Abstract
Materials like carbamide peroxide or hydrogen peroxide are commonly used for vital teeth bleaching. However, there have been concerns regarding their effect on composite-to-bleached enamel bonding strength. The study investigated the impact of organic and antioxidant agents on composite bond strength in bleached enamel with different dental adhesive solvents. Human third molar teeth were sectioned into buccal and lingual halves. The two main adhesive solvent groups evaluated were acetone and alcohol. Each main group was divided into six groups. The positive control group received no bleaching, the negative control group consisted of bleaching with no surface deoxidization; and other experimental categories involved post-bleach treatments with 95% ethanol, sodium ascorbate (10%), acetone solution, or sodium fluoride solution (1.1%). Following the surface treatment and enamel bonding procedure, nano-hybrid composite cylinders measuring 3 × 2 mm were directly cured over the bleached enamel substrate. The shear bond test was performed after 24-h storage and 12,000 thermocycles on a universal testing machine. In this study, one-way ANOVA was used along with Tukey’s HSD tests at a significance level of 0.05. The negative control groups showed significantly lower bond strength than the positive control group. Ethanol surface treatment had superior mean bonding strength in acetone and alcohol-based adhesive solvent groups. The utilization of sodium ascorbate for surface treatment resulted in a significant enhancement of adhesion between the composite material and bleached enamel surface. Sodium fluoride application showed no significant recovery in shear bond strength in both dental adhesive groups. It was concluded that hydrogen peroxide severely compromised the immediate bond strength of composite resin. Surface treatment of bleached enamel with ethanol, sodium ascorbate, and acetone solutions is an effective option for restoring bond strength.
Introduction
Contemporary society’s enhanced esthetic demands have resulted in the introduction of multiple esthetic dental interventions. The researchers report an association of white teeth with individual confidence and social recognition. 1 Tooth discoloration results from either intrinsic or extrinsic factors; depending on the accumulation of pigments within or outside the tooth structure. 2 The researchers attribute non-vital teeth discoloration to pulp injury, leaching of restorative materials, and incomplete pulp extirpation. 3 Teeth bleaching is the conservative method of treating discolored teeth compared to invasive treatments, like full veneer crowns or laminate veneers. 4
Carbamide peroxide or hydrogen peroxide is a regularly employed bleaching agent. 5 Reactive oxygen molecules, free radicals, and anions are released during the oxidation reaction that underlies the bleaching mechanism. They enter the enamel and dentin porosities and break down the double bonds in chromogens of organic molecules into carbon dioxide and water. 6 Researchers have considered it a safe and effective treatment for discolored teeth, 7 however, they reported various adverse effects including changes in enamel surface morphology, 8 composition, 9 surface micro-hardness, 10 and reduction in bond strength of adhesives. 11 Although the process of bleaching results in an instant and enduring alteration in the visual color and brightness of teeth, the hue tends to transform within 24–48 h. A decrease in the “b” value is noticed once the activity of free radicals from the oxidizing bleaching agents ceases. The persistent oxygen-free radicals resulting from bleaching agents’ disintegration inhibit adhesive polymerization, resulting in a reduction in bond strength. Altered enamel structure and composition also contribute to compromised bond strength between enamel and tooth substrate. 12 Additionally, remaining oxygen within the dentinal tubules hinders resin infiltration. Hence, researchers recommend delaying the composite restorations by 2–4 weeks to facilitate residual oxygen getting eliminated from the dental substrate. 13
However, a recommended delay time is not always feasible in clinical practice because of pressing esthetic demands from the patients. Hence, researchers suggested a few methods for reversing the restoration bond strength. These methods include the application of organic solvents, antioxidant agents, enzymes, and antiseptic agents. Rose and Bode 14 and Kum et al. 15 stated that alcohol and acetone could eliminate residual oxygen from bleached enamel, and reestablish bond strength to a large extent. The advantage of utilizing these compounds includes easy clinical availability and no requirement for special storage facilities. The use of antioxidants like sodium bicarbonate, sodium ascorbate, and grape-seed extract has been recommended to improve bleached enamel bond strength. 16 Moradian et al. 17 reported positive results using flavonoids such as 10% quercetin. Numerous fruits and vegetables contain the antioxidant quercetin. α-Tocopherol is the most common form of vitamin E and its antioxidant properties have been reported to promote partial reversal of bond strength. 18 These compounds reestablish the changed redox potential of the substrate by facilitating the polymerization of free radical resins bereft of premature interruption. 19 Alternatively, Cheng et al. 20 0.5 mm surface removal immediately after bleaching.
Hydrophilic and hydrophobic monomers comprise dental adhesive systems. While the hydrophobic groups allow for interaction and co-polymerization with the restorative material, the hydrophilic groups increase the wettability of dental hard tissues. The chemical configuration of bonding agents also includes polymerization initiators, stabilizers, solvents, and inorganic fillers. Resin infiltrates micro porosities created by acid-etching the enamel, forming resin tags that resemble prisms, thus achieving micromechanical retention. Similarly, the resin can penetrate acid-etched dentin, creating a novel structure that comprises a resin matrix reinforced by collagen fibrils. Dental adhesive could contain up to 80% of solvents by weight. 21 The regularly used solvents within dental adhesives are alcohol, acetone, and water. 22 They are volatile organic solvents assigned to facilitate the transportation of co-monomers and initiators within a dental substrate. 23 The solvents reduce the viscosity of monomers, hence expediting the infiltration into the demineralized tooth structure. 24 These solvents significantly vary in their physical and chemical properties. Acetone-based solvents are less viscous, but sensitive to water and have low hydrogen bonding potential.25,26 Whereas ethanol-based solvents possess high hydrogen bonding capacity and a high vapor pressure compared to water. Hence, it is prudent to assess the effectiveness of different approaches for reversing the bond strength of bleached enamel for various dental adhesives containing different solvents. The purpose of this study was to assess the effectiveness of various organic and antioxidant agents in restoring the restoration bond strength to bleached enamel with different dental adhesive solvents. The hypothesis that was tested was that the bond strength of composite resin in conjunction with different adhesive solvents to bleached enamel substrate is not affected by different organic and antioxidant agents.
Methods
The study proposal received authorization from the IRB, College of Dentistry (IRB/KKUCOD/ETH/2021-22/018) in compliance with ethical standards. Sixty intact human third molar teeth were procured from maxillo-facial surgery clinics. The donors provided their written agreement to utilize their teeth for study objectives. Dental extractions were performed purely for the purpose of treatment. Extracted teeth were cleared of periodontal ligaments and calculus with an ultrasonic scaler. For 7 days, tooth samples were kept in a 10% formalin solution and later immersed in distilled water at ambient temperature prior to investigation. Caries, fractures, previous endodontic treatment, and dental anomalies were among the exclusion criteria for tooth specimens. The root and cusps were sectioned at the CEJ and central fossa floor. The teeth specimens were subsequently separated into buccal and lingual halves, using a low-speed diamond disk saw (Isomet, Buehler Ltd., Lake Bluff, IL, USA) under water cooling. The sectioned portion of the teeth was implanted within auto-polymerizing clear PMMA resin (Major Base 20; Major Prodotti Dentari S.p.A., Montecalieri, Italy). Sectioned teeth samples were fixed with the assistance of a vertical holding machine (Dentsply, NEYTECH, Charlotte, NC, USA) to ensure the buccal and lingual half surfaces were in line with the external surface of the acrylic resin. The enamel surface was leveled through successive abrasion using waterproof 600 and 800-grit SiC discs (3 M, St. Paul, MN, USA). Enamel surface preparation was done by a single operator for 1 min.
Teeth samples were randomly allocated to two main groups (n = 60) according to acetone and alcohol dental adhesive solvents. Each of the main groups was subdivided into six subgroups based on surface treatment protocols. Ten were chosen as the sample size for each group based on previous research.27,28 The G* Power software (version 3.1, University of Düsseldorf), with an effect size (d) of 1.4, of 0.05, and 1-β (power) of 0.85, was used to determine the sample size. 29 Using the bond strength values of bleached to unbleached enamel surfaces, the effect size was estimated.30,31
Teeth sample groups
The study employed a randomized allocation of teeth samples into one positive control group and one negative control group, along with four experimental groups. The positive control group (n = 10), in which enamel surfaces were restored with composite without following the bleaching protocol (Figure 1). One negative control group (n = 10) with only a bleaching protocol, no further surface deoxidization treatment, and four experimental groups. Post bleaching protocol, the first experimental group (Gr-I, n = 10) was treated with 95% ethanol (Maxill Inc., 80 Elm Street, St. Thomas, CA), while the second group (Gr-II, n = 10) was treated with acetone solution (Acetone ACS, Henry Schein Dental, Melville, NY, USA). The third (Gr-III) and experimental groups (Gr-IV) were treated with 10% sodium ascorbate (Now Foods, Bloomingdale, Illinois, USA) and 1.1% sodium fluoride solution (DentiCare Pro-Gel, Henry Schein Dental, Melville, NY, USA), respectively.
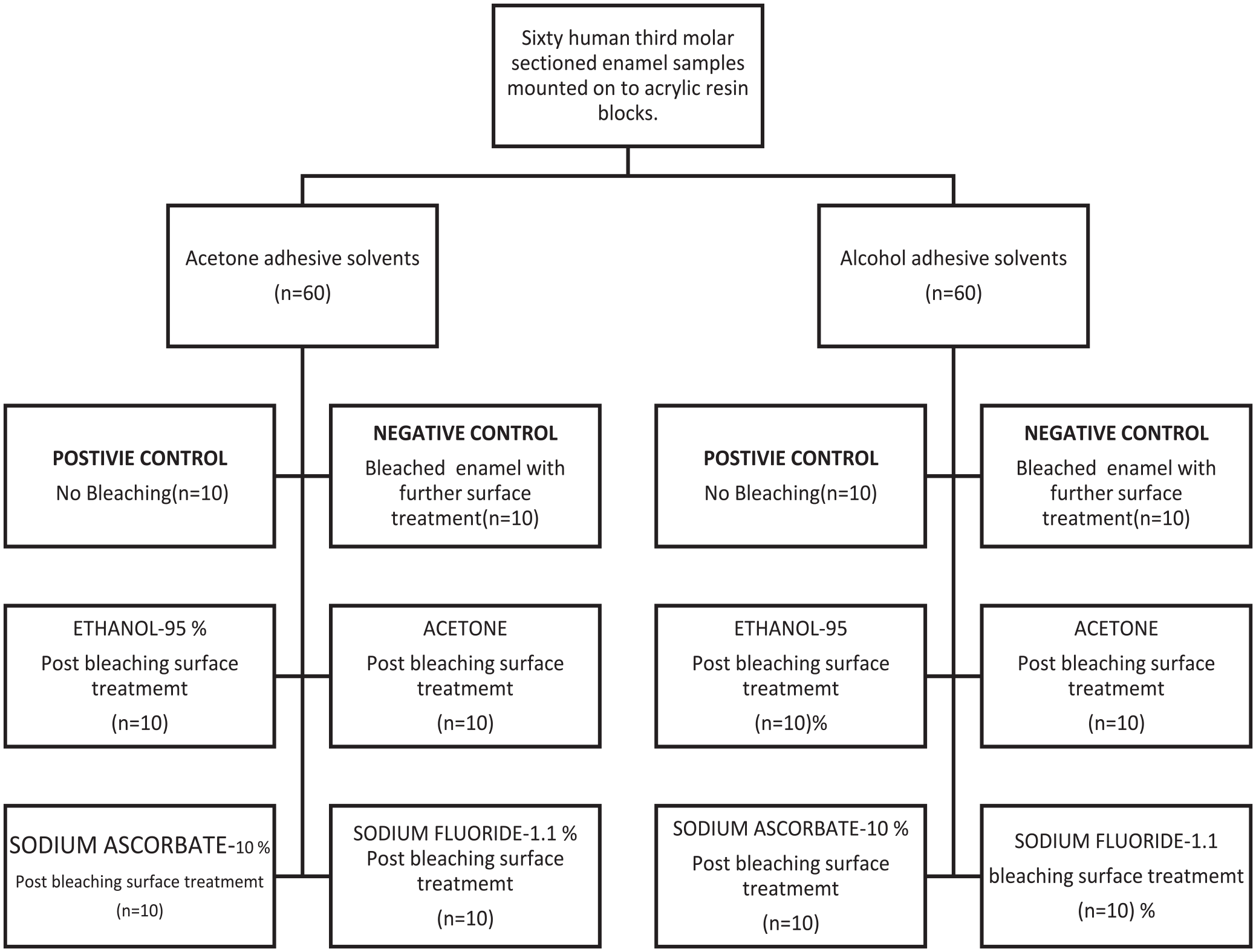
Sample distribution graphical scheme.
Bleaching process
A bleaching gel containing hydrogen peroxide at a concentration of 32%, with a thickness of 1 mm, (Flash, Whitesmile GmbH Birkenau, Germany) was administered onto the enamel surface and allowed to remain undisturbed for 15 min. The bleaching gel was stimulated using a flash chairside light (WHITEsmile GmbH, Weinheimer Straße 6, Birkenau, Germany) while concurrently agitated with a dental explorer to eliminate air pockets during the illumination process for 15 min. Using high-volume suction, the gel was removed after 15 min of bleaching. The bleaching procedure was repeated for three 15-min cycles, and at the end, the bleached surfaces were rinsed under running water. The teeth were subjected to two bleaching sessions, spaced 1 week apart. The samples were kept in distilled water at 37°C throughout the bleaching intervals.
De-oxidization and bonding protocol
Bleached enamel surfaces in the experimental Gr-I were treated by application of 95% ethanol for 3 min and later washed with running water. The Gr-II bleached enamel surfaces were conditioned with a 3-min acetone application and washed with water, and air-dried. To ensure surface moisture, a steady stream of solution was delivered using a syringe as the delivery method for 10 min at an infusion rate of 1 mL/min. In Group-III experimental groups, bleached enamel surfaces were de-oxidized by immersion in 10% sodium ascorbate solution for 10 min, subsequently rinsed with water, and dried with compressed air. Group IV samples were modified by the application of 1.1% sodium fluoride gel (1 mm thickness) for 15 min, washed with water, and dried on air.
Bonding protocol for acetone-based adhesive (i-BONDING LC, Medicinos Linija UAB, Siauliai LT, Lithuania): The enamel surfaces were treated with 0.2 mL of 37% phosphoric acid for 30 s (Total Etch, Ivoclar Inc., NY, USA), then washed with water and gently air dried. A micro brush was used to apply two coats of adhesive layers, followed by 10–20 s of air drying to remove all solvent from the surface and 20 s of 700 mW/cm2 LED light curing (Bluephase, Ivoclar Vivadent, Amherst, NY, USA). The alcohol-based (Tetric N-Bond Universal, Ivoclar Vivadent AG, Schaan, Lichtenstein) teeth samples were also etched with 0.2 mL 37% phosphoric acid (Total Etch, Ivoclar Vivadent Inc., Amherst, NY, USA) for 30 s. Afterward, the gel was washed off with water and air-dried. The etched surfaces were coated with two layers of adhesive and air-dried after 20 s. Lastly, the enamel surfaces were light-cured with 700 mW/cm2 LED light (Bluephase, Ivoclar Vivadent, Amherst, NY, USA) for 10 s.
Composite build-up and µSBS Test
Dentin adhesive-impregnated enamel surface was restored with a 3 mm × 2 cylinder-shaped (A3 shade) nano-hybrid composite (Tetric N-Ceram, Ivoclar Vivodent AG, Schaan, Liechtenstein). The composite resin cylinder size and location were standardized using a customized silicone putty index (Figure 2). The composite resin was placed in a silicone putty mold, a flat glass slab was placed over the upper portion, and a 700 mW/cm2 light-curing unit (Bluephase, Ivoclar Vivadent, Amherst, NY, USA) was used for 20 s to cure the resin. Micro-shear bond strength (µSBS) specimens were evaluated in compliance with the ISO/TS 11405:2015 standard. For 24 h, composite restored samples were kept in distilled water at 37°C. Subsequently, the specimens were exposed to 12,000 heat cycles (SD Mechatronik, Feldkirchen-Westerham, Germany) ranging from 5°C to 55°C while maintaining a 30 s immersion time.

Customized silicone mold for uniformity in the dimensions and placement of the composite cylinder.
Using a universal testing device (Figure 3), shear stress was applied to the junction between the bonded composite and the tooth until fracture. The 200-µM chisel-shaped head with a ramp speed of 1 mm/min was used for loading the shear stress, and peak load at bond failure was recorded in Newton (N). The fractured interfaces were analyzed by digital microscope (Hirox, Hackensack, NJ, USA) at a 25x magnification to classify the failure pattern as adhesive, cohesive, or mixed.

Shear bond strength testing in INSTRON machine.
Failures at the composite-dentin interface were classified as adhesive failures, while failures within composite resin were classified as cohesive failures. Failure in both the composite-dentin interface and the composite resin was recorded as a mixed failure.
Statistical analysis
For statistical analysis, IBM’s Statistical Package Software for Social Sciences 19 (Armonk, New York, USA) was used. The normality of the distribution of quantitative variables was calculated using the Kolmogorov-Smirnov tests. The data was examined through the application of one-way ANOVA and post hoc Tukey HSD tests. A p-value of 0.05 indicated a level of significance.
Results
The micro-shear bond strength of both acetone and alcohol solvent-based dental adhesives after various deoxidizing surface treatments is summarized in Figure 4. The results confirmed a significant reduction in bond strength post-dental bleaching. Among the acetone-based solvent group, the bond strength reduction was recorded from 82.73 (±8.82) N in the positive control group to 54.30 N in the negative control group. Analogous results were observed in the alcohol groups, with corresponding bond strength values of 96.46 (±7.52) N and 64.53 (±11.4) N for the positive and negative control groups. Ethanol surface treatment of bleached enamel displayed significant recovery of the SBS to 82.58 (±13.41) N in alcohol-based solvent and 80.56 (±12.88) N in acetone-based solvent groups. Sodium ascorbate surface treatment also displayed substantial retrieval of SBS in both alcohol and acetone solvent-containing dental adhesives, with 82.56 (±14.03) N and 79.54 (±11.24) N, respectively. Considerable recovery of SBS in bleached enamel with acetone surface treatment was also recorded with values at 78.95 (±10.70) N and 78.89 (±9.74) N for alcohol and acetone solvent groups, respectively. Only marginal retrieval of SBS was observed with sodium fluoride in both the alcohol (76.84 N) and acetone (63.97 N) groups. Inter-assay coefficient for P controls. Controls N, ethanol, acetone, S-ascorbate, and S-fluoride were 5.8, 15.2, 14.1, 11.4, 15.3, and 2.6, respectively. The corresponding values for the acetone solvent group were 8.6, 10.8, 12.2, 10.1, 11.2, and 8.6, respectively. The intra-assay coefficient for the alcohol group was 10.73, whereas it was 10.22 for the acetone solvent group. The Shapiro-Wilk test was conducted to ascertain the normality of data distribution (Value 0.5).

Micro-shear bond strength of acetone and alcohol based adhesive solvent groups.
A one-way ANOVA (Table 1) was performed to analyze the effect of different surface treatments of bleached enamel on the micro-shear bond strength. The micro-shear bond strength was influenced significantly by the different surface treatments in the acetone-based solvent group, F (5,54) = 13.828, p = 0.000. Alcohol-based solvent groups also showed a significant effect of surface treatment on micro-shear bond strength, F (5,54) = 9.245, p = 0.000.
One-way ANOVA evaluation of various surface treatments for micro-shear bond strength in acetone and alcohol based adhesive solvent groups.
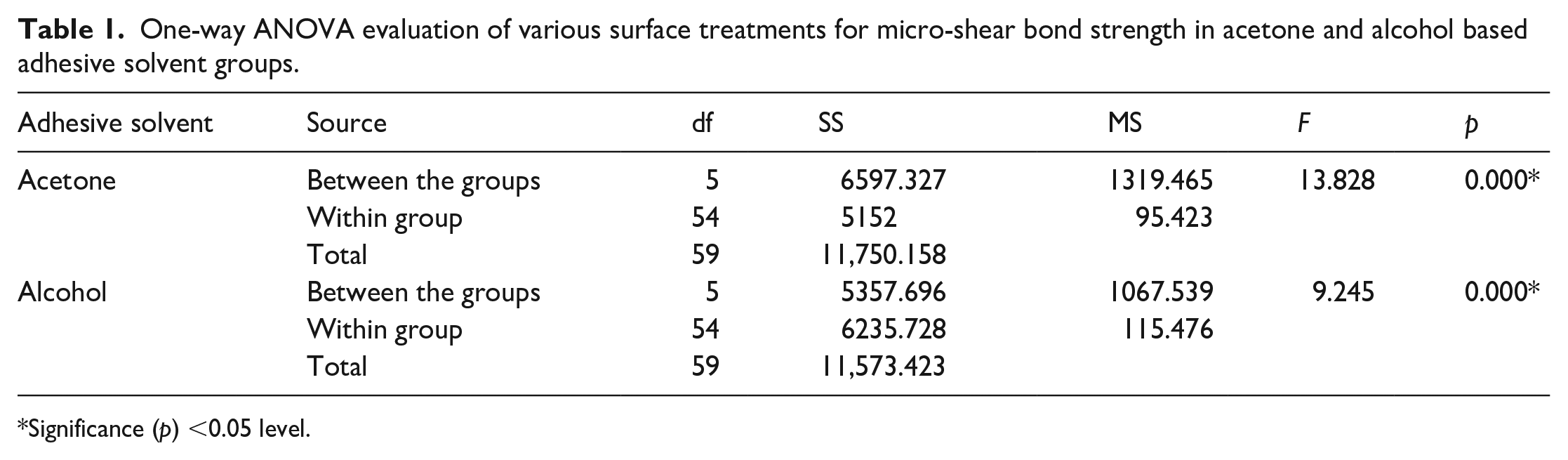
Significance (p) <0.05 level.
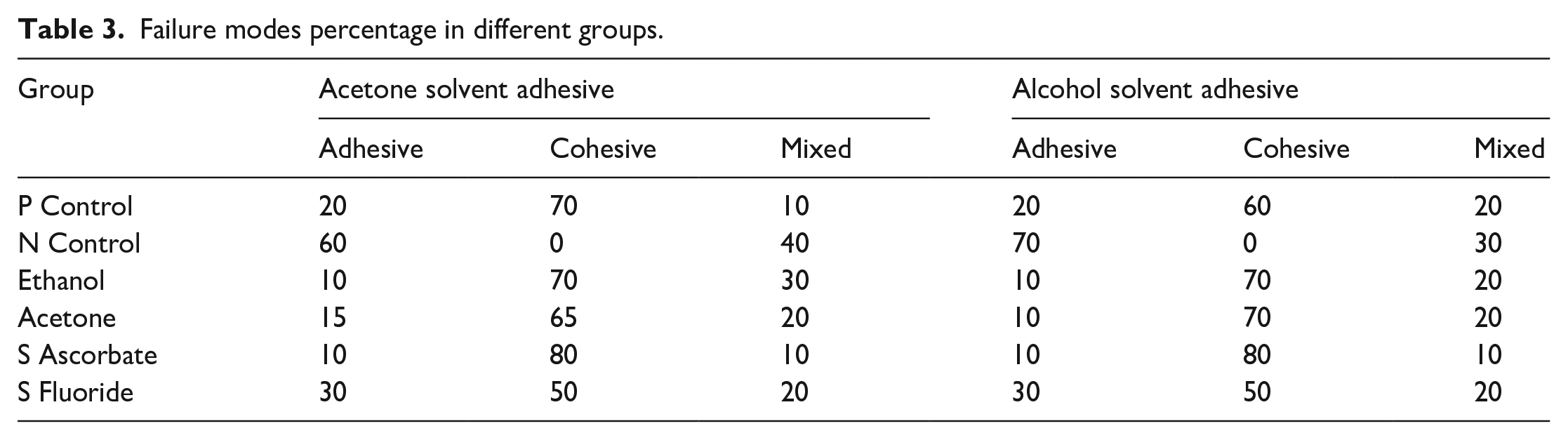
A Tukey HSD post hoc analysis (Table 2) of the Acetone group recorded significant differences between the majority of groups except between the positive control and the ethanol, acetone, and sodium ascorbate surface treatment groups. Among the alcohol solvent groups, a statistically insignificant difference was observed between the surface treatment groups of ethanol, acetone, and sodium ascorbate. The failure mode percentage (Table 3) was predominantly adhesive in the negative control group in both the alcohol solvent (70) and acetone solvent groups (60). The application of ethanol, acetone, and sodium ascorbate to the bleached enamel’s surface led to predominantly cohesive failures observed in both the alcohol and acetone solvent groups.
Tukey HSD post-hoc analysis of mean micro-shear bond strength amongst acetone and 2 alcohol solvent groups.
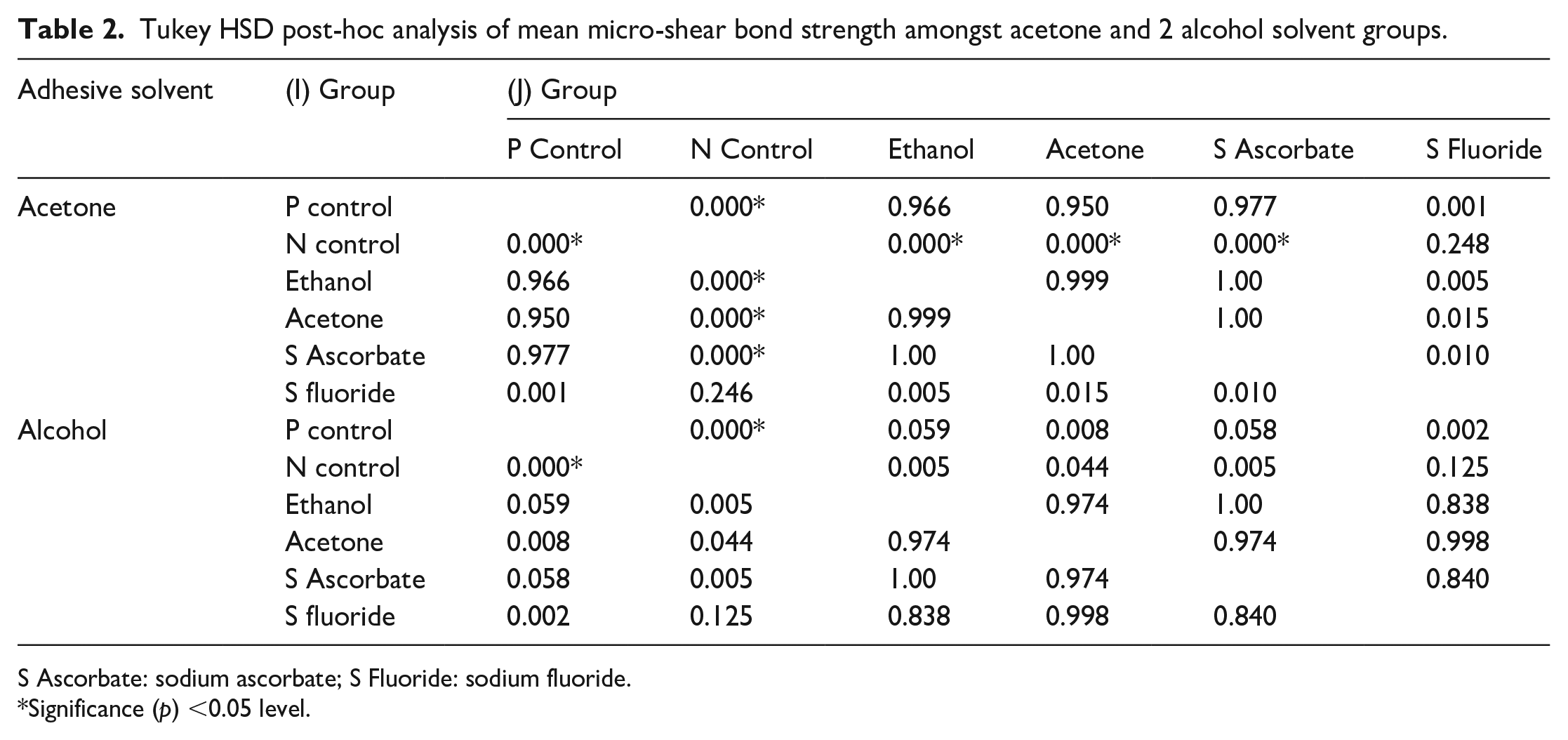
S Ascorbate: sodium ascorbate; S Fluoride: sodium fluoride.
Significance (p) <0.05 level.
Failure modes percentage in different groups.
Discussion
Bleaching is the most conservative treatment option to rehabilitate discolored teeth. Materials like hydrogen peroxide, carbamide peroxide, and sodium perborate are routinely utilized for bleaching. Post bleaching, the standard restorative protocol is to delay direct restorative procedures for 2–4 weeks. However, delayed restoration rehabilitation is impractical in an esthetic-conscious contemporary society. Hence, this in vitro study assessed the effect of different de-oxidizing agents on restoring the shear bond strength of composite restorations in combination with alcohol and acetone-based dental adhesives. According to the study results, the different anti-oxidizing agents had a significant impact on the composite SBS in relation to the bleached enamel surface. Hence, we rejected the null hypothesis that organic or antioxidant surface treatment would not affect the SBS of bleached enamel with composite resin.
The results of the study confirm the deleterious effect of 32% hydrogen peroxide bleaching on SBS between bleached enamel substrate and composite resin. Substantial reduction in SBS was recorded between positive groups and bleached surfaces. The SBS values for positive control and bleached surfaces in acetone solvent groups and ethanol groups were recorded at 82.73 (8.82) N, 54.30 (7.09) N, and 96.46 (7.52) N, 64.53 (11.4) N, respectively. The bleaching mechanism is predominantly achieved by the decomposition of hydrogen peroxide into water and nascent oxygen, hydroxyl, or peri-hydroxyl ions. Highly active free radical molecules react with pigments, splitting the link between an organic matrix of enamel/dentin and chromogenic molecules. The nascent oxygen further breaks down chromogenic molecules into smaller and less pigmented molecules. 32 Numerous factors contribute to the weakening of composite-resin bond strength on freshly bleached enamel. Researchers 33 proposed that residual peroxides and their products within the dental substructure inhibit dental adhesive polymerization. Dishman et al. 34 in their optical microscopic study observed that the scant, structurally incomplete, short adhesive rein tags within the bleached enamel surface. Titley et al. 35 believe that dentin and dentinal fluid absorb the free radical molecules and act as a reservoir of oxygen and peroxide. Pulpal microcirculation and surface diffusion deplete oxygen and peroxide levels at bonding interfaces. McGuckin et al. 36 and Lai et al. 19 found numerous trapped oxygen voids and bubbles within an adhesive layer during the polymerization reaction. García-Godoy et al. 37 and Muraguchi et al. 38 stated a substantial reduction in SBS values in bleached and unbleached enamel surfaces to composite resin. Meanwhile, few researchers 34 observed a substantial reduction in bond strength only with composite bonding immediately after the bleaching procedure. Lai et al. 39 reported a similar observation of a significant reduction in both acetone and ethanol-based adhesive bond strength to bleached enamel. Researchers recommended the application of adhesive systems containing organic solvents like alcohol 40 and acetone 41 to mitigate bleaching’s adverse effects on bond strength.
Both acetone and alcohol-based solvent groups displayed significant recovery in SBS strength with ethanol and acetone pre-treatment of the bleached enamel surface. The SBS values for acetone and alcohol-based solvent groups recorded 80.56 (12.88) N and 82.58 (13.41) N with ethanol pre-treatment, while corresponding SBS values for acetone pre-treatment were 78.89 (9.74) N and 78.95 (10.70) N. Kalili and Yoshida 42 reported that the surface treatment of bleached enamel with ethanol enhanced its bond strength to composite resin. They observed the improved SBS due to the reduction of residual water and oxygen with alcohol pre-treatment of bleached enamel. Analogous results of improved SBS with alcohol pre-treatment of bleached enamel surface are also reported by Torres et al. 43 and Whang and Shin. 44 Although the partial recovery in SBS was observed after ethanol and acetone pre-treatment, the SBS values did not recover equally to the un-bleached enamel surface. Perdigão et al. 45 attribute it to the more porous nature of bleached enamel, resulting in more water containing oxygen. Other researchers like Nascimento et al. 46 explained that the demineralization, denaturation of dental collagen, and corresponding reduction of surface energy resulting from bleaching also contribute to the irreversible compromise of bond strength to bleached enamel.
The potent antioxidant sodium ascorbate (10%) immersion for 10 min also resulted in significant improvement in the bond strength of composite resin to bleached enamel up to 79.54 (11.24) N and 82.56 (14.03) N in acetone and alcohol solvent adhesives. Sodium ascorbate is preferred to Ascorbic acid due to its neutral pH (7.4) and biocompatibility. Previous studies selected a 10% concentration and 10-min duration of sodium ascorbate due to its clinical applicability and reported effectiveness.11,47,48 The results agree with Ghaleb et al. 30 and Thapa et al. 49 studies. They demonstrated that the application of 10-min application of 10% sodium ascorbate significantly enhanced the bond strength between bleached enamel and composite resin. Improved bond strength with sodium ascorbate is ascribed to its potent antioxidant ability in quenching reactive free radicals. 50 It facilitates the polymerization of free radicals within adhesives devoid of premature disruption by reestablishing the modified redox ability of oxide adhesive substrates. 51
Sodium fluoride was selected in the study due to its routine application to dentin hypersensitivity as a result of tooth bleaching. According to the study, sodium fluoride was ineffective at reversing bleaching’s adverse effect on bond strength. The SBS values of the bleached enamel surface with sodium fluoride pre-treatment were recorded at 63.97 (7.54) N and 76.84 (3.75) N for acetone and alcohol solvent adhesives. The antioxidant capacity of sodium fluoride is attributed to its ability to inhibit the onset of dental caries and periodontal breakdown. Our study results agreed with Garcia et al. 52 ; they opined that sodium fluoride alone is not adequate to nullify the oxidative latent of 35% hydrogen peroxide.
The negative control groups with immediate composite resin bonding to bleached enamel recorded predominantly adhesive failures. This is probably due to the diminished polymerization of bonding agents in the presence of residual oxygen. Meanwhile, the ethanol, acetone, and sodium ascorbate pre-treatment groups recorded higher cohesive failures. The present study’s findings were consistent with Muraguchi et al. 38 and Kimyai et al. 53 results. The increased cohesive failures could be due to post-bleaching softening and increased tooth porosity. 54
The clinical applications of the research findings include the alcohol acetone sol-vent-based bonding agents’ bonding potential to bleached enamel could be enhanced with ethanol and acetone pre-treatment. Hence, immediate composite resin bonding devoid of significant loss of bond strength following bleaching is clinically feasible with the utilization of selective adhesive solvents and enamel surface pre-treatment. Limitations of the study were that it only examined the effectiveness of different pre-treatments on the enamel surface. While in clinical situations, restoration involves combined enamel and dentin surfaces. The limitation also includes the enamel substrate utilized in the research procured solely from the third molar dentition. There is no consensus among researchers regarding the duration of antioxidant application to bleached enamel for optimum results. Lai et al. 39 suggest a minimum of one-third of bleaching time, while Lima et al. 55 recommend 24 h of application time. Since these application times are clinically impractical, the present study followed 10 min of application time.
There will probably be an escalating demand for cosmetic teeth whitening procedures. Being mindful of patient expectations and considering the need for multidisciplinary treatment is crucial to achieving the desired cosmetic outcome. Some concerns related to teeth bleaching include the uncertain final shade and the possibility of color regression, as well as external cervical root resorption. 56 The use of hydrogen peroxide is subject to safety regulations in certain regions around the world. Therefore, alternative techniques such as micro-abrasions, 57 Nd: TAG irradiation, and cold atmospheric plasma 58 are being investigated. The focus is on avoiding thermo-catalytic methods to prevent harm to the dentin structure and external root resorption. Bleaching with low-concentration oxidizing agents 59 and other catalytic approaches 60 like lasers are also being explored.
Additional investigation is suggested to evaluate the consistency of the antioxidant’s efficacy, as well as the ideal concentration. This is to achieve a quicker and more effective neutralizing outcome. The impact of various neutralizing agents and bleaching techniques on microleakage during post-bleach restoration of enamel-dentin cavities in clinical settings requires further evaluation.
Conclusions
Within the limitations of this in-vitro study, we conclude that immediate composite resin bonding to bleached enamel resulted in a significant reduction of SBS in both alcohol and acetone solvent adhesive groups. The application of alcohol or acetone as a pre-treatment method to bleached enamel effectively reinstated the bond strength between the composite resin and bleached enamel. Application of 10% sodium ascorbate for 10 min resulted in enhanced bond strength between bleached enamel and composite resin. Sodium fluoride was not effective in counteracting bleaching’s negative influence on bond strength.
Footnotes
Author contributions
Conceptualization, data curation, writing – original review and editing S.B.H.; investigation, R.M.A., F.A.A., R.A.A.; S.C.; N.M.A.: Project administration, Resources. All authors have reviewed and approved the published version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors extend their appreciation to the Deanship of Scientific Research at King Khalid University for funding this work through General Research Project under grant number GRP/64/44.
Guarantor
SBH
