Abstract
Purpose
This study evaluated the elution of 2-hydroxyethyl methacrylate (HEMA) from 4 different adhesives, using high-performance liquid chromatography (HPLC).
Materials and Methods
The adhesives were applied on a bovine dentin surface and polymerized using an LED curing unit (n=5). After polymerization, specimens were stored in 75% ethanol solution (6 mL). Residual HEMA that was eluted from adhesives (after 10 minutes, 1 hour, 24 hours, 7 days and 30 days) was analyzed using HPLC. Statistical analyses were performed using 1-way ANOVA, Tukey HSD test and paired 2-sample
Results
There were statistically significant differences among adhesive systems for the cumulative released HEMA and among the time periods (p<0.05). Clearfil SE Bond showed the highest amount of HEMA released, while Easy Bond showed the lowest. Among the time periods, the highest eluted HEMA value was detected in 10 minutes for all adhesives, and elution continued for up to 30 days.
Conclusions
The HEMA eluted from adhesives was in different amounts, and the elution continued for a long time. The amount of eluted HEMA from adhesives used in this study was not viewed as critical for toxic reactions in biological tissues.
Introduction
Dental adhesives are extensively used in dentistry in a variety of applications. The main function of dentin adhesives is to create bonding between materials such as composite and the tooth structure. They exhibit perfect mechanical properties, rapid polymerization, aesthetic quality, ease of handling and the ability to bond to enamel and dentin surface micromechanically (1). Dental adhesives are divided into the categories of etch and rinse and self-etching systems. Self-etch adhesives make use of acidic monomers that can etch and prime at the same time, so that the separate etching and water rinsing stages can be removed. This system reduces the technique's sensitivity and increases the capability of bonding in clinical procedures by reducing chair time (2). Etch and rinse systems are characterized by the use of an acid that is applied to the enamel and dentin and then rinsed off. This is followed by the application of a primer/adhesive on demineralized dentin (3).
Dental adhesives include both hydrophilic and hydrophobic monomers that bond to the surface of vital dentin. The main monomer systems of dental adhesives are bisphenol A glycidyl methacrylate (Bis-GMA), urethane dimethacrylate (UDMA), triethylene glycol dimethacrylate (TEGDMA) and 2-hydroxyethyl methacrylate (HEMA) (4).
HEMA is a common component of most bonding agents used to reduce viscosity, prevent collagen collapse, restore the collapsed collagen network, improve the wettability of acid-etched dentin and increase bond strength (5-6-7).
Acrylates and methacrylates have cytotoxic effects on biological tissues. Hydroxyl groups increase the cytotoxicity of acrylates and methacrylates (8). Due to its sensitizing property, HEMA causes rubefaction, pruritus, indurations, persistent paraesthesia, hypersensitivity, discoloration of the fingertips and allergic symptoms (9-10-11).
The degree of conversion (DC) of a resin material depends on the chemical structure of the dimethacrylate monomer, atmosphere, temperature, light intensity and photoinitiator conditions. For ideal polymerization, monomers in the resin materials should be completely converted to polymer form. However, all dimethacrylate monomers show remarkable residual monomer in the final product, with a DC ranging from 55% to 80% under conventional curing techniques (12).
The degree of monomer–polymer conversion affects the properties of adhesive systems. When polymerization is incomplete, it results in residual monomer in the polymer chain, which may contribute to bonding agent degradation. Chemical degradation is usually caused by oxidation and hydrolysis (13). Oxygen prevents the polymerization of monomer through the formation of an inhibition zone on the surface of a resin in contact with air (14). Adhesive materials have more potential for forming an oxygen-inhibited layer on the surface, as they are cured, and during polymerization a completely uncured layer covers the outer surface. This air-inhibited layer has the greatest potential to degrade residual monomers. Monomer leaching from resin-based dental materials, especially from the oxygen inhibition layer, leads to mechanical degradation as well as decreasing the biocompatibility of the material (15).
Various extraction media have been used to evaluate the elution of monomers of resin materials, including culture medium, distilled water, artificial saliva and ethanol/water solution (16-17-18-19-20-21). When a solvent enters the polymer network, it causes an expansion of the structure, facilitates the extraction of unreacted monomers and promotes the dissolution of linear polymer chains. This expansion is facilitated when the density of cross-links is low. Thus, polymers with a high density of cross-links are more resistant to degradation due to the greater limitation of space and possible paths for the diffusion of permeants in the polymer structure (13). Chromatography is the preferred method for identifying and quantifying residual monomers released from resin materials. High-performance liquid chromatography (HPLC) analysis has been used in various studies to evaluate monomer release from resin materials (1, 16, 21-22-23-24). The aim of this study was to investigate the amount of residual HEMA eluted from adhesive systems, by HPLC.
Materials and Methods
Four adhesive systems were used in this study (Tab. I). The bovine incisor teeth were disinfected by storage in a 5% sodium hypochlorite solution for 24 hours and then stored in distilled water until use. For each tooth, the root system was removed from the cemento-enamel junction. Longitudinal dentin sections were prepared from bovine incisors using a diamond saw (Buehler, Lake Bluff, IL, USA) under water irrigation. The dimensions of the dentin section were length of edge 7 mm and 0.9-mm thickness. The dentin slices were rinsed with physiological saline and sterilized by autoclaving (121°C for 25 minutes). Five dentin sections were used for each adhesive (n=5). Then equal amounts of adhesives (2.5 mg) were applied on the surfaces of the sections and polymerized using an LED (1,200 mW/cm2, Elipar S10; 3M ESPE, St. Paul, MN, USA) according to the manufacturer's instructions. After polymerization, each specimen was immediately immersed in a light-proof glass bottle containing 75% ethanol and 25% deionized water (6 mL) at 37°C. During the period of immersion, the bottles were shaken for 5 seconds, and then the samples were taken from each bottle at 10 minutes, 1 hour, 1 day, 7 days and 30 days using disposable injectors. Residual HEMA (Sigma Aldrich, St. Louis, MO, USA) eluted from adhesives was analyzed by HPLC (Agilent Technologies Inc., Santa Clara, CA, USA) under the following conditions:
CHEMICAL COMPOSITION OF ADHESIVE SYSTEMS
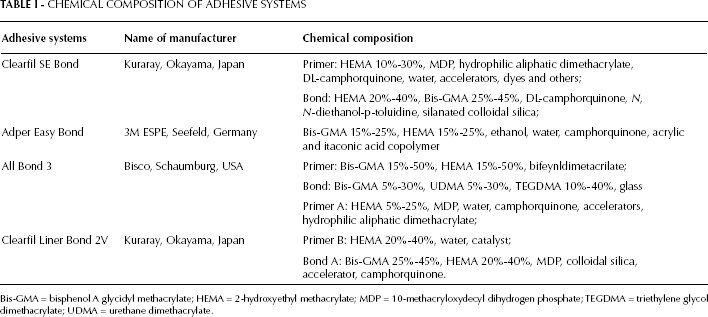
Bis-GMA = bisphenol A glycidyl methacrylate; HEMA = 2-hydroxyethyl methacrylate; MDP = 10-methacryloxydecyl dihydrogen phosphate; TEGDMA = triethylene glycol dimethacrylate; UDMA = urethane dimethacrylate.
Column: Steel column (Supelco SUPELCOSIL™, Bellefonte, PA, ABD), 250 mm in length, 4.6 mm in diameter, particle size of 5 µm;
Mobile phase: CH3CN 80% – H2O 20% (acetonitrile-water);
Flow rate: 1 mL/min;
Detection: UV: 208 nm;
Injection volume: 20 µL;
Temperature: Constant room temperature.
Calibration curves were drawn for HEMA at different concentrations, and the standard chromatogram was obtained. The concentrations of the leaching HEMA from adhesive systems were calculated as cumulative values using the coefficients obtained from a linear regression analysis of the results from the standard series. A linear calibration equation for HEMA is shown in Table II, as described by Altintas and Usumez (22).
LINEAR CALIBRATION EQUATION FOR 2-HYDROXYETHYL METHACRYLATE (HEMA)

λ = wavelength of UV light; r2 = correlation coefficients; y = area of HEMA peak; x = concentration of HEMA.
One-way analysis of variance was used to analyze the data on the quantity of HEMA released by adhesive materials. Multiple comparisons were analyzed using the Tukey HSD test. Comparison of different time periods was analyzed using the paired 2-sample
Results
Means and standard deviations for each group are presented in Table III. The cumulative amounts of HEMA released from adhesives were significantly different and ranked in the following order (from greater to less): Clearfil SE Bond > Clearfil Liner Bond 2V > All Bond 3 > Adper Easy Bond (p<0.05) (Tab. III). Among the time periods, the highest amount of released HEMA from adhesives was observed at 10 minutes (Fig. 1). The amount of elution continued to decrease up to 30 days. The highest amount of release was observed cumulatively at the 30 days, except in the case of All Bond 3.
HEMA CONCENTRATIONS ELUTED FROM THE ADHESIVE SYSTEMS IN 5 TIME PERIODS

Values are means (standard deviation). The concentration values are calculated as micromolar (μM). Groups with different letters are different (A, B, C, D, E, a, b, c, d. p<0.05); HEMA = 2-hydroxyethyl methacrylate.
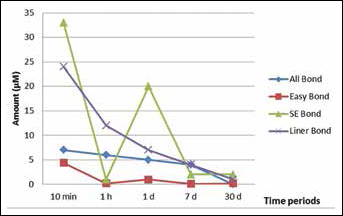
The mean amount of elution 2-hydroxyethyl methacrylate (HEMA) from adhesive systems per storage time.
Discussion
Adhesive systems form an integral part of present-day operative dentistry (25). It is important to analyze the chemistry and composition of these materials, since such characteristics have a direct effect on their clinical performance and biocompatibility. Substances eluted from the polymerized materials (residual monomers, impurities of monomers, additives and degradation products) may irritate the soft tissue, stimulate the growth of bacteria and promote allergic reactions (26).
HEMA is a water-soluble low-molecular-weight monomer that is included in adhesive formulations because of its good miscibility and wetting ability and to solvate hydrophobic methacrylates, which prevents phase separation between adhesive components. However, the high affinity of HEMA to water may negatively influence the mechanical properties of the adhesive system as a result of its intrinsic flexibility and inability to form cross-links by itself (27-28-29).
A powerful analytical technique called HPLC, used to analyze polymers including residual monomers, has been previously used in dental research (19, 21, 30). Distilled water, ethanol and saliva have been used in several studies as extraction solvents (16, 22, 23). An ethanol/water solution in a concentration of 75% is recommended by the US Food and Drug Administration as a clinically relevant food/oral-simulating liquid (31). One of the characteristic features of cross-linked polymers is that they are insoluble, but these polymers are capable of swelling in good solvents. Ethanol which is an aggressive solvent, penetrates the matrix and expands the space between polymer chains, and unreacted and leachable monomers may diffuse out (1). For these reasons, we used an ethanol/water solution as the extraction medium in this study.
Polydorou at al (17) investigated the elution of residual monomer at time intervals of 24 hours, 7 days and 28 days. Altintas and Usumez (22) evaluated the elution of HEMA at time periods of 10 minutes, 1 hour, 1 day and 3, 7, 14 and 21 days by HPLC. In the present study, to determine early and late elution of HEMA from adhesives, the time periods were set at 10 minutes, 1 hour, 1 day, 7 days and 30 days.
The degree of conversion of light-cured resin materials is 55%-80%, and this rate decreases to 35% in the oxygen-inhibited layer. Therefore this layer contains approximately 65% of unreacted monomers (16, 17). This information is supported by our results. Because in the present study, about 50% of the total HEMA released was detected within 10 minutes for all adhesive systems; this rate reached more than 90% in day 1. The higher amount of HEMA released in the first 10 min may be explained by the oxygen inhibition layer on the surface of polymerized adhesives, and the elution in the other storage periods may represent unreacted HEMA in the polymer chain.
A light-curing unit is essential for the success of adhesive restorations. Appliance variables, such as adequate light intensity, correct wavelength and energy density, are essential to achieve a proper depth of cure (24). The curing source must adequately polymerize the resin material and have a wavelength to initiate photo-curing of methacrylate groups and produce a highly cross-linked structure (32). Altintas and Usumez (22) reported that the mean concentrations of residual HEMA released from Single Bond and Optibond Solo Plus in 21 days were 1,097.0 µM and 3,385.2 µM, respectively. Moreira et al (33) evaluated the HEMA released from Adper Single Bond 2 cured inside a fabricated stainless steel circular matrix (8 mm x 1 mm) and reported that the highest amount of released was 930 µM. Cetingüç et al (34) evaluated the HEMA diffusion from Syntec Single and Clearfil SE Bond in young and old primary molars at a remaining dentin thickness of 1 mm and found that the amount of released HEMA ranged approximately from 400 to 470 µM in 3 days. These researchers did not specify the amount of adhesive used in each sample. In the current study, we applied 2.5 mg of each adhesive on the dentin surface. The elution values reported in the present study for both adhesives are lower than those reported in previous studies (22, 33, 34). This difference can be explained in terms of the lower quantity of adhesives that was applied on dentin surfaces and the stronger light source that was used for polymerization in the present study. This is because the degree of conversion of carbon–carbon double bonds also depends on the type, duration and intensity of the light source and on the properties of the resin system (35). The Elipar S10 LED curing unit had a relatively high mean power density (1,200 mW/cm2) which may have caused the high monomer conversion in the adhesive materials.
Large variations were observed for the eluted amount of HEMA between the adhesives. The Clearfil SE Bond and Clearfil Liner Bond 2V showed higher amounts of eluted HEMA than the All Bond 3 and Adper Easy Bond. This may be explained in that, according to the manufacturers’ data on the adhesives, Clearfil SE Bond and Clearfil Liner Bond 2V contain HEMA in both the bond liquid and primer liquid, and these adhesives contain a higher percentage weight of HEMA than All Bond 3 and Adper Easy Bond.
Incomplete polymerization of the resin materials and the leaching of monomers not only decrease the mechanical properties of the restoration, but can also negatively impact the biocompatibility of the material. Uncured monomers leached from resin materials may be eluted into salivary fluids and brought into contact with mucosal tissues causing allergic or adverse side effects in patients and dental staff (36). Previous studies have reported that HEMA's toxic effects on mammalian fibroblasts starts at 1,770 μM, and its toxic effects on monocytes starts at 2,500 μM (37, 38). In the present study, the lowest and the highest amounts of eluted HEMA were 4.4 and 57.9 μM, respectively. In this study, adhesives were applied on the dentin surface without restorations in order to obtain the maximum amount of HEMA that could be eluted from the adhesives. It was apparent that the quantity of HEMA released from the adhesives was not sufficient to be cytotoxic. It should be kept in mind that adhesive systems are not only composed of HEMA and that there is a possibility that several other components in adhesives can have detrimental effects (39). This in vitro approach is useful for the evaluation of HEMA release that can not be measured in vivo. The experimental setup did not consider in vivo conditions, and the measured elution of residual monomers can not be directly equated to the elution of residual monomers in vivo. Therefore, in vivo studies are still needed to evaluate residual monomers and their effects.
Conclusions
Among the time periods, the highest amount of HEMA released from all adhesives was observed at 10 minutes.
Clearfil SE Bond showed the highest amount of HEMA release, while Easy Bond showed the lowest.
The highest concentration of eluted HEMA detected was not viewed as critical for toxic reactions in biological tissues.
Footnotes
All authors declare that they have no conflict of interest.
