene addition, and attributed to MXene nanoscale properties including high surface area and surface reactivity, that improved contact as higher adhesion and less loosening. Findings are in line with Rashid et al., 32 and noticed an increase bond strength with nanomaterials addition. Similarly, Bohner et al., 21 and Franco-Luján et al., 60 mentioned the hydration mechanism and alteration in microstructure with decrease porosity to enhance strength. Gbureck et al., 61 Hofmann et al., 62 and Yin et al., 63 indicated the primary stage hydration process that improved the strength.
Distinct degradation behavior for control is supported by Tamimi et al. 18 and showed quick resorption by ordinary dissolution process. MXene has slowed down the degradation rate in MXene modified material, and is in agreement with Anasori et al. 64 that mentioned an increased stability, and an inhibitory behavior by limiting hydration and early chemical reactions within the cement matrix.
Subsequent to the mixing of powder of MCPM/TCP and citric acid solution, the established cement initiates a dissolution-precipitation procedure that lasts till the BrC cement is set. Through the mixing procedure, MCPM and TCP powders initiate to dissolve, then leach out Ca+2, and PO4¯ ions. 65 A gel formation takes place with supersaturation at the interface of powder/liquid and the BrC cement (dicalcium phosphate dihydrate) initiates to make nucleation and growth till the cement is all set and made hard. Incorporation of MXene, probably inhibits the dissolution process of TCP and provided stability66,67 and reduces the BrC precipitation,67,68 by enhancing TCP/DCPD ratio. Factors like added materials and any liquid utilized could substantially influence the setting time of BrC. 67 No studies have been performed till date on the setting time of MXene, so we compared with Mg based articles.
MXene significantly amplifies the antimicrobial properties and it stems from a significant surface area and reactivity as they can engage and hinder cellular processes of bacterial cell walls. The findings are in line with Kumar Balu et al., 69 who reported their direct interacting capacity against the bacterial cell membranes.
The acceptable biocompatibility report is supported by Nandi et al., 70 that shows low toxicity due to widespread specific area, hydrophilicity, greater electrical conductivity and ease of surface functionalization.
RUNX2 (transcription factor) regarded as primary marker of osteoblastic differentiation, 71 and an osteogenic inducer with significantly greater expression at 2 wt.% MXene. Additionally, OCN a late osteogenic differentiation marker,72,73 strengthened the osteogenic capability of MXene.73,74 ALP results agrees with Hurle et al., 74 and enhanced ALP-stimulated osteogenic differentiation.74,75 MXene elevated gene expressions (RUNX2, ALP, OCN) managed to stimulate the Wnt/β-catenin signaling pathway through increasing the Wnt-Frizzled complex binding, hence stabilizing HIF-1α. 76 Only MXene 2% concentration was utilized in our study to compare with BrC due to its effective mechanical properties, antibacterial activity and gene expression reports.
Comparing the bone regeneration of MXene relative to established bone grafts for bone lesions is crucial to assess its performance. A particular material utilized is brushite cement an artificial biomaterial that influences osteogenic activity through the path of Ca+2 ions and BMP-2 expression. Resulting in the stimulation of RUNX2, initiating osteoblasts differentiation, 77 and elevated OCN expression. 78 Similarly, Hydroxyapatite biomaterial elevates the osteogeic performance by means of ERK and p38 signal pathways, 79 despite low presentations of RUNX2, ALP and OCN. 80 Although, β-tricalcium phosphate boosts the bone formation and presentations of RUNX2 and OCN with low RUNX2 expression. 81
A novel version of BrC is made by adding different concentrations of MXene, the quantity of MXene is the ultimate variable influencing the properties. The invented material formed is MXene based brushite cement, with significant differences in mechanical, antibacterial, and biological properties representing a durable candidate for bone repair.
Conclusion
MXene enhanced the microcrystalline structure, mechanical strength, physical, and biological properties of brushite cement with acceptable biocompatibility. Increased primary and late osteogenic markers of osteogenic differentiation and a potential for promoting bone regeneration.
MXene holds tremendous potential as a natural additive and increasing the degradation period to weeks/months to properly evaluate properties. The optimized wide range concentrations of brushite-MXene material would indicate excellent potential for prospective utilization in bioactive, strengthened/stabilized, and antibacterial bone regenerating cement. Needs long term degradation study, biocompatibility testing with osteoblasts, broader concentrations, and in vivo studies in future to lay a foundation for its subsequent clinical application.
Footnotes
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work supported by Department of Dental Materials, Institute of Basic Medical Sciences, Khyber Medical University, Peshawar, KPK, Pakistan.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
