Abstract
Background
The purpose of this study was to analyze olfactory ensheathing cell (OEC) proliferation and growth on Biosilicate and collagen bioscaffolds, and to determine whether the application of laser phototherapy would result in increased OEC proliferation on the scaffolds. The use of bioscaffolds is considered a promising strategy in a number of clinical applications where tissue healing is suboptimal. As in vitro OEC growth is a slow process, laser phototherapy could be useful to stimulate proliferation on bioscaffolds.
Methods
OEC cells were seeded on the Biosilicate and collagen scaffolds. Seeded scaffolds were irradiated with a single exposure of 830-nm laser. Nonirradiated seeded scaffolds acted as negative controls. Cell proliferation was assessed 7 days after irradiation.
Results
OECs were successfully grown on discs composed of a glass-ceramic and collagen composite. Laser irradiation produced a 32.7% decrease and a 13.2% increase in OEC proliferation on glass-ceramic discs and on collagen scaffolds, respectively, compared with controls. Laser phototherapy resulted in a reduction in cell growth on the Biosilicate scaffolds and an increase in cell proliferation on collagen scaffolds.
Conclusions
These results were probably due to the nature of the materials. Future research combining laser phototherapy and glass-ceramic scaffolds should take into account possible interactions of the laser with matrix compounds.
Keywords
Introduction
Laser phototherapy at specific wavelengths and energy densities has been established in numerous studies as having the selective capacity to cause cell proliferation in vitro. Previously, our group demonstrated that osteoblast (MC3T3) cell proliferation and alkaline phosphatase (ALP) activity increased significantly after 830-nm laser irradiation at 10 J/cm but decreased with 780-nm laser phototherapy at the same energy density (1). Others have demonstrated similar results, with implications for clinical application of laser phototherapy for tissue regeneration (2, 3).
One such application is for spinal cord repair (4). Some authors have examined the effect of laser phototherapy on olfactory ensheathing cells (OECs) as a possible combination therapy for aiding the stimulation of spinal cord and vertebral spinal nerve sheath repair (5-6-7).
There are many other potential clinical applications in which laser phototherapy might enhance cell stimulation and tissue repair, such as for bone repair (8). Our group is investigating the effect of laser phototherapy on a range of cell lines seeded onto a variety of bioscaffolds as a potential method for assisting in tissue repair and healing (9).
The bioscaffolds being used by our group are innovative new compounds designed for use in clinical settings with the potential to be used in a range of applications such as bone healing and vascular support structures. Bioactive glass ceramic scaffolds (manufactured from Biosilicate®, patent application WO 2004/074199) have been showing stimulatory effects in bone cell proliferation in vitro studies (10) and in bone healing in models of bone defects in rats (11, 12).
Also, a specially modified collagen scaffold developed from the spongin skeleton of a particular species of marine sponge has been utilized in our current study. This scaffold appears to possess a number of unique properties such as a similar organization and composition comparable to that of human collagen Types XIII and IV that make it suitable for use as a biomaterial in tissue engineering applications (13, 14). This scaffold negates potential risks present with the use of bovine and equine collagens, such as the possibility of transmitting bovine spongiform encephalitis and other prion and viral pathogens (14). Furthermore, sponges are sessile, water-dwelling (mostly marine) and filter-feeding metazoans.
Hence, it is prudent to test the ability of these matrices not only to support a range of cell lines (in order to understand the capacity of the structures to support biological tissues) but also to determine if the scaffolds are compatible with laser phototherapy. In this context, the aims of the present study were to analyze the cell proliferation and the growth of the OECs on the bioscaffolds and to determine whether the application of laser phototherapy at 830 nm (continuous; 10 J/cm2 at diode) would result in the increase of OEC proliferation on the scaffolds and to compare any differences noted. To our knowledge, the present study was the first to examine the growth and proliferation of OECs on these 2 unique bioscaffolds to date.
Materials and methods
The OEC cell line was kindly donated by Prof. Alan Mackay-Sim of the National Centre for Adult Stem Cell Research, ESKITIS Institute for Cell and Molecular Therapies, Griffith University, Australia. This cell line is a transformed olfactory progenitor line from
Culturing and passaging of cell lines
The method of culturing and passaging has been described elsewhere (1, 12). Concisely, OECs were grown in sterile, serum-free Dulbecco's minimum essential medium (D-MEM; Gibco) supplemented with 25-mM HEPES, NT3 and 200 IU/mL penicillin with 200 μg/mL streptomycin (Invitrogen). Tissue culture assays and passages were performed under strict aseptic conditions in a biological safety cabinet (Model BH143; Gelman Sciences). Cell lines were grown in sterile 25-cm2 tissue culture flasks (Corning) in 5% CO2 in 95% air at 37oC in a humidified CO2 incubator and passaged every 4 days or as required using standard tissue culture protocols.
Bioscaffold fabrication and preparation
Glass-ceramic scaffold
A novel, fully crystallized bioactive glass-ceramic of the quaternary Na2O-CaO-SiO2-P2O5 system (Biosilicate®, patent application WO 2004/074199) was utilized in this study. This compound has been described previously (1). The glass-ceramic scaffolds (1 cm2) were prepared through the porogen agent addition method; this method consisted of the addition of a porogen agent (naphthalene) into a suspension containing powdered Biosilicate®.
Modified spongin collagen scaffold characterization
A specially modified collagen scaffold developed from the spongin skeleton of a particular species of marine sponge was utilized in our current study. Raw, sun-bleached and dried marine sponge (Family Spongiidae, genus
Seeding of Biosilicate and collagen scaffolds with mammalian cells
For each experiment, sterile, modified Biosilicate or collagen scaffolds were placed in wells of a sterile 6-well tissue culture plate (2 wells irradiated with laser and separated from 2 wells that contained scaffolds that acted as nonirradiated controls). Each of the wells was filled with 2 mL of fresh tissue culture medium (found to be optimal to cover the scaffold) prewarmed to 37oC. The scaffolds were then seeded with 1 × 106 freshly trypsinized OECs and incubated at 37oC in a humidified CO2 incubator (95% air with 5% CO2) for time periods ranging from 3 days to 3 weeks. Cells were cultivated at 1 × 103 cells per well. Cell proliferation and confluence over the seeded scaffolds and in control wells were monitored daily using an inverted light microscope, and spent medium was replaced as required for the duration of the trial period.
Laser irradiation
Twenty-four hours after the seeding of scaffolds, cells were exposed to laser irradiation. The wavelength used was 830-nm continuous (Smart Laser Medilaze; Adlaser Pty Ltd., NSW, Australia; 30 mW, GaAlAs, 10 mm2 beam area, 9.5o beam divergence, 33 second irradiation per spot). A single dose of irradiation was performed at a machine-set energy density of 10 J/cm2. To apply the laser, the irradiating probe was fixed perpendicularly above each culture well over the center of each bioscaffold, and at a standardized distance of 5 mm in order to irradiate as much of the scaffold surface as uniformly as possible. The power output of the laser unit was checked for stability prior to the experiments with a Nova laser power/energy monitor (Model 2A-SH; OPHIR Optronics Ltd.). Seven days after irradiation, cell proliferation was assessed. Each experiment was repeated in at least triplicate for statistical accuracy.
Scanning electron microscopy
Scanning electron microscopy (SEM) was utilized to better determine the microscopic structure of the glass-ceramic and collagen scaffolds after laser irradiation and to better visualize cell growth, migration patterns and morphology of cells growing on the 3-dimensional (3D) modified collagen scaffold and glass-ceramic scaffold. Pieces of 3D collagen and the entire discs of the glass ceramic scaffolds seeded with OECs cells were grown for a further 24 hours postirradiation, then fixed in a 4% glutaraldehyde in cacodylate buffer solution for at least 24 hours. Prior to SEM, the fixed specimens were washed 3 times for 10 minutes in cacodylate buffer, immersed in osmium tetroxide for 30 minutes, and then washed again in distilled water for 10 minutes. Finally the specimens were dehydrated in a graded series of alcohol solutions as follows: two 10-minute washes in 50% ethanol; two 10-minute washes in 70% ethanol; two 10-minute washes in 90% ethanol; two 15-minute washes in 100% ethanol; and two 15-minute washes in 100% amyl acetate. After the final wash, the specimens were critical point dried using a critical point drying apparatus (Denton Vacuum Inc.). The dried samples were then each mounted onto SEM stubs. The surface of each sample was then sputter-coated with gold particles using a Bio-Rad SC500 sputter coater at 15 mA for 2 minutes. The specimens were then placed into the vacuum chamber of a FEI Quanta 2000 scanning electron microscope and viewed at an accelerating voltage of 10 kV.
SEM photomicrographs were also examined in detail to determine if there were any morphological differences between cells seeded onto scaffolds and laser-irradiated (treated), and those grown on scaffolds without irradiation (negative controls).
Scaffold characterization
The total porosity of both bioscaffolds was determined by the immersion method, using Archimedes’ principle. The mean pore size was determined by the direct analysis of SEM images of the scaffolds embedded in epoxy resin (EpoxiCure™ - Buehler®) under vacuum.
Light microscopy
Biosilicate and collagen scaffolds were fixed in 4% glutaraldehyde in 0.1 M cacodylate buffer overnight. After fixation, scaffolds were placed in 1% osmium tetroxide for 60 minutes, and then washed in distilled water twice for 15 minutes. The samples were then placed in a 1%-2% solution of uranyl acetate for 30 minutes and then washed in distilled water for 5 minutes. After dehydration in a graded series of ethanol solutions, samples were permeated with Spurr's epoxy resin and polymerized in an oven at 70°C overnight. Sections were cut using an ultramicrotome. For light microscopy, samples were then stained with Toluidine blue dye, dried, and then stored in a cool dry place until viewing.
Cell proliferation assays
Cell proliferation was assessed by the CellTiter 96® AQueous One Solution Cell Proliferation Assay (Promega Corp.) following the manufacturer's instructions. This assay employs a colorimetric method based on the tetrazolium compound (3-(4, 5-dimethylthiazol-2-yl)-5-(3-carboxymethoxyphenyl)-2-(4-sulfophenyl)-2H-etrazolium, inner salt; MTS) for determining the number of viable cells in proliferation. The MTS tetrazolium compound is bioreduced by cells into a colored formazan product that is soluble in tissue culture medium. Cell proliferation was determined using a Spectromax 250 spectrophotometer (Molecular Devices) at an absorbance wavelength of 490 nm (12).
Statistical Analysis
Descriptive statistics were used to describe means and standard deviation (SD) of the percentages. A 2-way ANOVA test was used to assess the significance of differences between the percentage of increase or decrease of irradiated groups compared with negative (untreated) controls, and Duncan's test to identify the differences. A p value of 0.05 was considered statistically significance.
Results
Biosilicate scaffold characterization
Figure 1 shows an overview of the representative SEM sections of Biosilicate® scaffolds. Scaffolds macropores were oval-shaped and presented an average diameter of 53 µm (Fig. 1A). The oval shape is caused by a local deformation of the Biosilicate® block during the fiber burn-out (Fig. 1B). It can be noted that the scaffolds also constituted of a network of interconnected microchannels (Fig. 1C). The existence of these microchannels allowed the complete penetration of the resin inside the scaffold (Fig. 1D). The structure composed of Biosilicate® grains presented closed micropores smaller than 1 µm in diameter (Fig. 1E). In addition, it was possible to observe the presence of microcracks in the Biosilicate® grains (Fig. 1F). Nevertheless, the scaffolds had enough mechanical strength for handling and insertion into the bone defects.

Scanning electron microscopy.
The Biosilicate scaffolds showed a total porosity of 44% and macroporosity of around 15%. The remaining porosity corresponded to the sum of the interconnected microchannels and the closed pores. The results concerning porosity are show on Table I.
Physical characteristics of the Biosilicate scaffolds

Values are means ± standard deviation.
Collagen scaffold characterization
The collagen scaffolds had pore sizes ranging from 50 μm to over 1,000 μm and a mean major fiber diameter of approximately 20 μm (Tab. II). Also, the scaffold surface produced a smooth topography (Fig. 2A, B) rather than a spiral patterning. Furthermore, the modified fibrils appeared to possess greater interconnectedness, with the possibility that some fibrils had crosslinked with one another (Fig. 2C, D). The manufacturing process also appeared to remove contaminants from the scaffold, including bacteria, fungal spores, sponge cell reproductive bodies (gemmules) and silicate spicules.
Physical characteristics of the collagen scaffolds

Values are means ± standard deviation.
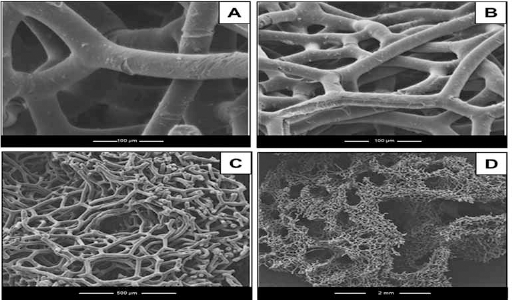
Scanning electron microscopy of the collagen scaffolds.
SEM evaluation of scaffolds seeded with cells
Figures 3 and 4 show a series of representative SEM images of the collagen and glass-ceramic scaffolds, with or without cells (24 hours after laser irradiation and control scaffolds at 24 hours). Figures 3A and 4A show the structure of Biosilicate and collagen scaffolds before the immersion, respectively. Also, examination of SEM revealed that both scaffolds were capable of supporting OEC growth, indicating that both scaffolds had the capacity to support the cell line utilized (Figs. 3B and 4B, for Biosilicate and collagen scaffolds, respectively). Figures 3C and 4C show the morphology of both scaffolds, without cells, after the immersion. On gross examination of the scaffolds at the conclusion of the experiments, we were able to distinguish a crystalline substrate in all culture wells (laser and controls) containing the glass-ceramic scaffold, consistent with our previous findings (Fig. 3B, C). Biochemical and morphological analyses of the crystalline substrate revealed the presence of possible minor remnant silicate as an artefact of the glass-ceramic scaffold manufacturing process (Fig. 3B, C). There was an absence of a similar substrate in the culture wells containing the collagen-derived scaffolds (Fig. 4B, C). However, despite this crystalline deposit in the collagen scaffolds, OECs grew consistently, and there did not appear to be any notable detrimental effects on cellular attachment, growth or physiology (Fig. 4B, C).

Scanning electron microscopy (SEM) images.
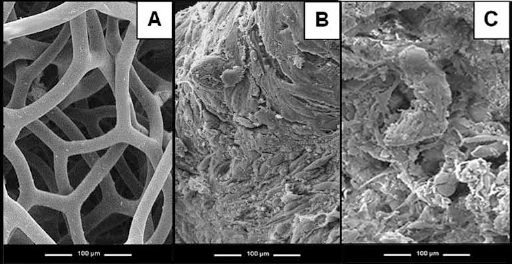
Scanning electron microscopy (SEM) images.
Cell proliferation
Laser irradiation at 830 nm (30 mW, 10 J/cm2 at diode) produced a significant 32.7% ± 5.5% decrease in OEC growth on the glass ceramic scaffold discs (mean absorbance ± SD = 0.192 ± 0.002) compared with control (nonirradiated) discs (mean absorbance 0.22 ± 0.002). The same laser parameters produced a significant 13.2% increase in OEC cell proliferation on the collagen scaffolds (mean absorbance 0.625 ± 0.007) (group 4) compared with nonirradiated control scaffolds (mean absorbance 0.552 ± 0.005) (group 3) (Tab. III).
Mean percentage change in stem cell proliferation on Biosilicate and collagen scaffolds, with and without laser
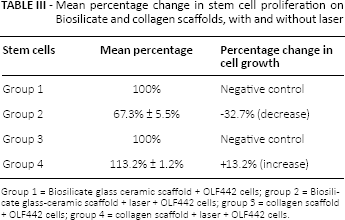
Group 1 = Biosilicate glass ceramic scaffold + OLF442 cells; group 2 = Biosilicate glass-ceramic scaffold + laser + OLF442 cells; group 3 = collagen scaffold + OLF442 cells; group 4 = collagen scaffold + laser + OLF442 cells.
Discussion
The results of this study demonstrated that the seeding techniques and methodology described were successful for both bioscaffolds. The scaffolds were capable of supporting progenitor-like OEC growth. In addition, we have demonstrated an inhibitory effect of laser phototherapy (830 nm at 10 J/cm2) on these cells when grown on the glass-ceramic scaffold but a stimulatory effect when grown on the collagen scaffold.
The ability of Biosilicate scaffolds to support osteoblast cells and to accelerate bone healing has been previously demonstrated, corroborating the results found in the present study (12, 15, 16). This fact may be explained by the ion dissolution which happens immediately after the contact of bioactive glass with fluids and is responsible for the stimulation of cell growth and proliferation (17).
Similarly, a successful growth of OECs was observed on the collagen scaffold. A study conducted by Green et al (18) demonstrated that collagen fibers could support the attachment, aggregation and proliferation of human osteoprogenitor cells. In their study, histochemical staining for molecular markers of bone differentiation indicated formation of new bone matrix in bone tissue grown on the spongin scaffolds.
Laser phototherapy produced a decrease in cell proliferation on the Biosilicate scaffolds. These results confirm our previous observations that irradiation by laser phototherapy at 830 nm (10 J/cm2 at diode) of osteoblasts on a glass-ceramic scaffold results in inhibition of cell proliferation compared with nonlaser controls (12). It is important to note that, although the glass-ceramic scaffold demonstrated the capacity to support cell growth and proliferation in a previous study (10), this is the first time the glass-ceramic disc has been assessed for its capacity to support OEC in combination with laser phototherapy. The glass-ceramic scaffold as used in this study was supplied as a solid disc, without the same open-pore structure demonstrated with the collagen-derived scaffold. The structure of the glass-ceramic scaffold and the breakdown crystals may have resulted in the loss of light fluence creating an environment in which the light was unable to penetrate the structure, or indeed where the light was reflected and refracted from the crystalline structure, thus rendering the laser phototherapy suboptimal or even supraoptimal, resulting in the inhibitory response noted herein.
Interestingly, an increased cell proliferation was observed in the irradiated collagen scaffolds. This observation is significant if one considers the recent interest in developing bioscaffolds for tissue regeneration purposes. Given that we used identical methods (including cell lines, preparation of cells and laser irradiation parameters and methods), clearly the type of scaffold on which the cells are grown can influence the response. Others have also noted the effects of laser phototherapy on bioscaffolds (19). The above observations raise the possibility that the structure of the scaffolds used in this study may have influenced the outcomes.
Our group has characterized a modified collagen-derived scaffold. It is spongiform in nature with larger pores and channels penetrating the surface of the matrix allowing the growth of cells on and throughout the scaffold (with the aim of promoting angiogenesis and mechanical strength in the clinical setting), thus making it conducive to tissue viability. These modified collagen scaffolds had pore sizes ranging from 200 μm to over 1,500 μm – much larger than those noted in other commercially available scaffolds. The mean major fiber diameter of the scaffolds was approximately 20 μm, and the surface of the fibers was quite smooth in topography. All of these factors are likely to have resulted in the promotion of cell growth as confirmed in the results of this study. In contrast, the glass-ceramic scaffold used in this study was supplied as a disc of bioactive material with a total porosity of 61% and mean pore size of 53 µm.
Given the stimulatory effect of the laser phototherapy on the collagen scaffold, it would appear that some form of physicochemical interaction in the glass-ceramic scaffold has led to the results noted here. Further study will need to be undertaken to identify what the interaction may have been.
Investigations into tissue growth, differentiation, repair and remodeling are employing in vitro and in vivo 3D tissue models and scaffolds as important research tools with implications to benefit the clinical arena (20-21-22). Moreover, there are now increasing reports to confirm that cell-extracellular matrix interactions influence cell growth and differentiation (23-24-25). The intended application of some of this research is to grow viable, functional tissue structures to support and/or replace dysfunctional or nonfunctioning tissues. The results of this study suggest that the scaffold properties need to be taken into account when using laser phototherapy in combination with bioscaffolds in future studies of tissue regeneration.
The results of this study support the concept that laser phototherapy can be used to stimulate the growth of cells on modified collagen bioscaffolds. It is well known that cell–tissue response is influenced by laser irradiation parameters (both wavelength and dose). The laser protocol used in the present study was chosen based on a previous study performed by our team which demonstrated that 830-nm laser at 10 J/cm2 was capable of increasing osteoblast cell proliferation in an in vitro study (1). However, based on the negative results found in the irradiated cells seeded in the Biosilicate scaffolds, future work now needs to consider the effect of different wavelengths and doses on the scaffolds. The potential for using laser phototherapy in combination with bioscaffolds has been established herein, paving the way to using laser phototherapy in future models of tissue regeneration.
Conclusions
A successful method for seeding progenitor-like olfactory neuronal ensheathing cells to bioscaffolds has been developed and described. The bioscaffolds used in these experiments had a demonstrated capacity to support the cell line utilized. Despite successful seeding of glass-ceramic discs and modified spongin collagen scaffolds by these progenitor cells, laser phototherapy (830 nm at 10 J/cm2 at diode) resulted in a reduction in cell proliferation in the glass-ceramic scaffolds compared with nonirradiated controls and an increase in cell proliferation in the collagen-derived scaffolds compared with nonirradiated controls. Interaction by laser with the crystalline structure of the glass ceramic compound is a possible, although unverified, cause of the inhibition of cell proliferation. Future investigations combining laser phototherapy and bioscaffolds (in particular, biosilicate and collagen matrices) should enable us to elucidate the possible interactions of the laser with the chemical components of a specific bioscaffold and the physical properties of the scaffold matrix.
Footnotes
Financial support: No financial support was received for this study.
Conflict of interest: There were no conflicts of interest.
