Abstract
BACKGROUND:
It has been documented that transient receptor potential melastatin 7 (TRPM7) plays a pivotal role in the development of multiple cancers. However, the role of TRPM7 in human colorectal cancer (CRC) is poorly understood. Therefore, the aim of this study was to investigate the expression and significance of TRPM7 in CRC.
METHODS:
In this study, TRPM7 expression was first investigated in Gene Expression Omnibus (GEO), and then validated it with the data from our medical center. CCK-8, colony survival, transwell, and flow cytometry assays were employed to evaluate the effects of TRPM7 knockdown on the CRC cell proliferation, migration, and invasion, as well as cell cycle and apoptosis.
RESULTS:
We observed markedly increased TRPM7 expression in CRC tissues. CRC patients with high expression of TRPM7 suggested deeper tumor infiltration, positive lymph node metastasis, distant metastasis, and advanced clinical stage. In addition, TRPM7 was also overexpressed in CRC cell lines. Downregulated TRPM7 in vitro suppressed CRC cell proliferation, migration, and invasion, as well as triggered cell cycle arrest at the G0/G1 phase, reduced the S phase, and promoted apoptosis. Importantly, decreased TRPM7 in CRC cells reversed the epithelial-mesenchymal transition (EMT) status, accompanied by downregulation of N-cadherin and upregulation of E-cadherin.
CONCLUSION:
Our study indicated that the expression of TRPM7 was positively correlated with tumor infiltration, lymph node metastasis, distant metastasis and clinical stage of CRC. Besides, decreased TRPM7 in vitro inhibited CRC cell proliferation, migration and invasion by modulating EMT.
Introduction
Colorectal cancer (CRC) is one of the leading causes of cancer-associated mortality, resulting in a heavy healthcare burden in China [1]. Although the survival of patients with clinically localized CRC has been steadily enhanced by effective surgical and chemotherapeutic treatments, the prognoses of those with metastatic CRC remain unsatisfactory [2]. The alteration of oncogenes and tumor suppressor genes mainly initiates multiple biological events involved in CRC progression [3]. Thus, it is of great significance to comprehensively understand the molecular mechanisms underlying the pathogenesis of CRC.
Transient receptor potential melastatin (TRPM) is a superfamily of cation-selective channels located on the cell membrane, consisting of eight members, namely TRPM1 to TRPM8 [4]. In our previous study, gene expression microarray analysis was used to analyze differentially expressed genes (DEGs) between CRC and paired adjacent normal tissues (PANTs), which revealed high expression levels of the TRPM7 gene in colorectal cancer (CRC) [5]. TRPM7 is a bifunctional ion channel with a functional serine/threonine-specific protein kinase domain, exhibiting abundant expression in various human cells [6]. As a protein kinase enzyme, TRPM7 phosphorylates itself and other downstream substrates. The activity of TRPM7 is important for the function of ion channels and various cellular processes, including growth, migration, and survival. As an ion channel, it is highly permeable to both divalent (Ca
Epithelial-mesenchymal transition (EMT) is a biological process comprising the trans-differentiation of epithelial cells to mesenchymal cells. Notably, EMT is pathologically involved in fibrosis and carcinogenesis. During EMT, cells gradually relinquish their epithelial characteristics and, simultaneously, acquire mesenchymal properties; this process is characterized by decreased expression of E-cadherin and increased expression of N-cadherin [13, 14, 15]. Additionally, EMT enables cell motility and invasiveness. Considerable research has demonstrated EMT as a critical step for invasion and metastasis in various cancers, including CRC [13, 16, 17].
To this end, the expression of TRPM7 was first examined in the Gene Expression Omnibus (GEO) database, followed by further confirmation at our institute. Subsequently, TRPM7 expression was assessed in clinical samples, followed by an investigation of the relationship between TRPM7 expression and clinicopathological factors. Moreover, we evaluated the effects of TRPM7 on proliferation, migration, and invasion in CRC cells, to further assess the involvement of EMT in these processes.
Materials and methods
Microarray data processing
Microarray datasets of CRC, including GSE41328, GSE39582, and GSE32323, were downloaded from the GEO database (
List of primary antibodies
List of primary antibodies
This study was strictly performed according to the Declaration of Helsinki; it was approved by the Research Ethics Committee of Zhongnan Hospital of Wuhan University (approval number: 201511). All CRC patients provided written informed consent after sufficient communication. Clinical samples were obtained from patients who had been pathologically diagnosed with CRC and underwent surgery at Zhongnan Hospital of Wuhan University.
The tissue microarray was prepared with six pairs of CRC tissue and PANT (more than 5 cm away from the cancer tissue) collected after surgery that took place between June 2012 and November 2012 [5]. A total of 103 CRC patients underwent surgical resection between January 2015 and June 2016; subsequently, their clinicopathological data were collected.
Cell culture
Human CRC cell lines HT29, DLD1, SW480, LOVO, and HCT116 and the human normal intestinal mucosa cell line NCM460 were commercially obtained from the Cell Center of the Chinese Academy of Sciences (Shanghai, China). The NCM460, SW480, HT29, DLD1, and HCT116 cells were maintained in Dulbecco’s modified Eagle’s medium (DMEM); the LOVO cells were cultured in Ham’s F-12K (Kaighn’s) medium at 37
RNA isolation and RT-qPCR
RNA isolation and subsequent RT-qPCR were conducted in line with previously described methods [19]. The primer sequences for TRPM7 were: 5
siRNA transfection
siRNA was transfected into the TRPM7-expressing cell lines HT29 and SW480 using lipoJetTM (SignaGen, China) in line with standard protocols. TRPM7-specific siRNA (siTRPM7) and control siRNA (siCON) were synthesized by GenePharma (Shanghai, China). The siTRPM7 sequence was 5
Cell viability
Cell viability was analyzed using the Cell Counting Kit-8 (CCK-8 kit, Dojindo, Japan) in line with previously described methods [17].
Clonogenic survival
A clonogenic survival assay was conducted as previously described [19].
Flow cytometry
Cell cycle and apoptosis assays were conducted via flow cytometry in accordance with previous protocols [19].
Transwell migration and invasion assays
Transwell chambers (Corning, USA), with an 8.0-
Increase of TRPM7 expression in GEO and TCGA databases in CRC. (A) DEGs in CRC tissues vs PANTs across each independent dataset. Each column represents a sample and each row represents the expression level of a given mRNA. The color scale represents the raw Z score ranging from blue (low expression) to red (high expression). (B) Venn diagrams of overlapping DEGs between GSE32323, GSE39582 and GSE41328.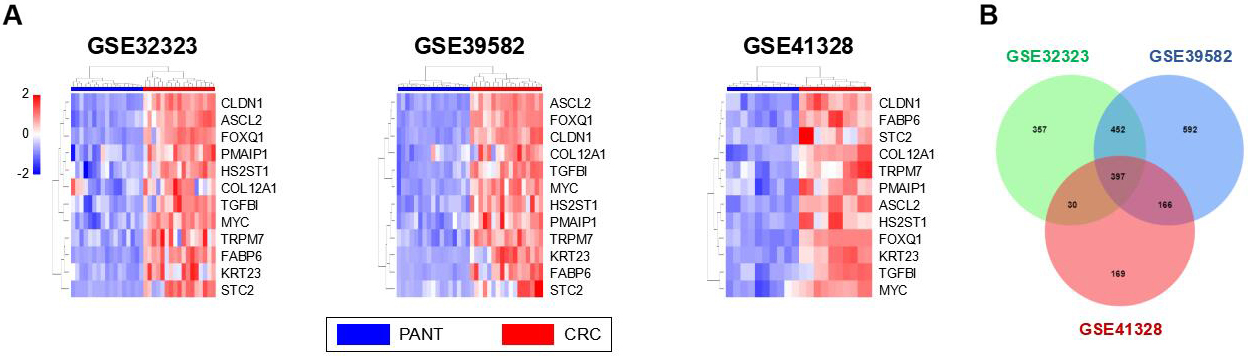
Total proteins were extracted from CRC tissues and cells using a RIPA lysis buffer containing a protease inhibitor (Sigma-Aldrich, USA). The specific procedure followed for the western blot analysis was the same as that used in our previous study [19]. The primary antibodies are listed in Table 1, and the appropriate horseradish peroxidase-labeled IgG secondary antibodies (1:10000, LK2001 or LK2003, Sungene Biotech, China) were used. The Enhanced Chemiluminescence kit (Bio-Rad, USA) was used to visualize the protein bands, followed by quantification with ImageJ software (version 1.46; NIH, Bethesda, MD, USA).
Immunofluorescence staining
Immunofluorescence (IF) staining was performed in line with standard protocols. CRC tissue specimens were fixed with 4% PFA at 4
High expression of TRPM7 validated by our own database in CRC. (A) DEGs in six pairs CRC and PANT. (B) Representative immunofluorescence staining of TRPM7 (red) in CRC tissues compared with that observed in PANTs. Nuclei were stained using 4’,6-diamidino-2-phenylindole (blue). (C) The levels of TRPM7 mRNA in 15 CRC tissues and adjacent normal tissues were detected using qRT-PCR. The housekeeping gene GAPDH was used as an internal control. *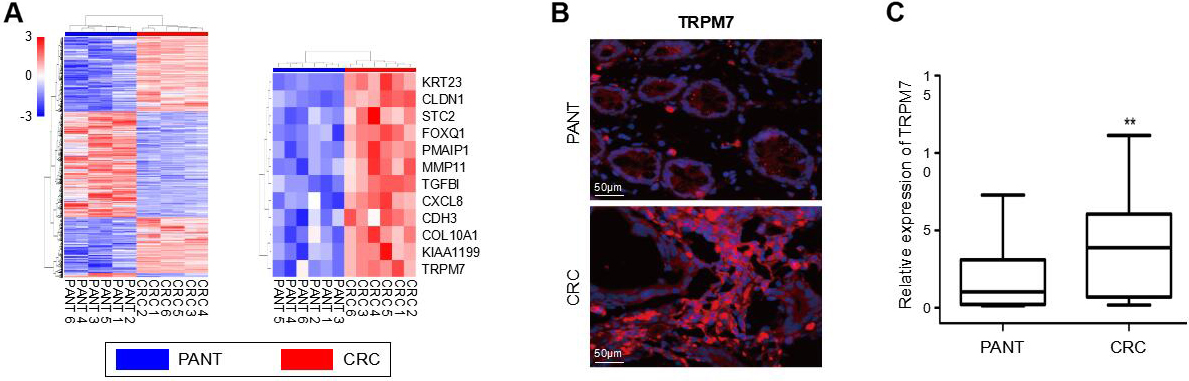
TRPM7 was upregulated in CRC cells and TRPM7 deficiency inhibited the proliferation, migration and invasion of CRC cells. (A) The mRNA levels of TRPM7 in five human CRC cell lines and the human normal intestinal mucosa cell line NCM460 were detected using qRT-PCR. **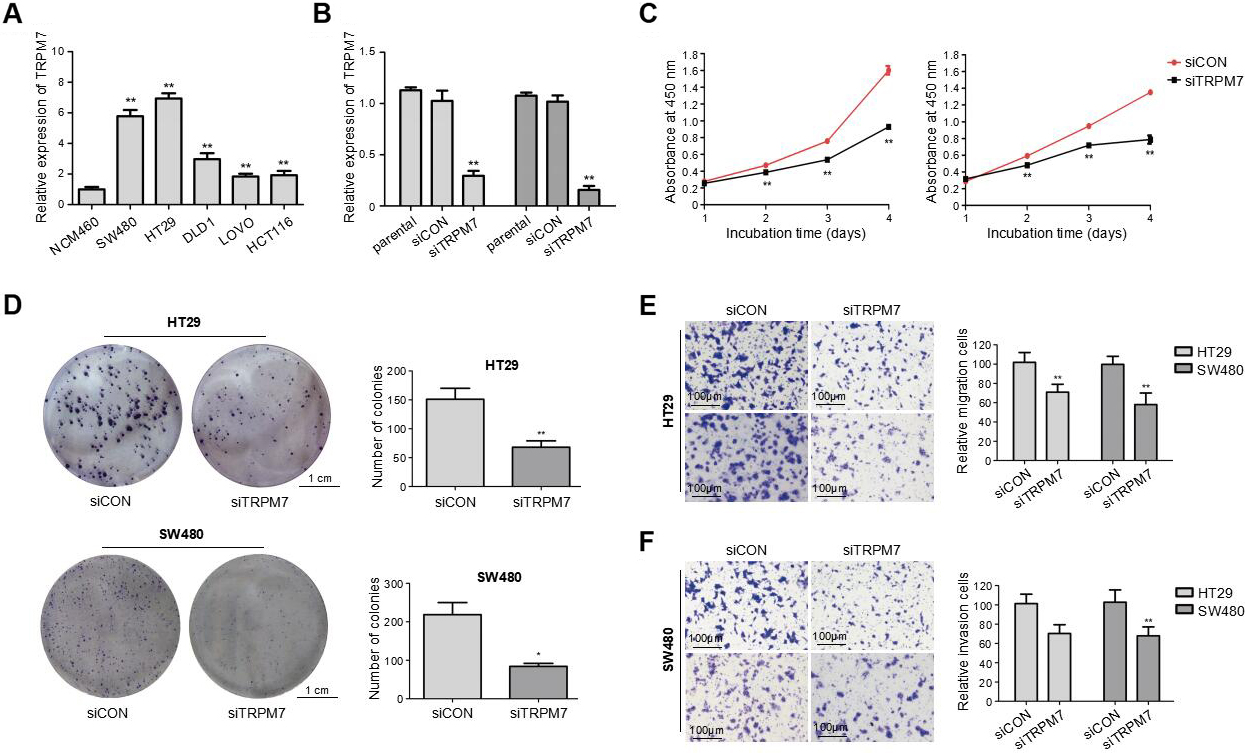
Statistical analyses were performed using R software (version 3.5.1, Vienna, Austria) [21, 22]. Data are shown as the mean
Results
Microarray datasets and differentially expressed genes
The GPL570 microarray platform was utilized for the mRNA expression profile datasets, including GSE41328, GSE39582, and GSE32323. In total, 44 CRC tissue samples and 44 PANT samples were analyzed. Using R software, 762, 1607, and 123 DEGs were identified among CRC tissues and PANTs from GSE41328, GSE39582, and GSE32323 datasets, respectively (
Correlations between TRPM7 expression level and clinicopathological variables of 103 CRC patients
Correlations between TRPM7 expression level and clinicopathological variables of 103 CRC patients
Note: Bold italics indicate statistically significant values (
In six pairs of specimens, microarray data from our medical center revealed the significant upregulation of TRPM7 gene expression in CRC tissues (Fig. 2A). Consistently, IF staining showed the upregulated expression of TRPM7 in CRC tissues, compared to the expression in PANTs (
Associations of TRPM7 expression with clinicopathological parameters in CRC
The correlation between TRPM7 expression and the clinicopathological features was analyzed in 103 CRC patients. CRC patients were categorized into two groups: those with low TRPM7 expression (
TRPM7 overexpression in CRC cell lines
Consistent with the results observed for the CRC tissue samples, mRNA expression of TRPM7 was significantly increased in the five CRC cell lines, especially in the HT29 and SW480 cells, in comparison with that in the human normal intestinal mucosa cell line NCM460 (
TRPM7 Knockdown triggered cell cycle arrest and promoted apoptosis in CRC cells. (A) Representative flow cytometry analysis of the cell cycle in CRC cells (HT29 and SW480) after transfection with siTRPM7 and siCON. Percentages (%) of cell populations at different stages of the cell cycle in HT29 and SW480 cells were statistically analyzed. *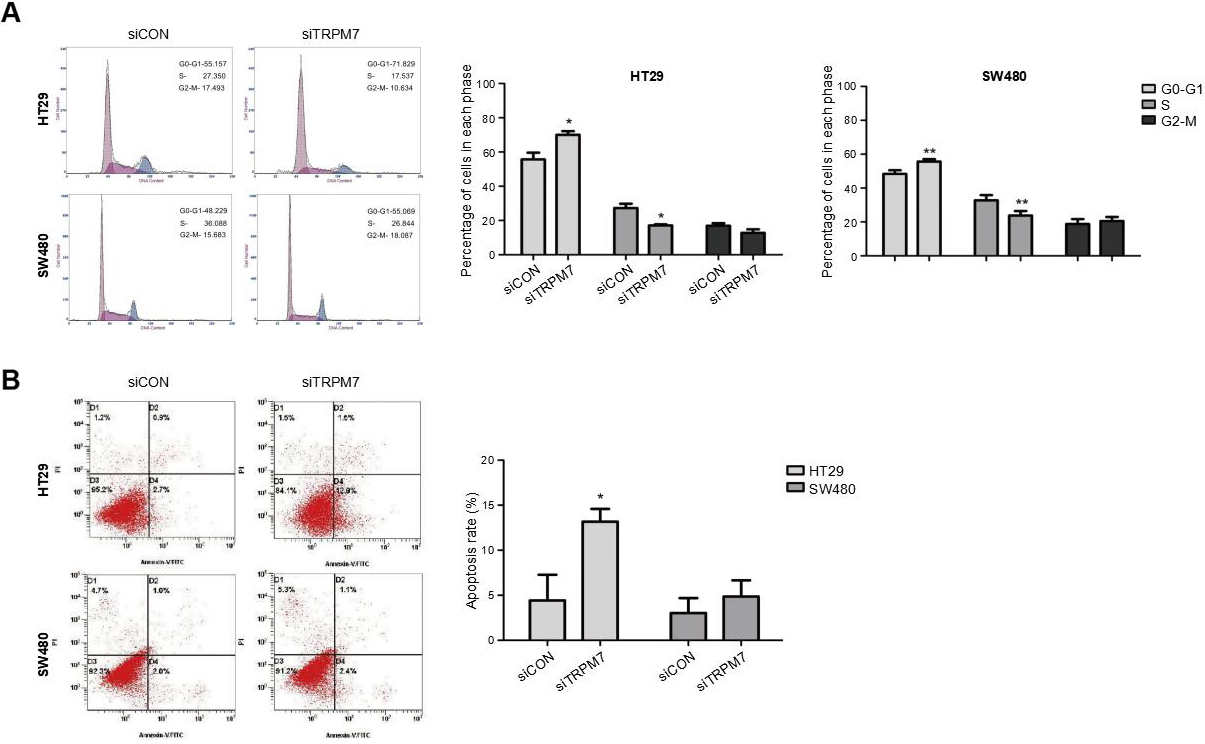
TRPM7 was related to the EMT pathway. (A) Representative immunofluorescence staining of TRPM7, E-cadherin, and N-cadherin (red) in CRC tissues compared with that observed in adjacent normal tissues. In CRC tissues, the expression of TRPM7 and N-cadherin was increased, whereas that of E-cadherin was reduced. Nuclei were stained using 4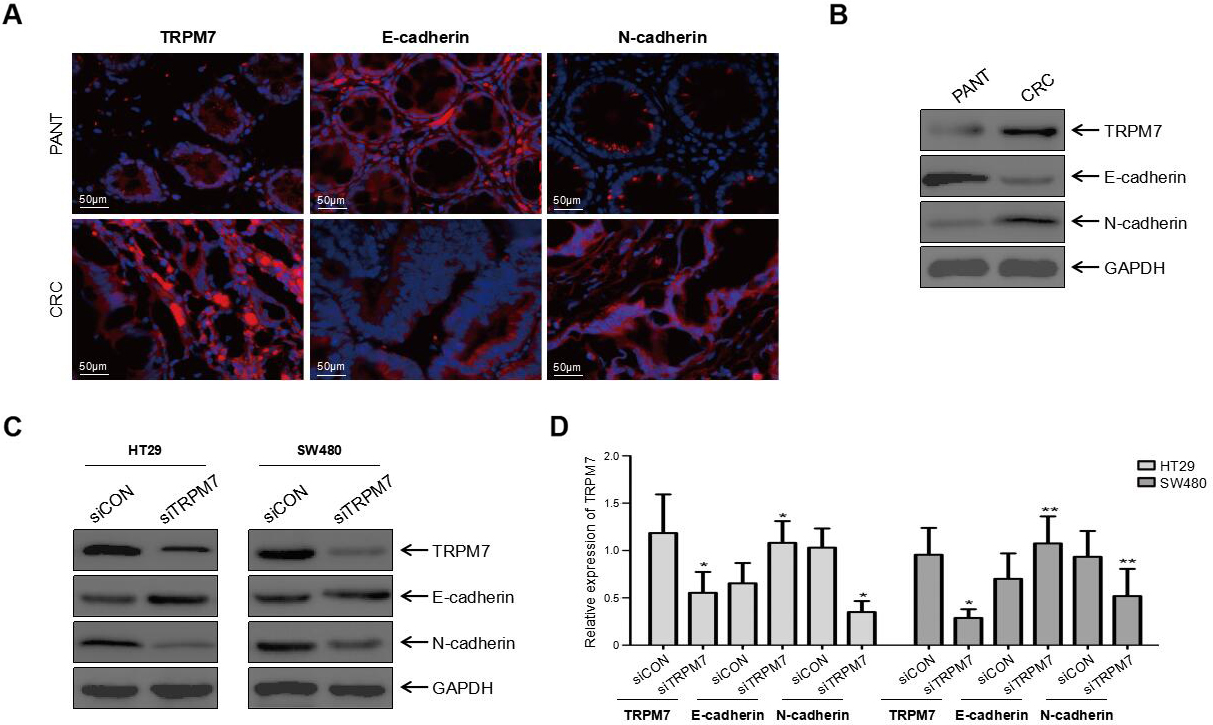
siRNA transfection was performed to knockdown TRPM7 in HT29 and SW480 cells, to investigate the functions of TRPM7 in CRC cell lines. The efficiency of the knockdown was confirmed through qRT-PCR (
Silencing of TRPM7 suppressed the proliferation of CRC cells
Results of the CCK-8 assay revealed that TRPM7 knockdown significantly suppressed the viability of CRC cells (HT29 and SW480) transfected with TRPM7-siRNA, in comparison with those transfected with siCON (
Decreased TRPM7 inhibited migration and invasion of CRC cells
Results from the Transwell migration assay suggest that siTRPM7 transfection drastically reduced the migration of CRC cells (
Downregulation of TRPM7 triggered cell cycle arrest at the G0/G1 phase, reduced the number of the cells in the S phase, and promoted apoptosis
To identify the mechanism by which TRPM7 knockdown attenuated cell proliferation, flow cytometry was utilized for cell cycle and apoptosis analysis in HT29 and SW480 cells transfected with siTRPM7. Consequently, TRPM7 deficiency triggered significant cell cycle arrest at the G0/G1 phase and reduced the number of cells in the S phase in HT29 and SW480 cells compared to the control group (
TRPM7 deficiency reversed the dysregulation of EMT markers
Epithelial-mesenchymal transition has been shown to accelerate the metastasis of CRC cells. Therefore, both IF and western blot analysis were utilized to investigate EMT markers in CRC tissues and cells, including E-cadherin and N-cadherin. In contrast to the downregulation of E-cadherin and upregulation of N-cadherin in CRC tissues (
Discussion
The TRPM7 gene has been implicated in numerous types of human malignancies [23], including esophageal [8], breast [9], gastric [10], bladder [11], and prostate cancers [12]. Moreover, a recent study conducted by Huang et al. [24] has revealed that the inhibition of TRPM7 suppressed CRC cell proliferation, implying that TRPM7 may serve as a cancer-promoting gene in CRC. However, there is no adequate evidence concerning the functional role of TRPM7 in CRC. Consequently, the present study was designed to explore the role of TRPM7 in CRC progression and the underlying mechanism. Our study revealed the significantly upregulated expression of the TRPM7 gene in CRC tissues in both the GEO datasets and our microarray data. Additionally, qRT-PCR data clearly demonstrated the significant upregulation of mRNA levels of TRPM7 in 15 CRC tissues in comparison with those of PANTs. These results were further validated by western blot analysis and IF staining. In exploration of the possible correlation between the mRNA expression of TRPM7 and clinicopathological features of CRC patients (Table 2), TRPM7 expression was positively related to tumor infiltration, clinical stage, lymph node involvement, and distant metastasis. However, TRPM7 expression was not significantly associated with gender, age, tumor location, tumor size, serum CEA, vascular invasion, or lymphatic invasion. Our results support the hypothesis that TRPM7 may serve as an oncogene in CRC.
Additionally, we further detected the mRNA levels of TRPM7 in different cell lines; these were higher in the five CRC cell lines than in the NCM460 cell line. In consideration of the dramatically upregulated TRPM7 expression in HT29 and SW480, the two cell lines were chosen for constructing TRPM7-deficient CRC cell lines using siRNA transfection. Consistent with the findings of a previous report [24], results from CCK-8 and clonogenic survival assays revealed that silencing of TRPM7 reduced the proliferation of CRC cells. Cell proliferation is largely affected by the cell cycle as well as apoptosis [25]. Thus, flow cytometry was used to analyze the cell cycle and apoptosis. As predicted, TRPM7 deficiency triggered significant cell cycle arrest at the G0/G1 phase and reduced the number of cells in the S phase in HT29 and SW480 cells. Moreover, the downregulation of TRPM7 induced the apoptosis of HT29 cells.
TRPM7 has been revealed to modulate the migration and invasion of human carcinoma cells [8, 9, 11, 12], indicating a potential role in cancer metastasis. Therefore, Transwell migration and invasion assays were performed to assess the impact of TRPM7 silencing on CRC cell migration and invasion. The results indicated that the knockdown of TRPM7 reduced cell migration and invasion in CRC cells. A plethora of studies has reported that EMT expedites cancer cell migration and invasion [12, 16, 17]. To unveil the mechanism underlying the regulatory role of TRPM7 in mediating CRC cell migration and invasion, IF staining and western blot analyses were performed to detect the expression of EMT markers, including E-cadherin and N-cadherin, in CRC tissues and cells (i.e., HT29 and SW480). In contrast to the downregulation of E-cadherin and upregulation of N-cadherin in CRC tissues, western blot analyses revealed that silencing of TRPM7 increased E-cadherin expression and reduced N-cadherin expression in CRC cells. These results suggested that TRPM7 deficiency may reverse the dysregulation of EMT markers. Further studies are warranted to address the limitations of the present study. For instance, investigation into the potential correlation between TRPM7 expression and the prognosis of CRC patients requires a longer follow-up time and a larger sample size. Moreover, pathways involved in the TRPM7 functional mechanism in CRC progression should also be elucidated in-depth.
Conclusion
In summary, our study revealed that high TRPM7 expression in CRC tissues is significantly related to deeper tumor infiltration, an advanced clinical stage, positive lymph node involvement, and distant metastasis. Moreover, silencing of TRPM7 in vitro suppressed CRC cell proliferation via the induction of cell cycle arrest at the G0/G1 phase, reduction in the number of cells in the S phase, and promotion of apoptosis. Furthermore, downregulation of TRPM7 in CRC cells inhibited cell migration and invasion by reversing EMT in vitro.
Footnotes
Acknowledgments
This work was supported by National Natural Science Foundation of China (81072152, 81770283), Natural Science Foundation of Hubei Province (2015CFA 027), Research Foundation of Health and Family Planning Commission of Hubei Province (WJ2015MA010, WJ2017M249), Clinical Medical Research Center of Peritoneal Cancer of Wuhan (2015060911020462) and Subsidy Project of No. 1 Hospital of Lanzhou University (ldyyyn2018-13).
Conflict of interest
The authors report no conflicts of interest in this work.
