Abstract
Background:
Light-activated resin-based dental fissure sealants are the most widely accepted among clinicians. The objective of this study was to determine monomer release of 3 different light-curing fissure sealants in vitro: Control Seal (Voco, Germany), Fissurit FX (Voco, Germany) and R&D Series Fıssured Nova (Imicrly, Turkey).
Methods:
Each material was polymerized with a curing light: Valo Cordless (Ultradent) LED in standard mode for 20 seconds (n = 27) and 40 seconds (n = 27) and in Xtra power mode (plasma arc) for 3 seconds (n = 27). High-performance liquid chromatography (HPLC) was used to measure the amount of monomers released over 1, 3 and 7 days.
Results:
Plasma arc groups showed the highest release of monomers at 1, 3 and 7 days in the 3 fissure sealant groups (p<0.001). The greatest release of bisphenol A glycidyl methacrylate (bis-GMA) determined for R&D Series Fissured Nova both with LED 20 seconds and LED 40 seconds and for Control Seal was with the plasma arc. With time, release of the monomer was reduced for all polymerization systems.
Conclusions:
Efficiency of the polymerization system and applying the recommended curing time for light-activated resin-based dental materials are very important to protect the patient from potential hazards of residual monomers.
Introduction
Although continuous attempts have been made to find a remedy, dental caries remains the most common chronic disease of children, and persists as a public health problem (1). Sealing pits and fissures in newly erupted molars is accepted to be one of the most effective approaches to prevent caries development and progression (1-3).
Resin-based fillings and glass ionomer cements are the most commonly used materials to seal pits and fissures. However, due to poor retention properties, glass ionomer cements have recently been used as a delivery system for fluoride release rather than for sealing of fissures and pits (4). In contrast, cavity development and caries progression are prevented by resin-based fissure sealants owing to their high retention rate (4-6). Sealants have been used vigorously in regular dental care by pediatric dentistry, and greater numbers of children are receiving fissure sealants every day after oral health screenings performed in schools.
The resin chemistry of sealants mainly relies on polymerization reactions of bisphenol A glycidyl methacrylate (bis-GMA), which contains bisphenol A (BPA) as a precursor.
BPA provides the source of the mechanical strength and rigidity for resin-based sealants, and resistance against biodegradation (7); however, BPA is released as an impurity after the synthesis process of BPA blends (8, 9). Release of BPA may cause adverse effects after interaction with the oral cavity, where chemical degradation, softening and corrosion alter the mechanical properties of resin composites (10). BPA also acts as an endocrine disruptor that mimics estrogen and alters hormonal function, which can adversely affect neurodevelopment, reproductive development and metabolic processes (11, 12). Several studies have shown that dental sealants or composites containing bis-GMA can leach BPA into saliva (13, 14). Biomarker studies have shown an increase in urinary BPA concentration after patients received bis-GMA-based dental sealants or composites that did not contain BPA as an active ingredient (14, 15). Public concern about products that contain BPA has increased, leading to calls for BPA-free consumer products. In light of this growing concern, and given the experience of public concerns regarding mercury in amalgams, the dentistry industry needs to be proactive in evaluating the potential adverse effects of BPA and BPA-derived components in dental materials.
Previous studies showed a mean of 13 µg, and up to 30 mg, in vitro BPA leaching after 24 hours of a full crown placement on molar teeth. Based on this information, BPA release was calculated to be 250 times smaller, or 10 times greater than the tolerable daily intake (TDI) which is 0.2 mg/kg for a person weighing 60 kg (16). Therefore, depending on the size and number of the resin-based dental restorations, patients are at risk in terms of significant BPA release (9). A recent review that focused on psychosocial health in children who received composite restorations and were therefore exposed to BPA derivatives showed increased levels of anxiety, depression, social stress and interpersonal relationship hurdles (17).
Since there are rising general health–related concerns related to BPA release, the purpose of the present in vitro study was to evaluate time-related monomer release from the different brands of commercially available resin-based fissure sealants over different exposure times.
Materials and methods
Sample fabrication
Three light-curing fissure sealants (Control Seal, Voco, Germany; Fissurit FX, Voco, Germany; and R&D Series Fissured Nova, Imicryl, Turkey) were used. Table I shows the chemical compositions of the resin materials used.
Monomer composition of resin-based dental materials studied
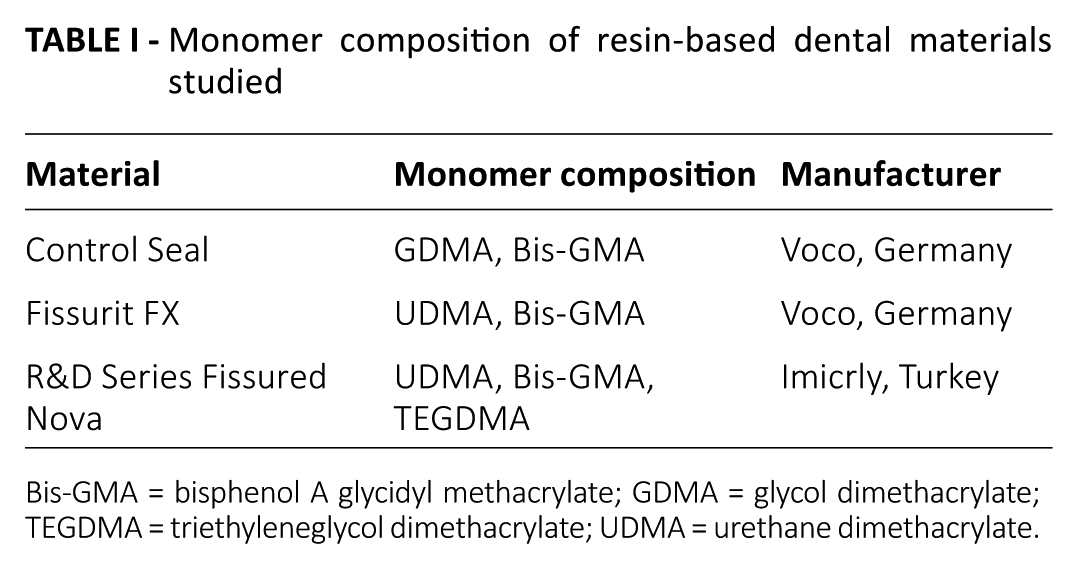
Bis-GMA = bisphenol A glycidyl methacrylate; GDMA = glycol dimethacrylate; TEGDMA = triethyleneglycol dimethacrylate; UDMA = urethane dimethacrylate.
Teflon discs of 10-mm diameter were prepared for each material, with a thickness of 1 mm (n = 81). To prepare flat disc surfaces, the surfaces of the glass slides were brought into contact with the filling materials from both sides of the moulds. Filling materials placed in Teflon discs were also covered with a strip band prior to the contact of the glass slides and LED exposure. Filling materials were polymerized with a Valo Cordless (Ultradent Products Inc, USA) in standard LED mode for 20 seconds (n = 27) or 40 seconds (n = 27) or in Xtra power mode (plasma arc) for 3 seconds (n = 27).
A constant distance between samples and the LED light was set using a glass slide with a 1-cm thickness. The tip of the light source was brought into close contact with the surface of the filling materials to maximize polymerization. Glass slides and strip bands were removed as soon as light curing was completed. The room temperature in the laboratory was kept constant at 23°C ± 1°C during all of the preparation steps of the resin discs.
Release studies and HPLC analysis
The cured discs were immediately placed in 5 mL of ethanol at 37°C for 7 days for analysis at 1, 3, and 7 days. Release of bis-GMA, urethane dimethacrylate (UDMA), glycol dimethacrylate (GDMA) and triethyleneglycol dimethacrylate (TEGDMA) from resin discs was analyzed using reverse-phase high-performance liquid chromatography (HPLC) (Agilent Technologies HPLC 1100 Series, USA). Detection was performed at 208 nm using a diode array detector. A C18 LiChrospher 100 analytical column (maintained at 25°C, 250 mm × 4.6 mm × 5 µm) with a particle size of 5 mm was used during the stationary phase of the analysis. When the mobile phase was switched, an isocratic solution of 75% acetonitrile at a flow rate of 1 mL/min was delivered to pass through the column, and triplicate measurements were performed using a 20-μL injection volume from the media of the samples after 1, 3 and 7 days. Identification of bis-GMA, UDMA, GDMA and TEGDMA was made based on the comparison of the column retention times of these monomers with the corresponding standards. A chromatogram of standard monomer samples is presented in Figure 1.
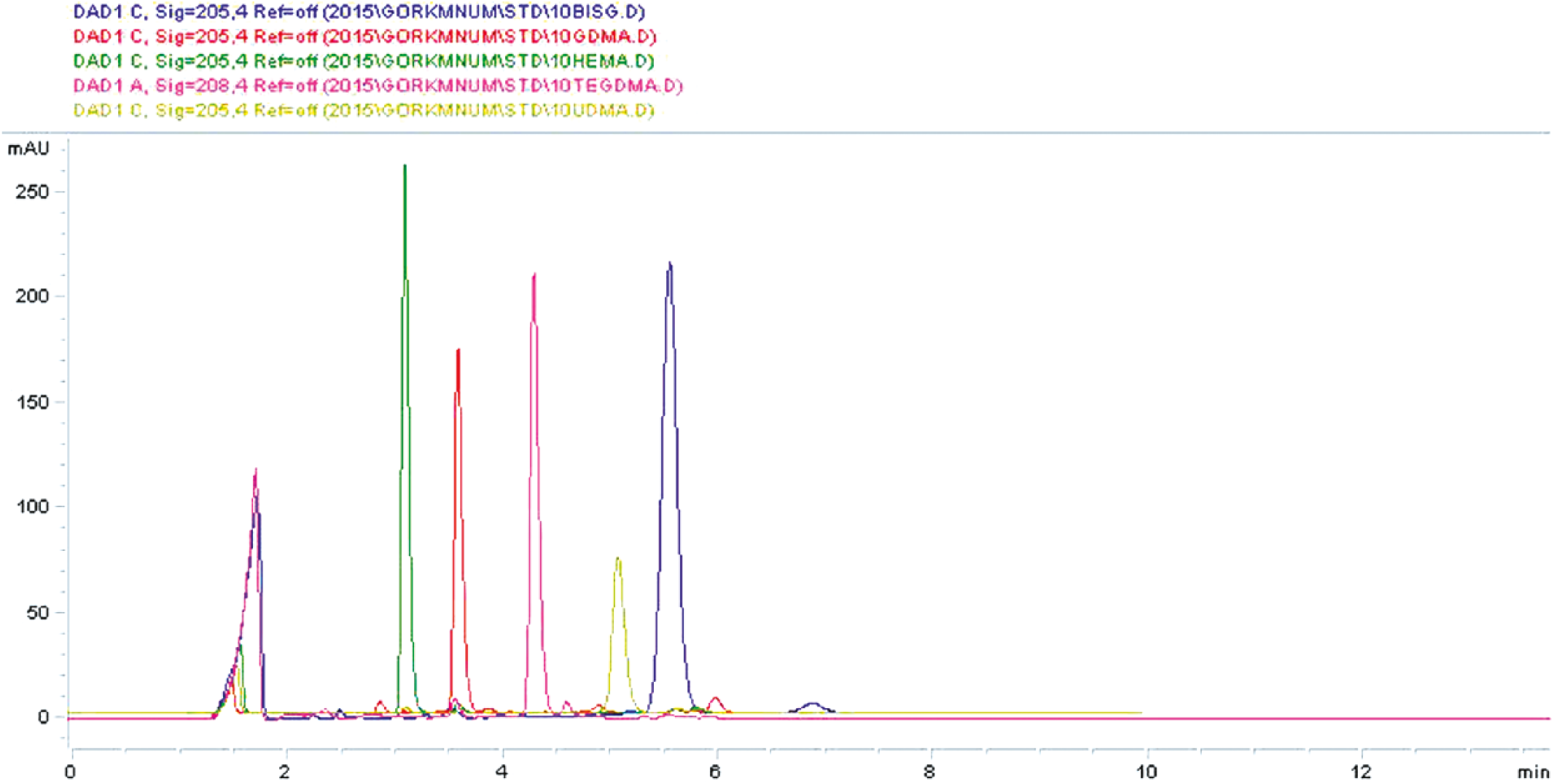
Chromatogram of standard monomer samples. BISG = bisphenol A glycidyl methacrylate; GDMA = glycol dimethacrylate; TEGDMA = triethyleneglycol dimethacrylate; UDMA = urethane dimethacrylate.
The SPSS software SPSS Inc., Chicago, IL, USA) was used in the statistical analysis of the data. Data were analyzed using 1-way ANOVA, Student’s t-test and the Duncan C-test. Data were considered statistically significant when p values were smaller than 0.05.
Results
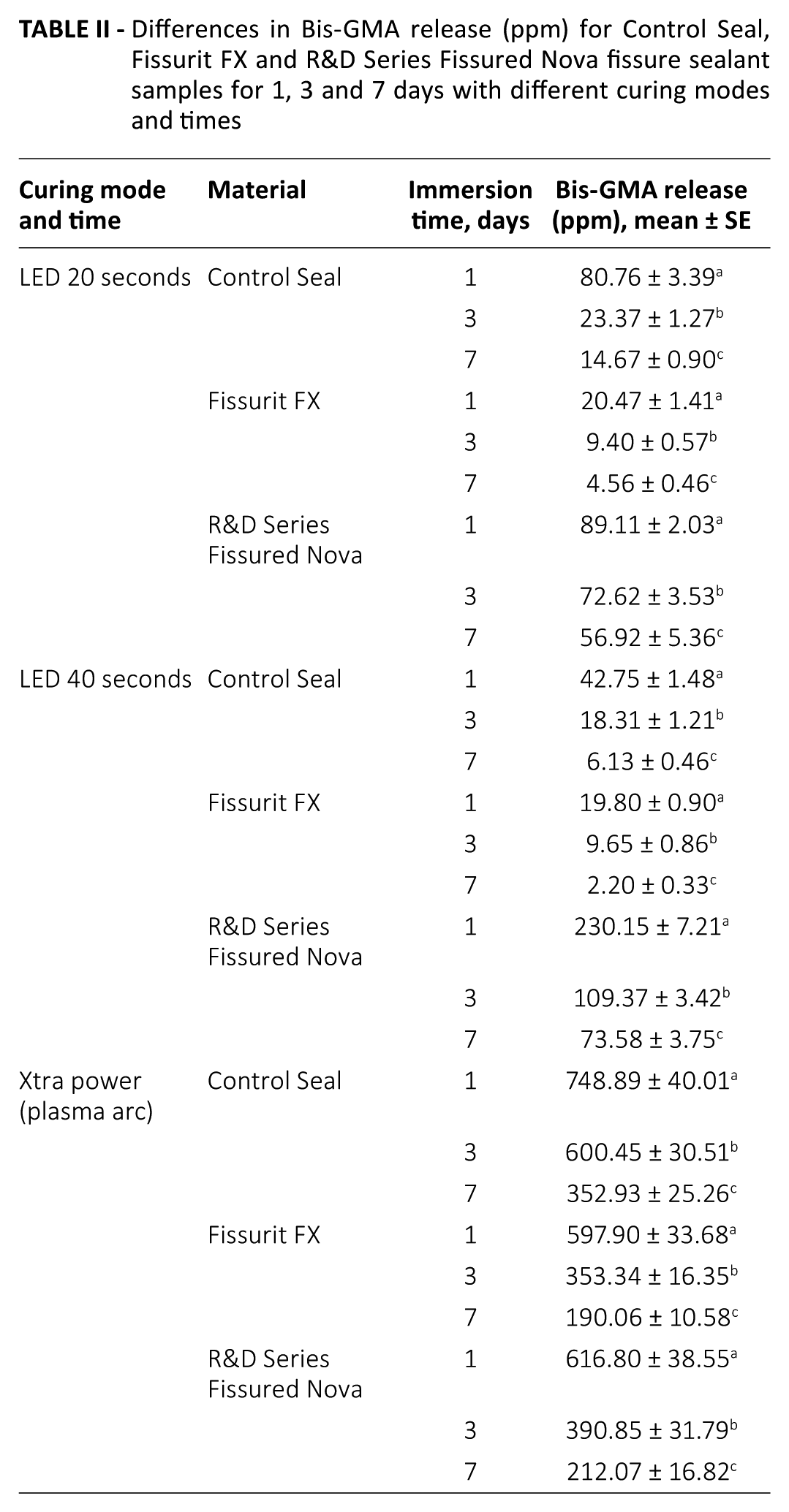
Table II shows the mean amounts of bis-GMA released from Control Seal, Fissurit FX and R&D Series Fissured Nova samples after 1, 3 and 7 days of immersion in ethanol. In the tables, a statistically significant differences is shown with the letter a, b, c in the same column. In the R&D Series Fissured Nova group, the release of bis-GMA was statistically significantly higher for LED 20 and 40 seconds, compared with Control Seal and Fissurit FX at all time points (p<0.001) (Tab. III). The Xtra power group showed the highest release of monomers at 1, 3 and 7 days in all 3 fissure sealant groups (p<0.001). The LED 40 second groups for Control Seal and Fissurit FX released the lowest amount of bis-GMA (p<0.001) (Tab. IV). With time, the release of monomers was reduced for all polymerization systems (p<0.001).
Differences in Bis-GMA release (ppm) for Control Seal, Fissurit FX and R&D Series Fissured Nova fissure sealant samples for 1, 3 and 7 days with different curing modes and times
Differences for 1, 3 and 7 days in Bis-GMA release (ppm) for Control Seal, Fissurit FX and R&D Series Fissured Nova fissure sealant samples with different curing modes and times
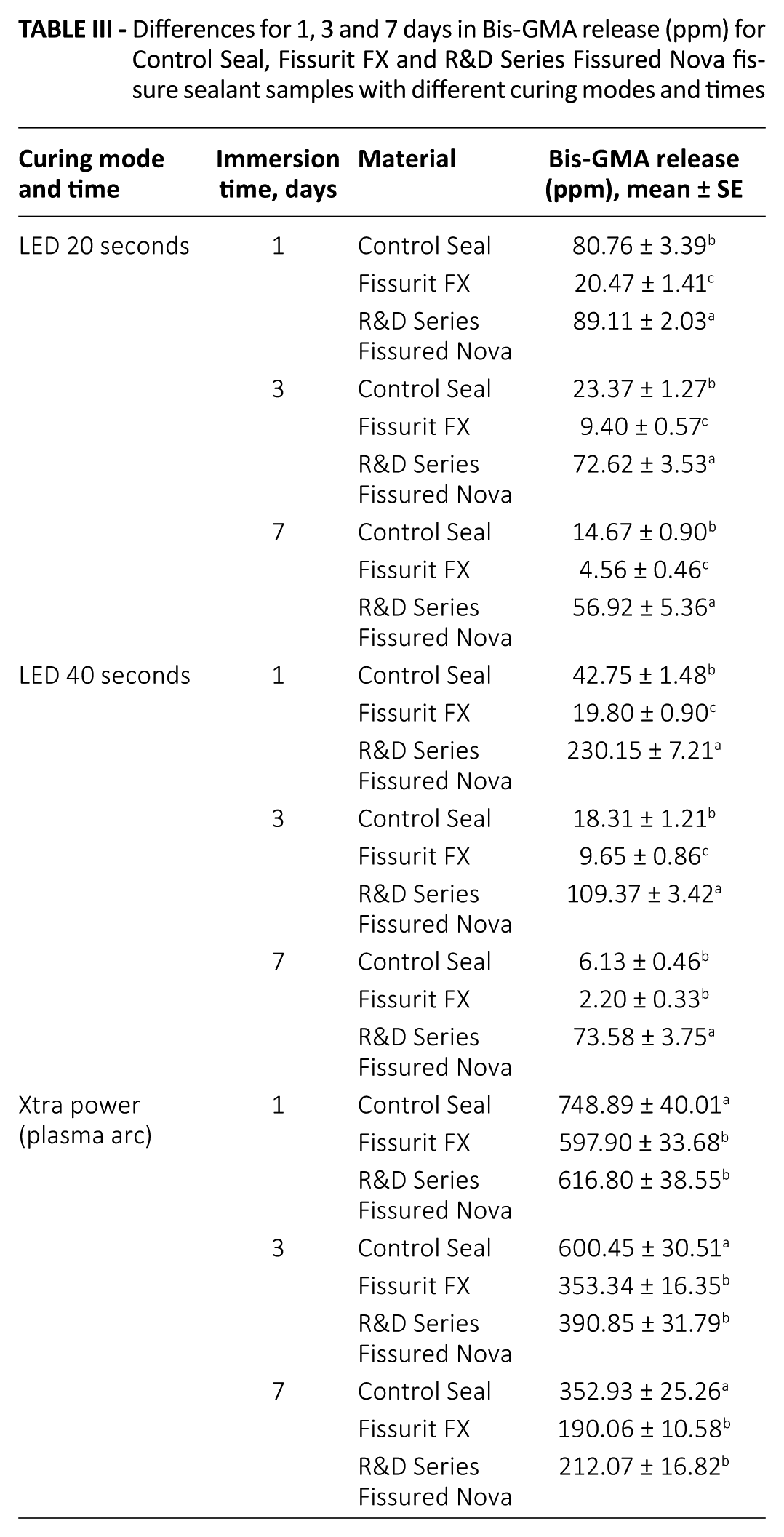
Differences in Bis-GMA release (ppm) for different curing modes and times for Control Seal, Fissurit FX and R&D Series Fissured Nova fissure sealant samples for 1, 3 and 7 days
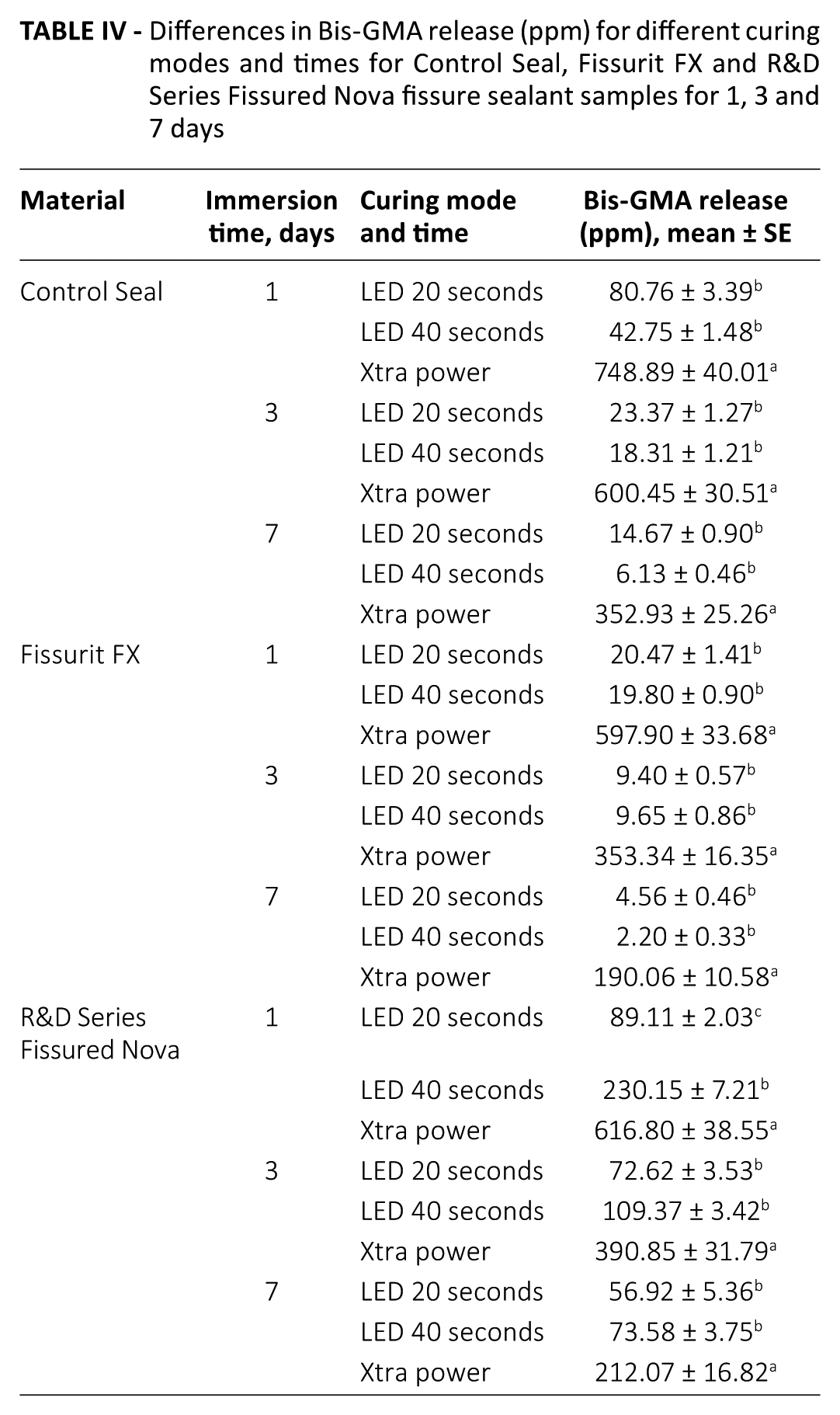
In the Fissurit FX and Fissured Nova groups, monomers of UDMA were released more in the Xtra power group than in the LED 20 and LED 40 second groups, and the release of monomers decreased over time (Tab. V). Regarding GDMA in the Control Seal and TEGDMA in the Fissured Nova groups, the Xtra power group released more monomers than the LED 20 and LED 40 second groups at all time points (p<0.05) (Tabs. VI and VII).
UDMA release (ppm) from Fissurit FX and R&D Series Fissured Nova fissure sealant samples in ethanol after 1, 3 and 7 days
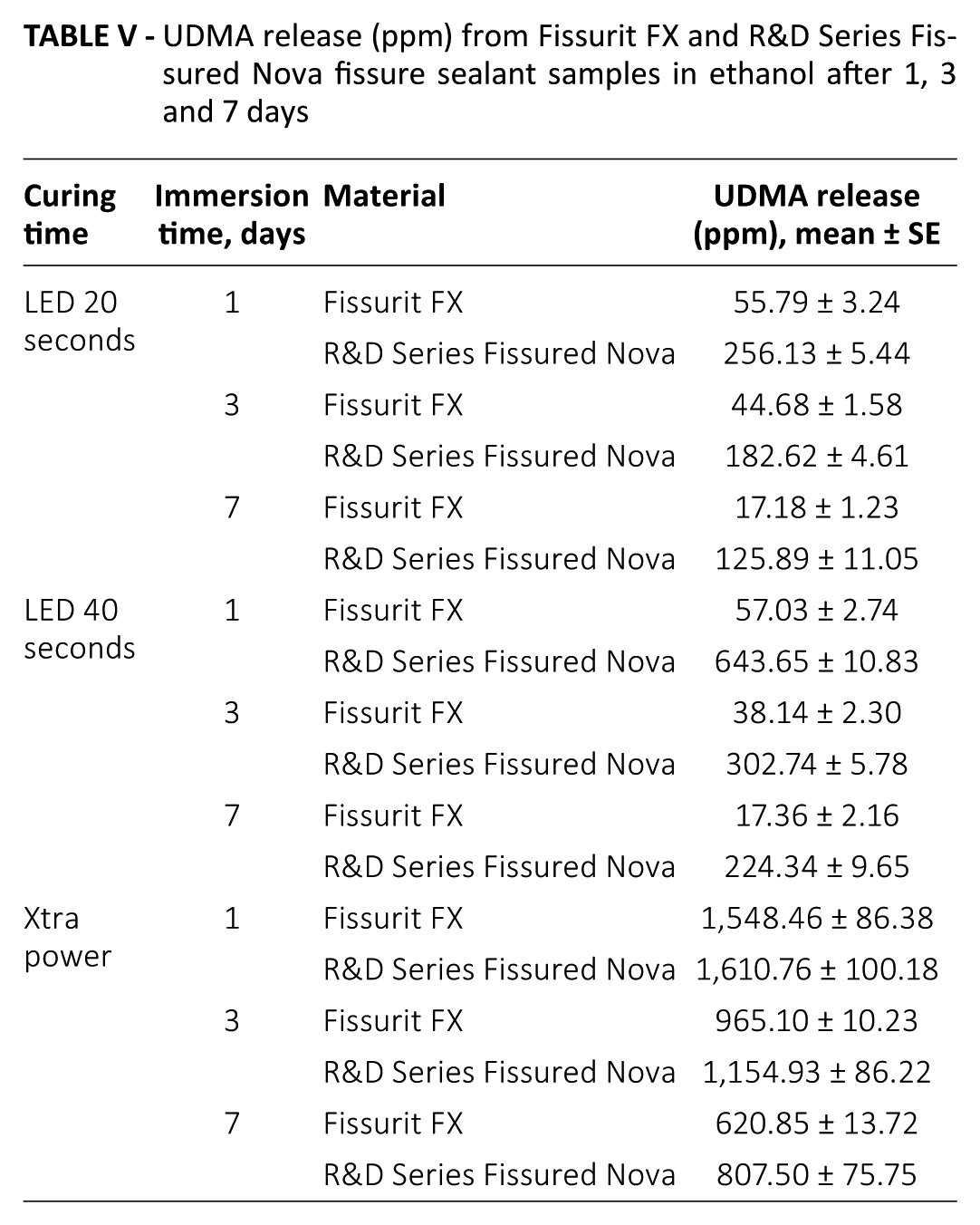
GDMA release (ppm) from Control Seal fissure sealant samples in ethanol after 1, 3 and 7 days
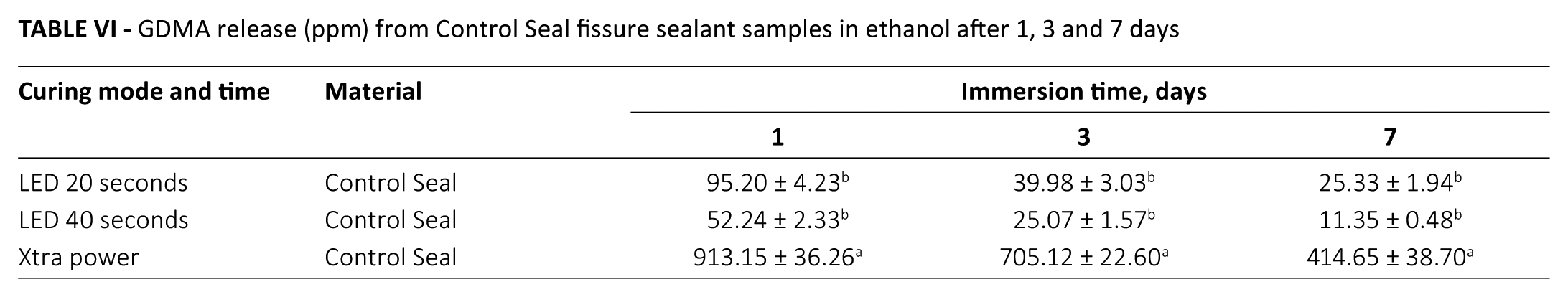
TEGDMA release (ppm) from R&D Series Fissured Nova fissure sealant samples in ethanol after 1, 3 and 7 days

Discussion
The effect of type of material (Control Seal, Voco, Germany; Fissurit FX, Voco, Germany; and R&D Series Fissured Nova, Imicryl, Turkey) and influence of light curing mode (standard LED 20 seconds, standard LED 40 seconds and Xtra power) were evaluated, for the release of bis-GMA, TEGDMA, UDMA and GDMA monomers from resin-based sealants at 1, 3 and 7 days.
Fissure sealants, which have been demonstrated to be efficient in preventing and arresting caries, act as a protective coating on permanent molars (1, 2, 18). Resin-based filling materials used to seal pits and fissures contain more resin matrix and smaller portions of filler particles compared with dental composites; as a result, they contain greater amounts of monomers proportionally.
Methyl methacrylate groups and the hydroxyl groups of BPA are bound with glycidyl groups in bis-GMA, which is the most common base of the resins. Resin-based filling materials also contain other monomers, such as TEGDMA and UDMA to maximize resin viscosity (19).
Following exposure to the substances in the oral environment, various compounds are released from the resin-based dental restorative materials into the surrounding tissues (20). For a decade, studies on the levels of BPA, bis-GMA and bisphenol A dimethacrylate (bis-DMA) in saliva after the placement of dental sealants and composites have shown mixed results. BPA or its derivatives were detected in some in vitro (21, 22) and in vivo (13, 23) studies, while some in vitro studies detected none after periods as long as 10 days after exposure to sealants (24). In the present study, 3 fissure sealants were studied after polymerization with LED and plasma arc modes and analyzed at 1, 3 and 7 days after application. Bis-GMA is a common monomer in pit and fissure sealants. Detectable levels of bis-GMA by HPLC were found with all materials. In this in vitro study, although the measurement of monomers showed reductions with time, monomers were still found in a detectable range.
Recently, a synthetic estrogenic activity was shown in the monomers released from dental resins (25). Thus, some researchers have switched their focus to factors which are contributing to the release of monomers from resin-based dental materials. Some factors that affect monomer release have been studied: chemical composition of the solvent, chemistry and molecular weight and volume of the released components and degree of polymerization (26).
The present study used ethanol to dissolve monomers into the media from resin-based sealants. The amount of elution of monomers was found to be greater than that found in previous studies (26, 27). This was likely because the resin discs were submerged in the organic solvent as soon as the polymerization step was completed. This finding is clinically relevant, because chemical curing of resin which incorporates more available monomers into its structure lasts for some time after the polymerization step is completed using the light source. This finding indicates that the monomer dissolution and release were greater in organic solvents than in pure water. While monomers dissolving in organic solvent demonstrate degradable components in resin materials, leaching of monomers by the dissolving effect of an organic solvent is not a phenomenon seen in clinical conditions (28).
It was thought that resin-based dental materials should be fully polymerized to prevent monomer release in the oral environment (29). However, complete polymerization and thus full monomer conversion is practically impossible, whereas monomer conversion varying only between 35% and 77% (5, 18). This phenomenon can be explained by the difficulty in controlling for many factors such as the wavelength and intensity of the LED source, duration of exposure, restoration thickness and light source distance, which determine the optimal conditions for polymerization (30). Despite various intensities and exposure times being used by researchers, an intensity of 300 mW/cm2 or greater at a wavelength of 450-500 nm is thought to be required for the full polymerization of a 2-mm-thick resin dental composite (31, 32).
In this study, we used a LED device with a wavelength range of 395-480 nm; we used standard power (1,000 mW/cm2) for 20 and 40 seconds, and the Xtra power mode (3,200 mW/cm2) for 3 seconds. The concentrations of monomers released (bis-GMA, UDMA, TEGDMA and GDMA) were significantly higher for the specimens cured with the Xtra power curing mode than for those cured with the LED for 20 and 40 seconds. Under these experimental conditions, Xtra power curing caused inferior polymerization of fissure sealants compared with the LED device for 20 and 40 seconds. Munksgaard et al (29) and Park et al (33) reported similar results, showing that plasma arc curing did not provide optimal curing when monomer release was analyzed by HPLC.
As a common and reliable separation method, HPLC analysis (34) was used in the present study to monitor monomer release from the resin-based sealants. Solvent chemistry and also the chemical composition of components that elute or degrade in resin determine the monomer release from the sealants (26). A study revealed that smaller monomers such as TEGDMA are eluted earlier and quicker than bigger monomers such as bis-GMA and UDMA (35). In the present study, the amount of TEGDMA released was higher than the amount of bis-GMA for the Fissured Nova sealant. This could result from differences in chemical properties and reactive potentials of TEGDMA and bis-GMA under the different curing methods (36).
The manufacturers’ recommendation for curing times was 20 seconds for the 3 fissure sealants used in the study. Although in the Control Seal (Voco, Germany) and Fissurit FX (Voco, Germany) groups, curing with the LED for 40 seconds resulted in less bis-GMA elution than did the recommended 20 seconds, the difference was not statistically significant. For the Fissured Nova (Imicryl, Turkey) group, the recommended 20-second curing time resulted in lower amounts of bis-GMA elution than did curing with the LED for 40 seconds.
Resin-based fissure sealants have demonstrated the greatest effectiveness in preventing and stopping the progression of decay. An increasing number of children are receiving sealants during pediatric dental care. There are precautionary measures that can be taken to reduce monomer release during application of resin-based fissure sealants. In summary, the present results showed that all fissure sealants release monomers, with reductions over time. Since the most intense BPA exposure happens postoperatively, certain precautions seem to be indicated for during the placement of resin-based dental materials, to minimize the BPA release (13, 37). Wiping the surface with cotton swabs or rinsing the mouth with water right after the dental procedure is one of the measures that is effective in removing the residual monomer. However, rinsing mouth might become challenging in paediatric patients, rinsing by the operator with using a water spray-suction system is preferred.
Based on the findings of the present study, it can be concluded that the production and use of dental materials free from BPA and its derivatives should be the priority of the dental sealant industry. Nevertheless, toxicity and related BPA hazard of the current alternatives should be tested to ensure the safe use of newly developed sealant materials. Rubber dam use, as with all dental operative procedures, isolates the operative field, and that limits potential exposure to monomers and other relevant dental materials.
Footnotes
Acknowledgements
The analyses of monomer release of fissure sealants with HPLC reported in this paper were performed by Handan Gaygısız at the Environmental Development Application and Research Center of the İzmir Institute of Technology.
Disclosures
Financial support: The authors have no funding to disclose.
Conflict of interest: The authors declare they have no conflicts of interest.
