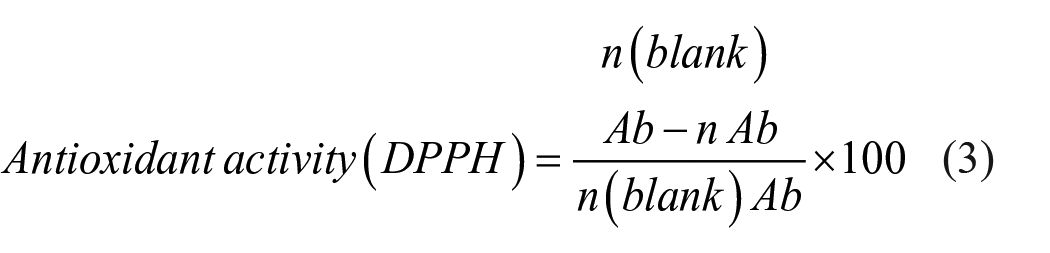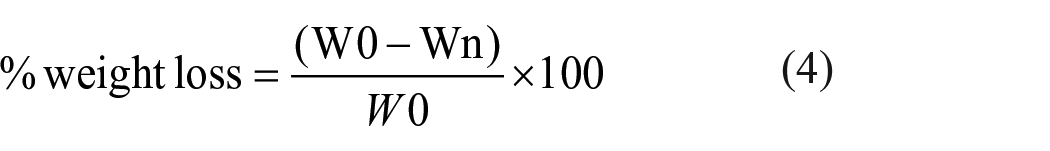Abstract
The research on tissue engineering applications has been progressing to manufacture ideal tissue scaffold biomaterials. In this study, a double-layered electrospun biofiber scaffold biomaterial including Polycaprolactone (PCL)/Collagen (COL) fibrous inner layer and PCL/ Momordica charantia (MC) and Hypericum perforatum (HP) oils fibrous outer layer was developed to manufacture a functional, novel tissue scaffold with the advantageous mechanical and biological properties. The main approach was to combine the natural perspective using medicinal oils with an engineering point of view to fabricate a potential functional scaffold for tissue engineering. Medicinal plants MC and HP are rich in functional oils and incorporation of them in a tissue scaffold will unveil their potential to augment both new tissue formation and wound healing. In this study, a novel double-layered scaffold prototype was fabricated using electrospinning technique with two PCL fiber layers, first is composed of collagen, and second is composed of oils extracted from medicinal plants. Initially, the composition of plant oils was analyzed. Thereafter the biofiber scaffold layers were fabricated and were evaluated in terms of morphology, physicochemistry, thermal and mechanical features, wettability, in vitro bio-degradability. Double-layered scaffold prototype was further analyzed in terms of in vitro biocompatibility and antibacterial effect. The medicinal oils blend provided antioxidant and antibacterial properties to the novel PCL/Oils layer. The results signify that inner PCL/COL layer exhibited advanced biodegradability of 8.5% compared to PCL and enhanced wettability with 11.7° contact angle. Strength of scaffold prototype was 5.98 N/mm2 thanks to the elastic PCL fibrous matrix. The double-layered functional biofiber scaffold enabled 92% viability after 72 h contact with fibroblast cells and furthermore provided feasible attachment sites for the cells. The functional scaffold prototype’s noteworthy mechanical, chemical, and biological features enable it to be suggested as a different novel biomaterial with the potential to be utilized in tissue engineering applications.
Introduction
Tissue engineering has three main components: cells, scaffold and biological signals. The biomedical field has observed excellent achievements with the manufacture of polymeric scaffold candidates. This encourages the researchers to study on the novel and modified functional biomaterials for tissue regeneration and wound healing. The skin is a highly organized organ that covers the outer surface of the body and prone to many damages and has many vital functions. 1 The most physically serious injuries are skin damage due to surgical operations, burns, and exposure to heat, electric shock, or corrosive chemicals followed by delayed wound healing. 2 Skin repair is defined as complicated synchronized biological activities involving numerous cells and cytokines. The wound dressing acts as a permeable layer that assists wound closure and protects the injured tissue from further external impacts and contaminants causing a prolonged healing period. The design of wound dressings is still a major challenge in healthcare marketing due to the limitations in developing a product that completely meets the ideal wound dressing requirements. The ideal wound dressings are preferred to absorb excess exudate, have good porosity for oxygen exchange, protect the wound site from bacterial infections and mechanical effects, keep the wound site moist to prevent dehydration, decrease the surface necrosis of the wound, enhance the cellular regeneration by stimulating cell proliferation, and be flexible for avoiding the wound from compression. 3
In recent years, several types of tissue scaffolds have been designed in the form of hydrogels, sponges, foams, films, and nanofiber mats, to be used as wound dressing. 4 Nanofibers have gained attention due to their great surface area to volume ratio, high porosity, and enhanced mechanical properties. They are bioinspired materials imitating extracellular matrix (ECM) structure to promote cellular proliferation, cell adhesion and absorbing exudate for tissue regeneration. 5 Electrospinning is an efficient and easy to method to manufacture nanoscaled fiber mats that enable delivery of bioactive agents like wound healing accelerators, inhibit the spread of external microorganisms and control fluid drainage owing to the excellent surface properties. 6
Poly-ε-caprolactone (PCL) is one of the biocompatible and biodegradable FDA approved synthetic polymers that can be simply electrospun and possess some important physicochemical features such as hydrophobicity, favorable mechanical properties, and low degradability rate. It has been widely investigated and used as a matrix enabling incorporation for the loading of natural or biological substances such as drugs, nanoparticles, herbal extracts and essential oils for tissue regeneration applications.5,7–9 However, the hydrophobicity and low degradation ratio of PCL led to demand for combination of excellent properties of fibers with the natural biopolymers to increase hydrophilicity for enhanced biocompatibility and to offer typical binding sites for cells to adhere. In this regard, collagen (COL) is the most abundant structural protein and biopolymer making up about 70%–80% of ECM and present as an excellent component for manufacturing composite scaffold with PCL. 10 It promotes skin regeneration by providing the attachment sites of vascular cells, removal of debridement, and re-epithelization. 11
Momordica charantia (MC), commonly known as bitter melon or bitter gourd, has the potential to be used for the treatment of viral infections, diabetes, inflammation, and cancer, besides microbial infections, owing to an anti-inflammatory, antioxidant, immune stimulant, immunoregulatory, antidiabetic, antiviral, hypoglycemic, antibacterial, antifungal, anticancer and antiulcer properties.12,13 These effects are mainly attributed to the biologically active phytocompounds such as fatty acids, proteins, polysaccharides, alkaloids, flavonoids, steroids, saponins, triterpenoids, and essential oils. 14 Momordica charantia seed oil consisting of stearic acid, linoleic acid, and, oleic acid applied as the wound dressing materials have been reported comprehensively. 13 The results confirmed that MC seed oil shows antidiabetic, antitumor, antioxidant, and antibacterial activities which are significant properties to accelerate wound healing.
Hypericum perforatum (HP), popularly called “St. John’s wort,” is a plant containing bioactive phytochemicals such as hypericin, hyperoside, isoquercetin, rutin, hyperforin, epicatechin, and essential oils, used in alternative medicine due to its therapeutic activities like sedative, anti-inflammatory, antimicrobial, anti-ulcer, antioxidant, analgesic and wound healing.6,15 The superior effects of that plant oil in enhancing wound healing have been reported to result from enhanced infection resistance, activation of fibroblasts, stimulated collagen synthesis, and collagen accumulation. 16 The most of the studies have been focused on the therapeutic effects of HP oil on wound healing and supported these beneficial characteristics in vitro and in vivo. One of these studies reported that an HP oil-loaded membrane exhibited increased antibacterial activity against S. aureus and E. coli and biocompatibility without causing any apoptotic or necrotic effects on the L929 mouse fibroblast cell line. 17 Additionally, other studies confirmed the antibacterial activity of HP and declared that it allows a high cell proliferation rate and cell attachment in vitro.18,19 Furthermore, HP extract in a niosomal topical drug delivery system exhibited increased cell proliferation rate, declined inflammation, enhanced re-epithelization, and reduced wound size when compared to the control groups. 20 Accelerated inflammatory response and improved healing performance of HP on diabetic wounds are reported in another in vivo study. 21
In this study, the aforementioned functional oils extracted from medicinal plants MC and HP were incorporated in PCL fibers as an outer layer to boost the biological activity of COL enriched PCL tissue scaffold layer and to fabricate a potential functional scaffold prototype for tissue engineering and wound healing applications. The literature knowledge reveals that both MC and HP oils have excellent biological functions and wound-healing activities. In this study, it was aimed to exhibit the biological contribution of the blend of oils to the biomaterial. The main approach was to combine the natural perspective using medicinal oils with an engineering point of view to fabricate a potential functional scaffold for tissue engineering and wound healing. Here it was intended to design a double-layered electrospun biofiber scaffold prototype consisting of the inner layer including COL and PCL mat and the outer layer comprising MC/HP oils blend in the PCL polymer matrix. In such a double-layered scaffold prototype, PCL/COL layer would enhance epithelialization by acting as a mechanically strong substrate for cell adhesion and proliferation as previously proposed.10,11 The novel blended oils including the outer layer would play a role in preventing oxidative stress to speed up the wound-healing process for tissue regeneration. It is anticipated that the scaffold prototype will pave the way for cutting-edge biomaterials research using polymers and phytochemicals to hasten tissue regeneration and wound healing
Methods
The MC and HP oils were analyzed concerning the European Pharmacopoeia monographs. The double-layered scaffold model was fabricated and characterized by Field Emission Scanning Electron Microscopy (FE-SEM), Fourier Transform Infrared Spectroscopy (FTIR), Thermogravimetric Analysis (TGA), mechanical tensile test and wettability test. Additionally, in vitro biodegradability test was conducted in PBS, bio-compatibility was evaluated on the L929 fibroblast cell line and antibacterial activity was tested against on E. coli and S. aureus.
Chemicals, reagents, and cells
Acetone (99.5%, Tekkim), cell proliferation kit (XTT based, Sartorius), 4′,6-Diamidine-2′-phenylindole dihydrochloride (DAPI, Sigma Aldrich), dichloromethane (DCM, 99.5%, VWR Chemicals), DMEM (Medium with high glucose, L-Glutamine, phenol red, without HEPES, Sigma-Aldrich, Life Science) ethanol (99%, ISOLAB), fetal bovine serum (qualified, heat inactivated, Gibco), formic acid (98%, Merck), gentamycin solution (Biological Industries), hexamethyldisilazane (HMDS, Sigma-Aldrich), Hypericum perforatum oil (Zade Vital Pharma Chemicals & Food Inc.), marine collagen type I (Zade Vital Pharma Chemicals & Food Inc.), Momordica charantia maceration oil (Zade Vital Pharma Chemicals & Food Inc.), polycaprolactone (PCL, Mw = 80.000 g/mol, Sigma-Aldrich), phosphate buffer solution (PBS, pH:7.4, Sigma-Aldrich, Life Science), tryphan blue solution (Sigma Aldrich), Rhodamine phalloidin (ThermoFisher Scientific), E. coli (ATCC 25922) and S. aureus (ATCC 29213) donated by Necmettin Erbakan University Medical Faculty and L929 fibroblast cell line were used for the experimental setup of the study.
Equipment
Electrospinning equipment consisting of a high voltage source (up to 40 kV, Eraktek Inovation), an aluminum rotary cylinder collector, and four syringe pumps (New Era Pump Systems, Inc.), Centrifuge (Nüve, Türkiye), Class II sterile cabinet (DEMAIR, Tip A), CO2 incubator (Nüve, Türkiye), Contact angle measuring device (Biolin Scientific, Attension Theta model), FE-SEM (ZEISS Gemini SEM 500, Germany), Fluorescent Microscope (BAB image system), FTIR (Shimadzu IRTracer-100, Japan), Inverted light microscope (Olympus, CKX53), TGA (Shimadzu Japan TGA-50), Tensile tester (Shimadzu AGS-X), 96 well plate reader (Elisa reader Thermo-Skyhigh) were used.
Production of biofibers by electrospinning method
The electrospinning was carried out in two steps which are preparing the PCL/COL and PCL/Oils solutions as the first stage and applying the electrospinning technique to form biofibers under the electrical field as the second step as given in Figure 1. In the preparation process of PCL/COL solution 15% w/v PCL and 15% w/v COL type I were dissolved in a solvent mixture of formic acid: acetone (70:30) v/v. Following, PCL/Oils electro-spinning solution was prepared by dissolving 10% PCL, 2.5% HP oil, and 2.5% MC oil in the 100% v/v DCM, as solvent. The electrospinning process was applied for both solutions of biofiber layers with parameters of 0.75 mL/h feeding rate, 25–27 kV applied voltage, and the distance of 12 cm between the needle tip (22 G, 0.413 internal diameter) and the collector with the rotational speed range of 500–600 rpm. The electrospinning was carried out at room temperature (~25°C) and 50% humidity. The double-layered biofiber dressing was formed by electrospinning of PCL/Oils solution onto the PCL/COL electrospun biofiber layer which was collected on the rotating drum previously. The thickness of both layers was standardized at 0.75 mm by using an equal volume of polymer solution during electrospinning and the thickness of each layer was measured with a micrometer. The double-layer scaffold membrane was dried under a vacuum.
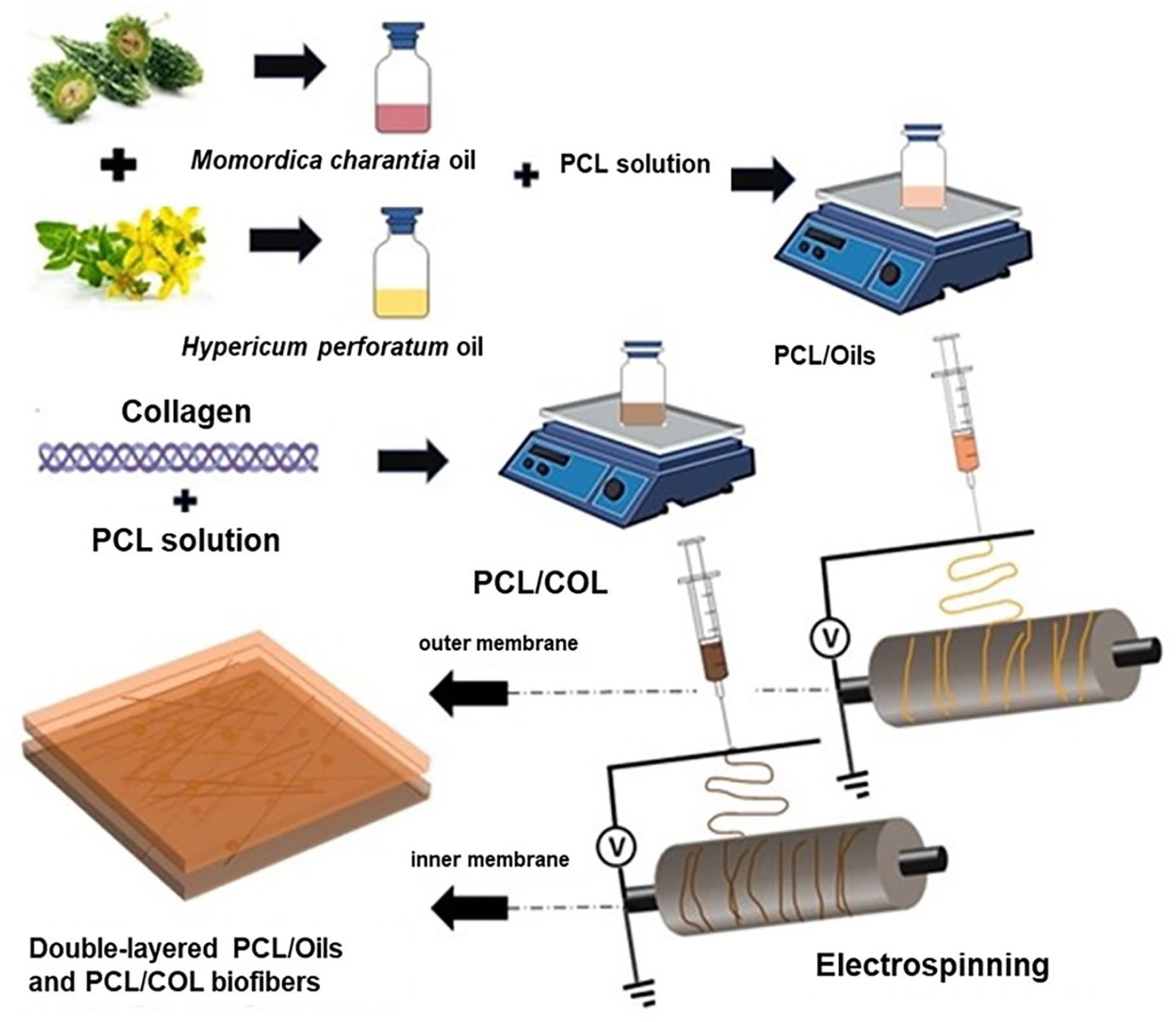
The schematic diagram of electrospinning process of double-layered PCL/Oils and PCL/COL biofibers.
Characterization
Oils
The analyses 2.4.1.1-2.4.1.7 were performed according to the European Pharmacopoeia monographs.
Free fatty acid
Free fatty acid amounts of the MC maceration oil and HP oil (10 g each) were dissolved in 50 mL of ethanol: petroleum ether (2:3 v/v). 1 mL of phenolphthalein was added as an indicator. Titration was done with 0.1 N KOH until the color turned pink. The results were evaluated using equation (1). 22
V; sample titration consumption (mL); P; 282 g/mol, molar mass in grams per mole of oleic acid; M is the exact concentration in moles per liter of titrated potassium hydroxide solution used; m; sample amount (g).
Fatty acid composition
Oil samples (60 mg each) were mixed with 2 mL of 2 N methanolic potassium hydroxide (KOH) solution by vortex. Then 2 mL of n-heptane was added to the sample solution and mixed by vortexing. The oil sample was centrifuged at 3000 rpm for 5 min. The supernatant (organic phase) was transferred into the vial after filtering. Analysis was done by gas chromatography (Shimadzu Japan GC 2010 Plus) at temperature: 250°C, Injection Volume: 1.0 µL. Relative retention times of methyl esters of fatty acids were determined by FAME standard mix (Sigma Aldrich Supelco 37 Component FAME Mix).
Peroxide number
MC oil and HP oil (5 g each) were mixed with 30 mL of glacial acetic acid: chloroform (1:1 v/v) solution. 0.5 mL of saturated potassium iodide solution was added. 1.0% starch solution was used as indicator. Titration was done with 0.01 N sodium thiosulfate solution. The results were evaluated using the equation (2). 23
V; sample titration consumption (mL), M; sample amount (g).
Density
Densities of MC maceration oil and HP oil samples were analyzed by densitometer (Rudolph Research Analytical DDM 2910 Automatic Density Meter, USA).
Refractive indices
The refractive indices of MC maceration oil and HP oil were analyzed by refractometer at 20°C (λ = 589.3 nm) (Rudolph Research Analytical J257 Automatic Refractometer, USA).
Phenolic content
Natural phenols are very diverse group of compounds carrying one or more phenolic group, the total phenolics are expressed as milligrams of gallic acid equivalents per gram of extract (mg GAE/g extract). MC maceration oil and HP oil blend (5 g each oil) was subjected to Folin’s assay Total amount of phenolic substance in oil samples were calculated through calibration curve and expressed as gallic acid equivalent (GAE).
Antioxidant activity
DPPH is a stable highly colored free radical that can split labile hydrogen atoms from phenolic antioxidants with concomitant formation of a colorless hydrazine. To measure the antioxidant activities of MC maceration oil and HP oil DPPH method was followed. L-Ascorbic acid was used as positive control due to its high free radical scavenging activity. Absorbance was measured at 517 nm. The results were evaluated using equation (3). The total antioxidant activity of each oil was expressed as the concentration of sample that reduce 50% of DPPH (IC50).
Double layered scaffold prototype
Chemical structure (FTIR analysis)
FTIR (Shimadzu IRTracer-100, Japan) was used to characterize the presence of PCL, oils, collagen the chemical groups in the biofiber scaffold layers. FTIR spectra of samples were recorded in the range of 350-4500 cm-1.
Morphological structure FE-SEM
The morphology and diameter of the electrospun biofibers were examined under scanning electron microscopy (FE-SEM, ZEISS Gemini SEM 500, Germany). Prior to FE-SEM observations, biofibers were cut into 1 cm2 pieces for iridium coating under pressure. At least 20 biofibers were randomly selected from the two FE-SEM images of each PCL/COL and PCL/OILS mat and the diameters were measure by Image J software to calculate the average value of the biofiber diameters. The frequency of fiber diameters for each layer was demonstrated as histogram plot.
Thermal behavior (TGA)
Thermal behavior was determined to measure the temperature dependent mass loss of biofiber scaffold layer. TGA was carried out under nitrogen atmosphere in the range of 0°C–500°C and at a heating rate of 20°C/min for 15 min. The thermal characteristics, thermal stability and thermal decomposition temperatures of the biofibers were determined and the thermal behavior relationships of the samples were discussed. TGA helps to find the temperature range in which the material does not lose weight, and this range represents the temperatures at which the material is stable the temperature at which it loses weight, shows the temperature range at which the material degrades. 5
Mechanical behavior
Tensile strength, elastic modulus, and elongation at break of all biofiber types were determined by tensile test according to ASTM D638-14 standard. In order to determine the mechanical properties of the biofiber mat, a tensile test was performed with a universal tensile testing machine (Shimadzu AGS-X). The scaffold layers were cut into 10 mm × 50 mm dimensions, the test was carried out at a tensile speed of 10 mm/min. The tests were performed at room temperature and were repeated five times for each type of fiber mat. Then, stress versus strain curves were plotted using the mean value of stress and strain of each layer and scaffold prototype. The elastic modulus of the fiber mats, which is determined by the slope of the initial linear part of the stress-strain curves, as well as the maximum stress, and percent elongation were reported.
Biodegradability, wettability, and in vitro biodegradation
Biofiber scaffold layers were cut in 2 cm × 2 cm dimensions and placed in the contact angle measuring device to examine the wettability of the biofiber layers. Measurements were made with the sessile drop technique by dropping approximately 5 μL of ultrapure water on the biofibers at room temperature. Average values were calculated for each sample. 5
Both PCL/COL and PCL/Oils layers of scaffold prototype were cut into 9 cm2 pieces to evaluate degradation behaviors. The samples were immersed in PBS. After the samples were weighed, each sample was put in a glass tube filled with 15 mL PBS at pH 7.4. The samples in tubes were incubated at 37°C during a week for degradation process and after each week, the samples were air dried for 2 days and weighed. This cycle was repeated 18 times and the time-dependent weight loss (%) were estimated from the residual weights measured for each sample in accordance with equation (4), where W0 is the initial weight and Wn is the weight recorded at the end of degradation periods.
Biocompatibility
Cell viability
The biocompatibility of the double-layered biofiber scaffold prototype was evaluated using L929 healthy fibroblast cells. The two sides of the scaffold were sterilized by UV radiation for 1 h and placed in flat bottomed wells in a 96-well plate. 100 µL of high glucose DMEM medium containing L-glutamine, 10% fetal bovine serum (FBS) and antibiotic was added on each well to condition the fiber scaffold. Then, cells were seeded on the scaffold with (5000 cells per well). Three different test plates were prepared for 24, 48- and 144-hours incubation. After incubation periods, XTT cytotoxicity (Sartorius) solution containing 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide to measure mitochondrial activity was added in to each well and incubated for 4 h. Absorption of the developed orange color from dissolved tetrazolium salts was measured at 450 nm in the 96 well plate reader to calculate the percentage of viability of cells in contact with scaffold. The cell viability for the untreated control wells was presumed as 100%. Results and standard deviations were calculated from at least eight wells for each time period and t-test was applied (p < 0.05). Based on the percent viability of the cells, the biocompatibility level of the scaffold was evaluated. A biomaterial exerted cell viability higher than 70% is assumed as biocompatible according to ISO 10993-5. 24
Furthermore, the cell viability was evaluated by tryphan blue cell exclusion assay. The scaffold was cut in 25 mm diameter and each side of the biofiber prototype was sterilized under UV irradiation. The scaffold was put in to 35 mm diameter cell culture dish and conditioned in complete DMEM cell culture medium (with 10% FBS) in CO2 incubator overnight. L929 cells, previously cultured, were trypsinized, counted by tryphan blue exclusion assay, seeded on the biofiber prototype (5x105 cells), and incubated for 72 h. The control cells were not incubated with scaffold. The dead cells and living cells were counted by tryphan blue assay. The viability of the control cells was assumed as 100%.
Cell attachment
The oil-loaded double layered PCL/COL scaffold was cut in 25 mm diameter and each side of the biofiber prototype was sterilized under UV irradiation. The scaffold was put in to 35 mm diameter cell culture dish and conditioned in a complete DMEM cell culture medium (with high glucose, L-glutamine, and 10% FBS) in CO2 incubator overnight. The biofiber scaffold was rinsed in PBS solution. L929 cells were seeded on the biofiber prototype (5x105 cells) and incubated for 24 h for attachment. The feasibility of biofiber scaffold for cell attachment was evaluated by both FE-SEM imaging and by fluorescent microscopy, Rhodamine phalloidin, DAPI staining. The biofiber scaffold incubated with cells was rinsed twice with 0.1 M sodium cacodylate buffer (pH 7.4) and fixed by immersion in 3% glutaraldehyde (in 0.1 M sodium cacodylate) for 2 h at room temperature. The sample was rinsed three times in distilled water and dehydrated in 50%, 75%, 90%, and 100% (v/v) ethanol for 20 min, respectively. The specimen was treated with 100 μL HMDS and dried in a fume hood overnight. The dried sample was coated with iridium and observed under FE-SEM. The attachment of the cells onto the scaffold was also confirmed by DAPI staining. The culture medium was discarded after 24 h incubation and the DAPI stain solution (300 nM, 1 mL) was added to the petri dishes. The nuclei of the cells attached overnight onto the biofiber scaffold were imaged under a fluorescent microscope, upon 5 min incubation with DAPI. Cell elongation on the fiber mat was visualized by Rhodamine phalloidin staining according to manufacturer’s instructions (Thermo Fisher Scientific) under fluorescent microscope.
In vitro antibacterial effect
The layer which contains the oil blend is suspected of an antimicrobial effect. The disk diffusion method was applied to evaluate the antibacterial activity of the PCL/Oils layer against E. coli and S. aureus. Both sides of the disks (12 mm diameter) were sterilized under UV irradiation for 20 min. E. coli (Gram-negative), and S. aureus (Gram-positive) bacterial strains were grown in nutrient broth by incubating at 37°C for 24 h. Both bacterial colonies were equilibrated at 0.5 McFarland. The bacteria suspension was spread on the Muller-Hinton agar medium on the petri dishes. Then, the sterilized oil-loaded scaffold samples were placed on the bacteria culture and were incubated for 24 h at 37°C. The experiments were performed three times. The inhibition zone diameters around disks were measured, and mean and standard deviations from the mean values were calculated using Image J software.
Results
Properties of the oils used in biofiber scaffold
The fatty acid values, peroxide values, densities, refractive indices, phenolic contents, and antioxidant activities of the MC maceration oil and HP oil are summarized in Table 1.
Properties of the used oils for obtaining new scaffolds.
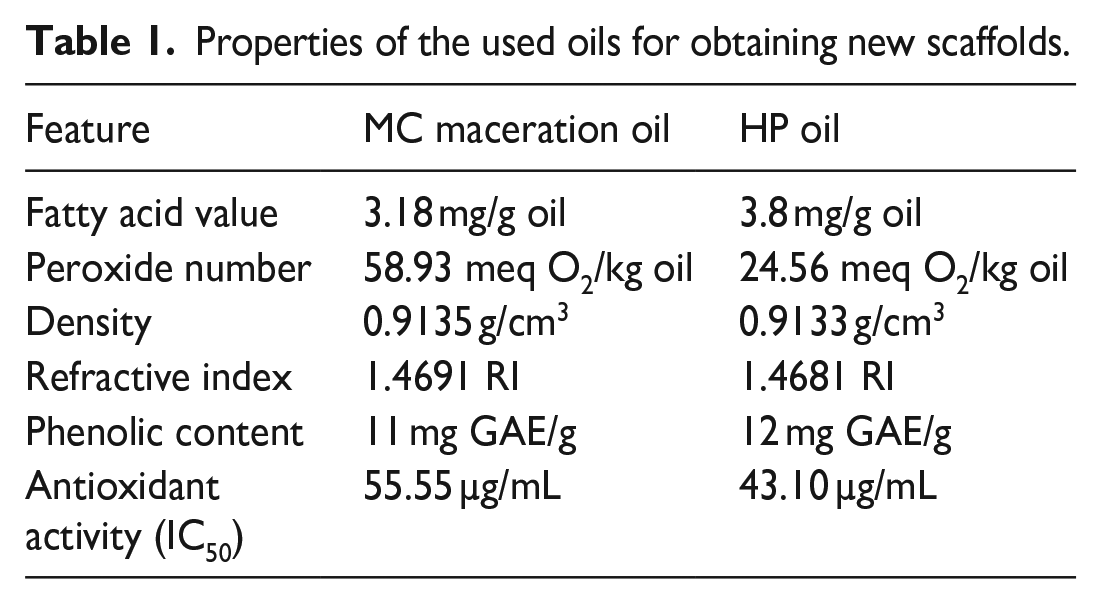
The features of the oils were found to be desirable according to the Turkish Food Codex. Acid values were calculated to be 3.18 mg/g oil and 3.8 mg/g oil for MC maceration oil and HP oil, respectively. The values are lower than 4.0 mg/g oil, which is the maximum limit specified in the Turkish Food Codex Communiqué on Oils Named by Plant (2012/29) Appendix-2. 25 Oxidation in oils results in undesirable taste and odor that adversely affects the nutrition and quality level of oils and causes toxicity. Oxidation in oils is measured by the peroxide number. Peroxide number is one of the most important quality criteria of vegetable oils. 26 Peroxide numbers of MC maceration oil and HP oil were found to be 58.93 meq O2/kg oil and 24.56 meq O2/kg oil respectively. Specific gravity is one of the physical parameters specific to the oils. It was found to be 0.9135 g/cm3 for MC maceration oil and 0.9133 g/cm3 for HP oil. The ratio of the speed of light passing through the medium (here it was water) and the speed of the light passing through the oil is called the index of refraction and depends on the physical structure of the oil. 27 Just like melting and boiling point, it is a constant specific to the substance type. The refractive indices of MC maceration oil, and HP oil were 1.4691, and 1.4681, respectively. Antioxidants are extremely beneficial for health, preventing or delaying oxidation processes and protecting cells from oxidative stress. 28 Phenolic compounds are secondary metabolites of plants and are described as free radical scavengers. The total phenolic content of the plant extracts was evaluated to determine the antioxidant capacity of the extract. 29 According to the total phenolic content analysis results, total phenolic values were calculated as 1.1 g GAE/100 g for MC maceration oil and 1.2 g GAE/100 g for HP oil. Free radical scavenging activities of L-ascorbic acid, MC, and HP oils were found to be 2.26, 55.55, and 43.10 µg/mL respectively. The oils exerted considerable antioxidant activities when compared to ascorbic acid which is known for high antioxidant activity. Bilia et al., and Günpınar et al. demonstrated that the wound healing effects of HP oil may be attributed to its antioxidant properties.30,31 Ilhan et al. declared that wounds healed in rats which may have resulted from the antioxidant properties of carotenoids and phytosterols in MC maceration oil. 32 The fatty acid content of MC, and HP oil was determined by gas chromatography and results are represented in Figure 2, Tables 2 and 3. The overall findings of the evaluation of MC and HP oil features indicate that the oils are feasible to be used in a biomaterial.
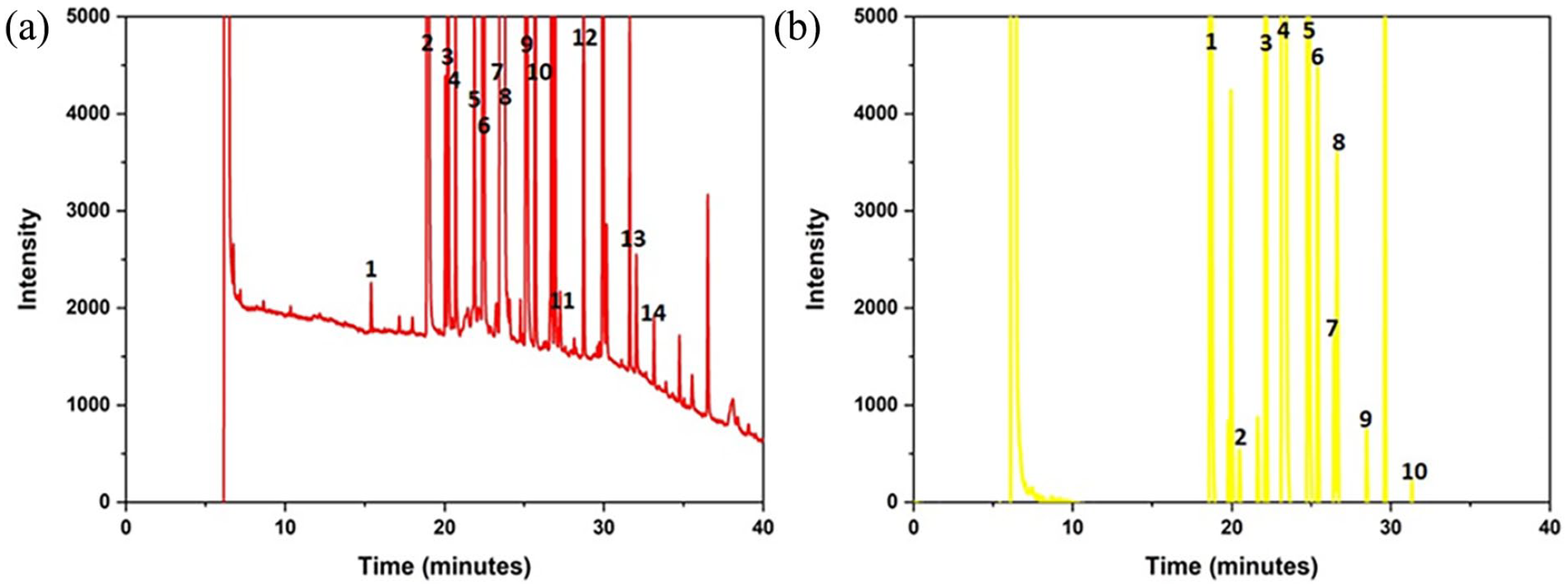
(a) MC maceration oil GC chromatogram and (b) HP oil GC chromatogram.
Composition of the Memoranda charantia maceration oil (GC measurements).
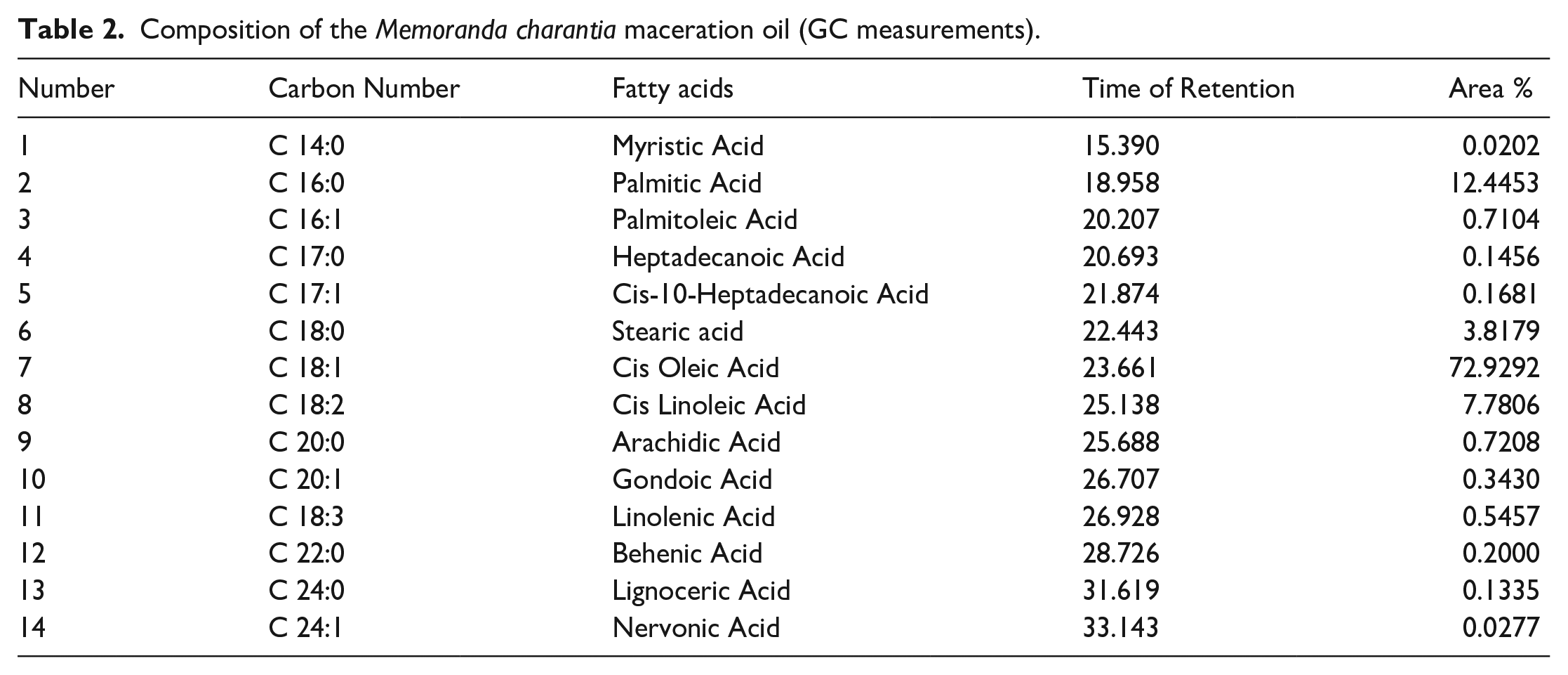
Composition of the Hypericum perforatum oil (GC measurements).
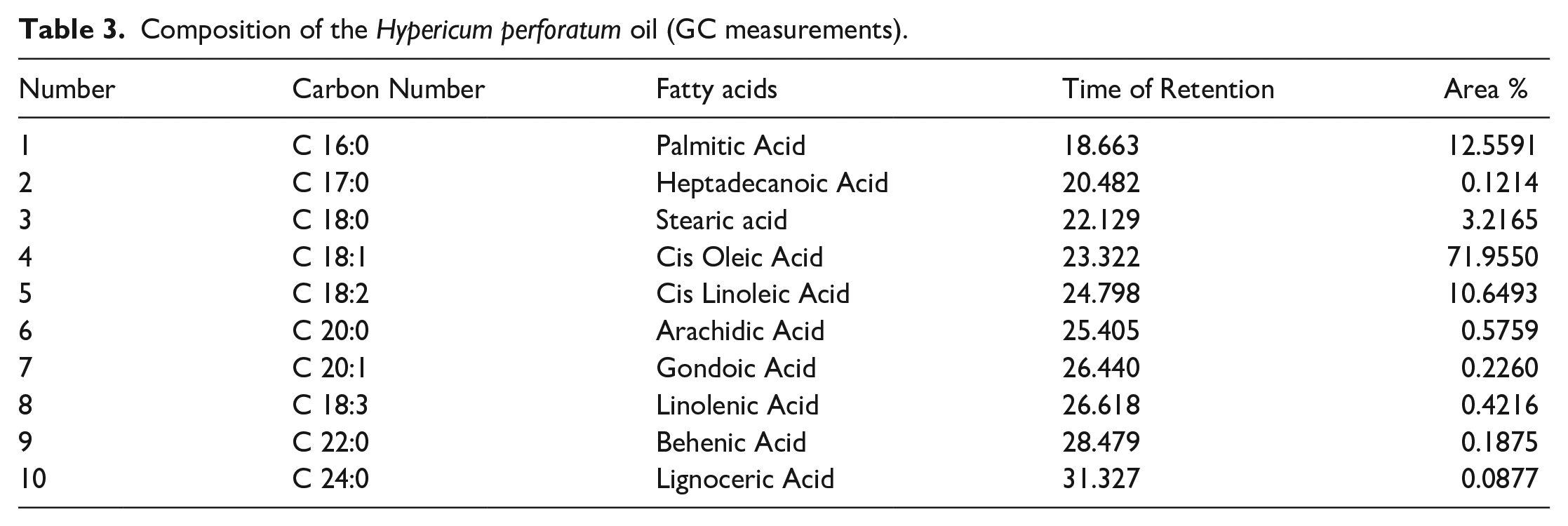
According to the GC analysis results, 14 different fatty acids were detected in MC maceration oil, and 10 different fatty acids were detected in HP oil. It was revealed that MC maceration oil and HP oil contains cis oleic acid, palmitic acid and cis linolenic acid, profoundly. The original GC chromatograms were presented as supplementary data S1 and S2.
Properties of the double layered scaffold prototype
Chemical structure (FTIR analysis)
MC and HP oils, PCL, COL and scaffold layers were analyzed through FTIR spectra. The FTIR spectrum plots of two different scaffold layers are shown in Figures 3 and 4. The characteristic peaks are from CH2 vibration at 732 cm−1, C–O–C symmetric bond at 961 cm−1, C–C bond at 1364 cm−1, C–O bond at 1140 cm−1, O–C–O bond at 1161 cm−1. The peaks 1239 and 1293 cm−1 represent the stretching of PCL in the crystal phase from C–C and C-O bonds respectively. The symmetrical deformation of the C–H bond at 1364 cm−1, C=O stretching vibration at 1720 cm−1, C–H2 symmetrical stretching vibration at 2865 cm−1, and C–H2 asymmetrical stretching vibration at 2942 cm−1.
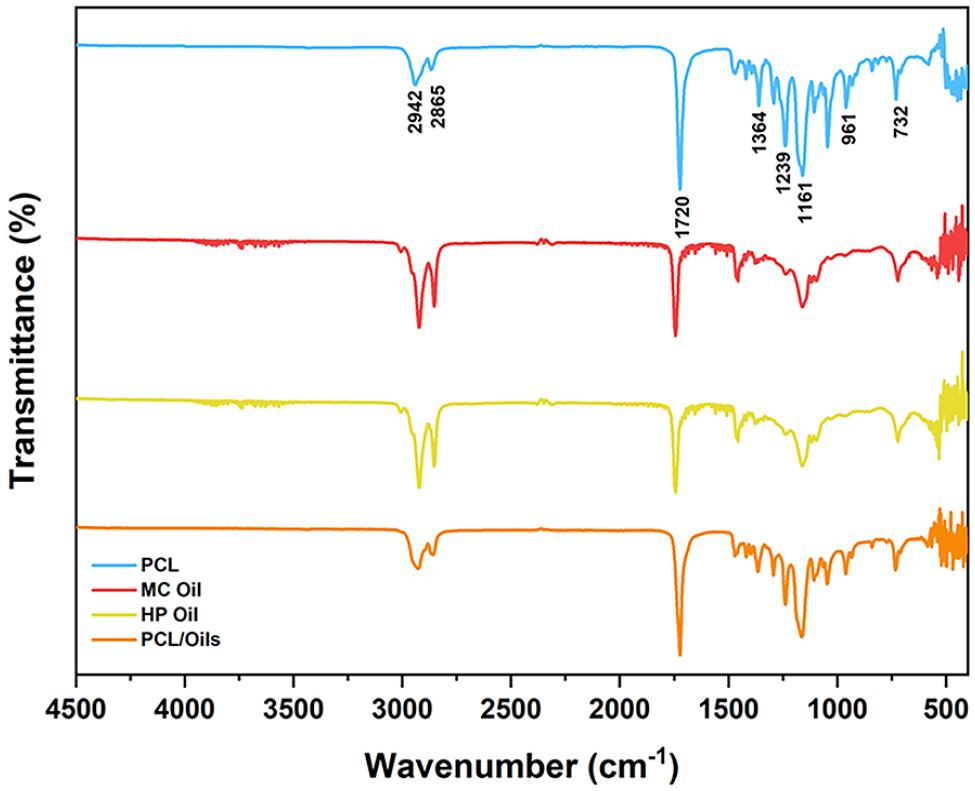
FTIR spectrum of the outer layer of biofiber scaffold prototype.
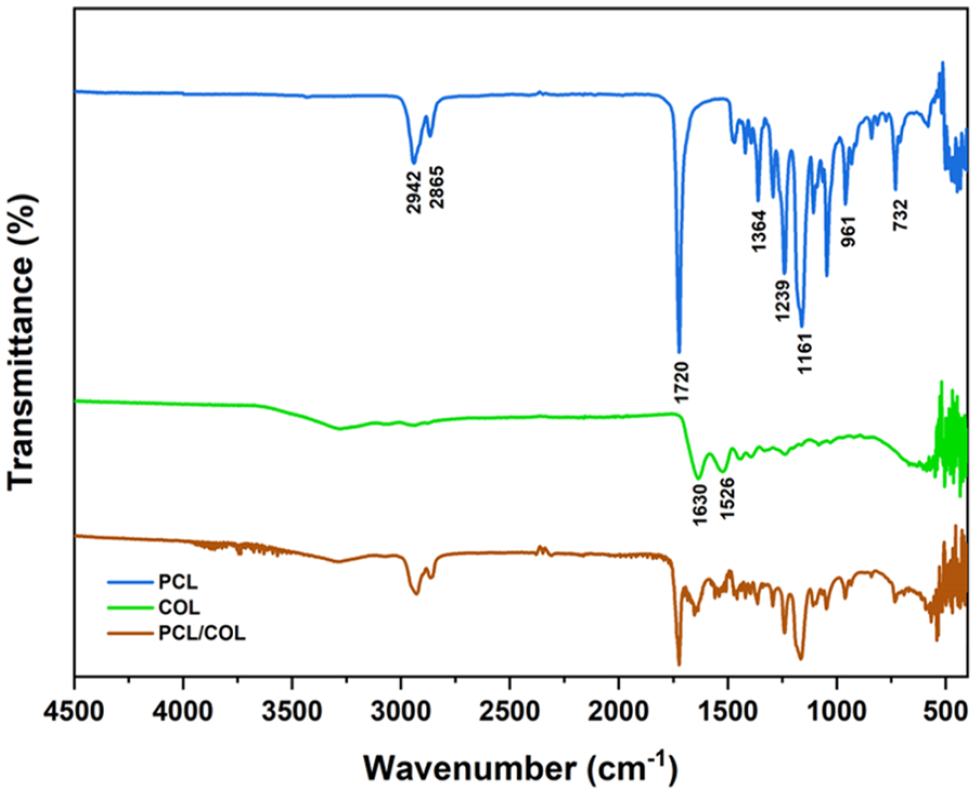
FTIR spectrum of inner layer of biofiber scaffold prototype.
When the FTIR spectra of PCL/Oils and that of bare oils are analyzed together in Figure 2, it is revealed that PCL/Oils biofiber exhibits a balanced structure without causing any degradation in functional groups of the oils.
In Figure 4, it is depicited that the transmittance percentage of PCL/COL biofibers decreased compared to PCL because the peaks of PCL polymer were suppressed by collagen. There was a clear difference in wave numbers between COL and PCL/COL due to the dominant peaks of PCL in the scaffold. The characteristic peaks of COL which are observed at 1630 and 1526 cm-1 verified the presence of COL in the PCL/COL scaffold. The characteristic peaks found in 1630, and 1526 cm-1 correspond to the peaks representing NH bending vibrations of amino groups in COL. 33
Morphological structure (FE-SEM)
The scaffold layers were monitored through FE-SEM imaging (Figure 5). Random and interconnected fibrous meshes were observed. SEM images illustrate that the biofiber mat layers were composed of beadles and smooth fibers. The diameters of PCL/COL fibers were ranging from 20 to 250 nm of which mean diameter was approximately 173 ± 54 nm. Mean biofiber size of PCL/Oils mat was 921 nm ± 328 nm which is calculated from the mean of fiber diameters ranging from 542 to 1580 nm. The frequency profiles of different sized fibers were demonstrated on the histogram plots for both layers (Figure 5(c) and (f)).
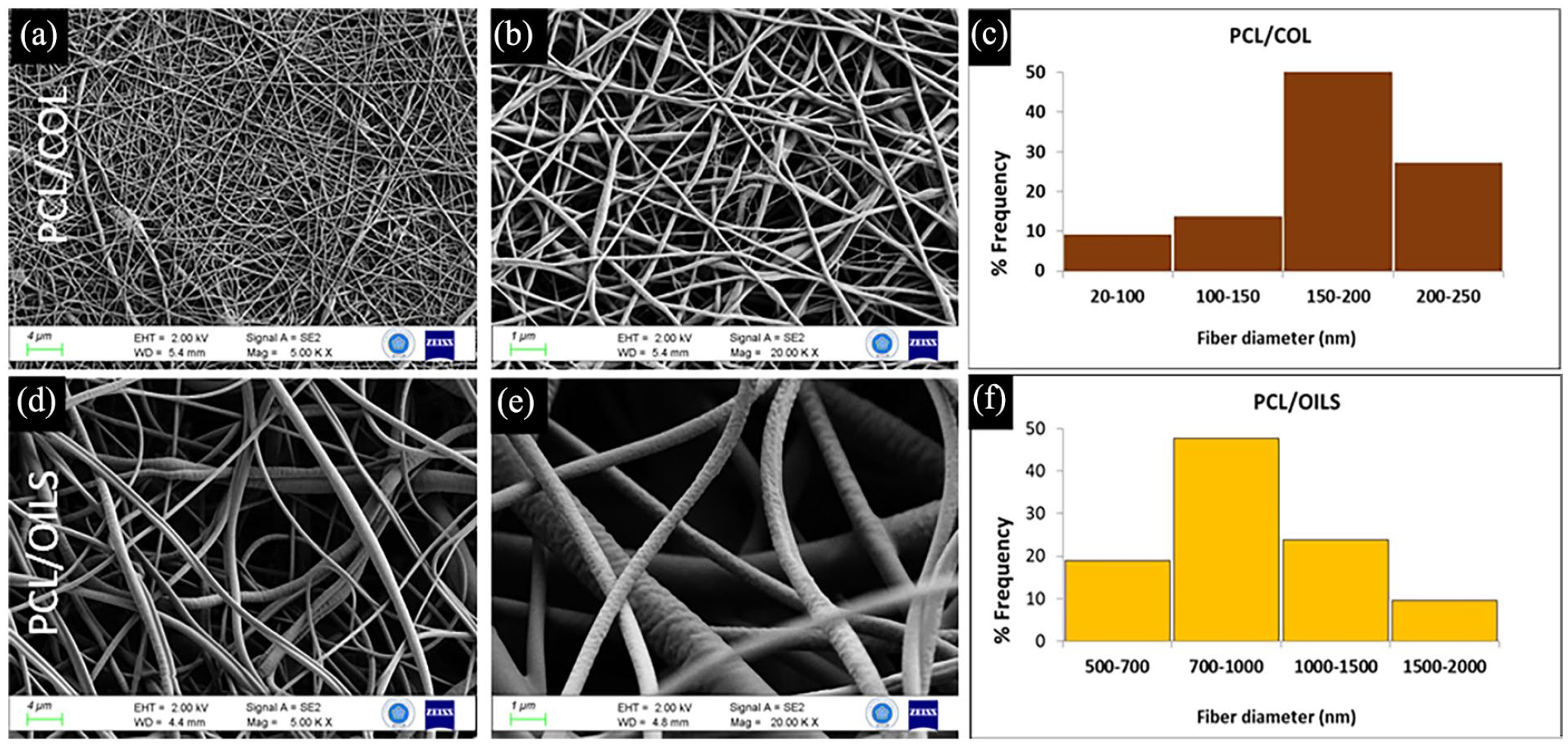
FE-SEM images of PCL/COL layer at (a) 4 kX and (b) 20 kX magnification and that of PCL/Oils layer at the (d) 4 kX and (e) 20 kX magnification. The distributions of fiber sizes are presented with the histogram graph for (c) PCL/COL and (f) PCL/OILS.
Thermal properties
The thermal stability of scaffold layers was inspected by TGA analysis. The thermal degradation curves of PCL/Oils, PCL/COL layers, and the double-layered scaffold prototype were demonstrated in Figure 6. Based on the graphs, the temperature range, thermal degradation range, and weight losses of the biofibers were calculated. Weight losses were realized at the temperature ranges of 350°C–440°C, 285°C–410°C, and 300°C–430°C for PCL/Oils, PCL/COL and scaffold prototype respectively. In fact, large scale thermal breakdown of the chains is thought to be the cause of the significant and dramatic weight loss of pure PCL that occurs in the temperature range of 300°C–425°C. 34 The decomposition temperature of COL was recorded as 275°C–310°C.35,36 According to our previous findings and literature, PCL experiences initial thermal degradation rate at temperatures between 360 and 380°C.5,37 Therefore, the PCL/COL biofibers show initial thermal decomposition temperature (285°C) between characteristic thermal decomposition temperature of PCL and COL.
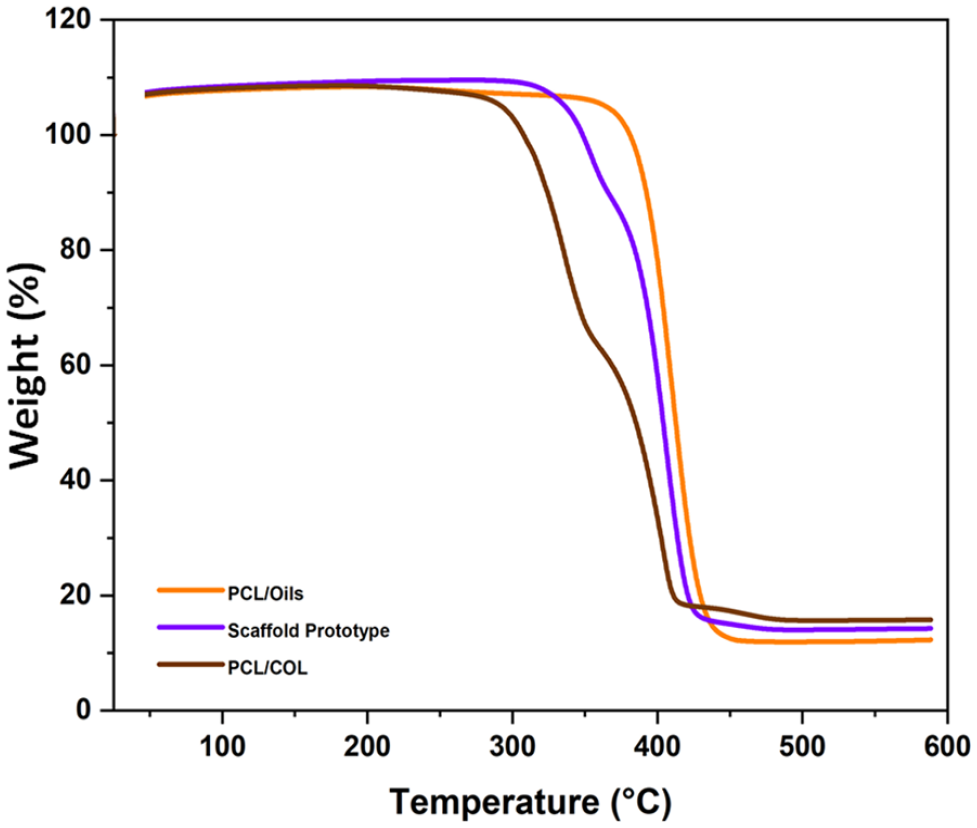
Thermogravimetric degradation curves of PCL/COL, PCL/Oils biofibers, and scaffold prototype.
Mechanical behavior
Tensile strength tests of the PCL/Oils and PCL/COL layers and scaffold prototype scaffold were carried out to unravel the mechanical properties of the functionalized biomaterials at a constant tensile speed. The curves of the variation of the tensile stress to strain are given in Figure 7. It is observed from these curves that the highest tensile strength was in the curve of the PCL/Oils layer sample as 6.40 MPa. The strength of the scaffold prototype was 5.98 MPa, while the strength of the PCL/COL fiber mat is 4.10 MPa. Furthermore, the tensile strength of the prototype was 45.85% higher than that of the PCL/COL fiber mat. The maximum strain values were found to be 142.26%, 72.10% and 159.35% for PCL/Oils, PCL/COL and scaffold prototype biofibers, respectively (Table 4).
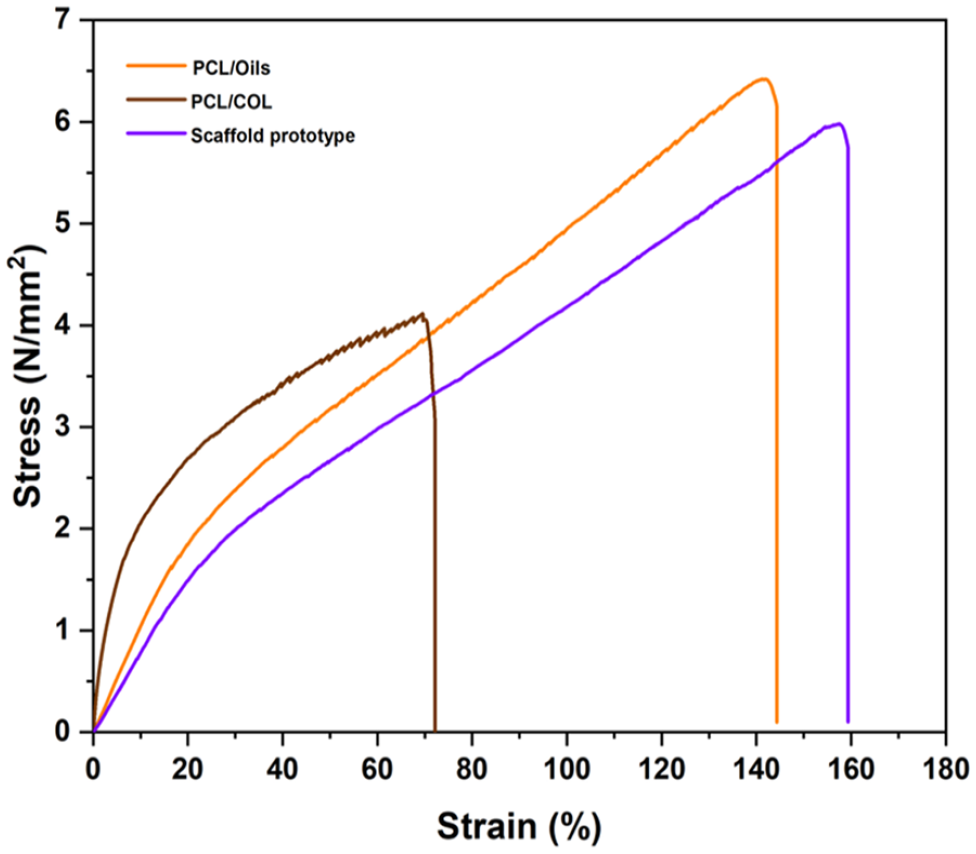
Stress-strain curves of biofibers.
Mean maximum stress, maximum percent elongation, and Young’s modulus values of biofibers.
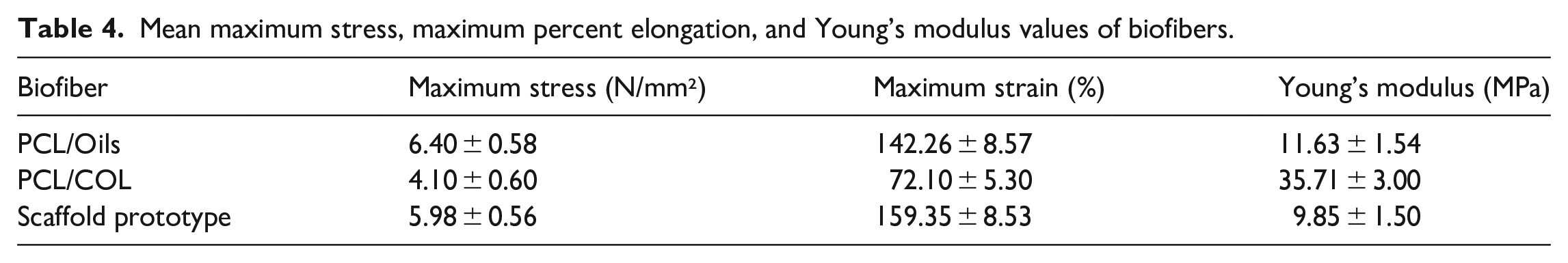
The Young’s Moduli of all types of fibers were calculated from the slope of the initial linear portion of the curves that are given in Table 4. Among them, the highest Young’s Modulus was obtained from PCL/COL fibers as 35.71 MPa. The Young’s modulus of the PCL/ COL layer is 207% greater than that of PCL/Oils biofibers and 263% greater than that of the scaffold prototype respectively. The addition of COL into PCL greatly increased the Young’s Modulus of PCL as we also stated earlier. 5 Collagen has a natural tendency to absorb fluids, increasing its modulus of elasticity. 38 Hereby, the scaffold prototype has enough mechanical stability to be used as PCL based functional scaffold component.
Biodegradability, wettability, and in vitro biodegradation
The results exhibited that PCL/Oils layer has a hydrophobic structure with an average contact angle of 97.8° (Figure 8). In our previous study, the contact angle of pure PCL biofiber was found to be 115.1° in the same experimental conditions. 5 PCL biofibers are hydrophobic in their pure form. Oils added to the PCL were found to slightly reduce the contact angle, but the material still retained its hydrophobic nature. The hydrophobic PCL/Oils layer of the scaffold could prevent contact between external factors (i.e. fluids, bacteria) and the wound by acting as a barrier.

Mean water-contact angles of (a) PCL/Oils and (b) PCL/COL biofiber mats.
PCL/COL inner layer has a hydrophilic structure with an average contact angle of 11.7°, owing to the hydrophilic behavior of collagen. Due to this feature, this layer which is desired to come into direct contact with the cells may ease the cell attachment and accelerate healing by triggering cell adhesion and cell migration.39–41
The weight loss of scaffold prototype layers was recorded by time in PBS for 18 weeks (Figure 9). The weight losses for PCL/Oils and PCL/COL layers were approximately 3.43% and 8.50% respectively. The results show that degradation rate of PCL/COL was about two folds more than that of PCL/Oils, which may be attributed the hydrophobic nature of oils blend.
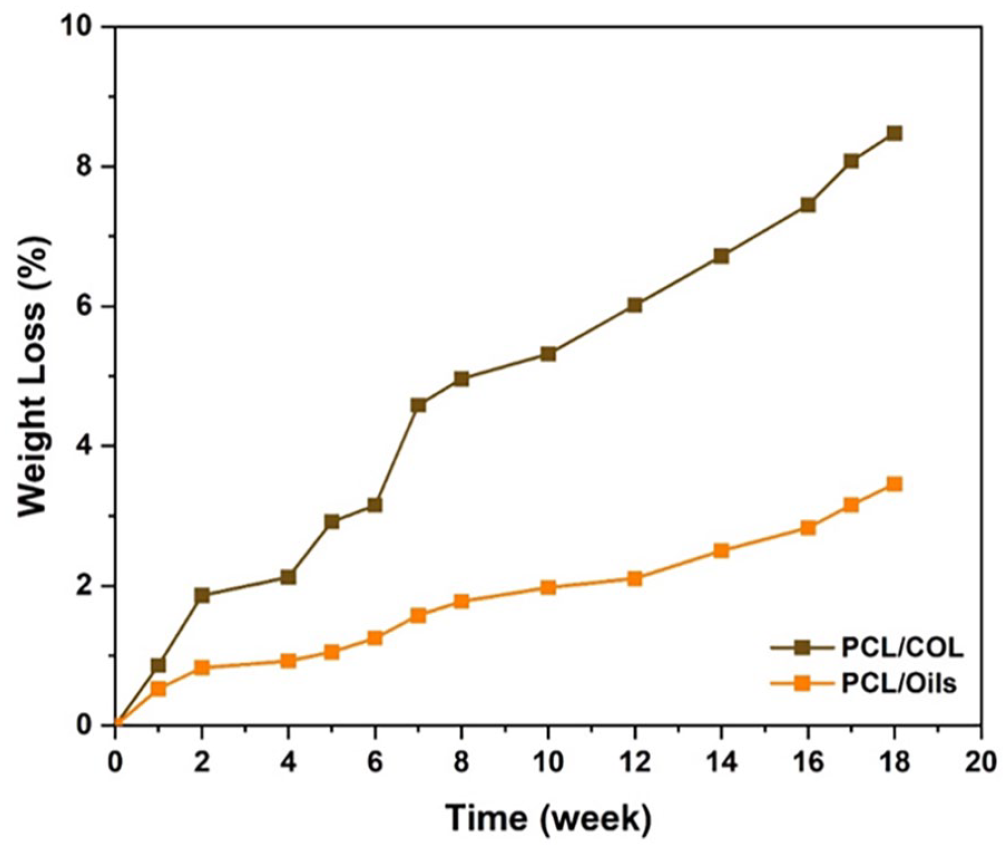
Weight loss due to in vitro degradation of PCL/Oils and PCL/COL layers in PBS.
Biocompatibility
Cell viability
L929 normal fibroblast cells were seeded on the scaffold prototype for 24, 72, and 144 h. L929 cells exhibited over 50% viability for contact periods and more than 70% viability after 72 h contact with scaffold prototype (Figure 10(a)). In this study, the viability of L929 cells were 60.4%, 80.2%, and 66.7% after 24, 72, and 144 h contact with the scaffold prototype respectively. This shows that the scaffold may be considered as biocompatible for 72 h contact. To confirm the viability directly by counting the viable cells, tryphan blue exclusion assay was conducted after 72 h incubation of the cells with the scaffold. The results indicated that the cell viability was 92% (92% ± 2.50%) when compared to untreated cells. The untreated control cells and the cells incubated for 72 h in culture plate in contact with scaffold were visualized under inverted light microscope. The control cells and the cells incubated 3 days in the extract of scaffold exhibited the similar confluency in the culture dishes (Figure 10(b) and (c)).
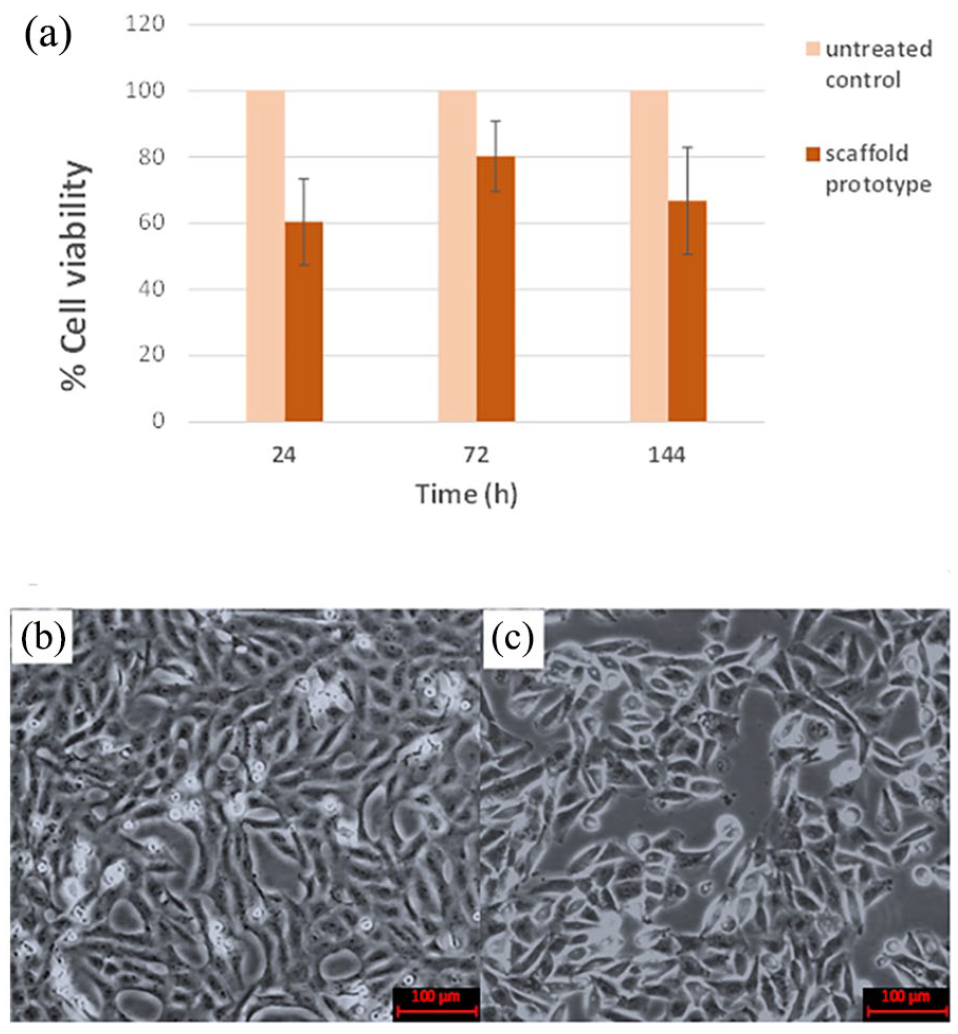
(a) Cell viability analysis upon contact with scaffold prototype (Exposure of the cells with biofiber mat prototype for 24, 72, and 144 h did not exert a significant difference between the cell proliferation of the groups.), (b) Untreated control cells, and (c) cells incubated in a medium in contact with scaffold and its extracts for 72 h.
Cell attachment
When the attachment behavior of the cells on the scaffold prototype model is evaluated, it was observed that fibroblast cells attached on and under the fibers of the mat prototype and cytoskeleton elongation was observed on the mat (Figure 11(a) and (b)). Chan et al. presented that the extent of cell elongation or spreading provides information on the biocompatibility of the scaffold. 42 In our work, the fibroblasts were able to live by elongating on and attaching to and under the fibers, by using them as scaffold and creating an extracellular matrix space for themselves. The viability of the attached cells on biofiber mat was confirmed with DAPI staining under the fluorescent microscope. The scaffold removed from the culture plate was visualized between the slide and coverslip. The nuclei of the attached cells were obviously detected under the microscope (Figure 11(c)–(e)).

The attachment behavior of L929 cells on scaffold prototype was visualized: (a) by fluorescent Rhodamine-phalloidin staining of cytoskeleton (100X) and (b) with FESEM by fixation of cells on to the fibers, 1 kX magnification. The living L929 cells were DAPI stained and visualized under fluorescent microscope. The specimens were: (c) untreated control cells, (d) cells treated with culture medium incubated with scaffold prototype (e) cells attached on the scaffold prototype (40X).
In vitro antibacterial effect
The antibacterial activity of PCL/Oils biofiber samples was evaluated using the disk diffusion method to demonstrate any effect of MC and HP oils on bacterial growth (Figure 12). The samples from the outer layer of the scaffold model exhibited 14.05 ± 0.05 mm and 12.70 ± 0.04 mm mean inhibition zone for E. coli, and S. aureus, respectively. The studies that examine the antibacterial activity of MC oil, have reported that MC oil has significant antibacterial effect on E. coli than other pathogens included S. aureus.43,44 The anti-bacterial property of HP oil was verified in several studies as well.18,45,46
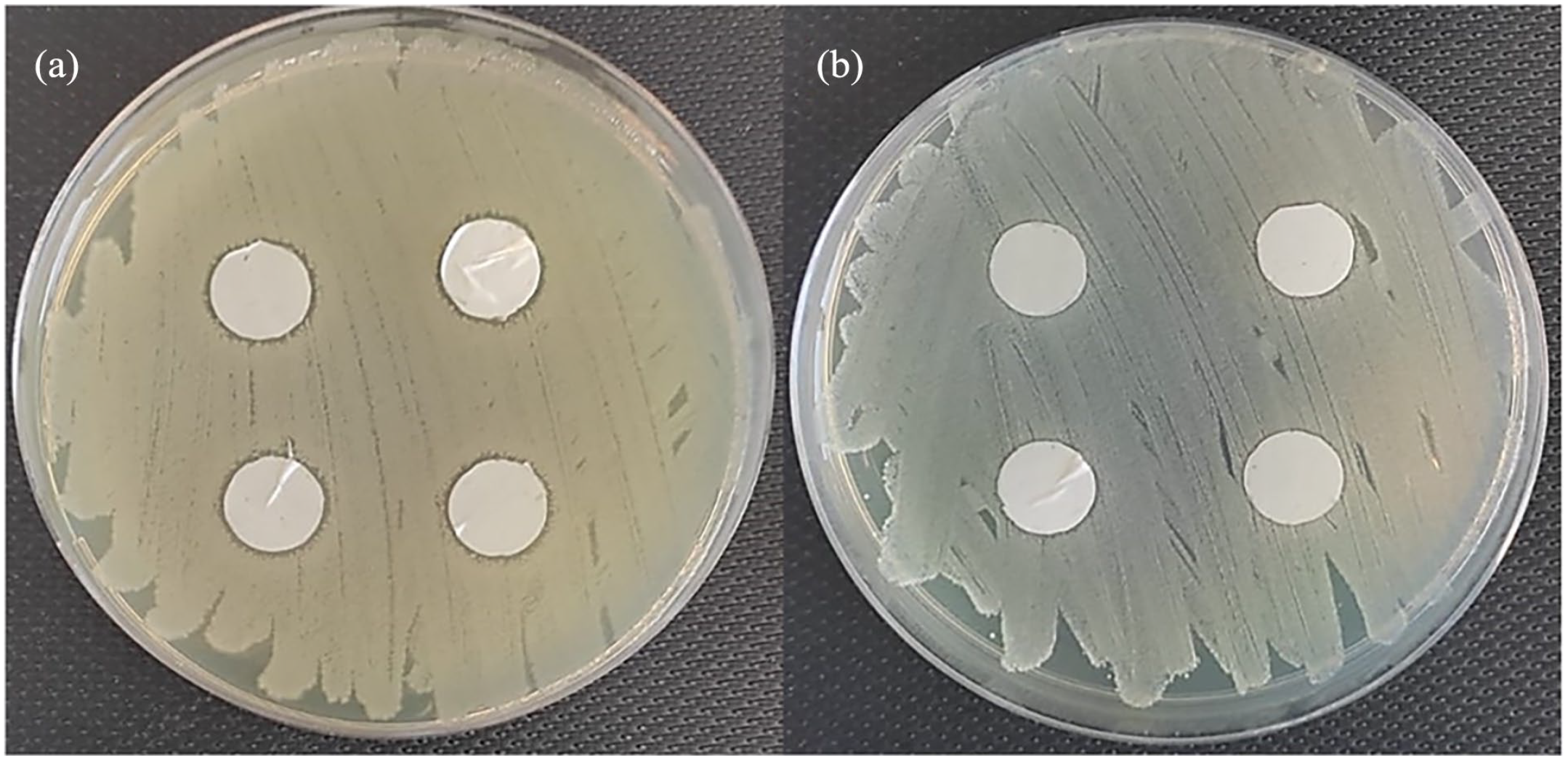
Antibacterial activity of PCL/Oils biofibers against (a) E. coli and (b) S. aureus.
Discussion
In this study, the double-layered electrospun biofiber scaffold that assemble the outer layer consisting of MC and HP oils loaded into PCL, and the inner layer including PCL and collagen constituting fibrous mat was fabricated intended to be used in tissue engineering applications that is, as scaffold. The oils and fibrous layers were characterized and proposed to have feasible surface morphology, mechanical properties, antioxidant properties and biological functions.
The acid values, peroxide numbers, refractive indices and specific gravities of the MC and HP oils were found to be desirable according to the Turkish Food Codex. 25 Antioxidants are extremely beneficial for health, preventing or delaying oxidation processes and protecting cells from oxidative stress. 28 Phenolic compounds are secondary metabolites of plants and are described as free radical scavengers. Total phenolic contents of MC and HP oils were found to be 11 mg/g and 12 mg/g GAE respectively. The HP and MC are important medicinal plants and the antioxidant activities of the extracts, obtained by various solvent extraction methods, were extensively worked on.30–32 Bayram et al. declared that total phenolic content of HP, extracted by various deep eutectic solvents, varied between 3.10 and 16.64 mg GAE/g. 47 Furthermore, in our work we found that the oils exhibited considerable antioxidant activities when compared to ascorbic acid which is known for high antioxidant activity. Bilia et al. and Gülpınar et al. demonstrated that the wound-healing effects of HP oil may be attributed to its antioxidant properties.30,31 Ilhan et al. declared that wounds healed in rats which may have resulted from the antioxidant properties of carotenoids and phytosterols in MC maceration oil. 32 According to our understanding, the combination of MC and HP oils is used for the first time in a biofiber mat. Antioxidant qualities are advantageous because they lessen the impact of oxidative stress on cells. Due to the characteristics of oils, it may be argued that the scaffold prototype has antioxidant action. A biomaterial for tissue engineering is desired to have this characteristic.
PCL/Oils layer includes MC and HP oils. The fatty acid contents of the oils were analyzed by gas chromatography and found to be constituting considerable amount of polyunsaturated fatty acids (PUFAs). Linoleic acid (n-6) and α-linolenic acid (n-3) protect and mature the outermost layer of the epidermis, the stratum corneum and inhibit the release of proinflammatory eicosanoids and cytokines. PUFAs are recommended to be used to treat skin diseases such as psoriasis. 48 Mohanty et al. declared that topical usage of curcumin-loaded oleic acid based polymeric bandages on rat models had sustained activity on wound healing. 49 In concordance with literature, it can be deduced from our results that the constituents of biofiber mat prototype may have beneficial effects on skin health and regeneration. PCL is a biocompatible polymer which is also a good carrier of drugs and bioactive molecules for wound dressings, that can be one of the reasons that encapsulation of MC and HP oils in a scaffold may allow controlled release of PUFAs and other antioxidant bioactive molecules to enable the scaffold to be functional biomaterial. The MC and HP oils utilized in the study added a new feature to the scaffold prototype in this situation. Recently, nanofiber biomaterials have been investigated as an excellent tool for conjugating bioactive molecules and acting as controlled drug delivery systems provided that release of molecules over a long time and can surmount the limitations of conventional treatments. 12
Furthermore, the scaffold layers were analyzed by FTIR spectroscopy. It is obviously seen that the incorporation of oils to the PCL biofiber didn’t cause any shifting in wavenumbers of the FTIR spectrum of PCL, just the transmittance peaks exhibited small differences in the same wavenumbers. For instance, in 1161 cm-1 the transmittance changed in PCL/Oils, the oils don’t have any functional group in the same wave number. This indicates that the oils contribute to the negligible shift in the peaks of the PCL FTIR spectrum without causing any chemical changes in the functional groups of the PCL structure which proves the physical loading of oils into the PCL polymeric matrix. Furthermore, Azzazy et al. previously claimed that FTIR shifts may be negligible in essential oil extract incorporated PLGA/PCL fibers due to the physical loading of plant oil within the polymeric matrix. 50 Moreover, FTIR results verified that the essential oil had the opportunity to interact with the carbonyl groups on PCL chains via hydrogen bonding. PCL/COL electrospun fiber mat was analyzed and FTIR peaks belonging to collagen (1630, 1526 cm-1) were observed, very similar to the findings of Gümüşderelioğlu et al. 51
The fiber sizes were examined by FE-SEM analysis. Both layers were fabricated from bead-free fibers. The SEM views reveal that the incorporation of oils into the fibers of the PCL/Oils layer resulted in a larger fiber diameter with respect to that of PCL/COL. The increase in fiber diameter may be due to the deposition of oils blend at the outer layer of fiber filaments as previously declared by El-Naggar et al. 52 Additionally, this may be attributed to the alteration in the solution characteristics caused by fatty acid inclusion and the oil interactions with the polymer chains that influenced the chain entanglements, responsible for fiber formation. As depicted in FE-SEM results the oils blend may entirely have been deposited on the fibers, forming widening in diameters.
Weight losses upon the temperature increase were inspected and found to be approximately 87.73%, 84.2%, and 85.7% for PCL/Oils, PCL/COL, and scaffold prototype respectively. The scaffold prototype exhibited a thermal degradation rate between those two layers, exhibiting the characteristics of both COL and oils. PCL/Oils layer started to decompose at higher temperatures than the other samples and it showed that PCL/Oils have the highest thermal stability. The sample with the lowest thermal stability was the PCL/COL because it started to degrade at a lower temperature compared to the other two samples. Furthermore, it is shown previously that the thermal stability of the polymer was increased with the addition of oils into polymer-based materials. 53 This rise in PCL/Oil degradation temperatures may be attributed to the barrier effect that polymer chains have on MC and HP molecules, which makes it more difficult to diffuse for volatile compounds, and this fact results in raises in the thermal stability of composites. 54 This is due to the cross-linking bonds that form between the components of fibers limiting the mobility of polymer chains and improving thermal stability. 38
When the mechanical behaviors of the layers were examined, it was revealed that the PCL/Oils biofiber layer had the maximum strength because the molecules that make up oil may have been strongly mechanically and physically connected with the thermoplastic PCL. The reason why the strength value of PCL/COL fiber mat is lower than PCL/Oils biofibers is due to the fact that collagen cannot form a good binding in thermoplastic PCL. Thus, it damaged the structural integrity of the PCL fibers, causing it to demonstrate lower strength.55,56 Although COL addition significantly decreased the strain value (Figure 7), it increased Young’s modulus by 207% in the elastic region compared to the PCL/Oils layer. The fiber diameter affects the tensile strength, especially for fibers with diameters below 700 nm, which can be explained by the fact that thinner fibers indicate greater Young’s modulus than thicker fibers.57,58 Therefore, the higher Young’s modulus in the PCL/COL layer can be attributed to the PCL/Oils layer’s mean fiber diameter (513 nm) being up to 3.79-folds higher than that of PCL/COL (135.3 nm) layer. In the elastic region of stress-strain curves, Young’s modulus and yield strength of the biofibers in PCL/COL increase as the fiber diameter decrease in comparison with PCL/Oils. Moreover, the ductility is decreased as a result of the decreased fiber diameter. 58 The lower ductility of the PCL/COL fibers compared to the PCL/Oils fibers can be explained by the fact that the PCL/COL fiber mat is composed of fibers with smaller diameter, and the COL reduces the continuity of the molecular chain of PCL polymer. The higher Young’s modulus and the decrease in strain value upon COL integration to the PCL were previously exhibited by Prabhakaran et al. study as well. 59 Moreover, aforementioned study confirms that the PCL/COL fibers (245 ± 80 nm) have a smaller mean diameter of fibers when compared with the mean diameter of PCL fibers (350 ± 83 nm).
Regarding biodegradability studies, the hydrophilic character of COL, which is advantageous for cell attachment, may be responsible for the higher weight loss on PCL/COL composite fiber layer. The reason for that enhanced biodegradation rate may be due to the disrupted polymer continuity of PCL by collagen causing to form gaps in the polymers. 60 In contrast, the outer layer of PCL/Oils showed less weight loss. In our previous study conducted at the same conditions, the 10-week degradation rate of the PCL fiber mat was found to be approximately 0.3%. 5 The results of this investigation show that the addition of oils increased the rate of pure PCL fiber breakdown. The inclusion of -OH ions and polar phytochemicals in MC and HP oils may provide an explanation for the increase in the biodegradation rate. 61 However, compared to PCL/COL layer, the hydrophobic feature of oils resulted in a reduced biodegradation rate. 62 The prototype layers showed an appropriate degradation profile that allowed designers of biomaterials for tissue engineering to get around certain difficult design constraints. It may be deduced that; the two-layer scaffold may degrade at a pace no faster than PCL/Oils layer.
Regarding the biocompatibility results, the in vitro biological characteristics of the scaffold exhibited favorable cell viability and attachment behavior of fibroblast cells. Going on to analyses of cell biology, it may be suggested that the biofiber scaffold prototype is biocompatible with healthy cells and enables cell adhesion. The antimicrobial activity of phytochemicals results from bacterial membrane damage, increased cell permeability, coagulation of bacterial proteins, and inhibition of ATP production, caused by phytochemicals. The oil-loaded layer was inspected for the possible antibacterial effect. The fibrous PCL/Oils layer has notable inhibitory zones, with mean inhibition zones for MC oil of 14.05 mm and HP oil of 12.70 mm. Zubair et al. published that 62.5 mg/mL MC seed oil exerted 12 mm inhibition zone against both S. aureus and E. coli. 44 In another study, it was found that HP extract inhibited the viability of S. aureus and E. coli with a maximum 21 mm and 19 mm zone diameters. 47 The differences in antibacterial activities may be resulted from the effectiveness of extraction method and source plant organ (i.e. seed, fruit, root). In this way, if the double-layered scaffold is intended to be used as a wound dressing, bacterial infections caused by external sources can be prevented thanks to the scaffold’s new oil-containing outer layer’s antibacterial properties.
Conclusions
The fabrication of a novel double-layered scaffold was described herein. As far as we know, research is being done on the development of scaffolds with superior and bioinspired features that support cell survival and tissue engineering. The anti-oxidant and anti-bacterial properties of the oils, as well as their high PUFA content, added value and uniqueness to the double-layered scaffold. Moreover, the PCL/Oil layer provided an opportunity to modify the mechanical and physical characteristics of the double-layered scaffold. To conclude, this novel double-layered fibrous scaffold prototype, with its applicable features, may be proposed as a candidate biomaterial for utilization as a scaffold for tissue engineering. Future evaluations of the prototype should include functional examinations on 3D wound models.
Supplemental Material
sj-pdf-1-jbf-10.1177_22808000231221067 – Supplemental material for In vitro assessment of Momordica charantia/Hypericum perforatum oils loaded PCL/Collagen fibers: Novel scaffold for tissue engineering
Supplemental material, sj-pdf-1-jbf-10.1177_22808000231221067 for In vitro assessment of Momordica charantia/Hypericum perforatum oils loaded PCL/Collagen fibers: Novel scaffold for tissue engineering by Emre Fatih Ediz, Cansu Güneş, Meltem Demirel Kars and Ahmet Avcı in Journal of Applied Biomaterials & Functional Materials
Supplemental Material
sj-pdf-2-jbf-10.1177_22808000231221067 – Supplemental material for In vitro assessment of Momordica charantia/Hypericum perforatum oils loaded PCL/Collagen fibers: Novel scaffold for tissue engineering
Supplemental material, sj-pdf-2-jbf-10.1177_22808000231221067 for In vitro assessment of Momordica charantia/Hypericum perforatum oils loaded PCL/Collagen fibers: Novel scaffold for tissue engineering by Emre Fatih Ediz, Cansu Güneş, Meltem Demirel Kars and Ahmet Avcı in Journal of Applied Biomaterials & Functional Materials
Footnotes
Acknowledgements
We gratefully acknowledge Zade & Zade Vital İbn-i Sina R&D Center for their support.
Contributorship
Conceptualization, E.F.E. and M.D.K.; methodology, M.D.K. and A.A.; software, E.F.E and C.G.; formal analysis, E.F.E, C.G and M.D.K.; investigation, E.F.E, C.G and M.D.K.; resources, M.D.K. and A.A.; data curation, E.F.E, C.G and M.D.K.; writing – original draft preparation, E.F.E, C.G and M.D.K.; writing – review and editing, M.D.K.; visualization, E.F.E. and C.G.; supervision, M.D.K.; project administration, M.D.K. and A.A.; funding acquisition, M.D.K. All authors have read and agreed to the published version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by the Horizon Europe project REGENEU with grant number 101079123. TUBITAK 2244 University Industry Collaboration Project (119C100) is also acknowledged for the PhD scholarship to the first author.
Guarantor
MDK.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.