Abstract
Background
The aim of this study was to evaluate the effect of increased exposure times on the amount of residual Bis-GMA, TEGDMA, HEMA and UDMA released from single-step self-etch adhesive systems.
Methods
Two adhesive systems were used. The adhesives were applied to bovine dentin surface according to the manufacturer's instructions and were polymerized using an LED curing unit for 10, 20 and 40 seconds (n = 5). After polymerization, the specimens were stored in 75% ethanol-water solution (6 mL). Residual monomers (Bis-GMA, TEGDMA, UDMA and HEMA) that were eluted from the adhesives (after 10 minutes, 1 hour, 1 day, 7 days and 30 days) were analyzed by high-performance liquid chromatography (HPLC). The data were analyzed using 1-way analysis of variance and Tukey HSD tests.
Results
Among the time periods, the highest amount of released residual monomers from adhesives was observed in the 10th minute. There were statistically significant differences regarding released Bis-GMA, UDMA, HEMA and TEGDMA between the adhesive systems (p<0.05). There were no significant differences among the 10, 20 and 40 second polymerization times according to their effect on residual monomer release from adhesives (p>0.05).
Conclusions
Increasing the polymerization time did not have an effect on residual monomer release from single-step self-etch adhesives.
Introduction
Currently, tooth-colored restorative materials are used for esthetic treatments more than ever before, and dental adhesives have become a complementary part of present day conservative dentistry (1). Dental adhesives are used extensively in dentistry in a variety of applications, as they exhibit perfect mechanical properties, rapid polymerization, esthetic quality, ease of handling and the ability to micromechanically bond to enamel and dentin surfaces (2). Dental adhesives are divided into the categories of etch and rinse and self-etching systems. Self-etch adhesives make it possible to use acidic monomers that can etch and prime at the same time, so that the separate etching and water rinsing stages can be eliminated. This system reduces technique sensitivity and increases the capability of bonding in clinical procedures by reducing chair time (3). All-in-one adhesives have further combined these bonding procedures into a single-step application. The increased concentration of acidic resin monomers in these systems enables them to etch the dentin and enamel simultaneously (4).
Dental adhesives include both hydrophilic and hydrophobic monomers that bond to the surface of vital dentin. The main monomer systems of dental adhesives are bisphenol A glycidyl methacrylate (Bis-GMA), urethane dimethacrylate (UDMA), triethylene glycol dimethacrylate (TEGDMA) and 2-hydroxyethyl methacrylate (HEMA) (5). The selection of the monomer strongly influences the reactivity, viscosity and polymerization shrinkage of the resin, as well as its mechanical properties, water uptake and swelling because of water in the resin (2, 6-7-8). Furthermore, the monomers can have cytotoxic, genotoxic, mutagenic or estrogenic effects and can cause pulpal and gingival/oral mucosa reactions (7, 9). The cytotoxic effects of monomers are the highest for Bis-GMA, followed by TEGDMA and UDMA, and finally HEMA. Bis-GMA has mutagenic effects on human genes (10). In a study conducted by Engelman et al (11), Bis-GMA at a concentration of >0.1 µM caused apoptosis in gingival fibroblasts. Stanislawski et al (12) showed the cytotoxic effects of TEGDMA on human fibroblasts. Bisphenol-A acts as an estrogen-like molecule and competitively binds to natural hormone receptors, which causes hormone-related toxic effects such as early puberty in females and feminization in males (13, 14). Due to its sensitizing property, HEMA causes rubefaction, pruritus, indurations, persistent paraesthesia, hypersensitivity, discoloration of the fingertips and allergic symptoms (15-16-17).
The degree of monomer-polymer conversion affects the properties of adhesive systems. When polymerization is incomplete, there are residual monomers present in the polymer chain, which can contribute to the degradation of the bonding agent. Chemical degradation is usually caused by oxidation and hydrolysis. When a solvent enters the polymer network, it causes an expansion of the structure and triggers the dissolution of the linear polymer chains (18). Oxygen prevents the polymerization of the monomer by forming an inhibition zone on the surface of the resin in contact with air (19, 20). However, if monomer conversion is not fully completed in the polymerization process, unreacted monomers are released into the oral environment (21, 22). There are several factors affecting the polymerization of the resin materials; including light intensity, exposure time, irradiant energy and curing units. Monomer elution from bonding systems is related to suboptimal polymerization (23-24-25). The null hypothesis was that increasing the polymerization time would affect the residual monomer release from dental adhesives. Numerous studies have been published about the degree of conversion of the monomers in adhesive systems (26-27-28-29); however, there have been few published studies about the effects of different polymerization times on monomer release (2). Therefore, the aim of this study was to examine the effects of increased exposure times on the amount of residual monomers released from single-step self-etch adhesive systems.
Materials and methods
This study was approved by the ethics committee of the Dental Faculty of Selcuk University. Two single-step self-etch adhesive systems were used in this study. The chemical compositions of these materials are listed in Table I.
Chemical composition of adhesive systems

Bis-GMA = bisphenol A glycidyl methacrylate; HEMA = 2-hydroxyethyl methacrylate; TEGDMA = triethylene glycol dimethacrylate.
Sample fabrication
Bovine teeth were freshly obtained from newly sacrificed cows in the slaughterhouse. The residual periodontal ligament or bone tissue, if present on teeth, was cleaned with a curette. The teeth were disinfected by storage in a 5% sodium hypochlorite solution for 24 hours and then stored in distilled water until use. For each tooth, the root system was removed from the cementoenamel junction. Longitudinal dentin sections were prepared from bovine incisors using a diamond saw (Buehler, Lake Bluff, IL, USA) under water irrigation. The dimensions of the dentin sections were an edge length of 7 mm and a thickness of 0.9 mm. The dentin slices were rinsed with physiological saline and sterilized by autoclaving (121°C for 25 minutes). Five dentin sections were used for each adhesive (n = 5). Then, equal amount of adhesives (2.5 mg) were applied to the dentin surface of the sections according to the manufacturer's instructions and polymerized using a light-emitting diode (LED; Elipar S10; 3M ESPE, St. Paul, MN, USA) for 10, 20 and 40 seconds. After polymerization, each specimen was immediately immersed in a light-proof glass bottle containing 75% ethanol and 25% deionized water (6 mL) at 37°C. During the period of immersion, the bottles were shaken for 5 seconds, and then the samples were taken from each bottle at 10 minutes, 1 hour, 1 day, 7 days and 30 days using disposable injectors. The residual monomers (Bis-GMA, TEGDMA, UDMA and HEMA) eluted from the adhesives were analyzed by high-performance liquid chromatography (HPLC; Agilent Technologies Inc., Santa Clara, CA, USA).
HPLC analysis
The extracts from the adhesives were analyzed by HPLC using an instrument equipped with a photodiode array detector (208 nm), an auto sampler, a pump and a C18 steel column 250 mm in length and 4.6 in diameter, with a particle size of 5 µm (SUPELCOSIL™; Supelco, Bellefonte, PA, USA). The mobile phase was a solution of 80% acetonitrile and 20% water that flowed at a rate of 1 ml/minute. The injection volume was 20 µL. Calibration curves were drawn for each monomer at different concentrations. In this way, standard chromatograms of Bis-GMA, UDMA, HEMA and TEGDMA (Sigma Aldrich, St. Louis, MO, USA) were obtained. The quantity of monomers leaching from the adhesives was obtained by a linear regression analysis of the results from the monomer standard series. A linear calibration equation for the monomers is shown in Table II.
Linear calibration equation for monomers
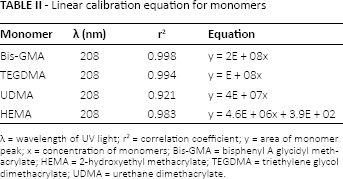
λ = wavelength of UV light; r2 = correlation coefficient; y = area of monomer peak; x = concentration of monomers; Bis-GMA = bisphenyl A glycidyl methacrylate; HEMA = 2-hydroxyethyl methacrylate; TEGDMA = triethylene glycol dimethacrylate; UDMA = urethane dimethacrylate.
One-way analysis of variance was used to analyze data on the quantity of monomers released by the adhesive materials after different polymerization times. Multiple comparisons were analyzed using the Tukey HSD test at a significance level of p<0.05.
Results
Bis-GMA, TEGDMA and HEMA were eluted from both adhesives, but UDMA was only eluted from Bond Force (Tab. III). Among the time periods, the highest amount of released residual monomer from adhesives was observed in the 10th minute, and the elution continued to decrease up to the 30th day (Figs. 1 and 2). The amounts of Bis-GMA, TEGDMA and HEMA eluted from Bond Force were significantly higher than those eluted from Adper Easy Bond (p<0.05). There were no significant differences among the 10-, 20- and 40-second polymerization times according to their effect on residual monomer release from adhesives (p>0.05).
Monomer concentrations eluted from the adhesive systems polymerized at 3 exposure times in 5 time periods
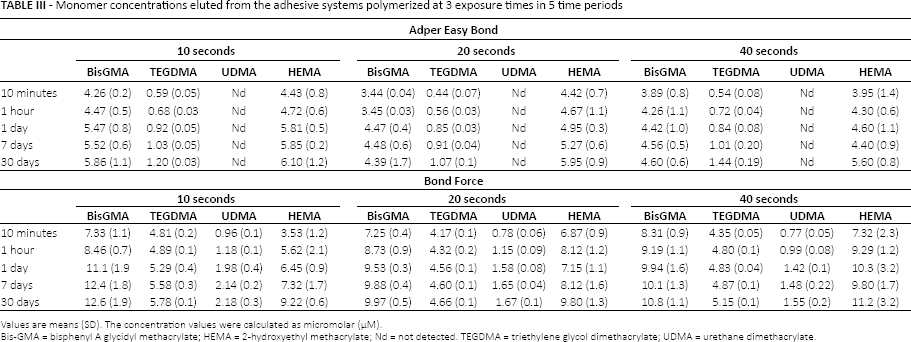
Values are means (SD). The concentration values were calculated as micromolar (μM).
Bis-GMA = bisphenyl A glycidyl methacrylate; HEMA = 2-hydroxyethyl methacrylate; Nd = not detected. TEGDMA = triethylene glycol dimethacrylate; UDMA = urethane dimethacrylate.
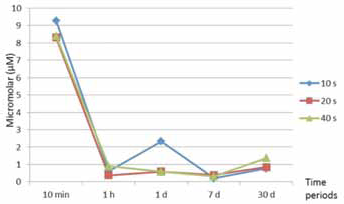
The mean amount of elution monomers from Adper Easy Bond, per storage time.
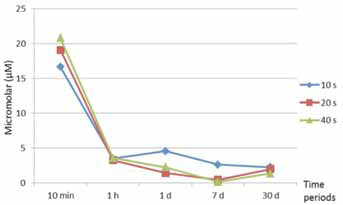
The mean amount of elution monomers from Bond Force, per storage time.
Discussion
Dental adhesives are extensively used in dentistry in a variety of applications. For this reason, it is important to analyze the chemistry and composition of these materials. Leached components from dental biomaterials, such as monomers or amines used for the initiation of polymerization, have been associated with a variety of cytotoxic reactions in tissues (7). Various solvents such as distilled water, ethanol and saliva have been used in several studies. Ferracane has suggested that intraoral fluids are probably some of the most aggressive organic solvents (18). The US Food and Drug Administration (US FDA) states that a 75% ethanol-water solution is clinically relevant as a food/oral simulation liquid. This solution has been used as an extraction solvent in several studies (2, 30-30-31-32). Cross-linked polymers are virtually insoluble but are capable of swelling in good solvents. The ethanol penetrates the matrix and expands the space between the polymer chains, allowing unreacted and leachable monomers to diffuse out (2). Thus, we used an ethanol-water solution as the extraction medium in this study.
Chromatography is the preferred method for identifying and quantifying residual monomers released from resin materials. HPLC analysis has been used in various studies to evaluate monomer release from resin materials (2, 30-31-32) because it is a very powerful separation method. HPLC is capable of detecting soluble and nonvolatile reactive compounds such as Bis-GMA, UDMA and TEGDMA (33). Therefore, HPLC analysis was used in the present study to evaluate monomer release from adhesive materials.
It is known that the degree of conversion of light-cured resin materials is 55%-80% and that this rate decreases to 35% in the oxygen-inhibited layer. Therefore this layer contains approximately 65% unreacted monomers (14, 33). This phenomenon is supported by our results; in the present study, the highest amount of monomer release in all adhesive systems was detected within a storage period of the 10th minute. This may be explained by the fact that the eluted monomers in the 10-minute period were based on oxygen-inhibited layers on the surface of polymerized adhesives and that the elution in the other storage periods may represent unreacted monomers that have been in the polymer chain.
An important factor affecting residual monomer release is the chemistry and size of the monomers in the resin materials. In the present study, the amounts of leached Bis-GMA and HEMA were found to be higher than the amount of leached UDMA and TEGDMA from adhesives. This might be because of differences in the chemical properties and reactive potentials of these adhesives (34). Sideridou and Achilias (2) reported that resins with lower degrees of conversion possibly contain higher amounts of residual monomers and also a larger amount of leachable monomers. Bis-GMA due to its rigid aromatic nuclei and hydrogen bonding ability with high strength demonstrates the highest viscosity value and glass transition temperature, leading to the lowest degree of double bond conversion (7). HEMA is a water-soluble monomer with low molecular weight, added in adhesive formulations due to its good miscibility and wetting ability, in addition to its ability to solve hydrophobic methacrylates preventing phase separations between adhesive components. However, the mechanical properties of the adhesive system may be negatively influenced by the high affinity of HEMA to water because of its intrinsic flexibility and inability to self-crosslink (35-36-37).
In this study, the amount of residual monomer eluted from the Bond Force was higher than that eluted from the Adper Easy Bond. This may be because of the differences in the amount of monomers in the resin matrices of these adhesives. Bond Force is composed of higher amount of monomers than Adper Easy Bond. The other reason may be the chemical structures of the monomers used for the preparation of the resins (2). Bond Force and Adper Easy Bond contain mainly Bis-GMA and HEMA, with camphorquinone (CQ) as the initiator; the curing process might be affected by the simultaneous presence of hydrophilic and hydrophobic constituents within the bonding agent (38). Ye et al (39) reported that poly(HEMA)-rich domains depart from poly(Bis-GMA)-rich areas, leaving heterogeneous polymers that cannot polymerize properly with CQ. It has been proposed that the degree of conversion might be improved by using hydrophilic photoinitiators in addition to standard CQ activation. It was observed in a study that the use of ethyl 4-dimethacrylaminobenzoate and diphenyl (2, 4, 6-trimethylbenzoyl)-phosphine oxide (TPO) improved the degree of conversion of hydrophilic domains in dental adhesive blends (38). Adper Easy Bond contains TPO (40), and it was demonstrated that a smaller amount of monomer was released compared with that from Bond Force. This may be related to improved polymerization as a result of the inclusion of this hydrophilic initiator (TPO) in its formulation. Another compositional difference that might be responsible for the different polymerization efficacy can be associated with silica filler. Silica fillers that are added to improve the mechanical properties of resins are included in the formulation of Adper Easy Bond, but are not in Bond Force. The viscosity of the resin system and also the polymerization can be improved this way (41). Finally, a higher degree of conversion and reduced residual monomer release can occur.
The elution values reported in the present study for both adhesives are lower than those reported in previous studies (30, 31). This difference can be explained by the lower quantity of adhesives that were applied on the dentin surface and the stronger light source that was used for polymerization in the present study. This is because the degree of conversion of carbon-carbon double bonds also depends on the type, duration and intensity of the light source and on the properties of the resin system (42-43-44). The Elipar S10 LED curing unit had a relatively high mean power density (1,200 mW/cm2), which may have led to the high monomer conversion in the adhesive materials.
Previous studies demonstrated that longer exposure times slightly improved the degree of conversion of the monomers in adhesive systems (26-27-28-29). Moreira et al (44) reported that increasing polymerization time decreased the monomer release from the Adper single bond 2. In the present study, no difference was found for 10, 20 and 40 seconds of polymerization of adhesives regarding monomer release. According to the manufacturer's instructions, 10 seconds of polymerization time with an LED unit is enough to achieve satisfactory polymerization of the adhesives. Increasing the polymerization time did not statistically decrease the monomer release from adhesives, so the null hypothesis was rejected.
Previous studies have reported that TEGDMA's toxic effects on human monocytes starts at 124.5 μM (45), Bis-GMA's toxic effects on mouse embryonic cells starts at 0.1 μM (5), UDMA's toxic effects on mammalian fibroblasts starts at 17.4 μM (17), and HEMA's toxic effects on mammalian fibroblasts starts at 1,770 μM and its toxic effects on monocytes start at 2,500 μM (46, 47). In this study, the lowest and the highest amount of eluted TEGDMA, UDMA, HEMA and Bis-GMA were 0.44-5.78 μM, 0.77-2.18 μM, 3.53-11.2 μM and 3.44-12.6 μM, respectively. It is apparent that the quantity of TEGDMA, HEMA and UDMA released from the adhesives is not sufficient to be cytotoxic. However, this quantity of Bis-GMA can be cytotoxic to biological tissues. In the present study, adhesives were applied on the dentin surface without restoration in order to obtain the maximum amount of monomers that could be eluted from the adhesives. Cytotoxic responses seem to be well correlated with substances eluted from the resins. It should be kept in mind that cytotoxicity potential is influenced by residual monomers as well as other additives such as initiators and stabilizers (48). This in vitro approach is useful for the evaluation of monomer release that can not be measured in vivo. The experimental setup did not consider in vivo conditions, and the measured elution of residual monomers measured can not be directly equated with the elution of residual monomers in vivo. Therefore, in vivo studies are still needed to evaluate residual monomers and their effects.
Conclusions
The present study concludes that released residual monomers were observed at all time periods and that the highest amount of monomers released from both adhesives were observed in the 10th minute. Bond Force showed higher amounts of monomers than Adper Easy Bond. Increasing the exposure time did not affect the amount of TEGDMA, UDMA, HEMA and Bis-GMA released from the single-step self-etch adhesives.
Footnotes
Financial support: This study was supported by Selcuk University Research Projects (11202017).
Conflict of interest: All authors declare that they have no conflict of interest.
