Abstract
Purpose
The aim of this study was to investigate the effects of laser therapy and Biosilicate® on the biomechanical properties of bone callus in osteopenic rats.
Methods
Fifty female Wistar rats were equally divided into 5 groups (n=10/group): osteopenic rats with intact tibiae (SC); osteopenic rats with unfilled and untreated tibial bone defects (OC); osteopenic rats whose bone defects were treated with Biosilicate® (B); osteopenic rats whose bone defects were treated with 830-nm laser, at 120 J/cm2 (L120) and osteopenic rats whose bone defects were treated with Biosilicate® and 830-nm laser, at 120 J/cm2 (BL120). Ovariectomy (OVX) was used to induce osteopenia. A non-critical bone defect was created on the tibia of the osteopenic animals 8 weeks after OVX. In Biosilicate® groups, bone defects were completely filled with the biomaterial. For the laser therapy, an 830-nm laser, 120 J/cm2 was used. On day 14 postsurgery, rats were euthanized, and tibiae were removed for biomechanical analysis.
Results
Maximal load and energy absorption were higher in groups B and BL120, according to the indentation test. Animals submitted to low-level laser therapy (LLLT) did not show any significant biomechanical improvement, but the association between Biosilicate® and LLLT was shown to be efficient to enhance callus biomechanical properties. Conversely, no differences were found between study groups in the bending test.
Conclusions
Biosilicate® alone or in association with low level laser therapy improves biomechanical properties of tibial bone callus in osteopenic rats.
Introduction
Osteoporosis is a systemic skeletal disorder characterized by low bone mass, deterioration of the microarchitecture of bone tissue and increased susceptibility to fragility fractures (1). Osteoporosis prevalence and fracture complications, such as pain and disability, depression, dependency and deterioration of the quality of life (2), are rapidly increasing worldwide and represent a severe health problem among elderly people (3).
In this context, there is a critical need for technologies that are able to enhance osteoporotic fracture healing. Our research group has developed a novel fully crystallized bioactive glass-ceramic of the quaternary P2O5-Na2O-CaO-SiO2 system (Biosilicate®, patent application WO 2004/074199). Biosilicate® has shown a stimulatory effect on bone cell metabolism (4). By comparing the growth of osteogenic cells on Biosilicate® and Bioglass® 45S5 disks (gold standard bioactive material) for a period of up to 17 days, Moura et al (4) found that Biosilicate® supported larger areas of calcified matrix, indicating that the full crystallization of bioactive glasses in a range of compositions of the system P2O5-Na2O-CaO-SiO2 may promote enhancement of in vitro bone-like tissue formation. Moreover, Granito et al (5) showed that the fully crystallized Biosilicate® also has good in vivo bone-forming and bone-bonding properties, supporting higher bone formation than the gold-standard Bioglass® 45S5.
Similarly, a significant body of evidence has been now accumulated demonstrating that low-level laser therapy (LLLT) also has a positive effect on bone tissue metabolism and on fracture consolidation (6-8). When a laser is applied to biological tissue, the light is absorbed by chromophore photoreceptors located in the cells. Once absorbed, the light can modulate cell biochemical reactions and stimulate mitochondrial respiration, with the production of molecular oxygen and adenosine triphosphate (ATP) (9, 10). These effects are known to increase the synthesis of DNA, RNA and cell-cycle regulatory proteins, therefore promoting cell proliferation (10). Different in vitro and in vivo studies have shown that LLLT was capable of increasing mitochondrial activity (11), bone nodule formation (12), bone sialoprotein and osteopontin expression and alkaline phosphatase (ALP) activity (13). Additionally, LLLT has been demonstrated to be able to accelerate the process of fracture repair in rabbits and rats, increasing the callus volume and bone mineral density (6, 14).
Other studies have investigated the effects of LLLT in animal models of bone disease. Our group showed that LLLT increased femoral strength, calcium content and bone density in osteopenic rats (15, 16). Moreover, Diniz et al (17) demonstrated that the association of bisphosphonate and laser can also increase the vertebral trabecular bone volume in osteopenic rats.
Despite the positive effects of Biosilicate® and LLLT on bone regeneration, it is still necessary to investigate in vivo biomechanical effects and dose-response characteristics of these 2 therapeutic modalities before they can be used with confidence to improve fracture healing. Based on the effects of LLLT or Biosilicate® used as single therapies, we hypothesized that the association of these 2 therapies could increase the strength of the tibial callus in osteopenic rats. In this context, this study aimed to investigate the effects of Biosilicate® and 830-nm laser on the biomechanical properties of tibial bone defects in osteopenic rats.
Materials and Methods
Biomaterial
High-purity silica and reagent grade calcium carbonate, sodium carbonate and sodium phosphate were used to obtain glass compositions: Biosilicate® parent glass. The chemicals were weighed and mixed for 30 minutes in a polyethylene bottle. Premixed batches were melted in a Pt crucible at a temperature range of 1250°C to 1380°C for 3 hours in an electric furnace (Rapid Temp 1710 BL; CM Furnaces Inc., Bloomfield, NJ, USA) at the Vitreous Materials Laboratory of the Federal University of São Carlos (São Carlos, SP, Brazil). Samples were cast into a 10×30 mm cylindrical graphite mold and annealed at 460°C for 5 hours. To obtain the fully crystallized Biosilicate® glass-ceramic, Biosilicate® parent glass cylinders underwent cycles of thermal treatment to promote their crystallization. The first thermal cycle was performed at a relatively low temperature, just above the glass transition temperature to promote volumetric nucleation of crystals. Afterward, the nucleated samples were submitted to further treatment at about 100oC above the nucleation temperatures. The detailed compositions and thermal treatment schedules to obtain the Biosilicate® glass-ceramic are described in the patent WO 2004/074199.
Surgical procedures
This study was conducted in accordance with the
Fifty female Wistar rats (12 weeks old, 250 g body weight) were submitted to ovariectomy (OVX) to induce osteopenia. OVX is the most common experimental model used for experimental osteoporosis research (15, 16, 18-20). This model mimics the bone loss and impaired fracture repair prevalent in postmenopausal women who are estrogen deficient and prone to osteoporotic fractures. Surgery was performed via bilateral translumbar incisions, under general anesthesia induced by intraperitoneal (IP) injection of Xilazin (Syntec®, 20 mg/kg, IP) and ketamine (Agener®, at 40 mg/kg, IP). The uterine tubes were ligated (catgut 4.0), the ovaries were removed and the incisions were closed (catgut 3.0). After surgery, all animals were conditioned during 8 weeks with the purpose of inducing osteopenia (15, 16, 18, 19).
Eight weeks after OVX, animals were randomly divided into 5 groups (n=10): osteopenic intact control (SC, intact osteopenic tibiae); osteopenic bone defect control (OC, osteopenic tibiae that were submitted to the surgical creation of bone defects without any treatment); osteopenic bone defect treated with Biosilicate® (B); osteopenic bone defect treated with laser at 120 J/cm2 (L120); and osteopenic bone defect treated with both Biosilicate® and laser at 120 J/cm2 (BL120).
A unicortical non-critical-sized bone defect was surgically created at the upper third of both tibiae (10 mm distal of the knee joint). Surgery was performed under sterile conditions, and general anesthesia induced by IP injection of Xilazin (Syntec®, 20 mg/kg, IP) and ketamine (Agener®, at 40 mg/kg, IP). The medial compartment of the tibia was exposed through a longitudinal incision on the shaved skin and muscle tissue. A standardized 2.0-mm-diameter unicortical bone defect was created by using a motorized drill (13,560 rpm) under copious irrigation with saline solution, exposing the medullary cavity, without affecting the opposite cortex. In Biosilicate® treated animals, the cavities were completely filled with the biomaterial. The cutaneous flap was replaced and sutured with resorbable polyglactin, and the skin was disinfected with povidone-iodine. Heating lamps were used to prevent hypothermia in the postoperative period, and animals were supervised until full anesthesia recovery. Nonsteroidal antiinflammatory drugs (NSAIDs) were for analgesia. The health status of the rats was monitored daily.
Low-level laser therapy
A low-energy GaAlAs (Teralaser; DMC® São Carlos, SP, Brazil), 830 nm, CW, 0.028 cm2 beam, 100 W/cm2, 120 J/cm2 (3.4 J) with an irradiation time of 34 seconds was used in this study (8). Laser irradiation was initiated immediately after the bone defect procedure, and it was performed on days 2, 4, 6, 8, 10 and 12 postsurgery. On day 14 after the injury, rats were euthanized with an IP injection of general anesthetic. The tibiae were defleshed and removed for analysis.
Biomechanical test
Biomechanical properties of the right tibia were determined by an Instron Universal Testing Machine (Model 4444; Instron Corp., Canton, MA, USA). Before the test, bones were thawed at room temperature. The indentation test was performed with a cylindrical indenter of 2.0-mm diameter that was applied to the center of the bone callus, at a constant displacement velocity of 1 mm/min. A 1-N pre-load was applied to avoid specimen sliding. A special device was used to locate the tibiae, prior to submitting their medial surface (repair area) to penetration. The indenter was allowed to penetrate the cavity to a depth of 1.5 mm. From the load–penetration curve, the maximal load (kN) and energy absorption (J) were obtained to a depth of 0.5 mm and 1.0 mm.
After that, the 3-point bending test with a-1 kN load (1-kN load cell; Model 4444; Instron Corp., Canton, MA, USA) was performed. Tibiae were placed on a 3.8-cm metal device, which provided a 1.8-cm distance between the 2 supports. The load cell was perpendicularly positioned in the anteroposterior direction at the exact site of the bone defect. A 5-N pre-load was applied to avoid specimen sliding. Finally, the bending force was applied at a constant deformation rate of 0.5 cm/min until fracture occurred. From the load–deformation curve, the maximum load at failure (N) was obtained.
Statistical analysis
The results are given as means and standard error (SE). The Kruskal-Wallis test was used to compare changes among the groups, and the Mann-Whitney
Results
The indentation test showed that, at a depth of 0.5 mm, the maximal load was higher in the SC and BL120 groups when compared with OC and L120 (Fig. 1A). The maximal load obtained from indentation test at a depth of 1.0 mm was higher in animals treated with Biosilicate® and Biosilicate® associated with laser therapy (BL120) in comparison with the OC and L120 groups (Fig. 1B). Maximal load was lower in the OC and laser-irradiated animals (L120) in comparison with the animals of the SC group (Fig. 1A, B).
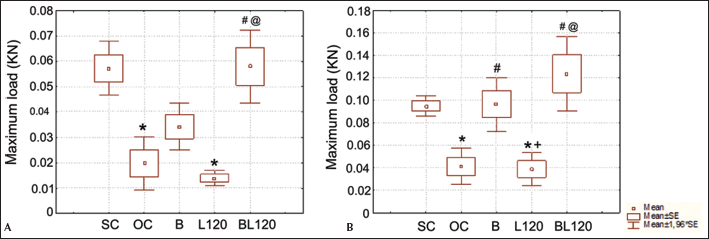
Maximal load obtained from the indentation test at a depth of 0.5 mm (
The energy absorption obtained from the indentation test at a depth of 0.5 mm was significantly lower in the OC, B and L120 groups when compared with osteopenic animals with intact tibiae (group SC; Fig. 2A). Bone callus from animals treated with both laser and Biosilicate® showed higher energy absorption in comparison with bone callus from animals that received only the laser therapy (Fig. 2A). Similarly, the indentation test at a depth of 1.0 mm showed that the energy absorption was higher in animals treated with Biosilicate® and irradiated with laser in comparison with the OC and L120 groups (Fig. 2B).
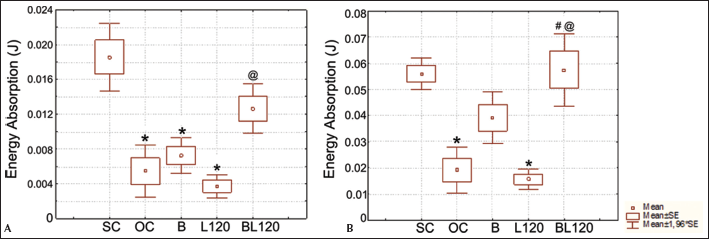
Energy absorption obtained from the indentation test at a depth of 0.5 mm (
The 3-point bending test, however, revealed no significant difference for maximal load or energy of absorption among the groups (Tab. I).
MAXIMAL LOAD AND ENERGY OF ABSORPTION OBTAINED IN THE 3-POINT BENDING TEST
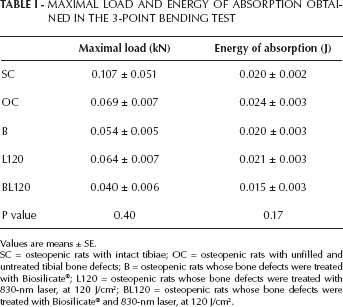
Values are means ± SE.
SC = osteopenic rats with intact tibiae; OC = osteopenic rats with unfilled and untreated tibial bone defects; B = osteopenic rats whose bone defects were treated with Biosilicate®; L120 = osteopenic rats whose bone defects were treated with 830-nm laser, at 120 J/cm2; BL120 = osteopenic rats whose bone defects were treated with Biosilicate® and 830-nm laser, at 120 J/cm2.
Discussion
Since previous in vitro and in vivo studies have demonstrated the high osteogenic potential of Biosilicate® (4, 5, 20), we hypothesized that the positive effects of this biomaterial could be improved by laser irradiation. In this context, the aim of this study was to evaluate if concomitant administration of Biosilicate® and LLLT was able to improve the biomechanical properties of bone callus in osteopenic rats.
We demonstrated that laser therapy, used at the dose of 120 J/cm2, was not effective in improving the biomechanical properties of the tibial callus in osteopenic animals. Interestingly, Biosilicate® and the associated treatment (biomaterial plus laser therapy) could improve biomechanical properties of the bone callus, with an advantage for the associated treatment.
Some studies have showed that laser therapy is able to increase bone cell proliferation and to stimulate cell osteogenic properties (21-23). LLLT creates a number of environmental conditions that accelerate the healing of bone fractures and defects. However, the exact mechanism of action is not fully understood. Probably, laser can stimulate mesenchymal cells or osteoblasts, increasing bone mass deposition and secretion of components of the matrix (24). Barbos Pinheiro et al (7) observed in a histological study that laser therapy accelerated bone repair and increased new bone formation. In addition, Lirani-Galvão et al (25) found that LLLT produced a significant increase in osteoblast numbers in tibial bone defects in rats. Similar findings were observed by Pretel et al (26). Luger et al (8) showed that laser therapy improved maximal load at failure and the structural stiffness of bone. The lack of effects in the laser-irradiated animals of the present study can probably be explained by the experimental postsurgery period (14 days) studied, which was not sufficient to induce modifications in callus bone strength. In addition, the disparate nature of the results may be due to a range of factors, including the treatment regime, the LLLT wavelength employed and/or differences in the model of osteotomy/fracture used.
Interestingly, Biosilicate® was able to increase biomechanical properties within the experimental period of the present study. These results are in agreement with those found by Granito et al, (5), which showed that bones with surgically created bone defects filled with Biosilicate® particulate exhibited large volumes of newly formed bone tissue and enhanced biomechanical properties.
Improved bone load-bearing capacity probably mirrors the huge amount and/or the spacial distribution of the newly formed bone into the defect site, the presence of the biomaterial particulate and the strong bond between them (5). Newly formed bone was observed surrounding Biosicate® and Bioglass® 45S5 particles in a direct contact with the material surface, in the study conducted by Granito et al (5). Moreover, interparticle spaces were also filled with new bone tissue, exhibiting trabeculae bridge formation between some particles, resulting in the glass particle incorporation within the bone structure. Both mineralized and osteoid volumes were increased by Biosilicate®, as well as the number of osteoblasts surrounding bone tissue. Therefore, bioactive glasses have osteogenic properties and can be used as a graft to heal bone defects (27), mainly due to their ability to adapt to the defect shape, their potential to stimulate osteogenesis and their capacity to bond to bone.
Despite the stimulatory effects of LLLT and bioactive materials on bone repair, few reports have studied the association of these therapies. Gerbi et al (28) investigated the influence of LLLT (4 J/cm2, 40 mW, every 48 hours for 15 days) on bone defects grafted with inorganic bovine material, and observed that the repair of the irradiated bone was characterized by an increase in bone formation and in the amount of collagen fibers around the graft. Positive biomodulatory effects may be reached by enhancing osteoblast adhesion and vessel migration toward the material surface. Bossini et al (29) showed that Biosilicate® associated with laser therapy was efficient to enhance bone formation. Moreover, LLLT with either 60 or 120 J/cm2 was able to increase collagen, Cbfa-1, VGEF and COX-2 expression in cells adjacent to Biosilicate®. Therefore, laser therapy and Biosilicate® could together improve the bone repair process by increasing bone formation, as well as COX-2 and Cbfa-1 immunoexpression, angiogenesis and collagen deposition in osteoporotic rats.
It is worth noting that the biomechanical analysis of osteopenic bone defects filled with particulate biomaterial and/or treated with LLLT was an efficient model. OVX is a reliable and widely used experimental model to induce osteopenia in rats, leading to a decrease of the bone mass and an increase of bone fragility (15, 16, 18, 19, 29). Noncritical bone defects were used to overcome the difficult of placing and retaining the biomaterial particles in the defect site. A cohesive mass was formed when Biosilicate® particles were placed in the bleeding site. Consequently, the particles could easily pack into the bone defect site and stay in place. The same phenomenon was reported by Oonishi et al (30) using Bioglass® 45S5 particles to fill noncritical bone defects in femoral condyles of rabbits. According to these authors, the cohesive mass is formed because rapid reactions on the material surface lead to the formation of a gel layer, and consequently, sufficient hemostasis is reached.
Finally, the 3-point bending test was used to determine the mechanical properties of the tibiae. This test revealed no significant difference for maximal load or energy of absorption among the groups, suggesting that the treatments with LLLT and Biosilicate® elicit changes in bone callus, but no effects in bone structure; a finding which is corroborated by a study by Ke et al (31).
In summary, our findings show that although it is well-known that LLLT exerts an osteogenic activity during bone repair, this therapy was not effective in increasing biomechanical properties in the present study. Interestingly, Biosilicate® and the association of Biosilicate® and LLLT increased maximal load and energy indentation.
Laser therapy may represent one of the most promising treatment modalities to improve the positive effects of bioactive materials on bone regeneration. Further long-term studies should be developed to provide additional information concerning the late stages of the bone matrix synthesis and resorption induced by Biosilicate® and laser therapy in osteopenic rats.
