Abstract
Background:
Laryngeal squamous cell carcinoma (LSCC) has a poor prognosis due to recurrence and metastasis. IQ-domain GTPase-activating protein 1 (IQGAP1), a scaffold protein, plays an important role in tumorigenesis and malignant development. In this study, we aimed to explore the role of IQGAP1 in LSCC.
Methods:
Expression of IQGAP1 in human LSCC specimens was assessed by immunohistochemistry. We also evaluated the roles of IQGAP1 in cell proliferation, migration and invasion and epithelial-to-mesenchymal transition (EMT) in Hep-2 cells.
Results:
The expression of IQGAP1 protein was significantly up-regulated in LSCC tissues compared with normal laryngeal tissues (p = 0.002). Furthermore, the knockdown of IQGAP1 in Hep-2 cells inhibited cell growth, migration and invasion. Moreover, we found that IQGAP1 silencing reversed EMT.
Conclusions:
These results show for the first time that IQGAP1 is up-regulated in LSCC tissues and plays an important role in LSCC cell proliferation and invasiveness, which indicates that IQGAP1 could work as an oncogene and may serve as a promising molecular target for treatment of LSCC.
Introduction
Laryngeal squamous cell carcinoma (LSCC) is one of the most frequent malignant neoplasms of the head and neck (1). Despite advancements in surgery, chemotherapy and radiation therapy, patients with LSCC have a poor prognosis because of recurrence and metastasis. To date, molecular mechanisms responsible for LSCC carcinogenesis and development have been comprehensively studied; however, there is still a lack of ideal biomarkers for predicting recurrence and metastasis of cancer. Therefore, much more needs to be done regarding recognition and identification of tumor diagnostic markers and novel therapeutic targets.
IQ-domain GTPase-activating protein 1 (IQGAP1) is a scaffold protein that is able to bind with multiple important cellular proteins through its multiple protein-interacting domains. The interactions of IQGAP1 with those critical molecules have been shown to regulate a multitude of cellular and biological functions (2-11). It has been reported that overexpression of IQGAP1 has been found in several types of human tumors, including ovarian cancer (12), lung cancer (13, 14), pancreatic cancer (15), hepatocellular cancer (16), colorectal carcinoma (17, 18), gastric cancer (19, 20), esophageal squamous cell carcinoma (ESCC) (21) and thyroid cancer (22). In addition, IQGAP1 overexpression is associated with histological differentiation and poorer prognosis (12-15, 18). However, IQGAP1 expression and its role in LSCC are not clear.
In the present study, we investigated IQGAP1 protein expression in LSCC tissues and the biological role of IQGAP1 knockdown by using short hairpin RNA (shRNA) expression plasmid in an LSCC cell line. Our results provide strong evidence that IQGAP1 protein is up-regulated in human LSCC tissues. Moreover, silencing of IQGAP1 expression in Hep-2 cells inhibits cell growth, migration and invasion by reversing the epithelial-to-mesenchymal transition (EMT). The data presented here indicate that IQGAP1 overexpression may play a role in occurrence and malignancy development of LSCC, and targeting IQGAP1 may be a potential strategy for treating LSCC.
Materials and methods
Tissue microarray and immunohistochemistry
The tissue microarray (TMA) included 30 LSCC specimens and 10 normal laryngeal tissues (Cybrdi Inc., Xian, China). The protocol of the study was approved by the ethics committee of Shanxi Medical University. Informed consent was obtained from all patients. Immunohistochemistry was performed as described previously (15). The primary antibody used was mouse-antihuman monoclonal antibody against IQGAP1 (1:1,000; BD Biosciences). IQGAP1 expression was evaluated independently by 2 experienced pathologists, without knowledge of the clinical information. For each section, 10 random non-overlapping fields were observed. The staining intensity was scored as 0 (negative), 1 (weak), 2 (medium) or 3 (strong). The extent of staining was scored as 0 (0%), 1 (1%-25%), 2 (26%-50%), 3 (51%-75%) or 4 (76%-100%), according to the percentage of the positively stained areas in relation to the whole tumor area. The product of the intensity score and the extent score was used as the final staining score. Specimens with scores of ≤4 were defined as negative, and specimens with scores of >4 were defined as positive.
Cell culture and stable shRNA transfection
The human laryngeal carcinoma cell line Hep-2 was purchased from BOSTER, Wuhan, China, and grown in RPMI-1640 medium supplemented with 10% fetal bovine serum (FBS) at 37°C in 5% CO2. Cells were transfected with shRNA against IQGAP1 RNA and control plasmid using Lipofectamine 2000 (Invitrogen). The stable clones were selected by culturing transfected cells in the presence of 400 μg/mL of G418 (Invitrogen) for 14 days.
Western blot analysis
The cells were lysed on ice in lysis buffer. Protein concentrations were determined by the Bradford method. Equal aliquots of protein were loaded on a sodium dodecyl sulfate–polyacrylamide gel. Following electrophoresis, the proteins were transferred to a nitrocellulose membrane (Millipore). The membrane was incubated with primary antibodies against IQGAP1 (1:5,000), E-cadherin (1:500; BD Biosciences), N- cadherin (1:500; BD Biosciences) and vimentin (1:500; Bioworld) overnight at 4°C and then with horseradish peroxidase-conjugated secondary antibody (1:3,000) for 1 hour. The immune complexes were detected using an enhanced chemiluminescence (ECL) reagent. β-Actin (1:5,000; Sigma) served as a loading control.
Reverse transcription polymerase chain reaction
Total RNA was extracted from the cells using Trizol reagent (Invitrogen). RNA (0.5 mg) was used as a template for reverse transcription reaction according to the manufacturer’s instructions (Takara, Dalian, China). The primer sequences for IQGAP1 were 5’-GGAGCACAATGATCCAATCC-3’ (forward) and 5’-ATGGTTCGAGCATCCATTTC-3’ (reverse). The primer sequences for the internal control GAPDH were 5’-GGCCTCCAAGGAGTAAGACC-3’ (forward) and 5’-AGGGGTCTACATGGCAACTG-3 (reverse). The polymerase chain reaction (PCR) conditions were as follows: 5 minutes at 95°C followed by 32 cycles of 94°C for 30 seconds, 58°C for 30 seconds and 72°C for 1 minute, and a final extension at 72°C for 10 minutes.
Immunofluorescence staining
Cells grown on glass coverslips were fixed and permeabilized with ice-cold methanol for 20 minutes. Cells were incubated with primary antibody against IQGAP1 (1:200) overnight at 4°C and subsequently incubated with TRITC-conjugated secondary antibody for 1 hour. The slides were incubated with 4’,6-diamidino-2-phenylindole (DAPI) for nuclear staining for 15 minutes and analyzed with a fluorescence microscope (BX50; Olympus Inc., Tokyo, Japan).
Cell proliferation assay
Cells were seeded in 96-well plates at a density of 2,000 cells per well. Two hundred microliters of 3-(4, 5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT; 5 mg/mL) was added to each well on days 1, 3 and 5. Then cells were incubated at 37°C for 4 hours, and 150 μL of dimethyl sulfoxide (DMSO) was added to solubilize the crystal product for 15 minutes, and absorbance was measured with a microplate reader (Bio-Rad) at 570 nm.
Colony formation assay
Cells were seeded into 60-mm plates at a density of 5,000 single cells and cultured for 2 weeks. Then the cells were fixed with chilled methanol for 30 minutes and stained with crystal violet for 20 minutes for colony counting.
Cell migration and invasion assays in vitro
Cell migration and invasion assays were investigated using Boyden chambers (Neuro Probe, USA). The upper and lower compartments of the chamber were separated with a polycarbonate filter (8-μm pore size). The cells (2 × 105/mL) suspended in 50 μL of serum-free medium were added to the upper chamber, and RPMI-1640 medium supplemented with 10% FBS was added to the lower chamber. For the invasion assay, the membranes were precoated with Matrigel (BD Biosciences). After a 24-hour incubation period, the cells remaining in the upper chamber were removed with a cotton swab, whereas the cells that migrated or invaded through the filters into the lower chamber were fixed and stained using 0.5% crystal violet in methanol. Five fields were randomly selected for photography, and cells were counted and averaged.
Statistical analysis
SPSS 17.0 version was used for statistical analysis. All data are presented as means ± standard deviation (SD). Fisher’s exact test was used to analyze the differential expression of IQGAP1 between laryngeal cancer and normal laryngeal tissue. Student’s t-test was performed to compare the statistical significance of the differences in data from 2 groups. A p value <0.05 was considered to indicate a statistically significant difference.
Results
Expression of IQGAP1 protein was elevated in LSCC tissues
To evaluate the expression level of IQGAP1 protein in LSCC tissues, we used immunohistochemistry to detect IQGAP1 in 30 LSCC and 10 normal laryngeal specimens. The staining of IQGAP1 protein was predominantly localized to the cytoplasm and cell membrane. There was weak (±) or no staining (-) in normal laryngeal tissues, whereas moderate (+) to strong (++) IQGAP1 immunostaining was observed in LSCC cells (Fig. 1). Further analysis showed that positive immunostaining for IQGAP1 protein was detected in 87% (26/30) of LSCC samples. Only 30% (3/10) of cases of normal laryngeal tissues were observed to have positive IQGAP1 expression. The protein levels of IQGAP1 were significantly up-regulated in tumor tissues compared with normal laryngeal tissues (p = 0.002) (Tab. I). The results suggested that IQGAP1 overexpression may play an important role in LSCC occurrence and progression.
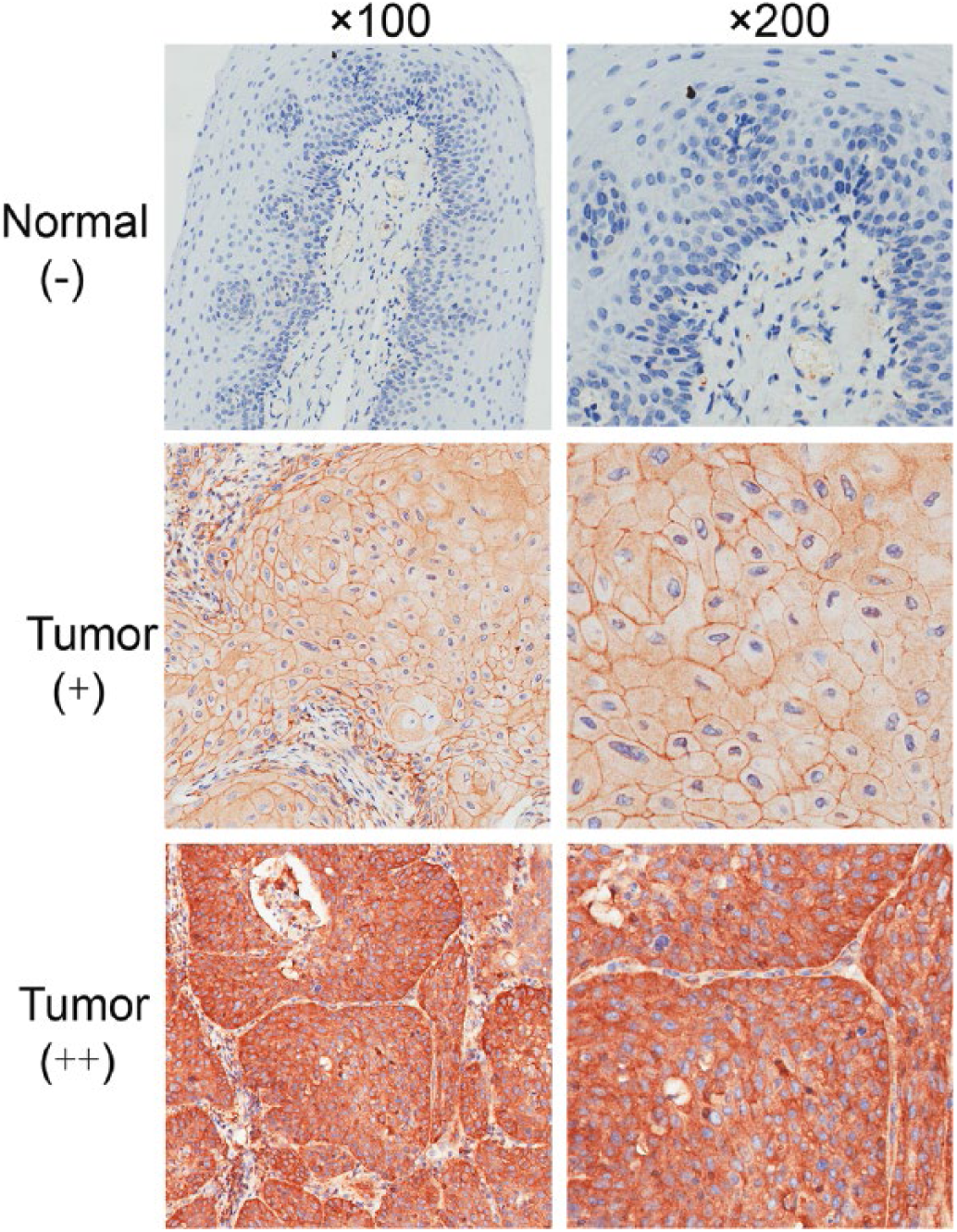
Representative immunohistochemical staining of IQGAP1 in laryngeal squamous cell carcinoma (LSCC) and normal laryngeal tissues. IQGAP1 expression was negative (-) in the normal laryngeal tissues, with moderate (+) and strong (++) staining of cytoplasm and cell membrane in LSCC tissues (original magnification ×100 and ×200).
Expression of IQGAP1 protein in LSCC and normal laryngeal tissues

LSCC = laryngeal squamous cell carcinoma.
Tumor tissue vs. normal tissue.
Stable knockdown of IQGAP1 expression in Hep-2 cells
To demonstrate the role of IQGAP1 in LSCC, Hep-2 cells were stably transfected with IQGAP1 shRNA or negative control shRNA. The efficiency of knockdown was confirmed by Western blot, reverse transcription polymerase chain reaction (RT-PCR) and immunofluorescence staining. Western blot and RT-PCR showed that IQGAP1 protein (Fig. 2A) and messenger RNA (mRNA) (Fig. 2B) expression were inhibited by up to 70%-80% in 2 stable IQGAP1 knockdown cells (termed shIQ1 and shIQ2) compared with control cells (termed shNC). Down-regulation of IQGAP1 was further confirmed by immunofluorescence staining. As shown in Figure 2C, IQGAP1 protein was mainly localized to the cytoplasm of tumor cells, and stain intensity of IQGAP1 showed an obvious decrease in shIQ1 and shIQ2 cells versus in shNC cells. These data confirmed that we successfully obtained stable IQGAP1 knockdown cells.
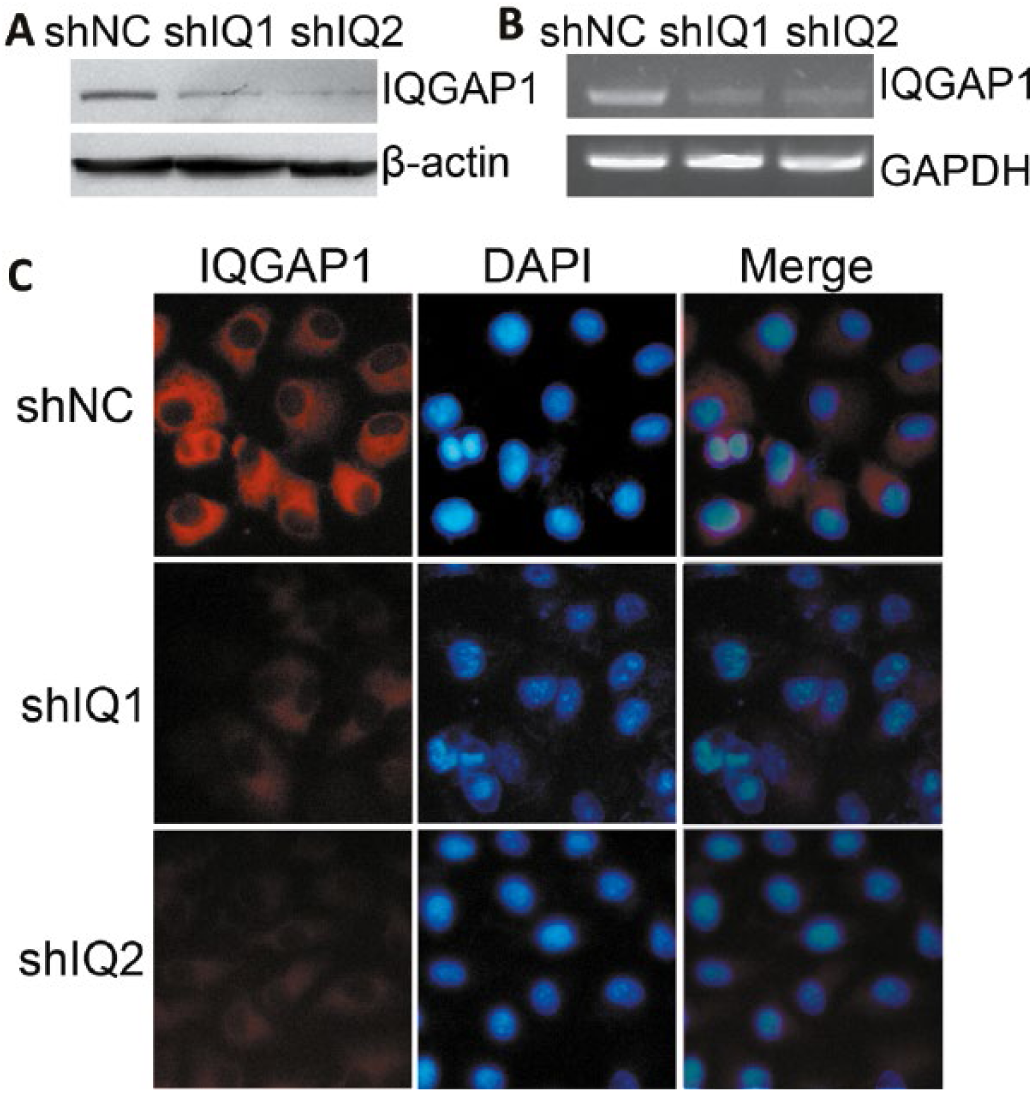
Stable transfection of IQGAP1 shRNA down-regulated IQGAP1 expression in Hep-2 cells. IQGAP1 protein (
IQGAP1 knockdown inhibited cell proliferation in Hep-2 cells
To analyze the function of IQGAP1 in LSCC cells, we examined the role of IQGAP1 knockdown in proliferation of cancer cells. Compared with shNC cells, shIQ1 and shIQ2 cells showed a significant inhibition of cell proliferation as determined by MTT assay (Fig. 3A, p<0.01). Similar results were obtained in the colony formation assay (Fig. 3B). The colony numbers were evidently decreased in shIQ1 and shIQ2 cells compared with shNC cells (Fig. 3C, p<0.01). These data revealed that IQGAP1 knockdown could inhibit proliferation in LSCC cells.
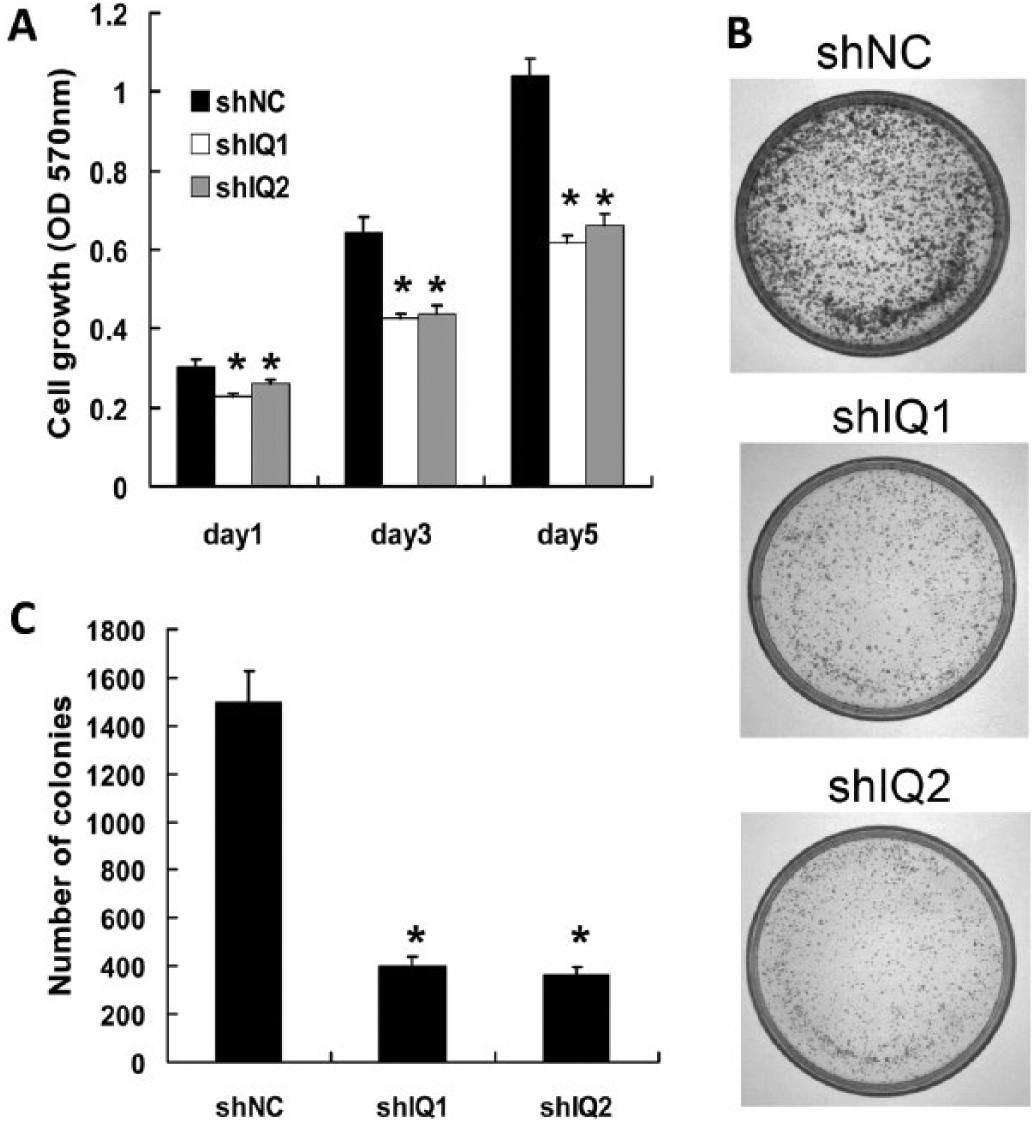
IQGAP1 knockdown inhibited cell proliferation in Hep-2 cells. (
IQGAP1 knockdown inhibited migration and invasion of Hep-2 cells
We further tested whether IQGAP1 knockdown would affect migration and invasion capabilities of Hep-2 cells. As shown in Figure 4A-D, the numbers of migrated (Fig. 4A, C) and invaded (Fig. 4B, D) cells was significantly reduced in shIQ1 and shIQ2 cells compared with shNC cells after 24 hours of incubation (p<0.01). These results strongly suggested that IQGAP1 plays an important role in malignant development of LSCC cells.
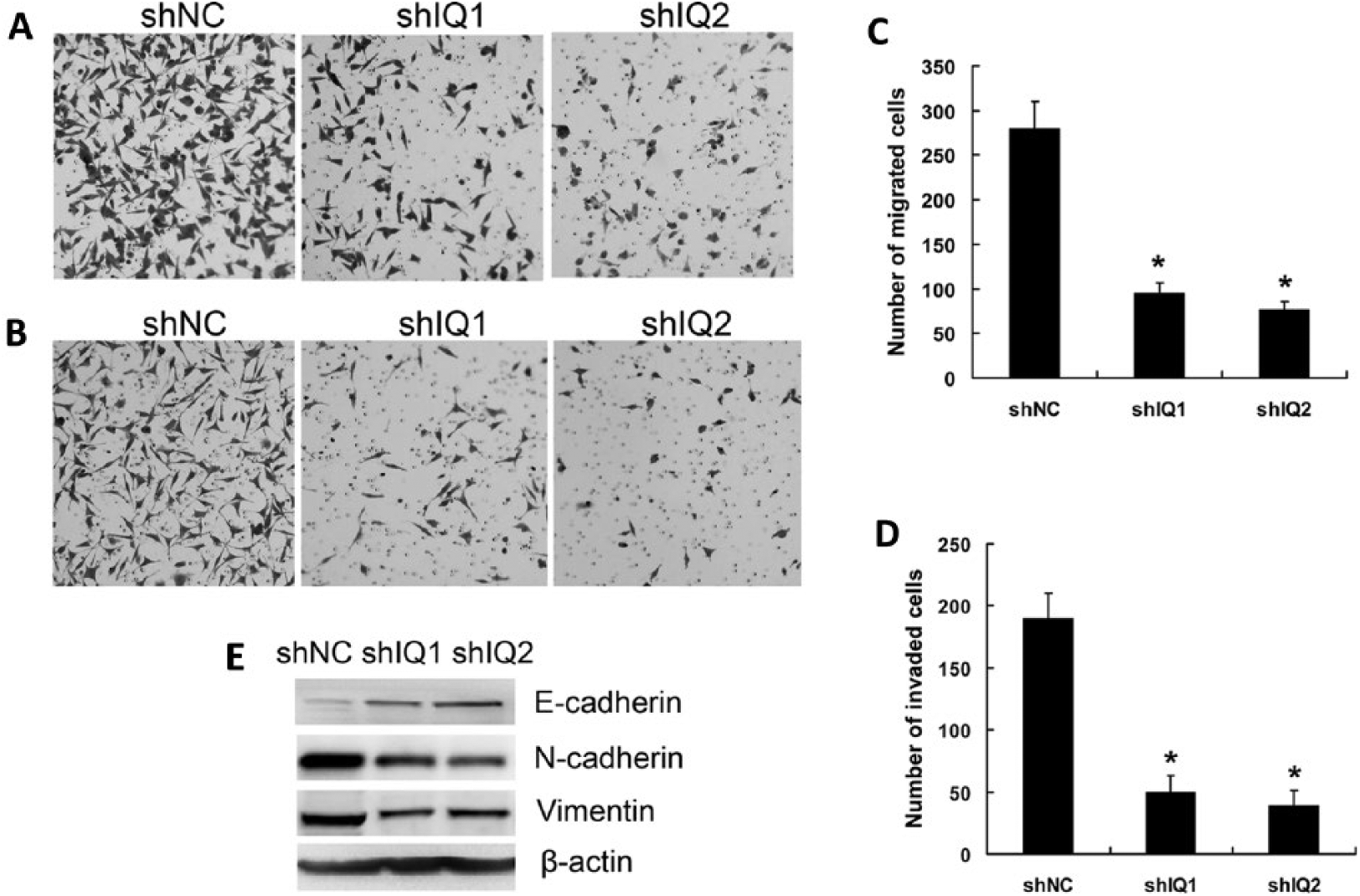
IQGAP1 Knockdown inhibited migration and invasion of Hep-2 cells by reversing epithelial- to-mesenchymal transition (EMT). Representative photographs of the migration (
IQGAP1 knockdown inhibited EMT
EMT plays an important role in the invasion and metastasis of various tumors (23). Because the down-regulation of IQGAP1 inhibited migration and invasion of LSCC cells, we sought to determine whether IQGAP1 suppression was sufficient to reverse EMT, by examining expression of epithelial and mesenchymal protein markers. Western blot analysis showed that knockdown of IQGAP1 in LSCC cells clearly up-regulated expression of epithelial marker E-cadherin, and significantly decreased expression of mesenchymal markers N-cadherin and vimentin (Fig. 4E). These data indicated that IQGAP1 knockdown attenuated EMT in LSCC cells.
Discussion
Most investigations have shown that development of LSCC is clearly a multistep process and associated with many molecular changes. It has been shown that tumor-associated genes, such as TGF-β receptor-II, CTNNA2 and CTNNA3 are frequently mutated in LSCC (24, 25). In addition, overexpression of the cellular protooncogenes STK15, OPN and cyclin D1 is found in this malignant disease (26-28). Therefore, abnormalities in oncogenes and tumor suppressor genes may contribute to LSCC progression.
Up to now, IQGAP1 overexpression has been widely shown to exist in many cancers. However, the biological roles of IQGAP1 in human LSCC are still unclear. In this study, we found expression of IQGAP1 protein was significantly up-regulated in LSCC tissues compared with normal laryngeal tissues, which is consistent with previous findings in most tumors (12-22). Then, we demonstrated a possible role for IQGAP1 in LSCC by RNA interference (RNAi), which has been widely used as an experimental tool in studying gene function. The results showed that the IQGAP1 protein and mRNA expression levels were reduced significantly after stable transfection with IQGAP1 shRNA. IQGAP1 knockdown clones were further confirmed by immunofluorescent staining. The results showed that IQGAP1 was in the cytoplasm of the tumor cells, and its expression was down-regulated by the specific shRNA against IQGAP1 in Hep-2 cells. Next, we performed MTT and colony formation assays to evaluate the effects of IQGAP1 knockdown on cell proliferation. The data demonstrated that IQGAP1 silencing led to a significant decrease of cell proliferation. This finding is consistent with the recent reports that IQGAP1 knockdown can inhibit cell proliferation of thyroid cancer (29), glioma (30), pancreatic cancer (31) and ESCC (21). These data suggest that IQGAP1 overexpression has an oncogenic role in LSCC.
To further understand the role of IQGAP1 in LSCC, we performed cell migration and invasion assays in vitro to evaluate the effects of IQGAP1 silencing on migratory and invasive potential of Hep-2 cells. Here we showed that knockdown of IQGAP1 expression inhibited cell migration and invasion ability. The result is consistent with several previous studies that have shown that IQGAP1 plays important roles in migration, invasion and metastasis of ESCC (21), thyroid cancer (22), ovarian carcinoma (13) and colorectal carcinomas (18, 19). Further studies are required to elucidate the molecular mechanisms of IQGAP1 affecting invasion and metastasis of LSCC.
EMT is a critical event in tumor development, which is characterized by loss of epithelial cell adhesion, and acquiring more migratory and invasive features leading to cancer cells departing from the primary tumor and invading surrounding tissue and distant organs (23). Therefore, elucidating the molecular mechanisms of EMT is important for understanding cancer invasion and metastasis, which may lead to the development of novel interventions for cancer metastasis. Activation of EMT is associated with altered expression of many proteins. The cell–cell adhesion between epithelial cells is principally mediated by E-cadherin. Reduction or loss of E-cadherin is considered a hallmark event of EMT. Concomitant with the loss of E-cadherin, cells gain mesenchymal properties that become apparent by the expression of mesenchymal markers such as N-cadherin and vimentin (23).
In this study, we showed that IQGAP1 knockdown up-regulated E-cadherin expression, and down-regulated vimentin and N-cadherin expression. This result indicated that IQGAP1 knockdown could inhibit the EMT process, thereby reducing the migration and invasion of tumor cells. In agreement with our results, Kuroda et al have reported that IQGAP1 overexpression reduced E-cadherin-mediated cell–cell adhesion by interacting with β-catenin in EL cells (3). It has also been reported that inhibition of IQGAP1 by RNAi reduces E-cadherin in MDCKII (32), ESCC (21) and thyroid cancer cells (22). Taken together, these data indicate that the inhibition of invasion and metastasis by IQGAP1 silencing might provide new treatment alternatives for cancer patients.
The cumulative evidence strongly shows that IQGAP1 is frequently up-regulated in many cancers. As a multifunctional scaffold protein, IQGAP1, with its interacting proteins, controls diverse signal transduction, which could lead to tumorigenesis and metastasis. For example, IQGAP1 interacts with Rac1, E-cadherin, β-catenin, RAF, MEK1/2 and ERK1/2 MAPK cascade kinases, which possess well-defined tumorigenic roles (2-11). Therefore, IQGAP1 is a potential tumor-required scaffold protein. Consistent with these observations, we found that IQGAP1 was overexpressed in LSCC and could regulate tumor cell proliferation and invasion. These data should lead to considerable attention to targeting IQGAP1 as a novel strategy to inhibit oncogenic signaling in cancer.
In conclusion, the expression of IQGAP1 protein is up-regulated in human LSCC. Knockdown of IQGAP1 suppresses proliferation, migration and invasion of LSCC cells by reversing EMT. To the best of our knowledge, this is the first report to elucidate a role for IQGAP1 in LSCC, suggesting that IQGAP1 has an oncogenic role in LSCC and might serve as a potentially valuable target for novel treatments in LSCC.
Footnotes
Disclosures
Financial support: This study was supported by grants from the National Natural Science Foundation of China (81372676) and the Postdoctoral Science Foundation of China (2014M551058).
Conflict of interest: The authors declare they have no conflicts of interest.
